Abstract
Over the past decade, cell therapy has emerged as a new approach to reversing myocardial ischemia. Several types of adult stem cells have been studied in both preclinical and clinical conditions for this purpose: bone marrow cells, circulating cells, and myoblasts. Nevertheless, the quest for the ideal “anti-ischemic” cell is still ongoing. Recently, the existence of a population of stem cells located in adipose tissue (adipose-derived stem cells) has been observed. These are able to differentiate into multiple cell lineages including cardiomyocytic differentiation. In this review we discuss the basic principles of adipose-derived stem cells (types and characteristics, harvesting, and expansion), the initial experimental studies, and the currently ongoing clinical trials.
Keywords
Adipose Tissue: A New Source of Stem Cells
Recent advances in the diagnosis and treatment of coronary artery disease have improved the prognosis of patients in the acute phase of the disease, the acute myocardial infarction (AMI). However, the loss of myocardial tissue and the ventricular negative remodeling process will lead this growing population of AMI survivors to the syndrome known as chronic heart failure (CHF). This major health problem and its enormous economic consequences have guided cardiovascular research towards new therapeutic approaches, among which cell therapy has been extensively studied as one of the more promising ones.
The quest for the best cell type is still ongoing. This ideal cell type should be capable of differentiating into functional cardiomyocytes and of forming new vessels to nourish the damaged area. Cell types from several different sources have already been tested in animal models, and bone marrow-derived cells and skeletal myoblasts have been used in clinical trials (13,28). Although results have shown an improvement in cardiac function due mostly to paracrine effects (8), formation of a new myocardial mass has only been established for embryonic stem cells.
Recently, it has been shown that in the adipose tissue stroma, besides committed adipogenic, endothelial, and pluripotent vascular progenitor cells, we can also find multipotent cell types (adipose-derived stem cells, ASCs). In this review we will focus on the types, isolation, characterization, and preclinical and clinical application of ASCs in the cardiovascular field.
Working in the Bench: Adipose-Derived Stem Cells Characteristics
Nomenclature
In recent years it has been shown that mesenchymal stem cells (MSCs) have an inherent ability for self-renewal, proliferation, and differentiation towards mature tissues depending on the microenvironment by which they are surrounded. Such characteristics, which define them as stem cells, make them very interesting for their use in regenerative medicine. It was originally thought that these MSCs were found exclusively in bone marrow, but many scientists have been searching for cells with a similar profile in other adult tissues (9,54,55) and have compared the characteristics of MSCs derived from different tissues (15,16,38,52).
The problem lies in the low number in which these cells are found in adult tissues, as well as in the difficulty in isolating and maintaining them in vitro. Adipose tissue has been deemed one of the most attractive tissues, as it is easily available after liposuction procedures. In addition, isolating MSCs from adipose tissue is a simple and reproducible process with an optimal cell yield given the amount of tissue from which they are derived (54).
In mammals there are two types of adipose tissue, brown (BAT) and white (WAT). BAT functions as an energy-dissipating organ while WAT serves as the principal energy storage for the organism. It has been reported that the heterogeneity to plasticity in adipose tissue depends on the type of tissue from which the stem cells are obtained (35). It has also been demonstrated the immense plasticity of stroma-vascular fraction (SVF) cells collected from inguinal fat in their ability to give rise to osteoblasts, endothelial cells, adipocytes, hematopoietic cells, and cardiomyocytes, compared to SVF cells derived from BAT. Thus, inguinal WAT has a larger resident population of stem cells and also greater plasticity, making it an optimal choice for cell therapy. Nevertheless, data confirming these theories in humans are not yet available. It seems that neither the type of surgical procedure nor the anatomical location from which the fat is collected affect the total number of viable cells obtained via SVF (31,42). However, it has been observed that factors such as age, the type of adipose tissue and its location, the surgical procedure used in obtaining the cells, and the cultivation conditions can affect the differentiation and proliferation capacity of ASCs (5,16,17,48,54,55).
There is no accordance when it comes to the nomenclature used in describing progenitor cells from adipose tissue-derived stroma, which can sometimes lead to confusion. Thus, the terms adipose tissue-derived stromal cell (ADSC), adipose stroma vascular cell fraction (SVF), and adipose-derived regenerative cells (ADRCs) correspond to cells obtained immediately after adipose tissue collagenase digestion; on the other hand, processed lipoaspirate cells (PLA) and plastic-adherent adipose-derived stem cells (ASCS) describe cells obtained after culture of the aforementioned ones. Other terms that have been used are multipotent adipose tissue-derived mesenchymal stem cell (AT-MSC), which includes SVF subpopulations, or even “preadipocytes” (adipocyte progenitors). Both cell types can be referred to as “adipose-derived stem cells” (ASCs), according to the International Fat Applied Technology Society Consensus (14). There are no standards regarding the minimum biological criteria that each cell type must meet to be classified as one type or the other. In the case of MSCs, the International Society for Cellular Therapy (ISCT) defines the minimal criteria to be met for cells to be considered MSCs as the following (6):1) adherence to plastic in standard culture conditions; 2) phenotype positive (≥95%+): CD105, CD73, CD90 and phenotype negative (<2%+): CD45, CD34, CD14 or CD11b, CD79α or CD19 and HLA-DR; 3) in vitro differentiation: osteoblast, adipocytes, chondroblast (demonstrated by staining of in vitro cell culture).
Adherence to these minimal criteria by scientists would help to standardize the protocols used in obtaining the cells and to the characterization of each cell type, therefore ensuring a more efficient progression in both preclinical and clinical trials.
Harvesting and Expansion
There are several expansion protocols and differentiation stages for cells obtained from adipose tissue, giving yield to a great diversity of results. Normally, ASCs are used in one of two stages of differentiation: immediately following tissue digestion with collagenase, referred to as the fresh fraction (SVF), or cells that have been expanded for three or four passages (PLA). It is also possible to find preclinical studies in which subpopulations of the SVF fraction are used: preadipocytes (41), multipotent adipose tissue-derived mesenchymal stem cell (AT-MSC) (2), etc.
SVF Isolation
The protocol used by different authors for the isolation of SVF is a modified version of the methods described by Zuk et al. (54,55). However, as previously noted, there are many protocols described for isolating the SVF and the following expansion (2,16, 19,24,31).
To isolate the SVF from adipose tissue, the tissue is washed with phosphate-buffered saline (PBS), and treated with collagenase under continuous shaking during a period of 30–60 min. Collagenase activity is usually neutralized with an equal volume of Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum (FBS). The digested solution is centrifuged at low speed for 10 min, the cell pellet resuspended in DMEM/10% FBS, and the resulting solution is filtered through a 40–200-μm nylon mesh filter. The solution is centrifuged again and the cells are resuspended in complete expansion medium.
All of these protocols must be carried out in extremely sterile conditions, and if at any point in time there were to arise a possible clinical application, these techniques would be continued in Good Manufacturing Practices (GMP) facilities. With this premise, many laboratories have limited use of these cells due to low availability and the high costs associated with the installation of GMP facilities. As a result, cell separation systems have been developed that optimize the time involved and the quality of the product, when deriving cells from adipose tissue under GMP conditions. An example is the Celution™ System (Cytori Therapeutics Inc., San Diego, CA, USA), a completely automatic cell isolation CE marked system with applications in cardiac pathologies. The Celution™ System enzymatically digests the adipose tissue in a single cellular solution, which is followed by a wash to separate the adult adipocytes and debris from the desired cell fraction, ASCs. This fraction contains adult mesenchymal-like stromal cells, endothelial progenitor cells, and other adipose tissue stromal cells (16,54,55). Once the ASCs fraction is isolated it is immediately delivered to the patient by percutaneous intracoronary or transendocardial injection (7) (Fig. 1).

An example of protocol using freshly isolated adipose-derived stem cells. After abdominal liposuction, fat tissue is digested and centrifuged with the Celution™ System (Cytori Therapeutics Inc., San Diego, CA, USA) to obtain stem cells that will be delivered percutaneously through transendocardial injections.
PLA Expansion
The PFA expansion protocols are just as diverse as the protocols for isolating SVF cells. As a general rule, nucleated cells in the SVF are counted and the viability of the cells is assessed using the trypan blue exclusion assay. The cells are cultured in complete expansion medium (DMEM/10% FBS/1% penicillin/streptomycin/1% L-glutamine) at varying cell concentrations, the initial passage of the primary cell culture being referred to as passage 0 (P0). The cultures are maintained in a 5% CO2, humidified incubator at 37°C. The nonadherent cells are removed at 12–24 h and fresh complete expansion medium is added. The medium is changed twice a week until observing 75–90% confluence, upon which the cells are passed with 0.5 mM EDTA/0.05% trypsin, incubated for 1–5 min at 37°C. The cells are plated at concentrations between 5,000 and 30,000 cells/cm2 (P1). The expansion can be continued, repeating the same process to obtain cells in different passages (P2, P3, P4, etc.) (Fig. 2).
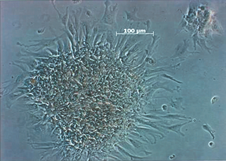
Autologous adipose-derived mesenchymal stem cells growing in colonies, in the sixth day of culture (magnification 10×). These cells were expanded from the adipose tissue stroma-vascular fraction (SVF).
Immunophenotype
Cytometry is the most frequently used tool in identifying distinct cell populations. Unlike other cell types such as hematopoietic cells, there are no specific markers for SVF or PLA cells. Because of this, stromal-associated and stem cell-associated markers are used for identification (Table 1). PLA cells express CD105 (endogline or MAb SH2), CD73 (ecto 5′ nucleotidase or MAb SH3), and CD90 (or Thy-1), markers that also identify bone marrow-derived MSCs. In fact, the expression of these antigens is one of the minimum criteria outlined by the ISCT for MSCs classification (14). Other surface antigens expressed by these cells include CD166 (adhesion molecule) or CD44 (hyaluronate receptor), as well as endothelial markers CD105 and CD146. However, PLA cells are negative for the following stem cell and hematopoietic lineage markers: CD34, CD133, CD117, and CD11b (granulocytes, monocytes, natural killer), CD14 (monocytes), CD19 (lymphocytes), and CD45 (pan-leucocyte) (2,24,31,40). They are also negative for histocompatible locus antigen-DR (HLA-DR), making these cells very attractive for allogenic clinical use due to low immunity reactions. Surprisingly, HLA-DR expression is detected within heterogeneous SVF cell populations, although a decrease is observed with subsequent passages. The expression of this antigen reduces the applications of these cells to strictly autologous use, because allogenic use would have a risk of rejection (22).
Cell Surface Characteristics of Adipose-Derived Stem Cells in the Two Stages of Differentiation: Stroma-Vascular Fraction (SVF) and Processed Lipoaspirate (PLA)

SP: subpopulations.
HLA-DR is not the only marker in adipose cells that varies in expression depending on whether the cells are freshly isolated or already expanded. The detection of other antigens also depends on the stage of the cell. The initial SVF cell population expresses high levels of CD166 (64%) and medium/low levels of CD44, CD29, CD73, CD90, and CD105 (5–54%). These values increase in passage 4 (P4) expanded cells to levels between 69% (CD166) and 98% (CD44). There is a subpopulation of SVF cells positive for stem cell-associated markers such as CD34 (60%), a marker that is eventually lost with expansion (24). The SVF cells also contain low/medium levels of subpopulations of hematopoietic lineages including both T and B lymphocytes (CD45+ CD34–CD3+; CD45+CD34–CD19+), granulocytes (CD45+ CD11b+), monocytes (CD45+CD14), as well as subpopulations that express markers found on endothelial cells (CD34+CD31+CD146+) and vascular smooth muscle cells (CD34dimCD31–CD146+) (31).
Differentiation
The stem cells found in adipose tissue have a large plasticity towards different tissues depending on the microenvironment that surrounds them. The first confirmation of multilineage differentiation observed in PLA cells was done using protocols previously described for MSCs (54). Currently, there are commercial media available for “in vitro” differentiation, being possible to differentiate PLA cells to adipocytes, chondrocytes, osteocytes, neurons, and cardiomyocytes.
This cardiomyogenic differentiation is carried out with confluent PLA cells. These cells are cultivated for 3–6 weeks with myogenic medium: DMEM, 10% FBS, 5% horse serum (HS), dexamethasone, hydrocortisone, and 1% antibiotic/antimitotic (26,54). Medium is changed twice a week and myogenic differentiation is confirmed by immunohistochemical staining for the muscle-specific transcription factors Myo-D1 and myosin heavy chain.
In 2004, the transformation of PLA cells to cardiomyocytes by adding 5-azacytidine to the culture medium was described (36). In the same year, another group obtained beating cells with characteristics typical of cardiomyocytes, cultivating SVF cells in MethoCult: 1% methylcellulose in Iscove's MDM with 1% BSA, 15% FBS, 2-mercaptoethanol, L-glutamine, human transferring, IL-3, IL-6, and SCF, without the addition of 5-azacytidine (33).
Translation with Adipose-Derived Stem Cells in Cardiovascular Diseases: Preclinical Background
Since the first experiences in isolation and culture of ASCs, numerous articles have been published studying their plasticity and applying them in both small- and large-animal models for many purposes in regenerative medicine. Focusing on the topic of this review—cardiovascular disease—an important number of animal studies have explored the use, behavior, and effect of ASCs in the three main scenarios of this pathology: AMI, CHF, and peripheral vascular disease (PVD).
Acute Myocardial Infarction
The first study in this setting investigated freshly isolated ASCs that were delivered into the left ventricular chamber (an approximation of intracoronary delivery) immediately after cryoinjury myocardial damage in rats (44). This study demonstrated for the first time myocardial engraftment of ASCs with expression of cardiac-specific products. In addition, functional and pathological assessments were done that showed significant improvements in global ejection fraction and in capillary density in the myocardial border area of treated animals compared with controls (45).
Cardiac stem cells (CSCs) isolated from BAT have also been investigated in acute myocardial ischemia. In an experimental AMI model in rats (49), CD29+ CSCs were injected at the infarct border area. A reduction in the amount of collagen with an improvement of ventricular function and remodeling by echocardiography were reported. This cardiac repair rate was of a higher efficiency compared with that in reports that used BM-MNCs, and was mediated through differentiation of CSCs into ECs, smooth muscle cells (SMCs), and cardiomyocytes (CMs), and through secretion of angiogenic and antiapoptotic factors [vascular endothelial growth factor (VEGF), b-fibroblast growth factor (b-FGF), and hepatocyte growth factor (HGF)]. To further investigate the mechanisms by which BAT-derived cells (BATDCs) contribute to myocardial repair, coculture experiments were performed by this group. First, they cocultured cord blood cells (CBCs) with BATDCs, and demonstrated that these educated CBCs were able to differentiate into CMs and to improve the cardiac function (50). Then they cultured CD133+ CSCs isolated from BAT with BM-MNCs to induce cardiomyogenic differentiation (51). When injected after an AMI in rats, these educated BM-MNCs effectively differentiated into CMs and improved cardiac function by means of differentiating into C31+ ECs and SMCs and producing VEGF and HGF.
Finally, ASCs have been used by intracoronary administration in a swine AMI model, with or without culture (1,46,47). In the first two studies freshly isolated ASCs were applied, thus improving left ventricle (LV) ejection fraction compared to the control group at 6-month follow-up (1,47). The study with cultured ASCs reported similar results (46). Four weeks after treatment, left ventricular perfusion, function, and remodeling were substantially improved by means of apoptosis inhibition and differentiation into ECs and SMCs (angiogenesis). This improvement was similar to that observed after intracoronary administration of BM-MNCs, though the latter did not have any effect on ventricular remodeling.
Chronic Heart Failure
Another type of cell, in this case CD29+/CD90+ MSCs from murine adipose tissue, were grown to create a monolayer sheet using temperature-responsive culture dishes. These monolayers were transferred and grafted onto scarred myocardium 8 weeks after AMI (25). This treatment resulted in decreased scarring and enhanced cardiac structure and function compared with controls, translating in a higher survival rate in the treated group. The improvement was explained by the authors by growth factor-mediated paracrine effects and by a decrease in left ventricle wall stress resulting from the thick MSC tissue, which included newly formed vessels, CMs, and undifferentiated MSCs.
ASCs have also been investigated in a CHF model in pigs by transendocardial injection with the NOGA™ navigation system (BDS, Cordis Corporation, Johnson and Johnson) (4). Four weeks after ameroid implantation, freshly isolated ASCs were delivered into the chronic ischemic tissue with preservation of wall thickness in the infarcted area in treated animals compared to controls. DAPI-labeled cells were seen in all animals 30 days after injections.
Finally, ASCs have been proved to provide a superior benefit in terms of cardiac function and tissue viability to cardiac predifferentiated ASCs and bone marrow mononuclear cells in a chronic model of myocardial infarction in rats (21).
In conclusion, ASCs have been proved to engraft and survive in the myocardium, having a beneficial effect on perfusion and structural and functional parameters through angiogenesis and paracrine mechanisms.
Peripheral Artery Disease
The first preclinical studies with ASCs in the cardiovascular application were performed by several groups in murine ischemic hindlimb models with CD31- ASCs (3,23,27,29,34,37).
In the first study, SVF cells from mice and humans were administered by intramuscular injection in the ischemic leg (34). An improvement in angiographic scores and cutaneous blood flow were observed in treated animals, the best results being obtained when culturing the cells before injection. Differentiation into endothelial cells (ECs) and vascular regeneration were invoked by the authors as probable mechanisms.
A different subpopulation of ASCs, CD34+/CD31–cells from the human SVF, were isolated and injected intravenously in nude mice, demonstrating an increase in blood flow and capillary density (23). Again, the incorporation of the ASCs into the mouse vasculature (vasculogenesis), possibly through differentiation into ECs, was suggested as the mechanism of this beneficial effect. Interestingly, both studies showed that ASCs effects on vascular regeneration were similar to that obtained with bone marrow-derived mononuclear stem cells (BM-MNCs), a conclusion later confirmed in the same model by other groups (29).
Another study with human ASCs showed that cultured Flk1+/CD34–/CD31– cells were able to differentiate into functional ECs (expressing human CD34) and to augment recovery of perfusion in ischemic hindlimbs when intravenously injected (3).
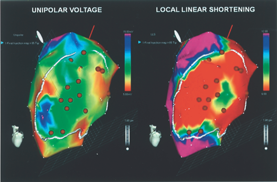
Electromechanical mapping of the left ventricle with the NOGA XP™ System (BDS, Cordis Corporation, Johnson and Johnson). Myocardial areas with low contractility and impaired endocardial voltage are identified as viable and targeted for cell injection (brown dots).

Flow chart of the PRECISE trial, the first-in-man experience with adipose-derived stem cells (ASCs) in patients with advanced coronary artery disease (CAD). LVEF: Left Ventricular Ejection Fraction; DSMB: Data Safety Monitoring Board; CCS: Canadian Cardiovascular Society; NYHA: New York Heart Association; ECHO: echocardiography; MRI: magnetic resonance imaging; SPECT: single photon emission computed tomography.
Two other studies underscored the importance of paracrine pathways with very similar models. Angiogenic cytokines such as VEGF and HGF were reported as ASCs products (27,30,37), able to contribute to the ASCs angiogenic properties.
Clinical Applications and Studies Going On
Conversely to preclinical studies, the field of human research with ASCs has been less explored. The first reported experiences were done in the plastic and reconstructive and in the digestive surgery settings.
ASCs were first used to stimulate bone repair in critical calvarial defects (18) and to heal chronic fistulas in Crohn's disease (11): in a phase I trial with patients with fistulas unresponsive to medical treatment, complete healing was achieved in 75% of the cases (12). These cells have been also applied in plastic surgery to promote retention of transplanted autologous fat tissue during surgical soft tissue augmentation (20,43). A recently initiated trial in Japan will investigate the application of ASCs for breast reconstruction after partial mastectomy due to cancer resection.
Turning to the cardiovascular scene, no clinical trials have been published yet with ASCs. Nevertheless, two currently ongoing trials are exploring the safety, feasibility, and efficacy of freshly isolated ASCs with the Celution™ system (Cytori Therapeutics Inc.) in both AMI and chronic myocardial ischemia patients. Both studies, the APOLLO and PRECISE trials, are being carried out in our institution at Hospital General Universitario Gregorio Marañón (Madrid, Spain) in collaboration with the Thoraxcenter (Erasmus MC, Rotterdam), which will also enroll patients for both trials.
The PRECISE trial is a prospective, double-blind, randomized, placebo-controlled, sequential dose-escalation trial that will randomize up to 36 patients with end-stage coronary artery disease not amenable for revascularization and with moderate-severe left ventricular dysfunction to receive freshly isolated ASCs or placebo in a 3:1 ratio. The cells are delivered via transendocardial injections after LV electromechanical mapping with the NOGA XP™ delivery system (BDS) (Fig. 3). Nineteen patients have been enrolled in our center so far and results are anticipated in 2009 (Fig. 4).
The APOLLO trial is aimed to investigate the same source of freshly isolated ASCs, but in this case in patients with AMI and LV ejection fraction impairment. It is also a prospective, double-blind, randomized, placebo-controlled, sequential dose-escalation trial that will include up to 48 patients. In the APOLLO trial ASCs are delivered through intracoronary infusion after appropriate infarct-related artery repair with stent implantation. The first patient has been included in our center and the enrollment is planned to be completed by next year.
Pros and Cons of Adipose-Derived Stem Cells
ASCs have emerged as a new and promising type of stem cells with two clear advantages over the previously used from bone marrow, blood, or skeletal muscle. Firstly, an easy and repeatable access that allows the harvesting of high amounts of adipose tissue by a minimally invasive method and secondly, an increased proliferative potential to expand themselves in culture, either because of properties of the cells or as a result of a greater frequency of stem cells within the population used to initiate the culture.
Moreover, ASCs can be efficiently cryoperserved with different solutions, keeping their viability and the adipo/osteogenic potential (32), and can be safely and efficiently transfected with Sendai virus (SeV) vectors for gene transfer (53).
An interesting point that deserves deep preclinical research is the immunogenicity of ASCs. As we have seen, class I and II major histocompatibility complexes (MHC) are expressed in only 1% of ASCs, and therefore it has been hypothesized that these could behave as universal donor cells and could be used for autologous and for allogenic transplantation.
Nevertheless, there are some scientific and medical questions related to ASCs that need to be answered before their clinical systematic application. First is the development of large-scale manufacturing techniques according to the GMP requirements. Thus, we will able to determine how to manipulate, store, and ship ASCs, always under GMP conditions to avoid contamination.
For safety purposes, the possibility of adverse events will need to be ruled out definitely. These include basically two concerns: hemostasis when harvesting adipose tissue in patients with AMI who are on antiaggregant and anticoagulant treatment (10), and the exceptional chance of long-term tumor development (39). No cases of arrhythmias have been reported in any of the studies performed so far.
Conclusions
As a summary, we have seen in this review that human adipose tissue is a novel promising alternative source of stem cells for cardiovascular repair that has showed encouraging results in the preclinical field. However, all this evidence needs adequate translation into humans. The ongoing PRECISE and APOLLO trials will shed light on the safety and efficacy issues of the cardiovascular application of these progenitor cells.
