Abstract
To evaluate DAPI (4′,6-diamidino-2-phenylindole) as a nuclear tracer of stem cell migration and incorporation it was observed the pattern of retinal integration and differentiation of mesenchymal stem cells (MSCs) injected into the vitreous cavity of rat eyes with retinal injury. For this purpose adult rat retinas were submitted to laser damage followed by transplantation of DAPI-labeled BM-MSCs grafts and double-labeled DAPI and quantum dot-labeled BM-MSCs. To assess a possible DAPI diffusion as well as the integration and differentiation of DAPI-labeled BM-MSCs in laser-injured retina, host retinas were evaluated 8 weeks after injury/transplantation. It was demonstrated that, 8 weeks after the transplant, most of the retinal cells in all neural retinal presented nuclear DAPI labeling, specifically in the outer nuclear layer (ONL), inner nuclear layer (INL), and ganglion cell layer (GCL). Meanwhile, at this point, most of the double-labeled BM-MSCs (DAPI and quantum dot) remained in the vitreous cavity and no retinal cells presented the quantum dot marker. Based on these evidences we concluded that DAPI diffused to adjacent retinal cells while the nanocrystals remained labeling only the transplanted BM-MSCs. Therefore, DAPI is not a useful marker for stem cells in vivo tracing experiments because the DAPI released from dying cells in moment of the transplant are taken up by host cells in the tissue.
Keywords
Introduction
To label cells without affecting their morphology or function is a critical issue for investigating cell behavior, growth, migration, and differentiation at a single-cell level. For all these purposes, researchers are always searching for a stable, nontoxic labeling that also will not leak, or be transferred from cell to cell either in vivo, in tracing experiments, or in vitro.
The blue fluorescent DAPI (4′,6-diamidino-2-phenylindole) nucleic acid stain preferentially binds to double-stranded DNA (31) to form a stable fluorescent complex that fluoresces approximately 20 times greater than DAPI alone. DAPI exhibits a preference for repetitive A-T base pairs in the minor groove (6,9). Although the dye is cell impairment, higher concentrations will enter a live cell. DAPI is a popular nuclear counterstain for use in multicolor fluorescent techniques, mRNA in situ hybridization, and in vivo cell tracking experiments.
Because stem cells can easily self-replicate and differentiate into lineage-specific cells, such characteristics provide several and important challenges for cell labeling. To date, thymidine analogs, such as 5-bromo-2-deoxyuridine, iododeoxyuridine, and tritiated thymidine, and DAPI, have been extensively used to label stem cells prior to transplantation. These dyes are used in order to allow the fate of the labeled cells into damaged areas to be determined (5,21,26).
Mesenchymal stem cells (MSCs) isolated from bone marrow of adult organisms are characterized as plastic adherent, fibroblastoid cells, which are able to differentiate into distinctive end-stage cell cycles such as ostoblasts, chondroblasts, and adipocytes. Cultured MSCs can be induced in vitro and in vivo to differentiate into nonmesenchymal derivatives such as neural cells, in a process called stem cell plasticity or transdifferentiation (19,22,32). Several reports have shown that bone marrow mesenchymal stem cells (BM-MSCs) differentiate into retinal neural cells in vivo and in vitro (11), and when implanted at a site of injury in experimental animal models, as they demonstrate the ability to migrate to the injury site, initiate tissue repair, differentiate into retinal cells, and restore function. Some of these studies used DAPI or thymidine analogs as nuclear markers to follow the migration of BM-MSCs and confirm their differentiation into retinal neurons after transplantation into injured retina (4,20). However, the specific mechanism responsible for the MSCs capacity of transdifferentiation in vivo remains unclear and recent reports shows that this “differentiation process” might be due to the fusion of the transplanted cells with resident cells (8) or even a false positive caused by DAPI uptake by the resident cells when this marker is released from dying transplanted stem cells.
In this study we took advantage of DAPI-labeled BM-MSCs transplantation model into the vitreous cavity of eyes with retinal injury to evaluate DAPI diffusion from labeled BM-MSCs that die in the transplantation process, releasing its chemical marker into the neighboring microenviroment and making possible its uptake by host cells, such as retinal neurons. Double-labeled BM-MSCs using DAPI and quantum dot, a cytoplasm marker with intense stable fluorescence that is only delivered into live cells by a custom targeting peptide that cannot be transferred to adjacent cells, were also used to confirm the fate of the transplanted cells.
Materials and Methods
Isolation and Culture of BM-MSCs
Bone marrow was collected from two 2-month-old isogenic Wistar rats by flushing their femurs and tibias with Dulbecco's modified Eagle's medium (DMEM; Gibco). Cells were incubated at 37°C and 5% CO2 for 48 h and nonadherent cells were removed. The cells, fibroblast-like in shape, were grown and expanded in flasks for several weeks, and exhibited typical characteristics of MSCs.
Establishment of a Laser-Injured Model
All experiments were performed according to the Ethical Principles in Animal Experimentation (CETEA/UFMG) and approved under the certificate number 3/2006.
Wistar rats (n = 18) were anesthetized with a dilution of ketamine (40 mg/ml) and xylazine (60 mg/ml), injected intramuscularly. Mydriasis was induced by anesthesia; no eyedrops were used for pupillary dilation. The retinal lesion was induced by an Nd-YAG laser, using an average of 0.5 mJ energy. A Goldman three-mirror lens was used to produce approximately 15–20 YAG laser shots around the optic disc. The lesions were all full-thickness retina and choroid disruptions, as observed by deep subretinal hemorrhage and sometimes mild vitreous hemorrhage after the spot was produced. Animals in which the laser caused massive vitreous hemorrhage (n = 3) were not used in the study.
Phenotypic Characterization of BM-MSC
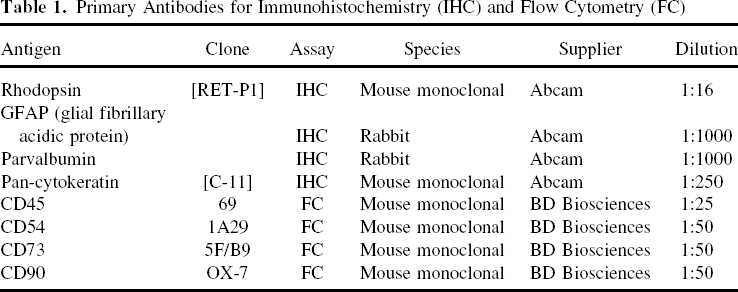
Primary Antibodies for Immunohistochemistry (IHC) and Flow Cytometry (FC)
BM-MSC Labeling and Viability
DAPI
After three passages, bone marrow-derived adherent cells were incubated with 50 μg/ml DAPI (Molecular Probes, Invitrogen, Carlsbad, CA) at 37°C and 5% CO2 for 2 h. BM-MSCs were treated with trypsin (Gibco) for 3 min to generate a single-cell suspension, washed three times with DMEM, resuspended in serum-free DMEM, counted, and stored on ice until transplantation. Fluorescence from DAPI labeling on BM-MSCs and cell viability were evaluated for 1, 2, 3, 4, and 8 weeks after cell labeling.
Quantum Trackers
Quantum trackers (Invitrogen) are microcapsules that express fluorescent nanocrystals called quantum dots (Qdots), which are delivered by a custom targeting peptide into the cytoplasm of live cells. Once inside the cells these nanocrystals emit a stable and intense fluorescence, which can be tracked for several cell generations, and do not leak out to be taken up by host cells in the tissue. Also, Qdots don not interfere with the self-replication process of stem cells. For BM-MSC (already labeled with DAPI) labeling with Qdot 655 we prepared a 10-nM solution containing 1 μl of reagent A (Qdot crystals) and 1 μ l of reagent B (targeting peptide). This solution was kept at room temperature for 5 min and then DMEM and a suspension of 1 × 106 BM-MSCs was added and incubated at 37°C for 60 min. The cells were washed three times with DMEM and stored on ice until transplantation.
Intravitreal Transplantation of MSCs
Rats (10–12-week-old Wistar SHR rats; n = 18) were anesthetized by intramuscular injection with a dilution of ketamine (40 mg/ml) and xylazine (60 mg/ml). A cell suspension (20 μl) containing approximately 4 × 105 DAPI-labeled BM-MSCs or double-labeled (DAPI and Qdot) BM-MSCs was slowly injected into the vitreous cavity via the pars plana using a 30-gauge needle 24 h after the retinal lesion was induced by the laser in nine animals (both eyes). Therefore, two groups of animals were studied: 1) rats with retinal injury and injection of BM-MSCs (n = 9); 2) rats with retinal injury without injection of BM-MSCs (n = 9).
Tissue Sectioning
The animals were sacrificed 8 weeks following transplantation. The eyeball was enucleated by performing a 360° limbal peritomy, isolating the extraocular muscles, and dissecting the optic nerve at the apex of the orbit. The eyes were fixed in Omnifix (FR Chemical Inc, Mount Vernon, NY). The slides were stained with hematoxylin/eosin, and visualized under a compound microscope to evaluate retina morphology.
Immunohistochemical Detection
All Paraplast-Plus-embedded blocks were cut, deparaffinized, rehydrated, and subjected to immunostaining with several antibodies specific for the retinal cells. Following blocking for 1 h at room temperature in phosphate-buffered saline (0.15 M PBS), 1% bovine serum albumin, and 2% normal bovine serum, ocular tissue sections were exposed to primary antibodies (Table 1) overnight at 4°C in a moist chamber. Control sections were treated identically, but the primary antibody was omitted. Sections were then washed three times for 5 min each in 0.15 M PBS solution. Secondary antibody (goat anti-rabbit and anti-mouse labeled with Alexa Fluor 488 (1:500, Molecular Probes, Invitrogen) was then applied for 1 h at room temperature. Sections were washed three times for 5 min each in 0.15 M PBS solution. The slides were viewed under a fluorescence microscope (Olympus®, Optical sectioning microscope attached to an Axioplan imaging Apotome® apparatus, Zeiss®, Germany). Colocalization of Alexa 488-labeled primary antibodies and DAPI was assessed by superimposing separate digital images of each fluorochrome.
Results
BM-MSC Characteristics and Labeling
After three passages in culture, the isolated cell population became homogeneous, showing a monolayer consisting of adherent cells displaying further traits of MSCs, including a typical fibroblast-like morphology and increased proliferation (Fig. 1A). Adhesion to the culture dish also served as a criterion to distinguish MSCs from free-floating hematopoietic cells. Figure 1B and C shows BM-MSCs nuclear labeling with DAPI (blue) and double-labeling with DAPI and quantum dot (red), respectively.
Morphologic analysis of cultivated and DAPI-labeled and double-labeled (DAPI and Qdot) rat bone marrow mesenchymal stem cells (BM-MSCs).(A) Phase contrast microscopy of rat BM-MSCs in the cell culture dish at passage 3, showing a stretched fibroblastic phenotype. The presence of round-shaped erythrocytes and nonadherent cells can be seen. After expansion to the third passage, a monolayer of adherent, fibroblast-like cells was labeled with DAPI. Fluorescence microscopy of adherent, fibroblast-like cells labeled with DAPI (B) or double-labeled with DAPI (nucleolus) and Qdot (cytoplasm) (C). Original magnification: 150×. Scale bars: 70 μm.
Phenotypic Characterization of BM-MSCs
After the third passage, BM-MSCs were also phenotypically characterized by flow cytometry using panels of antibodies and were defined as CD90+, CD73+, CD54+, and CD45~. These could be concluded because, from the cell population examined, 96.94% do not express CD45 and 86.77%, 93.99%, and 95.10% of cells that expressed CD90, CD73, and CD54, respectively (Fig. 2).
Phenotypical characterization of BM-MSCs. The panel of histograms shows the expression profile of BM-MSC surface markers CD45 (B), CD90 (C), CD73 (D), and CD54 (E). (A) The characteristic of the cell population evaluated, defined at the size versus granularity graph.
Incorporation and Distribution of the Grafted Cells
To verify the fate of DAPI-labeled BM-MSCs grafted in the laser-injured rat retina, the cells were injected into the vitreous space and evaluated 8 weeks after the grafting procedure. Retinal damage has an important role concerning the homing of transplanted cells in the retina, due to the high expression of chemoattractants by injured retinal pigment epithelium (RPE). Survival of the grafted cells was substantial in all transplanted eyes at the 8th week. At this point it was also observed that all cells in the neural retinal layers presented blue nuclei, characterized by DAPI labeling (Fig. 3). These findings could lead to the conclusion that the grafted DAPI-labeled BM-MSCs were able to fully integrate the laser-injured neural retina in only 8 weeks.
Immunohistochemical detection of DAPI-labeled BM-MSCs in injured retinal sections 8 weeks after intravitreal transplantation. The left panel of each row shows immunoreactivity for a particular marker in retinal sections with laser-induced injury without BM-MSC treatment (control). The right panel of each row shows immunoreactivity for a particular marker (green) in retinal sections with laser-induced injury and BM-MSC treatment. Eight weeks after transplantation, the grafted cells labeled with DAPI (blue) coexpressed the rod photoreceptor marker (green) (A, right panel) Rhodopsin, the bipolar and amacrine cell marker (green). (B. right panel) parvalbumin and the Muller cell and astrocyte marker (green) (C, right panel) GFAP. No grafted cells coexpressed the RPE marker (green) (D, right panel) Pan-cytokeratin. Note that only a small subset of grafted cells was GFAP immunoreactive (dashed arrows). Arrow in the right panel of (A) indicates the presence of grafted cells (blue) in the subretinal space. Micrographs of the control group did not show any nonspecific labeling or fluorescence expression. Scale bars: 20 μm. RPE, retinal pigment epithelium; ONL, outer nuclear layer; INL, inner nuclear layer; GCL, ganglion cell layer.
Immunohistochemical Analysis and Fluorescence Microscopy
Immunohistochemical analysis was then performed on sections in which DAPI-labeled BM-MSCs were presumed incorporated in the injured retina at 8 weeks after transplantation and in the control group to examine whether the grafted cells distributed into the retinal layers expressed retinal cell type-specific markers. The cell type markers used were rhodopsin for rod photoreceptor cells, glial fibrillary acidic protein (GFAP) for Muller cells and astrocytes, parvalbumin for bipolar and amacrine cells, and pan-cytokeratin for RPE (Table 1).
Most of the DAPI-labeled cells in the neural retina, which could be presumed to be the grafted BM-MSCs, incorporated into the outer nuclear layer (ONL) and expressed rhodopsin (Fig. 3A, right panel), and parvalbumin (Fig. 3B, right panel), suggesting their potential to differentiate into rod photoreceptors and amacrine or bipolar cells, respectively. A small subset of labeled cells in the inner retina was also labeled for the glial marker GFAP (Fig. 3C, right panel), suggesting that these cells could possess the ability to differentiate into astrocytes and Muller cells. Some DAPI-labeled cells were also observed in the subretinal space, but they did not express any retina-associated markers at any point (Fig. 3, right panel). In the control group sections (where the injured retina did not receive DAPI-labeled BM-MSC transplants), the retina-specific markers were evident and no DAPI-labeled cells were observed, ruling out the possibility of autofluorescence of the retinal cells (Fig. 3A–D, left panel). There was also no colocalization of DAPI-labeled cells with the RPE-specific marker (Fig. 3D, right panel), suggesting that the grafted cells did not reach the RPE layer.
The fact that most of DAPI-labeled cells in neural retinal tissue were able to coexpress its blue fluorescence in the nuclei with retinal markers, such as rhodopsin, parvalbumin, and GFAP could suggest a possible differentiation of the grafted cells into retinal neurons. However, further experiments are needed to confirm this possibility, because it is not likely that the grafted cells would repopulate the entire retinal tissue, as the immunohistochemical images shows most of the cells in the neural retina labeled with DAPI. Additionally, it does not seem possible that all the grafted mesenchymal stem cells would incorporate into neural retina and differentiate into retinal neurons—very specialized cells—in only 8 weeks.
To check the possibility of DAPI leaking or cellular fusion, we double-labeled BM-MSCs with DAPI and quantum dot, which due to the need of a targeting peptide cannot be transferred to host cells in vivo, and transplanted these cells into the vitreous space of injured retinas in the same condition as the DAPI-labeled cells only. Eight weeks after the grafting procedure, fluorescence images showed that most of the double-labeled BM-MSCs were still into the vitreous cavity, migrating towards the injury site, while the adjacent retinal cells in all layers presented a blue fluorescent label very similar to DAPI, but did not present any red labeling in their cytoplasm, characteristic from Qdot marker (Fig. 4). These experiments suggested that a large diffusion of DAPI occurred probably during the grafting procedure, when some of the labeled BM-MSCs die and release its chemical marker. After this procedure, we could also rule out the possibility of cell fusion between the grafted cells and retinal host cells, because all retinal cells presented only the blue DAPI labeling and no trace of Qdot fluorescence.
Migration of BM-MSC Qdot and DAPI-labeled BM-MSCs 8 weeks after transplantation. It can be noted a tissue disorganization (B). Hematoxilin-eosin) caused by the induced lesion (arrows). Some cells are migrating to the lesion (asterisks). The cells are doubled-labeled (QDot, red; DAPI, blue), confirming its origin (the transplanted cells) (A). Most of host cells are shown in blue but not in red, because of transference of DAPI from dead transplanted cells to host cells. Original magnification: 100×. Scale bars: 40 μm.
Discussion
Mesenchymal stem cells (MSCs) represent a stem cell population present in adult tissues that can be isolated, expanded in culture, and characterized in vitro and in vivo. Several researches continues to support the desirable traits of MSCs for development of cellular therapeutics for many tissues as they can be used for a broad spectrum of indications, including cardiovascular repair, treatment of lung fibrosis, and spinal cord injury (15). However, transplantation of BM-MSCs has generated unexpected phenotypes in vivo, including muscle cells (3), liver cells (16,25,30), brain cells (2,22), and others (14). This fact can be partially explained by several hypotheses.
Transdifferentiation or plasticity is one of the properties exerted by MSCs. However, the phenomenon of fusion between transplanted and host cells has been proposed as an explanation for the apparent plasticity of stem cells. Terada et al. (29) demonstrated that mouse BM-MSCs can fuse spontaneously with other cells and subsequently adopt the phenotype of the recipient cells. These data should be considered as an explanation for the differentiation of grafted BM-MSCs into unexpected cell types in vivo, reported by several studies. The fact that in various studies MSCs show a strong propensity to ameliorate tissue damage in response to injuries or diseases can be explained by the fusion of these cell with host adjacent cells, because “fused cells” can become a dominant population as they have a growth or survival advantage over their host cells by supplementing deficient genes. It is also assumed that the transplanted cells improve the functionally deficient cells as they act as a local cell delivery system for diffusible trophic factors.
Another aspect to be considered for the generation of unexpected phenotypes when BM-MSCs are transplanted into injured tissues is the widespread use of DAPI, organic dyes, BrdU, or other thymidine analogs as a unique marker of MSCs. These markers are often used solely as an approach to track the transplanted stem cells in vivo, distinguish the host cells form labeled cells, and assess their potential to integrate and differentiate in the developing or injured tissues. This is the case of a large amount of studies in which stem cells were transplanted into neural tissues and their differentiation into neural lineage cells demonstrated by the presence of these markers. These observations could suggest that MSCs may possess significantly greater plasticity than was previously realized, with the potential to differentiate into tissues from unrelated embryonic germ layers (11–13,18,24,34). This is a common procedure not only for ectodermic lineages differentiation process; several studies also took advantage of DAPI labeling to trace stem cells in vivo or to verify differentiation into mesoderm or endoderm lineages (7,17,23,28,35).
However, according to recent studies, some fluorescent dyes seems to leak from the cells and “contaminate” surrounding cells. To test DAPI leakage or transfer from cell to cell we used a strategy of stem cell therapy in injured retina. Several studies suggest that the retina is accessible to regenerative medicine strategies, including tissue repair using cell replacement. Specialized literature shows, in animal models, that visual function can be preserved, or possibly improved after transplantation of mesenchymal stem cells into injured or diseased retina. Regarding this possibility we injected BM-MSCs intravitreally into the rat eye 24 h after an Nd-YAG laser injury in the retina and observed that, 8 weeks after the procedure, the grafted cells dispersed into the host retina and assumed different patterns of distribution. Prior to transplantation, cells were labeled with DAPI or double-labeled with DAPI and quantum dots. Specific antibodies were used to determine the ability of grafted MSCs to morphologically differentiate within the environment of transplanted eyes. Colocalization of DAPI with one of the specific phenotypic antibody markers was used to evaluate neural differentiation of the grafted cells.
Immunohistochemical analysis with several retinal-specific markers revealed that the majority of the cells that presented DAPI labeling were also able to express rhodopsin, a rod photoreceptor marker, parvalbumin, a marker for bipolar and amacrine cells, and GFAP, a glial marker. It was also observed that most of the resident cells in retinal tissue presented a fluorescent blue nucleus, which suggests that all grafted cells not only repopulated the entire neural retina but also differentiated into highly specialized neural cells in only 8 weeks.
According to Kicic et al. (10), BM-MSCs are able to differentiate into photoreceptors in vitro and in vivo. Under specific culture conditions, BM-MSCs differentiated into photoreceptors in 8–10 days in vitro. However, the differentiation of BM-MSCs into photoreceptor in vivo, after transplantation into dystrophic rat retina, was observed in 12 weeks. Thus, it is doubtful that most of the grafted cells differentiated into photoreceptors, and were able to produce rhodopsin in 8 weeks. It is also questionable that the grafted cells could repopulate the entire neural retina. It is known that most of the cells die during the grafting procedure; in this way it is unlikely that the surviving subset of grafted cells would repopulate most of the neural retinal tissue in 8 weeks.
To test the above-mentioned hypotheses, we double-labeled BM-MSCs with DAPI and quantum dot, a nanocrystal that labels the cytoplasm, with a red fluorescence that cannot be transferred to host cells in vivo, due to the requirement of a targeting peptide. Eight weeks after the grafting procedure, fluorescent images showed that most of the resident cells in neural retina exhibited a blue fluorescence corresponding to DAPI in the nucleus, but did not show any red fluorescence characteristic by Qdot labeling. Also, most of the double-labeled cells were found in the vitreous cavity initiating a process of homing and migration towards the injury site, characterized by unorganized retinal layers. These results suggest that DAPI diffusion is responsible for the fluorescence exhibited by the host cells in neural retinal and that any kind of stem cell differentiation can be proved by the use of this marker only.
To date, although DAPI have been extensively used to track transplanted cells, false labeling of host cells is an issue when it comes to the use of this marker. Several dyes, such as thymidine analogs and organic dyes, also appear to have limitations. Recent studies are taking advantage of genetically encoded fluorescent proteins, such as green fluorescent protein (GFP), to track stem cells in vivo (33). Also, several reports are using various magnetic resonance (MR) contrast agents, such as ferumoxides (Feridex®)-protamine sulfate complexes to label stem cells for monitoring their temporal and spatial migration in vivo by magnetic resonance imaging (MRI) (1,11,27). So far these approaches have proved to be very effective, but still a reliable and long-term additional marker is necessary to validate any cell-based therapy when it comes to migration or differentiation properties.
Footnotes
Acknowledgments
This investigation received financial support from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES/Brazil), Fundação deAmparo à Pesquisa do Estado de Minas Gerais (FAPEMIG/Brazil) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq/Brazil). All experiments were performed according to the Ethical Principles in Animal Experimentation (CETEA/UFMG) and approved under the certificate number 3/2006. The authors would like to thank Gregory Thomas Kitten, Ph.D., from the Morphology Department for assisting in the immunohistochemical studies and Marco Antônio Màximo Prado, Ph.D., from the Pharmacology Department of the Biological Sciences Institute of Universidade Federal de Minas Gerais, Brazil for assisting with data acquisition.
