Abstract
Although the appearance of hepatic foci in the pancreas has been described in animal experiments and in human pathology, evidence for the conversion of human pancreatic cells to liver cells is still lacking. We therefore investigated the developmental plasticity between human embryonic pancreatic cells and liver cells. Cells were isolated and expanded from 7–8-week-old human fetal pancreata (HFP) and were characterized for the absence and presence of pancreatic and hepatic markers. In vitro expanded HFP were treated with fibroblast growth factor 2 (FGF2) and dexamethasone (DX) to induce a liver phenotye in the cells. These treated cells in various passages were further studied for their capacity to be functional in hepatic parenchyma following retrorsine-induced injury in nude C57 black mice. Amylase- and EPCAM-positive-enriched cells isolated from HFP and treated with FGF2 and DX lost expression of pancreatic markers and gained a liver phenotype. Hepatic differentiation was based on the expression (both at the mRNA and protein level) of liver markers albumin and cytokeratin 19. When transplanted in vivo into nude mice treated with retrorsine, both cell types successfully engrafted and functionally differentiated into hepatic cells expressing human albumin, glycogen, dipeptidyl peptidase, and γ-glutamyltranspeptidase. These data indicate that human fetal pancreatic cells have a capacity to alter their gene expression profile in response to exogenous treatment with FGF2 and DX. It may be possible to generate an unlimited supply of hepatocytes in vitro for cell therapy.
Introduction
Cell replacement therapies represent a possible treatment for liver diseases. Much of the research focus in cellular therapy has been based on the use of stem cells or stem cell-derived progeny for transplantation (14, 28). However, one alternative approach to cell therapy using stem cells is to induce the transdifferentiation of a liver phenotype from another a nonliver source (11). Recent studies have shown that it is feasible to convert one differentiated cell or tissue type into another type either by transduction of a transcription factor or by administration of selected growth or differentiation factors (1).
During embryonic development, the pancreas emerges from the dorsal and ventral domains of embryonic gut endoderm (4, 9). The ventral pancreas and liver arise from the same region of the developing embryo. It has been proposed that a common precursor cell exists and that signals from the cardiac mesenchyme are required for specifying the two tissues. In this model, the default state of the ventral foregut endoderm is to become pancreas, but a growth factor signal (FGF) from the cardiac mesenchyme diverts the fate of some cells towards liver (10). The common embryological origin of the ventral pancreas and the liver may be reflected in certain evolutionary and pathological states. Primitive invertebrates such as molluscs have a single organ, the hepatopancreas, which serves the functions of both organs (4). In addition, the caudal pancreas in the sea lamprey develops by transdifferentiation of cells in the hepatic duct (6). Thus, the partitioning of liver and ventral pancreas into separate organs may have evolved from a common cell population.
In addition, examples of liver cells appearing in the adult pancreas exist. For example, rats maintained on a copper-deficient diet develop pancreatic acinar cell atrophy. When this is followed by copper repletion in the diet, hepatocytes are formed in the regenerating pancreas (23, 24). When such pancreatic hepatocytes are transplanted to the liver of animals with an inherited metabolic deficiency of the liver, this leads to liver repopulation and restoration of metabolic liver defects (30). This effect was obtained with cells from the whole exocrine pancreas but not with purified ducts. This suggests that acinar exocrine cells may retain hepatogenic potential. Cells of a pancreatic ductal cell line differentiate into hepatocytes after subcutaneous or intraperitoneal transplantation. In some studies, acinar cells were considered to represent the hepatocyte precursors (25), whereas in other studies duct cells or “periductal” cells were thought to be their origin (15). Because liver and pancreas arise from adjacent regions in the embryo, it has been suggested that their specification differs by a single developmental decision, affecting the expression of a relatively small number of master switch genes (4, 9).
In vitro evidence for acino-hepatocytic transdifferentiation came first from the rat AR42J cell line and from organ cultures of early mouse pancreatic embryonic buds treated with dexamethasone (DX) (27, 29). CCAAT-enhancer binding protein-β (C/EBP-β) was demonstrated to represent the key transcription factor controlling the transdifferentiation of AR42J cells to hepatocytes. Later, it was found that adult rat pancreatic acinar cells in primary culture can indeed transdifferentiate into hepatocyte-like cells in the presence of DX (13). In these cells, the transcription factor C/EBP-β was upregulated in the presence of DX. The C/EBP family of transcription factors is known to be involved in the regulation of expression of multiple hepatocyte-specific genes (5). C/EBP-β is normally not expressed in the pancreas, but is known to play a role in liver regeneration and in the acute phase reaction (2, 8).
So far all the above work has been performed using either rodent pancreatic cells or pancreatic malignant cell lines. However, to date no information is available on the potential of primary human pancreatic cells to transdifferentiate towards a hepatic phenotype. This information is essential to the realistic development of a bioartificial liver for liver bridging support, cell therapy, or for extensive in vitro drug and toxicological testing.
Because fetal tissue is known to have high proliferative capacity, we investigated whether primary human fetal pancreatic cells (HFP) treated with DX and FGF could induce expression of liver-specific genes and differentiate into functional hepatocytes and cholangiocytes. We also tested the potential of cells to functionally engraft damaged liver parenchyma. We found that cells treated with DX and FGF started to express liver markers (both hepatocyte and ductal markers) and could engraft into chemically and surgically injured livers.
Materials and Methods
Isolation of Human Fetal Pancreata
Permission for the present study was granted from the local ethics committee at Karolinska University Hospital Huddinge. All women donating fetal tissue had been serologically screened for syphilis, toxoplasmosis, rubella, HIV-1, cytomegalovirus, hepatitis B and C, parovirus, and herpes simplex types 1 and 2. Written and oral informed consent was obtained. HFP tissue was obtained from aborted fetuses at 7–8 weeks of gestation in accordance with the Swedish guidelines. A modified vacuum curettage was performed (31). Gestational age was estimated according to specific anatomical markers (7). The abortions were performed in pregnancies with no apparent abnormalities, and no fetuses with anomalies were included. HFP was identified using stereoscopic microscopy and dissected out and placed in a sterile tube containing RPMI-1640 medium (Gibco, Invitrogen Corp. UK). A total of 10 different HFP from different donors were individually studied for the following analyses.
Culture, Phenotyping, and Proliferation of Human Primitive Progenitors From Fetal Pancreas
Following isolation, the explanted HFP tissue was placed directly into one well of a six-well tissue culture plate coated with collagen I (Biocoat, Becton and Dickinson, NJ, USA) and cultivated in Dulbecco's modified Eagle medium (DMEM, GIBCO, Invitrogen, Stockholm, Sweden) containing 10% inactivated fetal calf serum, 5% penicillin and streptomycin, 5% L-glutamine, 5% minimum essential amino acids (all from Biosource, Stockholm, Sweden), 10 ng/ml hepatocyte growth factor (HGF), 20 ng/ml epidermal growth factor (EGF) (R&D Systems, Abingdon, England), and 50 μg/ml gentamycin (Sigma). The medium was changed every 2–3 days for up to 14 days. Cells were allowed to migrate out from HFP tissue. At confluence, cells were split 1:2 with trypsin-EDTA (GIBCO) into collagen I-coated cell culture flasks using the above medium.
Flow cytometry was performed using an array of antibodies to specific markers expressed on pancreatic cells, hepatocytes, and bile ducts (16, 17) (Table 1). For certain markers intracellular staining was performed after cell permeabilization using 5% saponin in phosphate-buffered saline (PBS). Corresponding control isotypes were used for evaluation of nonspecific binding of monoclonal antibodies. The cells were analyzed on a Becton Dickinson flow cytometer (FACSorter). For detection of proliferation the cells were incubated with the thymidine analogue BrdU (30 mM, Sigma)) for 30 min, washed, and stained with FITC-conjugated anti-BrdU antibody (Sigma). The cells were analyzed by flow cytometry.
Frequency of Cells Expressing Hepatic and Pancreatic Cell Markers in Human Fetal Pancreas in Gestation Week 7–8

Treatment of Fetal Pancreatic Cells with Dexamethasone and Fibroblast Growth Factor 2
The synthetic glucocorticoid DX was added at a final concentration of 1 μM as a solution in ethanol to the culture medium of cells in passages 4 and 10. In addition, 30 ng/ml fibroblast growth factor 2 (FGF2, R&D Systems) was also added to these cultures. The medium was changed every 2–3 days. The cells were cultivated for 7 days after which the concentration of DX was decreased to 0.5 μM. The cells were further cultivated for an additional 20 days after which they were used for various analyses. In vitro expanded and DX- and FGF-treated cells in passages 4 (P4) and 10 (P10) were used for transplantation studies.
Immunofluorescence Analysis and Antisera
For immunofluorescent staining, single, double, and triple color staining was performed. For this, cells were cultured on glass slides, rinsed with PBS, fixed for 30 min with 4% paraformaldehyde in PBS, and then permeabilized with 0.1% (v/v) Triton X-100 in PBS for 30 min and incubated in 2% blocking buffer (Roche, Mannheim, Germany). The cells were then incubated sequentially with primary and secondary antibodies.
The primary antibodies were used as follows: rabbit anti-amylase (1:100, Sigma, Poole, UK), guinea pig antiinsulin (1:200, DAKO, High Wycombe, UK), mouse anti-glucagon (1:100, Sigma), rabbit anti-transferrin (1:100, DAKO), mouse anti-CK7 (1:200, Abcam, Cambridge, UK), sheep anti-albumin (1:100, AbD Serotec, Oxford, UK), rabbit anti-carbamoylphosphate synthetase I (CPS I) (1:300, a generous gift from Professor Wouter Lamers, Amsterdam), mouse anti-glutamine synthetase (GS) (1:300, Transduction Labs, Cowley, UK), rabbit anti-transthyretin (1:100, DAKO), sheep anti-uridine diphosphate glucuronsoyltransfase (UGT) (1:500, Cypex, Dundee, UK), mouse anti-C/EBP-β (1:100, Santa Cruz Biotechnology, Santa Cruz, CA). The secondary antibodies used were: goat anti-rabbit FITC, rabbit anti-guinea pig TRITC, horse anti-mouse AMCA, horse anti-mouse Texas Red, and rabbit anti-sheep AMCA (all at 1:100, Vector, Peterborough, UK). After immunostaining the slides were mounted in Gel-Mount™ mounting medium (Biomeda, Foster City, CA).
Only in the case of single color staining, the cell nuclei were stained with DAPI (blue). Images were collected using a Zeiss confocal LSM 510 and Leica DMRB compound microscopes.
Isolation of EPCAM-Positive Cells
Our initial experiments indicated the presence of CK7-positive cells in the fetal pancreatic cell cultures. In normal adult human livers, ductal cells are known to express CK7 and also the epithelial cell adhesion molecule (EPCAM). Because the phenotyping data of our cultured pancreatic cells indicated the presence of a low frequency of EPCAM-positive cells (approximately 2%; Table 1), we enriched for these cells in the hope of differentiating them to bile duct-like cells. EPCAM-positive cells were isolated from cultured pancreatic cells (see earlier section) in second passage using magnetic particles coupled with EPCAM antibodies (MiniMACs Miltenyi, Germany). The procedure was performed according to the manufacturer's instructions. The cells recovered at the end of the procedure were tested for viability and then cultured in DMEM medium. DX and FGF at concentrations given earlier were used for differentiation of these cells. Epithelial cells were detected by immunofluorescence using a nonconjugated anti-human cytokeratin 8, 18, and 19 antibody (Neomarker, USA), and a goat-anti-mouse IgG1 (FITC or Texas red) secondary antibodies (Jackson Immunoresearch, USA).
Mice
C57 black/nude female mice weighing 20–22 g were obtained commercially (Taconic M&B, Denmark) and served as recipients of human fetal pancreatic cells. The animals were housed at the animal laboratory at Huddinge University Hospital, Sweden, and received standard pellet chow and water ad libitum. Local Ethics Committee at Karolinska Institute approved all animal procedures and the mice were treated in accordance to Swedish regulations and laws for care and use of laboratory animals.
Liver injury was induced in mice (n = 30) by administration of retrorsine (Sigma Chemicals Co., Stockholm, Sweden). Two injections of retrorsine, 30 mg/kg each, were administered intraperitoneally, 2 weeks apart. This protocol is known to exert a persistent inhibition of hepatocyte cell division for several months (12). HPC were transplanted into the spleens of these animals 2 weeks after retrorsine treatment. Surgical procedure was performed under gas anesthesia with 1–1.75% of isoflurane (Fluovac Unit, IMS, Chechire, UK). Additional to chemical liver injury induced with retrorsine pretreatment, partial hepatectomy prior to transplantation was performed. The left median and the right middle lobes of the liver were removed with ligature technique via an upper abdominal transverse incision. After hemostasis control, intrasplenic transplantation of HPC was performed. Typically 2 × 106 DX- and FGF-treated (a mixture of equal numbers of amylase and EPCAM differentiated cells) (10 animals) or untreated cells (10 animals) in P4 or P10 suspended in 200 μl of DMEM medium were injected into the spleen of these mice over approximately 10–15 s. Ten mice were sham transplanted with just DMEM medium. After securing hemostasis, the abdominal incision was closed and the animals were monitored until recovery.
Preparation of Livers for Immunostaining
Mice were killed 4 weeks after transplantation and the liver and spleen were excised from each animal. Two or three biopsies from each liver of approximately 2 mm3 were shock frozen in liquid nitrogen and used to isolate RNA for RT-PCR analysis. The rest of the liver tissue was shock frozen for fluorescence and immunohistochemical analysis. Cryosections (5 μm thick) were air dried and fixed with cold 30% acetone in methanol for 10 min and further analyzed by immunohistochemistry.
Immunohistochemistry of Frozen Sections
For the detection of human cells in the mouse parenchyma an in situ hybridization technique described by Gupta et al., using a digoxygenin-labeled human DNA probe (Cytocell, Oxfordshire, UK), was used (3). Combination of in situ hybridization and histochemistry for hepatic markers was performed using a nonconjugated mouse antibody against human albumin (Nautec, Germany), and anti-CD26 (dipeptidyl peptidase IV, DPPIV). γ-Glutamyltransferase (γGT) and glycogen were demonstrated in situ as described previously (18, 26). Sections were counterstained with hematoxylin and mounted in mounting media (ImmunKemi, Stockholm, Sweden).
Morphometric Analysis
We screened 40 serial sections of each mouse liver for DNA probe-positive human cells. We also determined the number of transplanted cells and analyzed a minimum of 50 high-power fields in tissues from all transplanted animals. The number of transplanted human cells per cm3 was determined as described previously (21, 22).
Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR) and Real-Time RT-PCR
Total RNA was extracted from four human–mouse chimeric and three control murine liver tissues using the Micro-FastTrack RNA isolation kit (Invitrogen, Groningen, The Netherlands). We used human-specific primers to detect human albumin, CK19, and α-fetoprotein expression in the mouse liver. We selected primers using the Primer Express software version 2.0 (Applied Biosystems). Glucose-6-phosphate dehydrogenase (G6PD) was used as a housekeeping gene. Each set of primers was designed to target cDNA alone, not contaminating DNA. Primer sets were commercially synthesized by CyberGene (Huddinge, Sweden). Primer sequences were: CK-19 sense 5′-CCTGCGGGACAAGATTCTTG-3′ and antisense 5′-ACGGGCGTTGTCGATCTG-3′, expected product size 70 bp; α-fetoprotein sense 5′-GCAAAGC TGAAAATGCAGTTGA-3′ and antisense 5′-GGAAA GTTCGGGTCCCAAAA-3′, expected product size 129 bp; albumin sense 5′-GCTTTGCCGAGGAGGGTAA-3′ and antisense 5′-GGTAGGCTGAGATGCTTTTAAAT GT-3′, expected product size 88 bp; α1-antitrypsin sense 5′-CAGAGGAGGCACCCCTGAA-3′ and antisense 5′-AGTCCCTTTCTCGTCGATGGT-3′, expected product size 71 bp; G6PD sense 5′-TGCCCCCGACCGTCTAC-3′ and antisense 5′-ATGCGGTTCCAGCCTATCTG-3′, expected product size 76 bp.
PCR reactions were performed in duplicates in 96-well optical plates in a total volume of 25 μl. Each reaction contained 2.5 μl of cDNA, 12.5 μl SYBR Green Master Mix (Applied Biosystems), and 500 nM of each primer. Positive and negative controls were included in all runs. Thermal cycling conditions were 2 min at 50°C initially and 10 min at 95°C, as recommended by the manufacturer. Cycle conditions were 40 cycles at 95°C for 15 s and at 60°C for 1 min. The housekeeping gene, G6PD, was included as endogenous normalization control, which was used to confirm successful RNA isolation and reverse transcription, and the total amount of RNA in every sample. Real-time quantification of cDNA was performed using an ABI Prism 7000 Sequence Detection System (Applied Biosystems, Foster City, CA, USA) (16, 17). To calculate the relative gene expression, we employed the comparative ΔCt method (User Bulletin #2 ABI Prism 7700 SDS, Applied Biosystems). Results were calculated as the ratio between the gene of interest (CK19, albumin, α-fetoprotein, or α1-antitrypsin) and the endogenous control (G6PD) in the same sample.
Statistical Methods
The data are presented as mean ± SD. The significance of differences was analyzed with the Student's t-test and analysis of variance (ANOVA). A value of p < 0.05 was considered to be significant.
Results
Morphology and Phenotype of Untreated Human Fetal Pancreatic Cells
Human embryonic pancreatic cells were isolated and allowed to attach in culture. Cells rapidly attached to the underlying plastic and proliferated as detected using BrdU. Based on flow cytometric analysis, on an average 78.5 ± 10.6% BrdU+ cells were detected in passages 2–10 compared to 46.3 ± 11.2% BrdU+ cells in passages 11–20 (p < 0.01). In freshly isolated cells, hepatocyte markers (albumin, α-fetoprotein, carbamoylphosphate synthetase, glutamine synthetase, UDP-glucuronosyltranferase, and transferrin) were all undetectable (Fig. 1, Table 1). Phenotyping of the cell population migrating out of the explants showed expression of pancreatic exocrine markers (amylase positive) but not of insulin (Fig. 1, Table 1). Both epithelial (expressing CK7, CK8, and CK18) and some mesenchymal-like/fibroblast cell types (expressing CD90) were also expressed following isolation (Table 1).

Immunocytochemical staining of nontreated and dexamethasone (DX)- and fibroblast growth factor (FGF)-treated human fetal pancreatic cells (HFP). (A) Untreated HFP did not express any of the hepatic markers albumin or α-fetoprotein (upper panel). However, treatment of HFP cells with 1 μM DX and 30 ng/ml FGF for 2 weeks showed strong expression of albumin, a liver-specific protein (green) and the fetal liver marker α-fetoprotein (red) (lower panel). Cells are stained with DAPI, a reagent that stains the nucleus (blue). (B) Double or triple color staining showed that some of the untreated cells expressed amylase and a few cells expressed CK7 (upper panel). However, none of the untreated cells expressed transferrin, albumin, carbamoylphosphate synthetase I, glutamine synthetase, transthyretin, uridine diphosphate glucuronsoyltransfase, nor the transcription factor CCAAT-enhancer binding protein-β (upper panel). However, cells treated with DX and FGF for 2 weeks expressed, in addition to amylase (green, G) and CK7 (blue, B) (lower panel), other hepatic markers such as albumin (blue, B), carbamoylphosphate synthetase I (CPS green, G, seen as double positive bright blue cells), glutamine synthetase (GS, green, G, seen as double positive bright blue cells), transthyretin (green, G), uridine diphosphate glucuronsoyltransfase(UGT, blue, B), and the transcription factor CCAAT-enhancer binding protein-β (C/EBP-β, red, R), transferrin (green, G) (lower panel). Cells were negative for insulin (red, R). Scale bar: 20 μm.
Dexamethasone and FGF2 Converts Primary Fetal Pancreatic Cells to Hepatocytes
Because glucocorticoids can induce the expression of liver markers in the pancreatic cell line AR42J (19), and in adult rat pancreatic cell cultures (13), we wished to test whether addition of DX (in the presence of FGF) can have a similar effect on HFP. The fetal liver markers α-fetoprotein and albumin were expressed in DX- and FGF-treated cells (Fig. 1A). Addition of 1 μM DX and 30 ng/ml FGF2 to HFP cells (in P4 and P10) for 5 days increased the frequency of amylase-positive cells from 33.3 ± 2% to 56.3 ± 8% (flow cytometric analysis). There was also an increase in the number of the CK7-expressing ductal-like cells from 10 ± 2% to 33 ± 7% (flow cytometric analysis). However, we later observed changes in the morphology of the cells, which progressively flattened onto the substratum (Fig. 2A). After 2 weeks, the cells were extremely flattened and the cells gradually lost expression of amylase. At 3 weeks, these cells showed strong expression of albumin, a liver-specific protein (Fig. 1B). After 10, 15, and 20 days of culture in the presence of DX and FGF, approximately 60%, 74%, and 90%, respectively, of the cells were immunoreactive for albumin as detected by flow cytometric analysis. However, the viability of the cells decreased at 20 days of culture (viability ~70%). DX- and FGF-treated cultures were further tested for induction of other hepatic markers. We found that at least some of the flattened cells expressed transferrin, carbamoylphosphate synthetase, glutamine synthetase, transthyretin, UDP-glucuronosyltransferase, and the transcription factor CEBP-β (Fig. 1B, lower panel).

Morphological changes in cultured human fetal pancreatic cells. (A) Explants of HFP (8 gw) when plated in tissue culture plates attached and cells migrated out from the explants. (B) Approximately 3 weeks later the cells became confluent. (C) After 3 weeks, the cells changed morphology and became elongated and extremely flattened. Scale bar: 50 μm. (D) Freshly isolated EPCAM+ cells expressed the epithelial cell markers CK8 and CK18 but not CK19 (upper panel). However, treatment of EPCAM-positive cells with dexamethasone (DX) (1 μM), fibroblast growth factor (FGF2) (30 ng/ml) for 3 days resulted in expression of CK8 and CK18 as well as CK19 (lower panel). Negative control represents cells stained with only secondary antibody. Scale bar: 25 μm.
Isolated EPCAM Pancreatic Cells Express CK19 After Treatment with Dexamethasone
Freshly isolated EPCAM+ cells expressed CK8 and CK18 but not CK19 (Fig. 2D). However, treatment of these cells with DX (1 μM) and FGF (30 ng/ml) for 2–3 days induced expression of the ductal marker CK19 (Fig. 2D).
Detection of Human Cells and Transcription of Human Liver-Specific Genes in Mice Transplanted with DX-Treated Human Fetal Pancreatic Cells
We tested the in vivo functional potential of DX- and FGF-treated pancreatic and EPCAM+ cells expanded in culture (P4 and P10) by transplanting these cells into mice treated with retrorsine to induce acute liver injury. Two mice in the control and three in the test group died within 24 h after treatment. For simplicity, results from mice surviving at 4 weeks (n = 8 control; n = 17 test) after cell transplantation are presented. The species specificity of the human DNA probe is demonstrated by the positive result with human liver (Fig. 3A) and the negative immunohistochemistry with sham-transplanted livers from nude mice. Some positive cells were also found in mice transplanted with untreated pancreatic cells (Table 2).

Detection of transplanted human cells and transcription of human liver-specific genes in the mouse liver. (A) Using in situ hybridization [as described by Cho et al. (3)], the human DNA probe (black, arrows) stained normal human liver cells. However, livers of sham-transplanted did not stain positive, thus demonstrating the specificity of the probe. (B) Fourth (P4) and 10th (P10) passage dexamethasone (DX)- and FGF-treated human fetal pancreatic, when transplanted into retrorsine mice showed differentiation into hepatocytes and cholangiocytes (black staining; arrows). Scale bar: 40 μm. (C) Human cytokeratin 19 (CK19), α-fetoprotein (α-feto), albumin (alb), and α1 antitrypsin (anti-tryp) were detected, albeit weakly in animals transplanted with untreated HFP (Un) but not in the sham-transplanted mice (S) (upper panel). The same markers were significantly highly amplified (lower panel) in the livers of retrorsine-treated mice that received DX and FGF-treated human fetal pancreatic cells. A representative picture from two different mice transplanted with cells in P4 and P10, respectively (lower panel). (Also see Table 3). Glucose-6-phosphate dehydrogenase was used as the housekeeping gene.
Human Fetal Pancreatic Cell Engraftment in Retrorsine-Treated Mice at 4 Weeks After Transplantation

p < 0.001 for untreated FPC versus DX- and FGF-treated FPC.
DX- and FGF-treated pancreatic cells in P4 and P10 when transplanted differentiated into hepatocytes (Fig. 3B) and formed bile ducts at 4 weeks after transplantation, while untreated cells did not (Fig. 3B).
We analyzed livers of the mice sacrificed 1 month after transplantation of human fetal pancreatic cells by RT-PCR using primers specific for human liver-specific genes, including albumin, α1-antitrypsin, CK19, and AFP. G6PD was used as a control for the integrity of the RNA. The CK19, albumin, α1-antitrypsin, and AFP primers were species specific for human and did not amplify the respective mouse genes (Fig. 3C). Quantitative analysis of the genes for human CK19, AFP, albumin, and α1-antitrypsin in the mice receiving human cells is shown in Table 3.
Quantitative Analysis of Human Liver-Specific Genes Expressed in Retrorsine-Treated Mice Transplanted With Human Pancreatic Cells
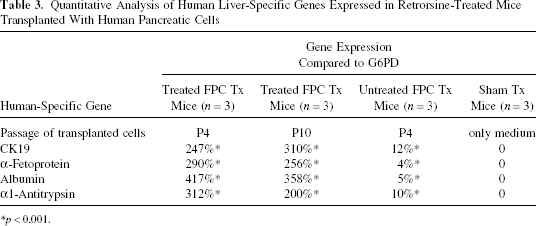
p < 0.001.
Expanded DX- and FGF-Treated Human Fetal Pancreatic Cells Successfully Differentiate Into Functionally Mature Hepatocytes and Cholangiocytes in the Livers of Retrorsine-Treated Mice
Human pancreatic and EPCAM+ cells were treated with DX and FGF for 2 weeks and then transplanted into retrorsine-treated mice. Transplanted human cells synthesized glycogen (Fig. 4) and integrated into the liver parenchyma based on coexpression in some cells of dipeptidyl peptidase IV (DPPIV) and the human DNA probe (Fig. 4). Some cells also integrated into the biliary tract [γ-glutamyl transpeptidase (GGT) and human DNA probe] (Fig. 4). The hepatocyte and biliary markers are expressed in the control sham-transplanted mice but not the human DNA probe (Fig. 4). A nonconjugated monoclonal antibody against human albumin was also used to examine the expression of human albumin in the transplanted cells. We found albumin-positive human transplanted cells (Fig. 4) in all of the DX- and FGF-treated FPC transplanted mice.

In vivo fate of in vitro expanded fibroblast growth factor 2 (FGF2)- and DX-treated human pancreatic cells. FGF2- and DX-treated human pancreatic cells 1 month after transplantation into retrorsine-treated mice contained glycogen (pink), dipeptidyl peptidase IV (red-brown), γ-glutamyl transpeptidase (red/brown cytoplasmic staining), and albumin (brown). Staining pattern for the same markers in sham-transplanted (receiving only culture medium) control mice is also shown. Glycogen, DPPIV, and GGT are present in all mouse and human hepatocytes, as expected. For albumin staining we used a human-specific antibody. Double-labeling of the human transplanted cells with the human DNA probe (arrow heads) visualized human nuclei (brown/black). Fourth and 10th passage human pancreatic cells when transplanted into mice with liver injury differentiated into mature hepatocytes. Scale bar: 40 μm.
To determine whether liver engraftment was comparable between human pancreatic cells in P4 and P10, we screened 40 serial sections of each mouse liver with the human DNA probe. In mice transplanted with P4 or P10 cells, we detected a threefold increase in the number of human cells compared to the number of cells transplanted initially, suggesting the cells expand in vivo (Table 2).
Discussion
Transdifferentiation of pancreatic cells to hepatocytes has been demonstrated in a number of model systems (13, 15, 23–25, 27, 29, 30). In this study we show for the first time that the combined treatment of human embryonic pancreatic cells with the synthetic glucocorticoid DX and FGF2 can induce the formation of hepatocyte-like cells. We examined the expression of pancreatic and hepatic markers and show that in the absence of extracellular factors, cells predominantly expressed the pancreatic marker amylase. In the presence of DX and FGF2 cells lost amylase expression and gained the hepatic phenotype. Our findings based on PCR and immunoflurosecence detection indicate that cells expressed liver genes involved in albumin production, transthyretin and the liver-enriched transcription factor C/EBP-β. Control cells did not express any of the hepatic markers.
Due to the nature of human pancreatic cells maintained in culture, we were interested to know whether the transformation of pancreatic cells to liver cells occurs at the level of a stem (progenitor) cell or differentiated cells. Although our data cannot exclude a stem cell origin, the results suggest that the hepatocytes arise from exocrine-like cells that are already present at the start of the culture because the majority of cells at the start of the experiments were amylase positive. Regarding the cell lineage, the results in the present study are similar to the situation with the AR42J cell line and adult rat pancreatic cells (both express an exocrine phenotype). In the human liver, pancreatic digestive enzymes (including amylase) are expressed during early fetal life (up to 25 weeks gestation), suggesting activation of pancreatic genes during normal liver development. Pancreatic endocrine cells are also found in the developing extrahepatic bile duct of the rat (24). Both the liver and pancreas arise from the embryonic foregut endoderm, and a number of studies have been conducted to investigate the potential of cells to interconvert (4, 6, 10, 33). Hepatic foci can be induced in the pancreas of rats or hamsters in response to various experimental protocols: for example, copper deprivation of the diet, after transplantation, or in transgenic mice overexpressing keratinocyte growth factor in the pancreatic beta-cells. The reverse transformation has also been observed in the liver of a cancer patient (32). Interestingly, in our experience, we found that fetal pancreatic cell cultures have a far greater proliferation capacity compared to fetal liver cells (unpublished data).
In the present study we used the retrorsine-induced acute liver injury model described previously (12). Retrorsine is a member of the pyrrolizidine alkaloid (PA) family of hepatotoxic chemicals. After an oral dose sufficient to cause death of half of the animals (LD50), severe centrolobular necrosis develops within 48 h of administration (20). In addition to acute toxicity, long-lasting effects on hepatocytes are found after a single high-dose injection or after low doses repeated over a long period of time. They are characterized by megalocytosis of hepatocytes, sometimes in association with bile-duct proliferation, fibrosis and vascular damage (20). Normally after partial hepatectomy in untreated rats, more than 90% of hepatocytes undergo mitosis within 48 h. However, the proliferative capacity of hepatocytes after a growth stimulus (partial hepatectomy or primary mitogens) is impaired for several months after retrorsine exposure (12). Thus, retrorsine treatment provides transplanted cells a proliferative advantage and was therefore a suitable liver injury model for studying repopulation with human transplanted cells.
The present study shows that pancreatic cells, present in the human embryonic pancreas, can be expanded and converted to hepatocyte-like cells by culture with DX and FGF2. These observations and those on the expression of pancreatic exocrine digestive enzymes in the liver may support the concept of a common cell lineage of liver and pancreas in humans. Converting pancreatic cells to hepatocytes offers an alternative to the isolation and culture of hepatocytes from liver. We have shown that the cells expressing a liver phenotype can also be transplanted into a mouse model of liver injury. The transplanted cells can integrate into the liver parenchyma. This model is also useful for determining the factors that regulate liver gene expression. Thus, transdifferentiation is a possible mechanism for the generation of cells for transplantation into patients with liver failure.
Footnotes
Acknowledgments
The present study was financed by grants from the The Medical Research Council No. K2005-06X-14004-02B, The Karolinska University Hospital Research Council, and The Lars Erik Gelins Foundations to S.S.H., and the Wellcome Trust to D.T. The authors declare that no conflict of interests exists.
