Abstract
The eye represents a highly specialized organ, with its main function being to convert light signals into electrical impulses. Any damage or disease of the eye induces a local inflammatory reaction that could be harmful for the specialized ocular cells. Therefore, the eye developed several immunoregulatory mechanisms which protect the ocular structures against deleterious immune reactions. This protection is ensured by the production of a variety of immunosuppressive molecules, which create the immune privilege of the eye. In addition, ocular cells are potent producers of numerous growth and trophic factors which support the survival and regeneration of diseased and damaged cells. If the immune privilege of the eye is interrupted and the regulatory mechanisms are not sufficiently effective, the eye disease can progress and result in worsening of vision or even blindness. In such cases, external immunotherapeutic interventions are needed. One perspective possibility of treatment is represented by mesenchymal stromal/stem cell (MSC) therapy. MSCs, which can be administered intraocularly or locally into diseased site, are potent producers of various immunoregulatory and regenerative molecules. The main advantages of MSC therapy include the safety of the treatment, the possibility to use autologous (patient’s own) cells, and observations that the therapeutic properties of MSCs can be intentionally regulated by external factors during their preparation. In this review, we provide a survey of the immunoregulatory and regenerative mechanisms in the eye and describe the therapeutic potential of MSC application for corneal damages and retinal diseases.

Keywords
Introduction
Various types of corneal injuries and retinal degenerative diseases, such as age-related macular degeneration (AMD), retinitis pigmentosa (RP), diabetic retinopathy (DR), and glaucoma, represent the main causes of decreased quality of vision and even blindness worldwide. However, despite extensive research, the effective treatment possibilities for these disorders are still very limited. A prospective option for the treatment has recently been offered by stem cell therapy, based on the local application of mesenchymal/stromal stem cells (MSCs). These cells have potent immunoregulatory properties and can inhibit a local harmful inflammatory reaction at the site of the disease. They are also the producers of numerous growth factors which could support the survival and regeneration of damaged ocular tissues. In addition, MSCs possess anti-apoptotic properties and have the possibility to differentiate into other cell types. For all these reasons, MSCs represent a promising tool for the treatment of so far uncurable ocular diseases.
The Eye and Its Structure
The eye is a complicated organ with the main function to capture a light signal using photoreceptors and to convert it into electrical impulses which are analyzed in the brain. Among the most important parts of the eye belong the cornea, which protects the eye against the outer environment and directs the light penetrating the eye, and the retina which converts light signals into electrical impulses (Fig. 1A).

Structure of the eye (A), cornea (B), and retina (C).
The cornea consists of three main layers (the epithelium, stroma, and endothelium), which are avascular in their central part and possess just a few cells of the immune system in the periphery (Fig. 1B). The low infiltration with cells of the immune system makes the cornea to be known as immunologically privileged tissue1,2. As a result of this immune privilege, corneal allografts survive in immunocompetent recipients without or with only weak local immunosuppression3,4. However, if the cornea is wounded by the injury or infection, the diseased site starts to be infiltrated with immune cells, which together with corneal cells produce a number of molecules which participate in the protection and regeneration of the cornea and support the wound healing response.
The retina is a highly specialized structure composed of several layers of morphologically and functionally different cell types (Fig. 1C). These layers are mutually interconnected and together convert light signals into electrical impulses which are transferred into the visual cortex of the brain. The cells of individual retinal layers produce numerous factors with immunoregulatory properties which contribute to the immune privilege of the eye 5 . These molecules orchestrate the balance between pro-inflammatory and anti-inflammatory reactions. Among the retinal cells with the highest secretory activity belong retinal pigment epithelium (RPE) cells which produce transforming growth factor (TGF)-β, interleukin (IL-1)-β; IL-6, IL-8, granulocyte-macrophage colony stimulation factor, monocyte chemotactic protein, and several other molecules6,7. Other retinal cell types, such as microglia, Miller glia cells, retinal ganglion cells, and ciliary body cells, also produce cytokines regulating inflammation or a suppressive reaction8–10 and thus participate in the establishment of ocular homeostasis and immune privilege 11 . The immune response in the eye is weaker than other organs due to presence of anatomic and structural barriers, a lower number of immune cells, and active suppression mediated by both ocular and immune cells. However, differences in the manifestation of immune-protective mechanisms exist between the cornea and the retina. In many cases, administration of antigen into the eye induces an aberrant immune response, described as immunological deviation or anterior chamber–associated immune deviation, which is characterized by the suppression of only some components of immune response, while other parameters of response remain preserved12–14.
Immune Cells in the Healthy Eye
In general, the healthy eye contains a relatively low number of immunocompetent cells that are kept by immunosuppressive mechanisms in a quiet state. Only very low numbers of T and B lymphocytes and low levels of cytokines are present in the vitreous humor of the healthy eye. Some dendritic cells and macrophages can be detected in peripheral regions of the cornea and in the retina. More frequent in the retina are cells of natural immunity. The most important among them are microglia, which represent a resident form of nonactivated tissue macrophages 15 . By their low but persistent production of various immunoregulatory molecules, these cells contribute to the local immune homeostasis and to the immunosuppressive environment of the eye 16 . However, the infiltration of the eye with immune cells and their activation are significantly increased when the eye is damaged or diseased.
Immune Reaction in the Diseased Eye
Any damage or disease of the eye is associated with elevated cytokine production, increased infiltration with immune cells, and with a local inflammatory reaction. This reaction can be detected by the presence of activated cells and by increased immunological parameters in the vitreous humor, cornea, retina, and in other diseased parts of the eye 17 . Nevertheless, the cornea and the retina represent morphologically and functionally different parts of the eye and different immunological mechanisms are involved in the protection of these structures against pathological disorders18,19. While damages in the ocular surface induce inflammatory reaction associated with the activation of T-cell populations and with the production of pro-inflammatory cytokines, retinal degenerative diseases activate innate immune response mediated mainly by monocyte, neutrophils, macrophages, microglia, or retinal cells. Because of the heterogeneity of ocular diseases and from the reason of different protective immune mechanisms, stem cell therapy should be rationally selected, and cells preconditioned into suppressive or effector phenotype should be therapeutically used 20 .
Although the cornea is considered to be an immunologically privileged site, any chemical21,22 or physical 23 insults or pathogenic infections 24 induce infiltration in the cornea and neovascularization. Tissue inflammation starts with the secretion of pro-inflammatory cytokines, chemokines, and other factors by activated corneal cells, and this reaction is potentiated by the recruitment of cells of the adaptive immune system which produce various pro-inflammatory molecules. Similarly, orthotopic transplantation of corneal graft induces immune response against antigens of the graft. The reaction is characterized by the infiltration and accumulation of immune cells which produce various cytokines, and among them the most important role is played by Th1 and Th2 cytokines secreted by T lymphocytes, and IL-1 produced by corneal cells25,26. This reaction can be attributed to the interruption of the immunological privilege, tissue neovascularization and to the production of chemokines, which attract immune cells.
Similarly, as in the case of corneal disorders, retinal degenerative diseases, such as AMD, DR, RP, or glaucoma, are also associated with a local inflammatory reaction and cytokine burn at the site of damage27,28. The high concentrations of cytokines produced by both immune and retinal cells can be harmful for retinal cells and can elevate the progression of degeneration29–31. Autoimmune disorders of the retina, such as uveitis or autoimmune retinopathy, are also characterized by an accumulation of activated T and B lymphocytes and by the enhanced production of cytokines32–36. Nevertheless, autoimmune diseases in the eye have different immune mechanisms than, for example, AMD. Uveitis has major components of adaptive immune response, whereas AMD and similar degenerative diseases primarily involve components of the innate immune system. The key role in pathogeneses of retinal autoimmune diseases plays IL-17A produced by Th17 cells37,38. The important role in the maintenance of retinal homeostasis is also played by retinal cells, such as RPE cells or microglia, which produce immunomodulatory molecules such as TGF-b2, IL-1, IL-10, IFN-β, and IL-11. It has been shown that immunoregulatory functions in the eye are decreased when the retina is pharmacologically damaged 39 .
Although various pharmacological interventions are effective in the therapy of inflammatory reaction in the eye, these treatments only temporarily inhibit immune reaction, but do not have longer effects and do not cure retinal damage. It has been shown that individual corneal or retinal diseases differ in the involvement of main components of the immune system, but some common characteristics of inflammation are found. Therefore, therapeutic approaches involving suppression of the local inflammatory reaction and simultaneously supporting the regeneration of ocular tissues are extremely demanding. Such demands can be fulfilled by MSC therapy which have both immunoregulatory and regenerative effects.
Mesenchymal Stem Cells
MSCs are multipotent stem cells which can be obtained from various types of tissues. They produced variety of immunoregulatory molecules creating regenerative microenvironment that has potential for tissue regeneration. MSCs are most frequently isolated from bone marrow or adipose tissue. In bone marrow, they are making and repairing skeletal tissues, such as cartilage, bone, and the fat in bone marrow. In adipose tissue, MSCs convert into lipid-accumulating fat cells. The great advantage of these cells is their relatively easy preparation, good growth properties during propagation in vitro, and the possibility to use them as autologous (recipient’s own) cells. Experiments in animal models and clinical observations have confirmed that MSCs do not induce tumor growth during long-term observation, and that MSC therapy is without significant negative side effects40,41. It has been shown that MSCs possess potent immunomodulatory, anti-inflammatory, and regenerative properties and are potent producers of cytokines and growth factors. These characteristics make them a promising candidate for cell therapy of inflammatory and degenerative ocular diseases.
Immunoregulatory Properties of MSCs
The immunomodulatory properties of MSCs are mediated by multiple mechanisms, including the production of immunoregulatory molecules, regulation by direct cell-to-cell contact, negative effects on antigen-presenting cells, and by the activation of regulatory T cells (Tregs) (Fig. 2). In general, MSCs inhibit immune reactions. In in vitro studies, MSCs inhibited T- and B-cell proliferation and the production of cytokines and suppressed the activity of cytotoxic T and natural killer (NK) cells42,43. In in vivo experimental models, administration of MSCs prolonged the survival of skin or corneal allografts44,45, decreased the incidence of graft-versus-host disease 46 , and suppressed the severity of autoimmune diseases47,48. It has been shown that more distinct mechanisms contribute to suppressive effects of MSCs. MSCs express indoleamine 2,3-dioxygenase, cyclooxygenase-2, TNF-α stimulated gene 6 protein, programmed death ligand 1, Fas-L molecule, and other immunosuppressive molecules49–51. In addition, MSCs secrete cytokines which influence immune reactions in both positive and negative manner. For example, they produce TGF-β and IL-6 which are the principal cytokines regulating, depending of their concentrations, the development of anti-inflammatory Tregs and pro-inflammatory Th17 cells 52 . We have shown that the production of individual cytokines by MSCs depends on the cytokine environment where MSCs reside 53 . Since the paracrine effect of MSCs based on the production of cytokines and growth factors was suggested as the main mechanism of their action, there are important observations that the secretory pattern of MSCs can be intentionally modified by their preconditioning with selected cytokines or other factors 20 . This observation suggests that the production of desirable cytokines and the functions of MSCs could be regulated by external factors and thus could be possible to prepare MSCs with improved therapeutic potential.

The main paracrine mechanisms of immunoregulatory effects of MSCs. Abbreviations: MSC, mesenchymal stem cell; DC dendritic cell; NK cell, natural killer cell; Treg, regulatory T cell; TGF-β, transforming growth factor-β; IL-6, interleukin-6; IL-10, interleukin-10; PGE2, prostaglandin E2; HGF, hepatocyte growth factor; PD-L1, programmed death ligand 1; CCL2, chemokine CCL2.
In addition to immunoregulatory properties, MSCs have anti-apoptotic effects. It has been demonstrated that increased levels of pro-inflammatory cytokines during inflammation in the eye can induce apoptosis of ocular cells 31 . It has also been shown that chronic inflammation is associated with endoplasmic reticulum stress which promotes the induction of apoptosis of corneal cells 54 . Therefore, the inhibition of a local inflammatory reaction by MSCs and attenuation of apoptosis might be beneficial in the treatment of ocular diseases. In this respect, it was shown that MSCs inhibit the expression of pro-apoptotic genes and decrease the number of apoptotic cells in the corneal explants cultured in the presence of apoptosis-inducing pro-inflammatory cytokines 31 .
Regenerative Properties of MSCs
MSCs are also potent producers of various growth factors which stimulate proliferation of ocular cells and thus support the regeneration of damaged or diseased ocular tissues. Among the growth factors which are produced by MSCs and that contribute to corneal or retinal tissue regeneration are hepatocyte growth factor, nerve growth factor, glial cell–derived neurotrophic factor, insulin-like growth factor-1, pigment epithelium growth factor, fibrocyte growth factor, platelet-derived growth factor, epidermal growth factor, angiopoietin-1, erythropoietin, vascular epithelial growth factor, TGF-β, and so on.55–57 Some of these factors are secreted by MSCs constitutively, while the production of others is increased after activation with pro-inflammatory cytokines. It has been suggested that the production of growth factors and their paracrine action are the main mechanisms of the tissue regenerative action of MSCs (Fig. 3). In addition, the enhanced secretion of some growth factors is observed following differentiation of MSCs into cells expressing corneal or retinal cell markers 58 . All these observations indicate that the preconditioning of MSCs with selected factors or their differentiation into cells expressing corneal or retinal cell markers could enhance their secretory activity and increase the therapeutic potential.

The main mechanisms of regenerative effect of MSCs for corneal and retinal disorders.
MSC-Based Therapy of Corneal Damages and Retinal Diseases
So far, abundant data on the use of MSC therapy for corneal or retinal diseases have been published. Various types of MSCs, such as bone marrow–derived, adipose tissue–derived, or MSCs isolated from other sources, have been tested. The cells for the therapy were used in a naïve form or preactivated with various cytokines or other agents. Also, different modes of their application have been compared. For the treatment of corneal disorders, topical applications using different carriers to fix the cells were most frequently tested. In cases of retinal diseases, intravitreal injection, subretinal implantation, or intravenous route have been compared. However, the optimal way of MSC application will depend on the type and the state of the disease.
Treatment of Corneal Damages
Any damage of the cornea, either by mechanical or chemical insults or by a pathological reaction, induces an inflammatory reaction at the site of injury and a rapid increase in the production of cytokines and chemokines by both cells of the cornea and the activated immune cells17,19,23. Therefore, the inhibition of a deleterious immune reaction and support for the surviving corneal cells by application of MSCs could represent an effective direction of the therapy. It has been shown that the treatment of diseased cornea with bone marrow–derived or adipose tissue–derived MSCs significantly suppressed the local production of pro-inflammatory cytokines, inhibited the expression of proapoptotic genes Bax and p53, and facilitated corneal regeneration 31 . If the corneal injury is more extensive and also involves the limbal region, where limbal stem cells (LSCs) reside, the corneal disease known as limbal stem cell deficiency (LSCD) occurs. In such cases, the damaged cornea cannot heal by autologous LSCs. The only therapeutic possibility is a limbal tissue transplantation 59 or transplantation of LSCs or MSCs 60 . Using an experimental rabbit model of chemically induced corneal damage, we demonstrated that MSCs can be as efficient in the treatment of LSCD as are LSCs 21 .
Treatment of Retinal Diseases
Retinal degenerative diseases represent the main cause of decreased quality of vision and even blindness. In addition to loss of retinal cells and to a progressive retinal degeneration, these disorders are accompanied by a local harmful inflammatory reaction. Therefore, the suppression of this reaction and support for the surviving retinal cells by MSCs were proposed as a promising way for the treatment of retinal disorders. The beneficial effects of MSCs on retinal diseases have been well demonstrated61–63. In a model of cytokine-induced ocular inflammation, we showed that the local production of pro-inflammatory cytokines by retinal cells can be significantly decreased by intraocular administration of MSCs 64 . The therapeutic potential of MSCs has been also documented in various experimental models of retinal degenerative diseases. For example, in a model of AMD, the intravitreal application of MSCs prevented pathological changes induced in the retina by the application of sodium iodate65,66. In experimental models of DR in mice with streptozotocin-induced diabetes, the application of MSCs had a positive effect on the retinal architecture and improved visual function measured by electroretinography 67 . RP is a group of retinal diseases characterized by a loss of photoreceptor cells. As the experimental model of this disease, mice with the rdl mutation causing the early loss of the photoreceptors are used. The treatment of these mice with MSCs supported the survival of photoreceptors and showed significant therapeutic benefits 68 . Another type of retinal degenerative disease is represented by glaucoma, which is a heterogeneous group of diseases caused by increased intraocular pressure and characterized by the progressive loss of retinal ganglion cells. To date, various experimental models have been established to imitate this type of retinal disorder and have been used to evaluate the therapeutic effects of MSC therapy 69 . For example, Mead et al. 70 showed that intravitreal administration of MSCs in a rodent model of glaucoma decreased ocular pressure and provided a neuroprotective effect. Similarly, as in other models of glaucoma, the neuroprotective effects of MSC-based therapy have been described71,72.
Altogether, several animal models of corneal disorders and retinal degenerative diseases have proved the therapeutic properties of various types of MSCs and warranted their testing in preclinical and clinical studies. The selected experimental models of corneal and retinal diseases, where therapeutic effects of MSCs were studied, are summarized in Table 1.
Selected Experimental Models of MSC-Based Therapy for Corneal and Retinal Disorders.

Abbreviations: MSCs, mesenchymal stem cells; AD-MSCs, adipose tissue–derived MSCs; BM-MSCs, bone marrow–derived MSCs.
Clinical Studies Based on the Application of MSCs for the Treatment of Corneal and Retinal Disorders
The encouraging results from experimental and preclinical studies84,85 have stimulated clinical trials to use MSCs in patients with corneal and retinal disorders60,63 The first studies showed important conclusion that the application of MSCs is not associated with serious complications40,41.
Corneal and Ocular Surface Disorders
There is currently a group of clinical trials evaluating the treatment of the cornea or ocular surface with MSCs. Stem cells are administered locally using different modes including topical eye drops, subconjunctival, or intrastromal injections. These trials aim to assess the efficacy of MSCs in treating conditions such as LSCD, ocular chemical burns, dry eye disease, keratoconus, or corneal dystrophies. Selected clinical studies using MSC application for the treatment of ocular surface disorders are shown in Table 2. The majority of trials emphasize the safety of the treatment and improvement of corneal architecture and visual acuity57,60.
Selected Human Clinical Studies Using MSC Therapy for Corneal or Retinal Diseases.
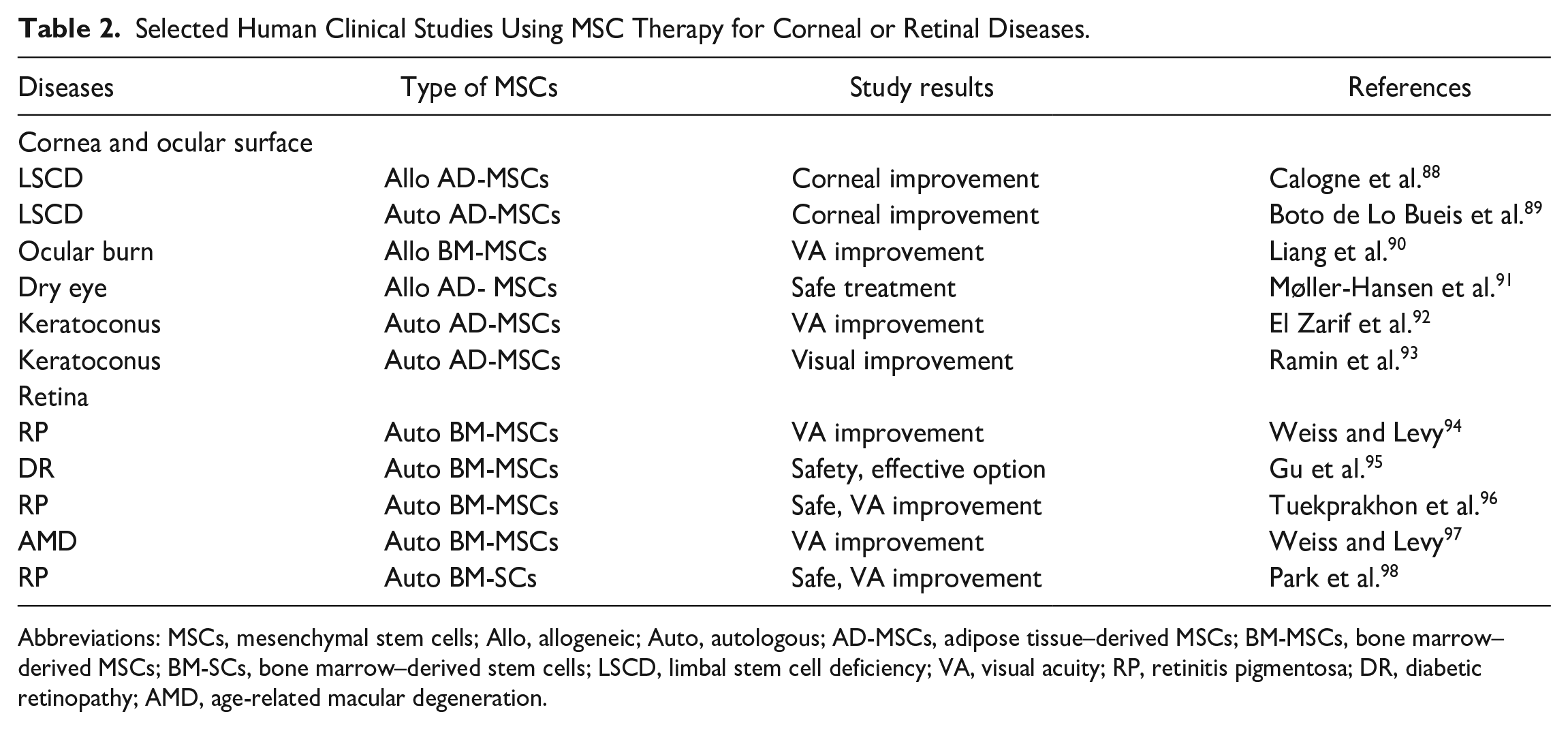
Abbreviations: MSCs, mesenchymal stem cells; Allo, allogeneic; Auto, autologous; AD-MSCs, adipose tissue–derived MSCs; BM-MSCs, bone marrow–derived MSCs; BM-SCs, bone marrow–derived stem cells; LSCD, limbal stem cell deficiency; VA, visual acuity; RP, retinitis pigmentosa; DR, diabetic retinopathy; AMD, age-related macular degeneration.
Retinal Diseases
The therapeutic effects of various types of MSCs for the treatment of retinal diseases were studied in patients with AMD, DR, RP, glaucoma, macular holes, optic nerve diseases, and other disorders. Although these disorders have different etiologies and various starting causes, cumulative damage, loss of specialized retinal cells, chronic inflammation, immune cell activation, and enhanced cytokine secretion represent the main characteristics of retinal degenerative diseases. For AMD is a typical loss of cells of the RPE which play a major role in photoreceptor nutrition. In DR, which is primarily caused by hyperglycemia in diabetes mellitus, the reduction in the number of pericytes and the decreased number of retinal neurons and glial cells result in the interruption of retinal integrity and in progressive loss of vision. RP is a genetic disorder which is caused by a progressive loss of the rod photoreceptor cells. On the contrary, a group of retinal diseases called glaucoma is characterized by increasing high pressure that causes a slowed exit of the aqueous humor through trabecular meshwork that causes gradual damage to the optic nerve and vision loss. Since the loss of specialized retinal cells and local inflammatory reaction are the main causes in the progression of the retinal degenerative diseases, the support for surviving retinal cells and the inhibition of inflammation could be the prospective approaches to treat these diseases. Recently, the most prospective treatment options are provided by MSCs63,86,87. Although most clinical studies are still running, the first published results in patients with RP, DR, and optic nerve diseases showed amelioration of retinal architecture and improvement of visual acuity94,95,99,100. Table 2 shows selected clinical studies using MSCs for the treatment of retinal degenerative diseases. However, further clinical trials made in larger cohorts of patients with diseased cornea or retina are still needed.
Perspectives of MSC-Based Therapy for Corneal and Retinal Disorders
Abundant data from experimental, preclinical, and first clinical studies have demonstrated the potential of MSC therapy for the treatment of a wide range of different types of ocular diseases and injuries. Recent trials have focused on the study of the safety of MSC therapy, on the possibility of activation or modification of MSCs prior to application, the optimization of the dose of cells, the identification of optimal routes for application, and on the possibility to replace the cells with their paracrine products. One of the most important issues concerning MSC-based therapy is the safety of MSC administration. The extensive analysis of results from studies using MSCs in more than 1000 patients confirmed that the therapy is safe and did not reveal a significant association between MSC administration and toxicity of the application, internal organ infection, tumor growth, or death 41 .
However, a lot of questions about the survival of therapeutically administered MSCs and about the mechanism of their action remain to be answered. Although numerous studies showed that MSCs are long-lived and their presence in recipients was demonstrated even a few months after the application81,101, other studies have suggested that in vitro expanded and transplanted MSCs are extremely short-lived102–104. Despite these observations, showing the rapid elimination of transplanted MSCs in vivo, there are abundant data demonstrating immunoregulatory and therapeutic effects of MSC administration for a long period of time after their application105,106. Since the long-lasting effects of MSCs were observed even in studies without the possibility to detect surviving MSCs, the effects of applied MSCs were explained by a rapid hit of MSCs (release of cytokines or exosomes, modulation of function of phagocytes) without the necessity for MSC survival107,108. This issue, that is, a short-time or a long-lasting survival of therapeutically administered MSCs, and whether the treatment is a short effect or a long-lasting cure, should be conclusively determined. Another issue that deserves attention is the use of xenogeneic (mainly human) MSCs in experimental animal studies (see also Table 1). Many of these studies have described the positive effects of human MSCs for the therapy of corneal defect healing73,75 and for the treatment of retinal disorders66,83 in rodents. However, the results of such experiments could be influenced by the interspecies barrier and by an incompatibility which limits the interaction between human MSCs and the cells of the immune system of the recipient. Lohan et al. 109 demonstrated that human MSCs injected into rats do not have the same therapeutic effect as rat MSCs have, and the immunoregulatory effects of human MSCs were strongly limited by the interspecies barrier. Norte-Munoz et al.110,111 compared therapeutic effects of intravitreally administered MSCs from syngeneic, allogeneic, and xenogeneic (human) donors in mice. The authors concluded that the specific functional and anatomical retinal response strongly depends on MSC transplantation modality, and that syngeneic MSCs may be more beneficial than even allografts, and that xenografts were the most detrimental. Therefore, the experiments based on the use of human MSCs in rodent models should be taken with precaution, and it is difficult to translate the results from such experimental studies into clinical practice, where autologous or allogeneic MSCs are used. Some of these problems with cellular incompatibility could be overcome by the use of cell-free MSC products (supernatants, vesicles) obtained after cultivation of MSCs112,113 or using severely immunodeficient mouse strains 114 or humanized mouse models 115 . Nevertheless, it is apparent that more distinct mechanisms are responsible for the MSC actions. They include the production of various cytokines and growth factors, the differentiation potential, the hit and die mechanism, the secretion of vesicles, the activation of immunoregulatory cell populations, and the anti-apoptotic effects. In addition, it could be taken into account that the immunoregulatory and regenerative effects of MSCs are dependent on the cytokine environment, where MSCs are applied20,53.
Although too many questions concerning MSC therapy remain to be answered, the beneficial effects of MSC therapy for various ocular diseases are well documented, and thus this approach deserves further attention.
Conclusion
Stem cell–based therapy represents the most promising approach for the treatment of currently uncurable ocular disorders. Corneal damages and retinal degenerative diseases are generally associated with immune cell infiltration, a local inflammatory reaction, cytokine burn, and with a progressive loss of specialized ocular cells. Therefore, effective treatment should be focused on inhibition of the deleterious inflammatory reaction and on the support for the surviving specialized ocular cells. These requirements are fulfilled by MSCs, which produce numerous immunoregulatory and growth factors and can thus regulate the immune response in the disease eye and support ocular tissue regeneration. This conclusion is supported by findings on numerous experimental and preclinical models and clinical trials. Although the results published so far are very encouraging, this cell therapy requires further experimental and clinical studies. Nevertheless, this therapeutic approach could lead to improvement of the quality of life in patients with corneal and retinal diseases.
Footnotes
Author Contributions
V.H. proposed the concept and conceptualized the study. V.H. and B.H. analyzed data and wrote the original draft. E.J. and K.P. performed the literature review and contributed to writing. K.P. prepared figures. All authors read and approved the final version of the manuscript.
Ethical Approval
This study was approved by our institutional review board.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Consent for Publication
All authors agree to submit the manuscript to the journal.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. This work was supported by the Programme Johannes Amos Comenius, the Ministry of Education, Youth and Sports of the Czech Republic – project with registration number CZ.02.01.01/00/22_008/0004562 (ExRegMed).
