Abstract
Bone marrow-derived mesenchymal stem cells (MSCs) could exert a potent immunosuppressive effect, and therefore may have a therapeutic potential in T-cell-dependent pathologies. In the present study, we aimed to determine whether MSCs could be used to control graft-versus-host disease (GvHD), a major cause of morbidity and mortality after allogeneic hematopoietic stem cell transplantation (HSCT). MSCs were isolated from Lewis rat bone morrow and then cultured in 10% FBS DMEM at 37°C for 4 weeks. The enriched conventional MSCs and macrophages were purified by auto-MACS. Cloned MSCs were obtained by cloning using the limiting dilution method and expanded up to more than 6 months. The identity of MSCs was confirmed by their typical spindle-shaped morphology and immunophenotypic criteria, based on the absence of expression of CD45 and CD11b/c molecules. Both types of MSCs were also tested for their ability to differentiate into adipocytes. We showed that MSCs, like macrophages, exhibit immunomodulatory properties capable of inhibiting T-cell proliferation stimulated by alloantigens, anti-CD3e/CD28 mAbs, and ConA in a dose-dependent manner in vitro. After performing adoptive transfer, MSCs suppressed systemic Lewis to (Lewis × DA)F1 rat GvHD. In contrast to the immunosuppressive activities of conventional MSCs, the cloned MSCs enhanced T-cell proliferation in vitro and yielded no clinical benefit in regard to the incidence or severity of GvHD. Therefore, these rat models have shown intriguing differences in the suppression effects of lymphocyte proliferation and GvHD prevention between short-term cultured conventional MSCs and cloned MSCs.
Keywords
Introduction
Allogeneic hematopoietic stem cell transplantation (HSCT) is the most efficient treatment for many hematological malignancies and for primary immunodeficiencies (5,19,20,53). HSCT relies on the elimination of the hematopoietic compartment by high-dose chemotherapy and irradiation, and the reconstitution of a new hematopoietic system provided by the donor hematopoietic stem cells (49,50). However, the transplants also contain mature T cells that can induce graft-versus-host disease (GvHD), a life-threatening complication of allogeneic HSCT (9). These donor T cells are strongly activated after the recognition of alloantigens presented by the recipient antigen-presenting cells (APCs), and infiltrate several target organs, such as skin, liver, and the gastrointestinal tract, where they exert cytotoxic effects (8,44). Therefore, the major goals of HSCT are to modulate alloreactivity by donor allogeneic T cells without causing GvHD, and to preserve the graft-versus-leukemia and graft-versus-infection effects.
Mesenchymal stem cells (MSCs) reside in bone marrow (BM) as a nonhematopoietic cell population, and are characterized by their ability to self-renew and differentiate into mesenchymal tissues, such as bone, cartilage, or adipose tissue (7,13,31,39). Several reports suggest that MSCs might exert a potent immunosuppressive effect in vitro and in vivo, and therefore may have therapeutic potential in T-cell-dependent pathologies (27,53). Recent MSCs are also an interesting new therapeutic approach in solid organ transplantation owing to immunomodulatory properties demonstrated in vitro and in vivo (26,29,37,51). Cell-based tolerogenic therapy has gained momentum in recent years with the identification of endogenous suppressor cells capable of inhibiting the lymphocyte effector functions (10). The majority of these studies have shown that MSCs can inhibit the activation of T-cell proliferation and secretion of cytokines. These include interleukin-(IL-2) and interferon-γ (IFN-γ) induced by mixed lymphocyte reactions (MLRs) (4,28,38,51), mitogens (14,28), and receptor engagement (1,23). In vitro studies indicate that soluble factors secreted by MSCs may be the primary mode of immunomodulation (21,51); however, cell contact-dependent mechanisms of suppression may also coexist (2,12). Regardless of the mechanism of action, the efficacy of MSC transplantation in severe acute GvHD in clinical trials demonstrates the potency of these cells as immunosuppressive agents (27,41). These immunosuppressive properties of MSCs open attractive possibilities in the field of solid organ transplant or HSCT. The purpose of this study is to establish whether MSCs might be used to control GvHD, a major cause of morbidity and mortality after allogeneic HSCT.
Materials and Methods
Animals
Adult male Lewis (RT1l), DA (RT1a), and (Lewis × DA)F1 rats were purchased from Shizuoka Laboratory Animal Center (Shizuoka, Japan) and used at 4–6 weeks of age. The animals were maintained under standard conditions and fed rodent food and water according to the laboratory animal care principles and the guide for the care and use of laboratory animals established by our institution.
Isolation and Culture of MSCs From Adult Rat Bone Marrow
Adult Lewis rat bone marrow cells were collected by flushing the long bones with DMEM medium (1.0 g/L glucose without L-glutamine and phenol red; Nacalai Tesque, Inc. Kyoto, Japan) and all cells were cultured in 150-mm cell culture dish in DMEM supplemented with 10% fetal bovine serum (FBS; Invitrogen, Tokyo, Japan), 2 mM l-alanyl-glutamine (GlutaMAX™, Invitrogen), antibiotic-antimycotic mixed solution (penicillin, 100 U/ml; streptomycin, 100 μg/ml; amphotericin B, 250 ng/ml, Nacalai Tesque). The culture medium was changed every week. On the day 14, adherent cells were trypsinized (5.0 g/L trypsin, 5.3 mmol/L EDTA solution), harvested, and then plated into five new 150-mm dishes. On day 28, all adherent cells were trypsinized and harvested for cell sorting. The morphology of the adherent cells was assessed by observation with a phase-contrast microscope (Olympus, Tokyo, Japan).
MSC Purification and Cloning
For the isolation of the purified conventional MSCs and macrophages, the above suspended 4-week cultured adherent bone morrow-derived cells were stained with phycoerythrin (PE)-conjugated anti-rat CD11b (OX-42, Serotec, Oxford, UK) and CD45 (BD Pharmingen, San Diego, CA) followed by anti-PE MicroBeads (Miltenyi Biotec, K.K. Tokyo, Japan) and then sorted on an auto-MACS system (magnetic activated cell sorter; Miltenyi Biotec). The negative fraction, enriched MSCs, and positive fraction, enriched macrophages were evaluated by FACScan (fluorescence-activated cell sorter; Becton Dickinson) and more than 98% purity of the MSC, 85% purity of the macrophages population were obtained. The cloned MSCs were obtained by cloning using limiting dilution method and then were passage up to more than 6 months.
Adipocyte Differentiation of Cultured MSCs
MSCs were cultured in six-well plates with MSC culture medium until they reached confluence. For differentiation, MSC were induced to differentiate into adipocyte in 10% FBS DMEM medium (Nacalai Tesque), adipocyte differentiation supplement (containing insulin, transferrin selenious acid and dexamethasone; DS Pharma Biomedical Co., Ltd. Osaka, Japan), and 2 mM l-alanyl-glutamine (GlutaMAX™, Invitrogen). Cells were cultured for 21 days, and then switched to fresh medium for 3–4 days. Induced adipocytes were visualized after fixation in 10% formaldehyde buffer (Nacalai Tesque) for 10 min followed by washing with PBS. After washing with 60% isopropanal (Wako) for 1 min, they were stained with Oil Red O (Muto Pure Chemicals Co., Ltd. Tokyo, Japan) for 10–20 min.
Flow Cytometric Analysis
MSCs were collected and suspended in PBS and then incubated at 4°C for 20 min with an optimal concentration of PE-conjugated anti-rat CD11b (OX-42, Serotec), CD90 (OX-7, Serotec), CD45 (OX-1, BD Bioscience, San Diego, CA), CD54 (1A29, BD Bioscience), RT1B (OX-6, BD Bioscience), RT1A (OX-18, BD Bioscience), CD80 (3H5, BD Bioscience), and CD86 (24F, BD Bioscience) diluted with PBS containing 1% fetal calf serum. Flow cytometric analysis was performed using a Becton Dickinson Immunocytometry System and a FACS Calibur cytometer (San Jose, CA). Data acquisition and analysis were performed using the Becton Dickinson CellQuest software.
T-Cell Purification and Proliferation Assays
Spleens from naive Lewis rats were harvested and single-cell suspensions were prepared by passing the tissue through cell strainers (70 μm; Falcon, Franklin Lakes, NJ). The cell suspension was overlaid on Ficoll Isopaque (Lympholyte Rat, Cedarlane Lab. Ltd., Ontario, Canada) and centrifuged at 3000 rpm for 25 min at 24°C. The cells of the interface layer were harvested and washed twice in PBS. Erythrocytes were lysed by hypotonic shock, and the remaining cells were washed with GIT medium (NIHON Pharmaceutical, Tokyo, Japan), followed by PanT MicroBeads (Miltenyi Biotec), and positively sorted on an auto-MACS (Miltenyi Biotec). Enriched PanT cells population were evaluated by FAC Scan (Becton Dickinson), and more than 98% purity of the PanT cells population were routinely obtained.
Enriched PanT cells (1 × 105) were mixed with Lewis enriched MSCs, macrophages or cloned MSC cells (1 × 103-1 × 105) and cultured with DA spleen CD11b/c+ cells (APCs, 1 × 104) in a flat-bottom 96-well white plate (Corning International K.K. Tokyo, Japan) at a final volume of 200 μl/well of the GIT medium containing 50 mM 2-mercaptoethanol (Wako) in humidified atmosphere at 37°C for 5 days. To evaluate and assess the response to mitogenic stimulations, purified PanT cells were cultured at 37-C for 3 days with concanavalin A (ConA; Wako) at 2 μg/ml or anti-CD3e (G4.18, BD Pharmingen), anti-CD28 monoclonal antibody (JJ319, BD Pharmingen) at 1 μg/ml, and various numbers (1 × 103–1 × 105) of Lewis enriched MSCs, macrophages, or cloned MSC cells. The proliferation of T cells was measured with cell proliferation ELISA kits (Roche Diagnostics Gmbh, Penzberg, Germany) (4). Briefly, the cells were labeled with 5-bromo-2-deoxyuridine (BrdU) solution at 10 μl/well and incubated for an additional 2 h at 37°C. After centrifugation, the supernatant was removed, 200 μl/well of fixation and DNA denaturation (FixDenat) solution was added to the cells and they were reincubated for 30 min at 15–25°C. The cells were cultured for 90 min with anti-BrdU-peroxidase solution and subsequently washed three times. After adding substrate solution at 100 μl/well, the BrdU incorporation was measured with a chemiluminescence reader (Wallac ARVOTM SX; PerkinElmer, Inc., Wellesley, MA) and the data were processed using Wallac1420 manager software (PerkinElmer).
Total Lymph Node (LN) Lymphocyte Purification and Assay of Systemic GvH Reactivity
For the preparation of purified LN cells, naive Lewis rat LN were harvested and gently ground with frosted objective slides in PBS. They were filtered through 200-mesh nylon filter (Tokyo Screen, Ltd, Tokyo, Japan) for single-cell suspension preparation and then separated by Lympholyte-Rat (Cedarlane Laboratories). Total LN single-cell suspensions (1.0 × 107) were intravenous injected into 4-week-old Lewis × DA F1 rats after whole-body 6-Gy γ-irradiation on day -2. MSC cells were administered to the host rat via tail veins at day -1 and day 6. The rats were weighed on alternate days during the active phase of the GvHD. Animals undergoing typical GvHD showed rapid weight loss and commonly described signs of the disease, including ruffled fur, reddening of the skin, a hunched posture, and ultimately death.
Statistical Analysis
A statistical evaluation for graft survival was performed using the Kaplan-Meier's test. Values of p < 0.05 were considered to be statistically significant. All in vitro experiment data were representative of three independent experiments and expressed as the mean ± SD.
Results
MSC Morphology and Proliferation
Whole mononuclear fractions of BM cells were cultured. Over the first 2 weeks, almost all of the suspended cells died, leading to a very small viable cell population. The adherent cells were replated by trypsinization when the cells reached 70–80% confluence. The MSCs were identifiable as colonies of adherent cells with a fibroblast appearance at the end of the 4 weeks (Fig. 1). After sorting and repeated subculturing, the cultures contained a homogenous layer of fibroblast-like cells (Fig. 1B). These cells can be maintained in continuous culture for more than 6 months. Initially, the growth rate was slow, but it gradually increased. The total expansion over 18 passages was extrapolated to be in the range of 1012 cells (Fig. 1C).
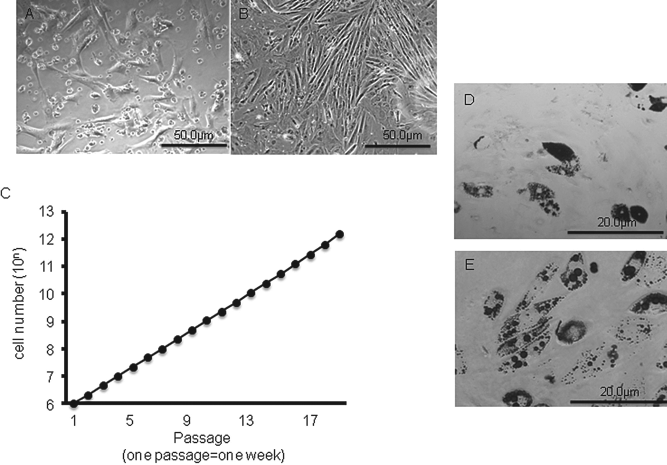
Mesenchymal stem cell (MSC) morphology and proliferation. Established, confluent MSCs in the culture displayed a typically homogeneous fibroblast-like pattern. Phase-contrast view of conventional MSCs cultured after sorting at 4 weeks (A) and cloned MSCs at 6 months (B). (C) Growth curve of bone marrow (BM)-derived cloned MSCs. The total expansion over 18 passages was extrapolated to be in the range of 1012 cells. Both MSCs (D) and cloned MSCs (E) formed lipid-filled adipocytes detected by Oil Red O staining. Data are consistent with the outcomes from similar experiments. Scale bars: 50 μm (A, B), 20 μm (D, E).
Adipogenic Differentiation Potential of BM-Derived MSCs
After 3 weeks of culture in the adipogenic induction medium containing insulin and dexamethasone, both conventional (Fig. 1D) and cloned (Fig. 1E) MSCs formed adipocytes with lipid cytoplasm, as shown by positive staining with Oil Red O.
Characterization of MSCs and Macrophages by Flow Cytometry
We determined the immunophenotype of the sorted CD45 and CD11b negative fractions, enriched conventional MSCs and positive fractions, and enriched macrophages at 4 weeks and compared them by flow cytometry to those of the monolayered single colony-derived adherent cells. The morphology of MSCs was larger than that of lymphocytes, so we required significant adjustment of flow cytometry gating established for lymphocytes (data not shown). The immunophenotypical profile of the macrophages' fraction expressed significantly higher CD45, CD11b, CD90, CD54, and OX-18 and lower OX-6, CD80, and CD86 molecules, as shown in Figure 2. Both types of MSCs were CD45 negative, and this finding is consistent with a nonhematopoietic origin and also confirms that either hematopoietic or mononuclear cells had been depleted from the culture. In line with this finding, no macrophages (CD11b cells) could be detected in the culture. Finally, both MSCs were found to be CD90 and CD54 positive. With respect to the markers known to strongly participate in immune activation, both MSCs were positive for major histocompatibility complex (MHC) class I (OX-18), but did not express MHC class II (OX-6) or B7–1 (CD80), as shown in Figure 2.

Characterization of MSCs and macrophages by flow cytometry. The cells were labeled with phycoerythrin (PE)-conjugated antibodies and examined by flow cytometry. Histograms demonstrating the expression of surface molecules were plotted against the control (anti-IgG). The immunophenotypical profile of the macrophages expressed higher CD45, CD11b, CD90, CD54, OX-6, and OX-18, and lower CD80 and CD86. Both MSCs expressed higher CD90, CD54, OX-18, lower CD80, and no expression of CD45, CD11b, OX-6, and CD86. Data are consistent with the outcomes from three related studies.
Conventional MSCs Have a Regulatory Activity, but Cloned MSCs do not
To specifically examine the suppressor cell activity of the conventional MSCs, cloned MSCs, and macrophages, the cells were isolated from Lewis naive rats as described above. Suppression assays were performed with increasing numbers of conventional MSCs, cloned MSCs, and macrophages to test their suppressive activity using anti-CD3e/CD28 or ConA along with autologus feeder APC cells. Both conventional MSCs and macrophages were able to suppress proliferation of naive T cells in a dose-dependent manner (Fig. 3, left and middle columns) at a ratio of 1:1 and 1:10 (Fig. 3). Similar to macrophage cells, conventional MSCs inhibited the proliferation of naive T cells stimulated with alloantigen by allogeneic APCs. Unlike the immunosuppressive activities of the conventional MSCs and macrophages, the cloned MSCs enhanced T-cell proliferation stimulated not only by ConA but also alloantigen in vitro (Fig. 3, right column). These data demonstrate that the immune suppression function of cloned MSCs is completely different from that of conventional MSCs.
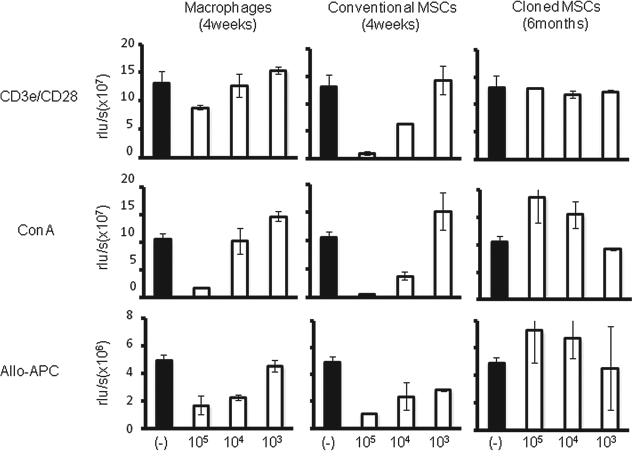
Conventional MSCs have regulatory activity in inhibiting T-cell proliferation induced by various stimuli, but cloned MSCs do not. MSCs, like macrophages, can inhibit T-cell proliferation induced by anti-CD3e/CD28 mAb, concanavalin A (ConA), and allogeneic stimulator cells in a dose-dependent manner. In contrast, the cloned MSCs enhanced the T-cell proliferation stimulated by ConA and alloantigen (allo-APC). Data are representative of those obtained from three independent experiments. rlu/s, relative light units/second.
Conventional, not Cloned MSCs, Suppressed GvH Reactions
Based on the above in vitro findings, a clinically relevant in vivo model was used to investigate the role of treatment with MSCs on allogeneic responses. The systemic GvH reactivity of the conventional MSCs, cloned MSCs, and macrophages treatment was tested by injecting these cells (1 × 107 each) via the tail veins at two different time points (namely, day 1 and day 6 after the LN-derived T-cell infusion) of the (Lewis × DA)F1 hybrid recipients (Fig. 4A). As shown in Table 1 and Figure 4B, the (Lewis × DA)F1 recipients of 1.0 × 107 nave Lewis LN lymphocytes developed host weight loss and definite lethality (survived to 24.3 ± 2.7 days). The conventional MSC treatment significantly inhibited GvHD mortality (55.6 ± 10.4 days) compared to the recipients of only the naive LN cells (p = 0.0001). Furthermore, the macrophage treatment also significantly inhibited GvHD mortality (49.7 ± 13.3 days) compared to the recipients of only the naive LN cells (p = 0.0001). Consistent with in vitro observations, cloned MSCs had no protective effect compared to the no-treatment group (30.4 ± 11.3, p = 0.4277). In the cloned MSC treatment group, extensive perivascular infiltration of the mononuclear cells and tissue damage were observed in histological studies of the skin, tongue, lung, and liver from the naive lymphocytes transferred to the host on day 14. In contrast, relatively few infiltrating mononuclear cells and almost a normal tissue architecture were observed in the conventional MSC and macrophages treatment groups (data not shown).

Treatment with conventional MSCs, but not cloned MSCs, suppressed graft-versus-host (GvH) reactions. (A) Experiment design. (B) Transfer of the naive Lewis lymph node (LN) T cells (1 × 107) to (Lewis × DA)F1 rat led to host's weight loss and definite lethality in a 6-Gy-irradiated rat GvHD model (circles). Nonetheless, the groups of conventional MSC-treated (triangles) and macrophage-treated (squares) host rats significantly suppressed the GvH reaction. Conversely, there were no protective effects in the cloned MSCs treatment group (X).
MSC Treatment Suppressed Systemic Graft-Versus-Host Disease

Model of systemic graftversus-host disease (GvHD) was performed by transferring 1.0 × 107 naive Lewis lymph node (LN) cells to (Lewis × DA)F1 rats after 6.0 Gy radiation. The p-value was compared with control (no treatment) group rats and determined by the Kaplan-Meier's test. MSCs, mesenchymal stem cells.
Discussion
MSCs have gained increasing interest for their application in cell-based therapies and tissue engineering approaches (11,17,24). This is based on their potential for self-renewal, multilineage differentiation, and easy accessibility (35). MSCs were first described as a nonhematopoietic stem cell population within adult bone marrow that supports the constant generation of hematopoietic cells. MSCs can give rise to a typical set of stromal elements, such as bone, cartilage, and adipose tissue (35). Another therapeutic potential for MSCs is an immunosuppressive effect in vitro. Data show that human, nonhuman primate, and mice MSCs can inhibit T-cell proliferation induced either in a MLR or by nonspecific mitogens (4,28,38,51). This suppression occurs regardless of the MHC of MSCs, or stimulator and responder lymphocytes. The immunoregulatory effect of MSCs seems, at least in part, to be mediated by the production of cytokines, such as IL-6 (15), IFN-γ (22,43), transforming growth factor-β1 (TGF-β1), nitric oxide (40), human leukocyte antigen-G5 (45), and hepatocyte growth factors (25). Data suggest this to be dependent on the induction of T-cell apoptosis related to the conversion of tryptophan into kynurenine by indoleamine 2,3-dioxygenase expressed by MSCs in the presence of IFN-γ (36). MSCs are especially interesting for transplantation medicine. Data indicate that they escape immunity and downregulate the immune response (3). Therefore, in the present study, we aimed to establish an animal model to evaluate the use of rat MSCs for the prevention or treatment of GvHD.
Since MSCs can rapidly differentiate in culture, and the culture of BM-derived MSCs is frequently contaminated by hematopoietic progenitors that tend to over-grow in cultures, we first developed sorting and culture conditions that produced high numbers of highly purified MSCs. However, it should be noted that, unlike conventional MSCs, the expansion of cloned MSCs was a very long and cumbersome process. Nevertheless, cloned MSCs that we used in the experiment always exhibited a spindle-shaped fibroblastic morphology and could differentiate into adipogenic lineages (Fig. 1B, C, E). The phenotypic character of MSCs is conserved among different species, including mice (23,40) and humans (1,14,30). Our data are consistent with this observation. Our rat conventional and cloned MSCs had the same phenotype since they expressed MHC class I molecules but lacked expression of MHC class II and mature leukocyte antigens (Figs. 1 and 2). The phenotypic pattern of our MSCs supported the working hypothesis that they would have low immunogenicity.
Natural suppressor cell activity is defined as the ability of unprimed effector cells to suppress the response of lymphocytes to immunogenic and mitogenic stimuli without MHC restriction (34,42). These cells have been found in mouse (16), rabbit (46), and human BM (32). The lineage of natural suppressor cells remains controversial. They have variously been characterized as having a T lymphocyte, early natural killer (NK), null cell, or macrophage lineage (42,54), and have also been shown to prevent the occurrence of acute GvHD when cotransplanted with allogeneic murine donor BM (33, 48,52). To distinguish and specifically examine the suppressor cell activity of the MSCs, we isolated the conventional MSCs and macrophages at the short-term culture period and cloned MSCs from Lewis naive rats as described above. Suppression assays were performed with increasing numbers of conventional MSCs, cloned MSCs, and macrophages to test their suppressive activity using anti-CD3e/CD28 or ConA along with autologus feeder APC cells. Consistent with the previous reports (4,28,38,51), both conventional MSCs and macrophages were able to suppress proliferation of naive T cells in a dose-dependent manner. Like macrophages, conventional MSCs inhibited the proliferation of naive T cells stimulated with alloantigen by allogeneic APCs at a ratio of 1:1 and 1:10 (Fig. 3). In classic mixed-lymphocyte reactions, conventional MSCs suppressed T-cell proliferation independent of MHC matching between T cells and MSCs (14). The mechanisms for MSC-induced modulation of the T-cell alloresponse have been considered, including the influence of MSCs on distinct sets of APCs, such as monocytes, dendritic cells, or natural killer cells (1,6). The effects on APCs are therefore thought to cause a deviation in the immune response via pathways of indirect antigen presentation in favor of immune tolerance. Data from our present study confirm the ability of conventional MSCs to suppress T-cell proliferation in vitro in the rat system. In contrast to the immunosuppressive activities of conventional MSCs and macrophages, the cloned MSCs enhanced the T-cell proliferation stimulated not only by ConA but also alloantigen in vitro (Fig. 3). These data demonstrate that the immune suppression function of cloned MSCs is completely different from that of conventional MSCs. The function of the MSCs therefore remains controversial and needs to be carefully analyzed and elucidated in the future.
The present in vitro results and previous reports show that MSCs are important to induce transplantation tolerance (4). Therefore, we expected systemic injection of MSCs to be effective on the GvHD host's survival. As with macrophages, we found that conventional MSCs exhibited a significant effect on the GvHD host's survival compared to the recipients of only the naive LN cells (Table 1, Fig. 4), thus suggesting a potentially active process of regulation within the graft. In contrast, and consistent with in vitro observations, cloned MSCs showed no protective effect compared to a no-treatment group. In the cloned MSC treatment group, we observed extensive perivascular infiltration of the mononuclear cells and tissue damage in histological studies of the skin, tongue, lung, and liver from the naive lymphocytes transferred to the host on day 14 (data not shown).
One possible explanation for these phenomena is that long-term culture may change some unknown factors, although we found no differences in the surface molecular and morphological features of conventional and cloned MSCs (Fig. 2). Recent publications shown that mouse MSCs suppress lymphocyte proliferation in vitro, but fail to prevent GvHD (47). Even using short-term cultured rat MSCs is ineffective for prolonging cardiac allograft survival and it also tends to promote rejection (18). These outcomes are consistent with our findings, namely that enhanced T-cell proliferation stimulated by ConA and alloantigen in vitro and infused cloned MSCs have no protective effect from GvHD. Therefore, the mechanism for the function of MSCs requires further investigation.
In conclusion, we demonstrated that, unlike conventional MSCs, cloned MSCs not only enhanced T-cell proliferation in vitro but also yielded no clinical benefit on the incidence or severity of GvHD. Therefore, our rat model showed differences in the suppression of lymphocyte proliferation and GvHD prevention between the short-term cultured conventional MSCs and cloned MSCs. Our findings suggest that the mechanisms of functional MSCs still remain to be identified. Our data also add valuable information to this controversial field.
Footnotes
Acknowledgments
This study was supported by research grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan (a Grant-in-Aid 20390349, 2109739), and Ministry of Health, Labour and Welfare of Japan (HS KHC1025). The authors declare no conflict of interest.
