Abstract
We have previously shown that populations of skeletal muscle-derived stem cells (MDSCs) exhibit sex-based differences for skeletal muscle and bone repair, with female cells demonstrating superior engrafting abilities to males in skeletal muscle while male cells differentiating more robustly toward the osteogenic and chondrogenic lineages. In this study, we tested the hypothesis that the therapeutic capacity of MDSCs transplanted into myocardium is influenced by sex of donor MDSCs or recipient. Male and female MDSCs isolated from the skeletal muscle of 3-week-old mice were transplanted into recipient male or female dystrophin-deficient (mdx) hearts or into the hearts of male SCID mice following acute myocardial infarction. In the mdx model, no difference was seen in engraftment or blood vessel formation based on donor cell or recipient sex. In the infarction model, MDSC-transplanted hearts showed higher postinfarction angiogenesis, less myocardial scar formation, and improved cardiac function compared to vehicle controls. However, sex of donor MDSCs had no significant effects on engraftment, angiogenesis, and cardiac function. VEGF expression, a potent angiogenic factor, was similar between male and female MDSCs. Our results suggest that donor MDSC or recipient sex has no significant effect on the efficiency of MDSC-triggered myocardial engraftment or regeneration following cardiac injury. The ability of the MDSCs to improve cardiac regeneration and repair through promotion of angiogenesis without differentiation into the cardiac lineage may have contributed to the lack of sex difference observed in these models.
Introduction
Heart disease is the leading cause of death in the world, and a recent estimate from the American Heart Association stated that 1 in 3 American adults have a form of heart disease (30). Heart disease includes both congenital and acquired cardiomyopathies as well as other types of damage, including ischemic injury from myocardial infarction. For most patients, the final therapeutic option for end-stage heart failure is heart transplantation, although this is not always a viable option due to low organ availability and the comorbidities associated with long-term immunosuppression (3). Limited donor organ availability and the low regenerative capacity of the adult heart following injury (8) has stimulated both basic science research and clinical trials to validate tissue engineering approaches to repair the injured and failing human heart.
Cellular cardiomyoplasty is an emerging therapeutic option that has gained recognition as a strategy to repair damaged myocardium and potentially reverse many debilitating heart diseases (3). To date, a wide variety of cell types have been examined for suitability for cardiac repair, including skeletal myoblasts (31), smooth muscle cells (11), fibroblasts (14), resident myocardial progenitors (20,26), mesenchymal stem cells (1,17), hematopoietic stem cells (2,22), and embryonic stem cells (19). There have also been numerous clinical trials using cell transplantation to repair the ischemic heart, but there were low efficiencies of transplanted cell survival and concerns regarding increased risk of clinical arrhythmias, which continue to be under investigation.
Recently a population of murine muscle-derived stem cells (MDSCs) has been shown to have an improved regenerative capacity in bone and skeletal/cardiac muscles when compared to satellite cells or myoblasts (25,29). MDSCs are a unique cell population whose characteristics, including marker profile, proliferation and differentiation kinetics, and regenerative capacity, are distinct from myoblasts (9,15). After implantation into ischemic hearts, MDSCs display high levels of engraftment that persist over time, induce neoangiogenesis, prevent cardiac remodeling, and elicit significant improvements in cardiac function (25). A human counterpart to MDSCs has been isolated based on cell surface markers with the human muscle-derived cells coexpressing myogenic and endothelial markers. These cells have been shown to be better than purely myogenic or endothelial cells for both skeletal muscle repair (37) and cardiac repair following myocardial infarction (23).
The variable results noted following cellular transplantation in most organ systems could be due to a variety of factors, including differences in cell populations and treatment regimens, as well as other intrinsic characteristics, including the sex of the cells and recipient. The association between sex and cardiac morbidity and mortality is well established. The prognosis for males with heart disease is worse than in premenopause females (16, 36), and cardiovascular disease in women is less common and has a later average age of onset (4,33). Sex-related differences in stem cell behavior have been noted for the efficiency of endothelial progenitor cells colony formation, mesenchymal stem cell activation and function, MDSC-triggered skeletal muscle repair, and stress-triggered MDSC gene expression (6,7,10,13). These results suggest that while the microenvironment plays a critical role in stem cell repair, differences in inherent stem cell characteristics, including sex, could account for some of the outcome variability noted in clinical trials. Characterizing subtle but clinically relevant factors that influence the biology of cells used for cardiac therapy is especially important to optimize clinical translation.
Our laboratory has recently shown that female MDSCs are superior to male cells for repairing skeletal muscle (10). Transplantation of wild-type female cells resulted in greater number of dystrophin+ myocytes in the gastrocnemius muscle of mdx mice (model for Duchenne muscular dystrophy lacking expression of dystrophin), regardless of the sex of the recipient (10). Female recipients were also shown to have more dystrophin+ myocytes after transplantation than male recipients with either cell sex. When exploring MDSC-induced osteogenesis, male MDSCs displayed more robust osteogenic and chondrogenic differentiation in vitro and in vivo versus female cells, regardless of the sex of the recipient (5,18). Based on these results, we hypothesized that there would also be a sex difference in MDSC-induced cardiac regeneration, which was supported by a recent study demonstrating sex differences in mesenchymal stem cells for cardiac repair (6). In the current study we investigated transplantation efficiency of MDSCs into both mdx mice and a SCID mouse model of myocardial infarction to determine if donor cell sex or recipient sex would influence cardiac engraftment and regeneration/ repair. Our results indicate that cell and host sex is not a critical factor for cardiac engraftment or repair using MDSCs.
Materials and Methods
MDSC Isolation
Muscle-derived stem cells were isolated from the skeletal muscle of 3-week-old normal C57BL mice (Jackson) using the modified preplate technique as previously described (9,12,29). Mice were anatomically sexed prior to isolation. Both male and female MDSCs (five populations of each) were cultured in proliferation media (PM) containing Dulbecco's modified Eagle medium (DMEM) (Invitrogen), 10% fetal bovine serum (FBS) (Invitrogen), 10% horse serum, 1% penicillin/ streptomycin, and 0.5% chick embryo extract (Accurate Chemical).
Intramyocardial Cell Transplantation to mdx Mice
The use of animals and the surgical procedures performed in this study were approved by the Institutional Animal Care and Use Committee of the Children's Hospital of Pittsburgh (protocol 14-06). The mdx mice (C57BL/10ScSn-Dmd mdx ) were bred in our institution's animal facility. Cell transplantation into mdx hearts was performed as previously described with less than 1% mortality (27,28). Briefly, each heart was exposed via a left thoracotomy, and 1 × 105 MDSCs in 10 μl phosphate-buffered saline (five separately isolated populations of male and female cells) were injected into the left ventricular free wall of male or female mdx mice (12–16 weeks of age, N = 6–12 animals per MDSC population). Two weeks after injection, the mice were sacrificed, the hearts harvested and flash-frozen in liquid nitrogen-cooled 2-methylbutane, and then cryosectioned to 10 μm thickness.
Cell Transplantation to Infarcted Myocardium
Fifty male immunodeficient C57BL/6J-Prkdc scid mice were bred for the current study. Infarcted mice were randomly allocated by a blinded investigator between the treatment groups (saline, female MDSCs, or male MDSCs), and 3 × 105 cells total in 30 μl saline were injected into the left ventricular free wall of male SCID mice (13–16 weeks of age) 5 min after the permanent ligation of the left coronary artery (25,27). Six weeks after cell injection, echocardiography was performed by a blinded investigator on the left ventricular short axis view at the midpapillary muscle level as described previously (25, 27) to assess cardiac function, and the mice were euthanized and the hearts harvested, flash-frozen in 2-methylbutane, and cryosectioned (25,27).
Engraftment
Cryosections were fixed in 4% paraformaldehyde and then stained to determine donor cell engraftment. For the noninjury model in mdx mice, the number of dystrophin+ myocytes was determined as described previously (9, 15). Briefly, sections were stained for dystrophin with a rabbit anti-dystrophin primary antibody (1:400; Abcam) and Alexafluor 488 donkey anti-rabbit secondary antibody (1:200; Molecular Probes). Nuclei were revealed with 4′6-diamidino-2-phenylindol (DAPI) stain (100 ng/ml; Sigma) and sections were mounted with Gel Mount Aqueous Mounting Medium (Sigma). The engraftment capacity was determined by counting the number of dystrophin+ fibers. In the infarction model, heart sections were stained with mouse anti-fast skeletal myosin heavy chain (fsMHC) antibody (1:400; Sigma) and Alexafluor 555 donkey anti-mouse secondary antibody (1:300; Molecular Probes) to examine engraftment of the different cell populations. Engraftment was determined by assessing the area of fsMHC+ cells normalized to the area of the entire muscle section (9).
Identification of Endothelial Cells Within the Engraftment of Transplanted Cells
The number of CD31+ cells in the engraftment area was determined by double-staining tissue sections with rat anti-CD31 primary antibody (1:300; Sigma) and rabbit anti-dystrophin primary antibody (1:400; Abcam), and Alexafluor 488 donkey anti-rabbit secondary antibody (1:200; Molecular Probes) and Alexafluor 555 donkey anti-rat secondary antibody (1:300; Molecular Probes) for the mdx mouse model. The rat anti-CD31 primary antibody (1:300; Sigma) and mouse anti-fsMHC antibody (1:400; Sigma) were used for the infarcted samples (25,27,28). Blood vessel formation within the cell-injected areas was determined by the number of CD31+ cells associated with dystrophin+ cells for the mdx model or fsMHC+ cells for the infarction model.
Collagen Staining
Infarcted heart sections were fixed with 1% gluteraldehyde for 2 min and stained with the Masson Modified IMEB Trichrome Stain Kit (IMEB, CA, USA). Trichrome staining was performed according to the manufacturer's guidelines and as previously described (25, 27). The sections were then assessed for the percentage area of collagen in five sections per heart normalized to total muscle area within the section using ImageJ software (NIH).
Staining for Cardiac-Specific Markers
Sections from mdx samples were stained with rabbit anti-dystrophin primary antibody (1:400; Abcam) and Alexafluor 488 donkey anti-rabbit secondary antibody (1:300; Molecular Probes) and goat anti-cardiac troponin I (1:25,000; Scripps) with rabbit anti-goat Alexafluor 555 (1:200; Molecular Probes). Sections from the infarcted samples were stained with mouse anti-fsMHC (1:400; Sigma) and Alexafluor 488 donkey anti-mouse secondary antibody (1:300; Molecular Probes) and goat anti-cardiac troponin I (1:25,000; Scripps) with rabbit anti-goat Alexafluor 555 (1:200; Molecular Probes) and the number of double-positive cells were counted in three high powered fields per heart.
Analysis of Angiogenic Factor Secretion
Five male and female MDSC populations were plated in PM at 50,000 cells/well in six-well collagen type 1-coated plates. Twenty-four hours later, the medium was switched to DMEM with 1% penicillin/streptomyocin. Cells were then cultured for an additional 24 h and the medium was collected and flash-frozen until analysis. ELISA for mouse VEGF (R&D Systems) was performed according to manufacturer's instructions and as previously described (27). The VEGF levels were normalized to cell number as measured by hemocytometer.
Cell Survival Following Oxidative Stress
Cells were plated in PM at 1,000 cells/well in a 24-well collagen type 1-coated plate. Twenty-four hours later, the media was switched to PM with propidium iodide (PI, 1:500, Sigma) or PM containing 350 μM hydrogen peroxide with PI. The plates were then placed onto a previously described live cell imaging system (Automated Cell, Inc), and fluorescent and brightfield images were taken every 10 min in three fixed locations per well. These images were analyzed using Image-Viewer software (ACI). Cell proliferation was determined by counting the number of cells present in the brightfield images at 12-h time points for 60 h. Cell survival was determined by counting the number of PI-positive cells in the fluorescent images at each 12-h time point.
Microscopy
Florescence and brightfield microscopy were performed using either a Nikon Eclipse E800 microscope or Leica DMIRB inverted microscope equipped with a Retiga digital camera and Northern Eclipse software (version 6.0, Empix). Image analysis was performed using Northern Eclipse software or Image J software (available from NIH).
Statistical Analysis
Data are summarized as mean and SD. Statistically significant differences between groups were determined using a t-test for mdx studies (Microsoft Excel) or one-way or two-way ANOVA (SigmaStat) as appropriate for the infarction model. In the event significant differences were detected using ANOVA, the appropriate multiple comparisons test was used for post hoc analysis (Tukey test).
Results
Sex-Related MDSC Fate Following Implantation Into the Nonischemic mdx Heart
Survival and Engraftment of MDSCs in the mdx Heart
To determine whether cell and recipient sex were critical determining factors in engraftment efficiency of dystrophin+ cells in the myocardium, five male and five female MDSC populations were injected into the hearts of both male and female mdx mice. Two weeks after cell transplantation, dystrophin+ myocytes were observed within the hearts of all animals injected with MDSCs (Fig. 1A and B). The regeneration index (RI) revealed variability in MDSC engraftment in cardiac muscle, with cell populations varying between approximately 100 to over 2,000 dystrophin+ cells. Sex-matched experiments showed no sex-based difference in RI. Male cells/recipients had 1,309 ± 295 dystrophin+ cells and female cells/recipients had 1,010 ± 181 dystrophin+ cells. Sex of the recipient also showed no preferential effect on MDSC engraftment within the heart: 1,086 ± 172 dystrophin+ fibers in male recipients versus 1,069 ± 146 dystrophin+ fibers in female recipients (Fig. 1C). When the sex of the cell was matched with that of the host, the number of dystrophin+ myocytes was slightly increased (sex matched: 1,167 ± 178, sex mismatched: 838 ± 107, p = 0.12). Very low levels of differentiation of implanted MDSCs toward a cardiac phenotype, averaging less than 1% of injected cells, were seen with all MDSC populations, regardless of sex or number of dystrophin+ myocytes, which is consistent with previously published results (25,28). Overall, there was no difference in the number of engrafted dystrophin+ myocytes based either on the sex of the MDSCs or the sex of the recipient.
Engraftment in muscular dystrophy model. (A, B) Representative images of dystrophin stain showing both low (A) and high (B) engraftments of dystrophin+ myocytes. Scale bar: 100 μm. (C) Number of dystrophin+ myocytes in each group, showing that there is no sex difference based on cell or host sex.
Sex Does Not Influence Vascularization of MDSCs in the mdx Heart
MDSC implantation triggers angiogenesis during cardiac remodeling following injury (25,27). At 2 weeks post-cell transplantation, we measured the number of endothelial cells within the dystrophin+ cell engraftment zones for all animals injected with MDSCs (Fig. 2A and B). There was no difference between male and female cells (male cells: 17 ± 3 CD31+ structures, female cells: 14 ± 2, Fig. 2A). No differences were noted between the sex of the MDSCs or recipient with respect to the number of CD31+ structures in the dystrophin+ areas (male cells: 17 ± 3, female cells: 13 ± 2, p = 0.26, male recipient: 15 ± 3, female recipient: 14 ± 3, p = 0.725, Fig. 2B). We also noted no correlation between the density of dystrophin+ myocytes and the number of endothelial cells (endothelial cells in high dystrophin+ regions: 17 ± 3, endothelial cells in low dystrophin+ regions: 12 ± 2, p = 0.23, Fig. 2B). Thus, angiogenesis within the implanted region did not appear to be affected by the sex of MDSCs and recipient mice or the efficiency of engraftment of MDSCs.
Blood vessel formation in the heart in muscular dystrophy model. (A) No significant difference in blood vessel formation in the engraftment of dystrophin+ myocytes is seen with cell sex alone or (B) cell sex and recipient sex. (C) Representative image showing blood vessels within the dystrophin+ myocytes (scale bar: 50 μm). Blue: DAPI, red: CD31, green: dystrophin.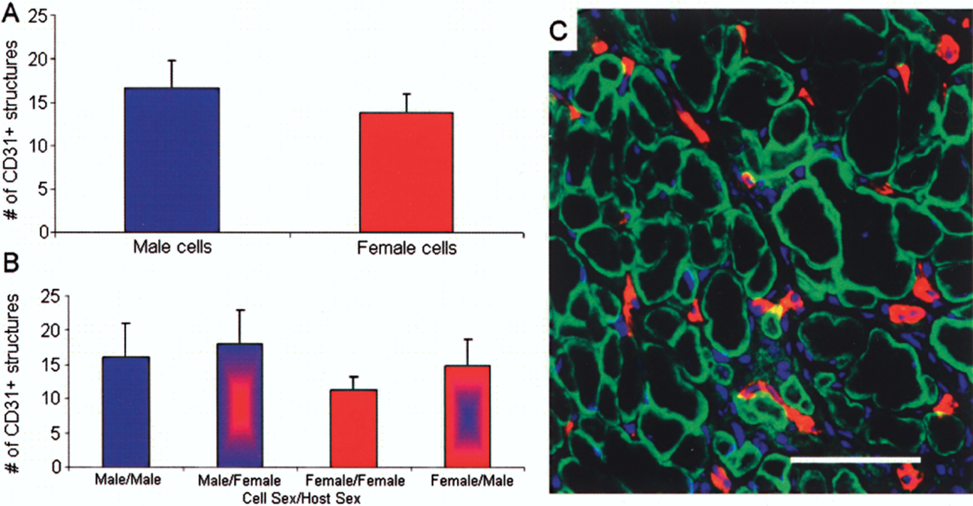
Sex-Related MDSC Fate in a Myocardial Infarct Model
Cardiac Function
We measured cardiac function using transthoracic echocardiography in SCID mice 6 weeks following myocardial infarction and MDSC implantation. Animal groups were divided into recipients treated with male MDSCs, female MDSCs, or saline alone. We noted no difference in left ventricular fractional area change (FAC) between cell-injected hearts based on MDSC sex; however, male and female cells were significantly better than saline controls (23.2 ± 1.3% for male cells, 21.5 ± 1.1% for female cells, and 13.1 ± 0.8% for saline, p < 0.05) (Fig. 3A). Postinfarction fractional shortening (male cells: 20.6 ± 1.2%, female cells: 19.1 ± 0.5%, and saline: 14.4 ± 0.8%), another measure of left ventricle (LV) contractility, and end diastolic area, which measures dilatation of the heart (male cells: 15.0 ± 0.7 mm2, female cells: 14.6 ± 0.6 mm2, and saline: 17.8 ± 1.3 mm2, p < 0.05) (Fig. 3B) displayed similar trends.
Cardiac function in ischemic heart. No difference is seen between the sexes in terms of cardiac function. When looking at fractional area change, all groups were significantly better than the saline control group (p < 0.05) (A). The same trend was seen with end diastolic area, with both male and female cell-injected hearts displaying less dilatation than saline (p < 0.05) (B).
Cardiac Scar Tissue Remodeling
The amount of collagen formed following myocardial injury influences systolic and diastolic function as well as functional recovery (35). Using standard measures of cardiac scar formation (collagen staining by Masson's trichrome and calculation of the ratio of healthy LV to LV scar), we noted that saline-injected hearts had very large scar tissue area ratios 6 weeks following myocardial infarction (65.0 ± 5.1%) (Fig. 4). MDSCs were found to have superior engrafting abilities compared with saline injected; however, sex did not influence postinfarction scar inhibition, as all cell-injected groups had significantly smaller scar tissue area ratios (male cells: 35.2 ± 5.4%, Fig. 4B; female cells: 35.7 ± 6.9%, Fig. 4C; p < 0.05 compared to saline, Fig. 4D).
Scar tissue area fraction. (A, B, C) Representative images of Masson Trichrome stains (collagen: blue, muscle: red) (A) saline-injected group, (B) male MDSC-injected group, and (C) female MDSC-injected group. Thinner walls and increased collagen content are visible in the saline-injected group. (D) No difference between the cell-injected groups, although all cell-injected hearts had significant lower levels of scar than the saline control (p < 0.05).
Sex-Related MDSC VEGF Secretion
We determined VEGF secretion into cell culture supernatant for 24 h of serum starvation due to the critical role of VEGF in postinfarction angiogenesis. No difference in VEGF secretion between male and female MDSCs was noted (male: 623 ± 200 pg/ml/106 cells, female: 752 ± 143, p = 0.58) (Fig. 5A). When subjected to oxidative stress after treatment with hydrogen peroxide, all cells increased their production of VEGF, regardless of cell sex (male cells: 1,387 ± 389 pg/ml/106 cells, female cells: 1,240 ± 233).
VEGF secretion and angiogenesis in infarcted heart. (A) VEGF secretion into cell culture supernatant demonstrated no sex-based difference. (B) At 6 weeks, no difference in blood vessel formation is seen in the fsMHC+ myocytes based on cell sex (N = 9–10 per group). (C) Outside of the engraftment areas, both male and female cells had induced more blood vessels than saline controls, but the difference was not significant.
Angiogenesis
No difference in blood vessel formation was noted in the engraftment areas based on MDSC sex (male cells: 10 ± 1 CD31+ structures, female cells: 11 ± 1, p = 0.37) (Fig. 5B). Outside of the engraftment area, there was again no difference between MDSC sexes. However, there was a trend that MDSC injection, regardless of cell sex, induced greater levels of neovascularization than saline injection at the 6-week time point (PBS: 34 ± 4 CD31+ structures, male cells: 43 ± 7, female cells: 54 ± 9) (Fig. 5C). These results support the role for MDSCs in inducing neovascularization versus a recipient response.
Cell Survival After Oxidative Stress
MDSCs have a superior ability to survive oxidative stress compared to myoblasts (25), and this seems to be related to the higher regeneration capacity in skeletal and cardiac muscle. Both male and female MDSCs display similar survival characteristics in response to oxidative stress (Fig. 6), which correlates to the similar levels of survival and cardiac engraftment for male and female cells seen in vivo in both the mdx and myocardial infarction (MI) mouse models.
Cell survival under oxidative stress and area of fsMHC+ myocytes in infarcted heart. (A) No difference was seen in the survival of male and female MDSCs in vitro after treatment with hydrogen peroxide (350 μM). Proliferation rates were comparable between the sexes (data not shown). (B) No difference is seen in engraftment at 6 weeks between cell sex or area of fsMHC+ myocytes.
Donor Cell Engraftment and Differentiation
We noted a lack of difference in the degree of MDSC engraftment in postinfarction SCID LV myocardium based on the sex of implanted MDSCs at 6 weeks (male cells: 27.2 ± 3.8%, female cells: 25.2 ± 3.9%) (Fig. 6B). As noted in previous studies, we found less than 1% of implanted MDSCs expressed cardiac markers (male cells: 0.2 ± 0.2%, female cells: 0.3 ± 0.1%), with the vast majority maintaining expression of skeletal muscle markers (25,27). The engraftment and regeneration seen in the infarction model differed from the noninfarcted mdx model, and that is likely due to the time points examined, the different number of injected cells, and presence of injury in the infarction model.
Discussion
Cell transplantation is considered to be a potential therapy for cardiac repair after injury; however, there is still debate as to how the repair takes place and what factors play a critical role. Some groups have demonstrated that stem cells can differentiate into cardiomyocytes in situ (19), thereby enacting repair, whereas others have shown that paracrine mechanisms provide cardioprotection (32,34). Up until recently, cell sex was not a factor for consideration in cell transplantation, and results seem to be dependent on injury model, cell type, and time points examined (4,6). Previous results from our laboratory indicate that cell sex should be considered for muscle, cartilage, and bone regeneration. These experiments demonstrated that female MDSCs are better at engrafting into skeletal muscle, while male cells are more osteogenic and chondrogenic (5,10). Other groups have shown that cell sex is also a factor with mesenchymal stem cell activation and cardiac repair, with female cells improving heart function to a greater extent (6,7). Based on these results, we hypothesized that female muscle-derived cells would be superior for cardiac repair.
We observed no difference in the mdx model in the engraftment capacity of MDSCs in the heart based on sex. This result is in contrast to previously reported results on MDSC transplantation into skeletal muscle, and we hypothesize that this difference may be due to the different muscle environment found in the heart, including cyclic mechanical strain and the biochemical makeup of infarcted myocardium. We have demonstrated that transplantation of both male and female murine MDSCs attenuated functional deterioration after myocardial infarction, and we observed that cardiac repair using MDSCs does not appear to be dependent on cell sex. There was no difference when examining cardiac function after cell transplantation, engraftment capacity, scar tissue formation, or coexpression of cardiac phenotype and induction of angiogenesis between the male and female cells. These results suggest that the recovery of postinfarction LV function following MDSC implantation is not due to MDSC sex.
The classic idea for the mechanism of cardiac cell therapy is that delivery of the appropriate stem cells would repair the damaged heart via active myocardial regeneration from transdifferentiation of the administered stem cells to the cardiac lineage (24). However, increasing evidence has led to the recognition of another mechanism of cardiac repair: the paracrine effect. In line with this mechanism, MDSCs seem to enact repair through paracrine mechanisms by increasing survival and repair of the recipient cardiac tissue rather than by differentiating into cardiomyocytes. Regeneration of cardiac tissue without cardiac differentiation has also been seen with bone marrow and hematopoietic stem cells used to treat ischemia reperfusion injuries and myocardial infarctions (22,34). Previous results have highlighted the importance of VEGF secretion in cardiac repair (27), and because both male and female MDSCs secrete similar levels of VEGF, this could help reconcile, at least in part, the lack of a sex difference seen in terms of capillary density in this study. When treating ischemic heart disease, perfusion of the affected tissue is an essential part of any therapy. The improved regenerative capabilities of MDSCs might be attributed to higher expression levels of antioxidants or increased VEGF production (27). In the heart, cells do not need to differentiate into cardiomyocytes to increase heart function after injury, and this leads to the hypothesis that sex-based differences in MDSCs may only be seen when differentiation toward a specific lineage is required. This idea is supported by sex differences observed with MDSC transplantation for skeletal muscle, where the cells had to differentiate toward skeletal muscle lineages to promote the healing process.
There are many issues to consider when examining the influence of sex on stem cell potential, particularly for cardiac repair. Intrinsic and extrinsic cellular mechanisms regulate the balance of self-renewal and differentiation in stem cells (21). Although we did not find sex differences with MDSCs for cardiac repair, other groups have shown that source sex is relevant for mesenchymal stem cells (6). Although it was not examined in the mesenchymal paper, we cannot exclude the possibility that a sex difference was seen with these cells due to differential differentiation toward a cardiac phenotype. In our current study, we implanted MDSCs isolated from 3-week-old mice, which could have influenced the results, although cells of the same age were used for previous sex studies in skeletal muscle, cartilage, and bone without negative effect.
In summary, there appears to be no sex-based difference for cardiac repair using MDSCs. Further work needs to be done to determine if this lack of effect is related to age of cells or recipients, as well as to clarify the role differentiation plays in sex-related differences in repair. The lack of a sex difference in MDSCs for cardiac repair is promising for clinical translation. For autologous use, these results demonstrate that the potency of the cells will theoretically be equal for both male and female patients. However, autologous transplantation it is not always feasible, and either sex of MDSCs can be used with beneficial effect.
Footnotes
Acknowledgments
This work was supported by grants to Dr. Johnny Huard from the MDA, the NIH (IU54AR050733-01, HL 069368), the PTEI, the Donaldson Chair and the Hirtzel Foundation at Children's Hospital of Pittsburgh, and the Mankin Chair at the university of Pittsburgh. The work was also supported by a predoctoral fellowship from the NIH to Lauren Drowley (T32 EB001026-05). We would like to thank Dr. Burhan Gharaibeh and Dr. Theresa Cassino for outstanding advice and technical support. During the performance period of this research project, Dr. Huard served as a consultant to Cook MyoSite, Inc.
