Abstract
A major factor limiting the engraftment of transplanted stem cells after myocardial infarction is the low rate of retention in the infarcted site. Our long-term objective is to improve engraftment by enabling stem cells to recognize and bind infarcted tissue. To this end, we proposed to modify the surface of embryonic stem cells (ESCs) with the C2A domain of synaptotagmin I; this allows the engineered stem cells to bind to dead and dying cardiac cells by recognizing phosphatidylserine (PS). The latter is a molecular marker for apoptotic and necrotic cells. The C2A domain of synaptotagmin I, which binds PS with high affinity and specificity, was attached to the surface of mouse ESCs using the biotin-avidin coupling mechanism. Binding of C2A-ESCs to dead and dying cardiomyocytes was tested in vitro. After the surface modification, cellular physiology was examined for viability, pluripotency, and differentiation potential. C2A covalently attached to the ESC surface at an average of about 1 million C2A molecules per cell under mild conjugation reaction conditions. C2A-ESCs avidly bound to dying, but not viable, cardiomyocytes in culture. The normal physiology of C2A-modified ESCs was maintained. The binding of C2A-ESCs to moribund cardiomyocytes demonstrates that the retention of transplanted cells may be improved by conferring these cells with the ability to bind infarcted tissue. Once established, this novel approach may be applicable to other types of transplanted therapeutic cells.
Keywords
Introduction
Implantation using cardiomyogenic precursor cells to improve the prognostic outcome of postmyocardial infarction (MI) patients is an attractive therapeutic option (3,4,7,8,10,12,13,15–18,20–22,25). To this end, cell therapies administered intravascularly are applicable to reperfused acute MI cases, as these are minimally invasive and do not involve perforation of the necrotic tissue (1,2,6,9,11,24,26,29).
It is critical that a sufficient numbers of cells are re-ained in the infarct site after transplantation to achieve successful cardiac repair. During coronary delivery, infused cells are dispersed throughout the microcirculatory bed in the selected perfusion territory via an intracoronary catheter. It is generally presumed that the infused cells are lodged in the tissue during first passage when the cell density is at the maximum (19). Despite such a highly concentrated cell preparation, only a small fraction of intracoronary-delivered cells are retained in the infarct region due to a lack of effective retention mechanisms. A substantial improvement in existing strategies is needed in order to attain a sufficiently high retention rate.
This study explores a novel approach that may lead to improved retention after implantation; we aimed to generate engineered stem cells that recognize and bind to dead and dying cells. A near-universal molecular marker for both apoptosis and necrosis is the exposure of phosphatidylserine (PS) on the cell surface (5,9,23, 27). This event, in turn, provides an ample and well-defined avenue for the targeted delivery of diagnostic and therapeutic agents using PS binding vectors (30–32). In the current communication, we report that the surface of therapeutic cells can be engineered using a PS binding vector, which is the C2A domain of synaptotagmin I. In an in vitro proof of concept, the C2A-modified stem cells bind specifically to dead and dying, but not viable, cardiomyocytes.
Materials and Methods
Recombinant C2A was overexpressed in E. coli as a glutathione-S-transferase (GST) fusion protein and purified with relatively high yield (70–90 mg/L culture) (31). After labeling with green fluorescein isothiocyanate isomer I (FITC), the conjugate has a FITC-to-protein molar ratio of 1.1 ± 0.2. For simplicity, the FITC-labeled C2A-GST is named C2A-FITC.
Preparation of Mouse Embryonic Stem Cells (ESCs)
A mouse ESC line (JxL1p2 ESCs) was recently derived from blastocysts resultant from crossing 129×1/SvJ x 129S1/SvlMJ mice and used at low (<10) passage. Pluripotency was maintained, as determined by Oct-4 immunostaining, by expansion on MEFs attached to gelatinized cell culture dishes in medium supplemented by 10% FBS plus 1x LIF. Upon expansion to 70% confluency on p35 culture dishes, cells were harvested by addition of trypsin. The volume of medium was then raised to 6 ml, and ESC groups were dissociated using a syringe attached to a 10-ml pipette. To separate harvested ESCs from MEFs following trypsin treatment and dissociation, the cells were collected by centrifugation, resuspended in 4 ml ESC medium, and submitted for Histopaque density-gradient separation, as described previously (14).
C2A Conjugation to ESC Surface
C2A-FITC was attached to the surface of viable ESCs by using an indirect conjugation method involving the high-affinity interaction between biotin and avidin (Fig. 1). C2A-FITC was covalently attached to neutravidin via a bifunctional linker succinimidyl-4-(N-maleimido-methyl)cyclohexane-1-carboxylate (SMCC). Neutravidin-SMCC was purchased from Pierce Chemical. C2A-FITC and neutravidin-SMCC were dissolved in phosphate buffer (100 mM phosphate, pH 7.4) at respective final concentrations of 0.8 and 1.1 mg/ml. The reaction was allowed to proceed at room temperature for 20 min, before quenching with 4 molar excess cysteine. The presence of FITC-C2A-neutravidin was confirmed as a high-molecular weight conjugate using SDS-PAGE (4–12% gradient).

Schematic illustration of ESC surface modification. The ESC surface is first biotinylated using Sulfo-NHS-biotin. C2A is covalently conjugated with neutravidin before attaching to the ESC surface.
To biotinylate the cell surface, ESCs were washed twice in Hank's balanced salt solution (HBSS), and resuspended at 2 × 106 cells/ml in HBSS. A concentrated stock solution of Sulfo-N-hydroxysulfosuccinimide ester biotin (Sulfo-NHS-biotin) was freshly prepared at 1.12 mg/ml in DMSO. To each milliliter of cells, 5 ml of Sulfo-NHS-biotin stock solution were added. The reaction mixture was incubated for 2 min at room temperature with occasional gentle inversion. Nonreacted Sulfo-NHS-biotin was removed by washing the cells in HBSS three times and centrifugation at 800 x g for 1 min each. The final biotinylated ESCs were resuspended in HBSS.
C2A-ESCs were produced by incubating biotinylated ESCs with FITC-C2A-avidin at room temperature in HBSS for 5 min with gentle agitation. The nonbound FITC-C2A-avidin was removed by two cycles of washing in HBSS and centrifugation at 800 x g. The FITC-C2A-avidin-conjugated cells were examined under a fluorescent microscope. The presence of fluorescent signal on the cell surface was confirmed by confocal microscopy using a Leica TCS SP2 Laser Scanning confocal microscope.
Quantification of Conjugation Chemistry
The average number of C2A molecules attached to each cell was quantified as follows: C2A-conjugated cells were counted to obtain the cell density, and were dissolved in a lysis buffer (100 mM Tris, 70 mM NaCl, 1% Triton-X100, pH 7.4). The fluorescent emission at 515 nm was measured using 490 nm excitation. A calibration plot was established where the emission values were measured from serial dilutions of a C2A-FITC sample with known protein concentration. Linear fitting indicated that the calibration plot was in the linear range, with an R value of greater than 0.99. The concentration of C2A-FITC in the cell sample, and therefore the average number of C2A molecules per cell, was calculated based on the calibration plot. The experiment was repeated three times, and the data were expressed as the mean with standard deviation.
C2A-ESC Binding to Dead Cardiomyocytes
Cardiomyocytes were isolated from adult rat heart and cultured as described in prior studies (28). Procedures involving animals were approved by the Institutional Animal Care and Use Committee and were performed under the guidelines issued by the National Institute of Health. Cultured cardiomyocytes were treated with doxorubicin at a final concentration of 10 μM for 48 h to induce cell death. The viability of myocytes was assessed by taking an aliquot of the cells, using a commercial apoptosis labeling kit involving Annexin V-FITC and propidium iodide (Sigma, St. Louis, MO). Annexin V-positive cells are apoptotic, and double-positive cells are secondary necrotic or necrotic. The loss of viability in cardiomyocytes was consistent with an amorphic appearance accompanied with a lack of rhythmic contractile activities. C2A-ESCs (~106) were added to culture dishes containing dead or viable cardiomyocytes (~105) and incubated for 5 min at RT with gentle agitation. The cells were then examined under a fluorescent microscope.
Immunohistochemistry Study of ESC Physiology
Following overnight attachment to p100 cell culture dishes coated with 0.1% gelatin, the cells were washed and fixed in 4% fresh paraformaldehyde at for 15 min. Cells were immunostained to detect expression of the following lineage markers: Oct-4 (pluripotency), brachury (T; early mesoderm), and Nkx-2.5 (precardiac mesoderm). Specifically, ESCs were washed with PBS and placed in blocking solution (1% goat serum in PBS) for 1 h at RT. Primary antibody (Oct4; Santa Cruz sc-9081, 1:500), Nkx2.5 (R&D Systems, AF2444, 1:500), or Brachyury (Santa-Cruz, 17745, 1:500)) was added in blocking solution and incubated overnight at 4°C. The next day, cells were washed in PBS 3x, 10 min each, and secondary antibody (Alexa-fluor 488-conjugated donkey anti-rabbit IgG for Oct4, 1:500, or Vectastain anti-goat IgG for Nkx2.5 and Brachyury) was applied for 1 h at RT in blocking solution. Cells were washed with PBS. Finally, the cells were counterstained with DAPI (1:10,000) for 15 min at RT. Oct4-positive cells were visualized using the green fluorescence channel. Nkx2.5 and Brachyury signals were visualized using a horseradish peroxidase signal amplication system with diaminobenzodine (DAB) as the colorimetric substrate.
Differentiation Potential of mESCs
To examine the differentiation potential of ESCs after C2A modification, cells were placed in suspension by culturing in 10 ml ES medium without LIF on uncoated petri dishes. In this condition, ESCs do not attach to the plate; they form aggregates, which develop into embryoid bodies (EBs). Differentiation was assessed in the resultant EBs by observing the spontaneous appearance of beating cardiomyocytes during the 12 days after modification; this was confirmed by fixing and removing sections from the EBs, following by immunostaining to detect sarcomeric α-actin (primary antibody Sigma #A-2172 at 1:800; secondary antibody #55498 FGAM IgM at 1:200).
Results and Discussion
The aim of the current study was to produce ESCs that have an affinity toward infarcted myocardium with the long-term goal of improving the engraftment rate and therapeutic efficacy of cell transplantation for cardiac repair.
C2A was attached to the surface of mouse ESCs by taking advantage of the high-affinity interaction of the avidin-biotin system. The distribution of the green fluorescent signal on C2A-FITC-modified ESCs appeared to be homogeneous (Fig. 2). The presence of C2A specifically on the cell surface was confirmed using confocal microscopy, revealing the absence of internalized fluorescent signal within the cells (Fig. 2). On average, there were 924,644 ± 19,241 C2A molecules attached to each cell, according to the current protocol. No membrane leakage in conjugation-modified ESCs was evident in comparison to control cells; dye exclusion assays with propidium iodide were used. C2A-labeled ESCs remained as single cells with no aggregation.
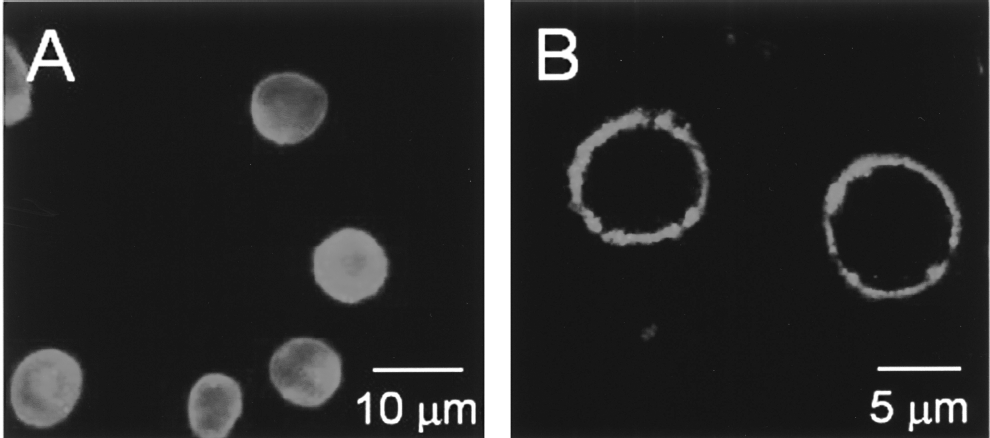
Covalent conjugation of C2A-FITC to the surface of ESCs. (A) Conventional fluorescent microscopy showing that C2A-labeled ESCs remain in a single-cell dispersed state without aggregation. (B) Confocal microscopy showing that the C2A moiety is specifically located on the plasma membrane.
Coincubation of C2A-labeled ESCs with cardiomyocytes resulted in avid binding between C2A-ESCs and the dead cells. However, this was not the case with the viable cardiomyocytes (Fig. 3). Multiple C2A-ESCs were often bound to a single amorphic cardiomyocyte; only a few ESCs remained free. The binding between C2A-ESCs and their target cells was sufficiently strong, such that gentle agitation of the culture dish did not cause dissociation. Doxorubicin treatment is an established procedure to quickly induce apoptosis in cardiomyocytes, and provides a convenient in vitro tool for the proof of concept that the engineered stem cells are capable of binding to dead/dying cardiomyocytes by recognizing exposed anionic phospholipids. The externalization of anionic phospholipids in the plasma membrane is a near-universal marker for cell death, regardless of the cause. The same molecular marker for dead/dying cells is present at high levels in ischemically damaged heart tissue, and will provide abundant binding sites for these engineered stem cells (31,32).

Binding between C2A-ESCs (small oval shapes) and moribund cardiomyocytes. (A–C) Moribund cardiomyocytes bind C2A-ESCs (arrowheads). Moribund and dead cardiomyocytes are spheroid and amorphic, while viable cardiomyocytes (arrow in D) maintain a distinct rectangular appearance. (D) Viable cardiomyocytes (arrow) do not bind C2A-ESCs.
C2A-ESCs retained viability, pluripotency, and differentiation potential. The C2A-modified ESCs, when recultured, reestablished adhesion to culture plates. Col-onies of ESCs started to form within 16 h, and the size of resultant colonies was similar to that of untreated ESCs. Immunohistochemical staining showed that modified ESCs were positive for Oct4 and negative for Brachury and Nkx2.5, confirming maintenance of pluripotency and absence of differentiation. Surface-conjugated ESCs were able to form embryoid bodies (EBs) wherein sarcomeric α-actin-positive cells were positively identified using immunohistochemistry in a fashion that did not differ from untreated controls; hence, the differentiation potential of C2A-modified ESCs was maintained. The cellular indices after C2A modification remain comparable to the native ESCs; it was confirmed that the conjugation reaction condition was sufficiently mild without significantly altering cellular physiology.
The concept of infarct-avid stem cells is being tested in vivo as part of an ongoing experiment using a rat model of myocardial ischemia and reperfusion. Preliminary data have demonstrated consistently that the retention of intravascularly delivered stem cells is significantly improved using the surface modification approach. Given the numerous variables that need to be optimized, the in vivo application is anticipated to undergo an extended period of improvement and refinement.
Overall, the current study takes an initial step toward addressing the challenge of low cell retention following transvascular infusion of stem cells by employing a chemical approach to render ESCs infarct avid. By using surface modification rather than overexpression of an exogenous receptor transgene, the genetic material of ESCs remains unaltered, thereby avoiding possible epigenetic issues. It is noteworthy that under sufficiently mild condition reaction conditions, C2A-modified ESCs exhibited no significantly altered physiology. The current data establish an in vitro proof of concept that the cell surface can be chemically modified to confer binding affinity with high specificity to infarcted tissue for targeted transplantation. ESCs having a binding affinity toward dead and dying tissues will not only help target transplanted stem cells to the infarct site, but will also improve their rate of retention and engraftment within the infarcted tissue. Once fully established, the concepts and conjugation chemistry techniques reported here should be applicable to modifying various stem cell types for therapeutic implantation to regenerate a wide variety of tissues.
Footnotes
Acknowledgments
Financial support from the American Heart Association, Advancing Healthier Wisconsin Award, and the National Institutes of Health (1R21HL090891, NIH HL079277) is gratefully acknowledged.
