Abstract
Chronic endometritis (CE) is closely linked to the reproductive failure. Exosome (Exo)-based therapy is proposed as an encouraging strategy in inflammation-related disorders; however, little work has been devoted to its usage in CE therapy. An in vitro CE was established by administration of lipopolysaccharide (LPS) in human endometrial stromal cells (HESCs). The cell proliferation, cell apoptosis, and inflammatory cytokine assays were performed in vitro, and the efficacy of Exos derived from adipose tissue–derived stem cells (ADSCs) was evaluated in a mouse model of CE. We found that Exos isolated from ADSCs could be taken up by HESCs. Exos elevated the proliferation and inhibited apoptosis in LPS-treated HESCs. Administration of Exos to HESCs suppressed the content of tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and interleukin-1β (IL-1β). Moreover, Exos exposure repressed the inflammation stimulated by LPS in vivo. Mechanistically, we demonstrated that Exos exerted their ant-inflammatory effect via miR-21/TLR4/NF-kB signaling pathway in endometrial cells. Our findings suggest that ADSC-Exo-based therapy might serve as an attractive strategy for the treatment of CE.
Introduction
Chronic endometritis (CE) is a persistent inflammatory disease that is characterized by the infiltration of plasma cells to the endometrial stroma 1 . Chronic endometritis is one of the major factors that account for the unexplained infertility, in vitro fertilization (IVF) failure, and recurrent failure in early pregnancy in women 2 . Antibiotic therapy is commonly used for the treatment of CE, but its efficacy is unsatisfied. Therefore, developing novel strategies for the prevention and treatment of CE is still needed.
Mesenchymal stem cells (MSCs) are a group of adult tissue progenitor cells that have the potential of self-renewal and multilineage differentiation 3 . A recent study shows that transfer of endometrial mesenchymal stem cells (enMSCs) meliorates CE and improves the repeated implantation failure 4 . Increasing evidence reveals that MSCs exert their function via transporting exosomes (Exos), extracellular vesicles, as well as cytokines to host cells 5 . Exos are nanovesicles secreted by different cell types, which transfer proteins, DNAs, and RNAs into the extracellular space and thus participate in a number of biological processes, such as cell survival, immunity, and inflammation6–8. The use of Exos avoids a number of shortcomings of stem cell therapy 9 and thus is suggested to be an attractive approach for the treatment of multiple diseases10,11.
Adipose tissue–derived stem cells (ADSCs) are mesenchymal cells derived from adipose tissues, which possess the capacity of regenerative properties as MSCs 12 . A previous study reported that in dairy cows, treatment with bovine ADSCs inhibits lipopolysaccharide (LPS)-induced inflammation response via suppressing the levels of tumor necrosis factor-alpha (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6) in endometrial epithelial cells 13 . Multiple studies have reported that Exos derived from ADSCs play a crucial role in the inhibition of inflammatory activity14–16. However, the therapeutic effect of Exos derived from ADSCs (ADSC-Exos) on CE remains unknown.
In this study, we sought to investigate the effect of ADSC-Exos on LPS-induced inflammation in endometrial cells both in vitro and in vivo, and explore the possible mechanisms underlying their action.
Materials and Methods
Cell Culture
Human endometrial stromal cells (HESCs) and human ADSCs were obtained from the American Type Culture Collection (ATCC, USA) and Cyagen Biology (Guangzhou, China), respectively. Human endometrial stromal cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM)/F12 (Thermo Fisher Scientific, Waltham, MA, USA), which contains 2% fetal bovine serum (FBS; Thermo Fisher Scientific) and 1% penicillin-streptomycin (Thermo Fisher Scientific), while ADSCs were cultured in DMEM medium (Thermo Fisher Scientific) that contains 10% FBS and 1% penicillin-streptomycin. Both HESCs and ADSCs were maintained at 37°C in an incubator with 5% CO2.
Extraction of Exos From ADSCs
Exosome extraction was carried out with the medium of ADSCs by ultracentrifugation. The centrifugation of medium was performed as follows: 3,000 × g for 15 min and then 20,000 × g for 45 min at 4°C. The supernatant was collected and filtered. Then, the supernatant was centrifuged again at 110,000 × g for 70 min at 4°C twice. The exosome pellet was resuspended with phosphate-buffered saline (PBS) and stored at −80°C for further experiments.
Transmission Electron Microscopy
Transmission electron microscopy was used to visualize the morphology of isolated Exos. A drop of Exos in PBS solution was placed onto a copper grid. Exos were washed with PBS and then fixed with glutaraldehyde solution (3%) for 15 min. After another wash with PBS, Exos were incubated with uranyl acetate solution (2%) for 5 min. Finally, Exos were visualized using a transmission electron microscope and the Exo size was measured using a dynamic light scattering.
Internalization of Exosomes
To analyze whether Exos could be taken up by HESCs, Exos were firstly labeled with KH67 fluorescent dye (Sigma-Aldrich, St. Louis, MO, USA). Then, 2 μg KH67-labeled Exos were added to HESCs that were cultured in 6-well plates. Twenty-four hours later, HESCs were fixed with 4% paraformaldehyde and stained with 4′,6-diamidino-2-phenylindole solution. The staining of HESCs was examined using a confocal microscope (Carl Zeiss, Oberkochen, Germany).
EdU Proliferation Assay
The EdU (5-ethynyl-2′-deoxyuridine) staining assay was performed to detect the proliferation of HESCs. Human endometrial stromal cells (5 × 104) were seeded in a 24-well plate and cultured overnight. 500 ng/ml LPS (Sigma-Aldrich) and/or 1 μg Exos were added onto HESCs. Twenty-four hours later, HESCs were washed with PBS once and stained with the BeyoClick™ EdU-488 Cell Proliferation Assay Kit (Beyotime, Shanghai, China), according to the manufacturer’s instructions. DNA staining was performed with DAPI solution. Human endometrial stromal cells were visualized using a fluorescence microscopy.
Luciferase Reporter Assays
To detect the direct interaction between miR-21 and TLR4, luciferase reporter assay was conducted. TRL4 3′-untranslated region fragments with wild-type or mutant miR-21 binding site were cloned into the luciferase reporter vector to construct TRL4 wild type 3′-untranslated region or TRL4 MU 3′-untranslated region. Human endometrial stromal cells were co-transfected with TRL4 wild type 3′-UTR (or TRL4 mutant 3′-untranslated region) and miR-21 (or control miRNA) using the Lipofectamine 2000 reagent (Invitrogen, CA, USA). After transfection for 48 h, the firefly and renilla luciferase activity were detected with the dual-luciferase reporter assay kit (Promega, Madison, WI, USA), according to the manufacturer’s protocol.
Apoptosis Analysis
Flow cytometry was performed to analyze the apoptotic rate of HESCs. Human endometrial stromal cells (2.5 × 105 cells/well) were cultured in 6-well plates overnight before treatment. Human endometrial stromal cells were treated with 500 ng/ml LPS alone or together with 1 μg Exos for 48 h. Then, the medium was removed and HESCs were collected. After centrifugation, the cell pellet was washed with PBS once. Staining of HESCs was conducted using the Annexin V-FITC apoptosis detection kit (BD, Franklin Lakes, NJ, USA) following the manufacturer’s introduction. Stained cells were analyzed using the flow cytometer (BD Biosciences).
Cell Viability Assay
Cell Counting Kit-8 assay was carried out to determine the viability of HESCs. HESCs were seeded in a 96-well plate and treated with different concentrations of LPS (0–500 ng/ml) for 48 h. At the indicated time point, 10 μl of Cell Counting Kit-8 solution (Seyotin, Guangzhou, China) was added to each well and the cells were incubated for 2–4 h. Finally, the optical density (OD) at 450 nm was examined by a microplate reader (BioTek, Vermont, USA).
Quantitative Real-Time PCR
Total RNA from HESCs or endometrial tissues was isolated using the Trizol reagent (Thermo Fisher Scientific). Complementary DNA (cDNA) synthesis was performed with total RNA using the Invitrogen TM Superscript III First-Strand Synthesis System (Seyotin). For the detection of miR-21, cDNA was reversed using a TaqMan™ Advanced miRNA cDNA Synthesis Kit (Seyotin). Real-time polymerase chain reaction (PCR) was carried out using the PowerUp SYBR Green PCR Master Mix (Seyotin). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal control for mRNA expression and U6 was used as an internal control of miR-21 expression. Relative gene expression levels were measured using a 2–ΔΔCT formula. The primers used were as follows: TLR4-F, ACTTGGACCTTTCCAGCAAC and TLR4-R, TTTAAATGCACCTGGTTGGA; TNF-α-F, TGACGGGCTTTACCTCATCT and TNF-α-R, TGATGGCAGACAGG ATGTTG; IL-6-F, AACGAGTGGGTAAAGAACGC and IL-6-R, CTGACCAGA GGAGGGAATGC; β-actin-F, CCAAGGCCAACCGTGA GAAAAT and β-actin-R, CCACATTCCGTGAGGATCTTCA; IL-1β-F, ATGAAG AGCTGCATCCAACA and IL-1β-R, ATGGAAGACATGTGCGTAGG.
Western Blot Analysis
HESCs and Exos were collected, washed with PBS, and lysed in radioimmunoprecipitation assay (RIPA) lysis buffer. The concentration of different samples was detected by the bicinchoninic acid method. The protein sample was separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gel, followed by transfer onto a polyvinylidene fluoride (PVDF) membrane (Seyotin). The membrane was incubated with TBST (TBS-Tween 20) solution containing 5% non-fat milk for 1 h at room temperature. Then, the membrane was incubated with primary antibodies at 4°C overnight. On the next day, the membrane was washed three times with PBS and incubated with horseradish peroxidase (HRP)-conjugated secondary antibodies for 90 min at room temperature. Protein bands were visualized using the ECL Western Blotting Substrate (Seyotin). The used antibodies were as follows: CD9, 1:1,000, ab92726, Abcam; CD63, 1:1,000, ab134045, Abcam; TNF-α, 1:1,000, 17590-1-AP, Proteintech; IL-1β1:1,000, #12242, Cell Signaling Technology; IL-6, 1:1,000, ab6672, Abcam; GAPDH 1:2,000, 60004-1-Ig, Proteintech; TLR4, 1:1,000, ab13556, Abcam; p-p65, 1:1,000, #3033, Cell Signaling Technology; p65, 1:1,000, #8242, Cell Signaling Technology.
DNA Transfection
PCDNA3.1-Myc-TLR4 overexpressing plasmid was purchased from Genecopoeia. DNA transfection was carried out using the Lipofectamine™ 3000 reagent (Thermo Fisher Scientific).
Animal Experiments
A CE mouse model was established to detect the function of Exos in the inflammation response in vivo. Kunming female mice (8–10 weeks old, 25–30 g) were purchased from the Chinese Academy of Sciences (Beijing, China) and cultured under a 12 h light/12 h dark cycle. The mice were randomly divided into three groups with 10 mice per group (control, LPS, and Exos). The control group received 20 μl of PBS by uterine perfusion, while the LPS group received 20 μl of LPS (2.5 μg/μl). Twelve hours post-perfusion, mice in the control and LPS groups were intraperitoneally injected with 20 μl of PBS, and mice in the Exos group were intraperitoneally injected with 20 μg of exosomes. Twelve hours later, mice were sacrificed and the uteri were isolated for further experiments. All animal procedures were conducted according to the animal guidelines approved by the Institutional Animal Care and Use Committee of Daoke Medical Technology (Guangdong) Co., LTD.
Immunohistochemistry (IHC)
Mouse endometrial tissues were fixed in 10% buffered formalin and embedded in paraffin. Paraffin-embedded tissues were cut into 4-μm-thick sections. Sections were then dewaxed, dehydrated, and antigen-retrieved in citrate buffer. After blocking in 5% bovine serum albumin, sections were probed with TNF-α, IL-1β, and IL-6 antibodies. Sections were washed three times with PBS and incubated with biotinylated secondary antibody. After three washes with PBS, sections were incubated with HRP-conjugated streptavidin (Thermo Fisher Scientific), followed by incubation with diaminobenzidine solution. Sections were counterstained with hematoxylin.
Statistical Analysis
Data were represented as mean ± standard deviation (SD). Statistical analysis was carried out using the SPSS 15.0 software (SPSS, USA). Comparisons among different groups were accessed by one-way analysis of variance (ANOVA). Values of P < 0.05 were considered statistically significant.
Results
Identification of Exos Derived From ADSCs
Exos were isolated by ultracentrifugation using the supernatant of ADSCs. The morphology of Exos was analyzed using transmission electron microscopy and a spherical morphology was detected (Fig. 1A). The mean size distribution of Exos was 118.9 nm (Fig. 1B). Western blot was performed to detect the exosome makers, CD9 and CD63. As shown in Fig. 1C, in comparison with the control cells, the expressions of CD9 and CD63 were highly increased in Exos. We then investigated whether Exos could be taken up by HESCs. Exos labeled with PKH67 green fluorescent dye was incubated with HESCs for 24 h and immunofluorescence staining was performed. As shown in Fig. 1D, green fluorescent spots were observed in HESCs, indicating that Exos were internalized by HESCs.
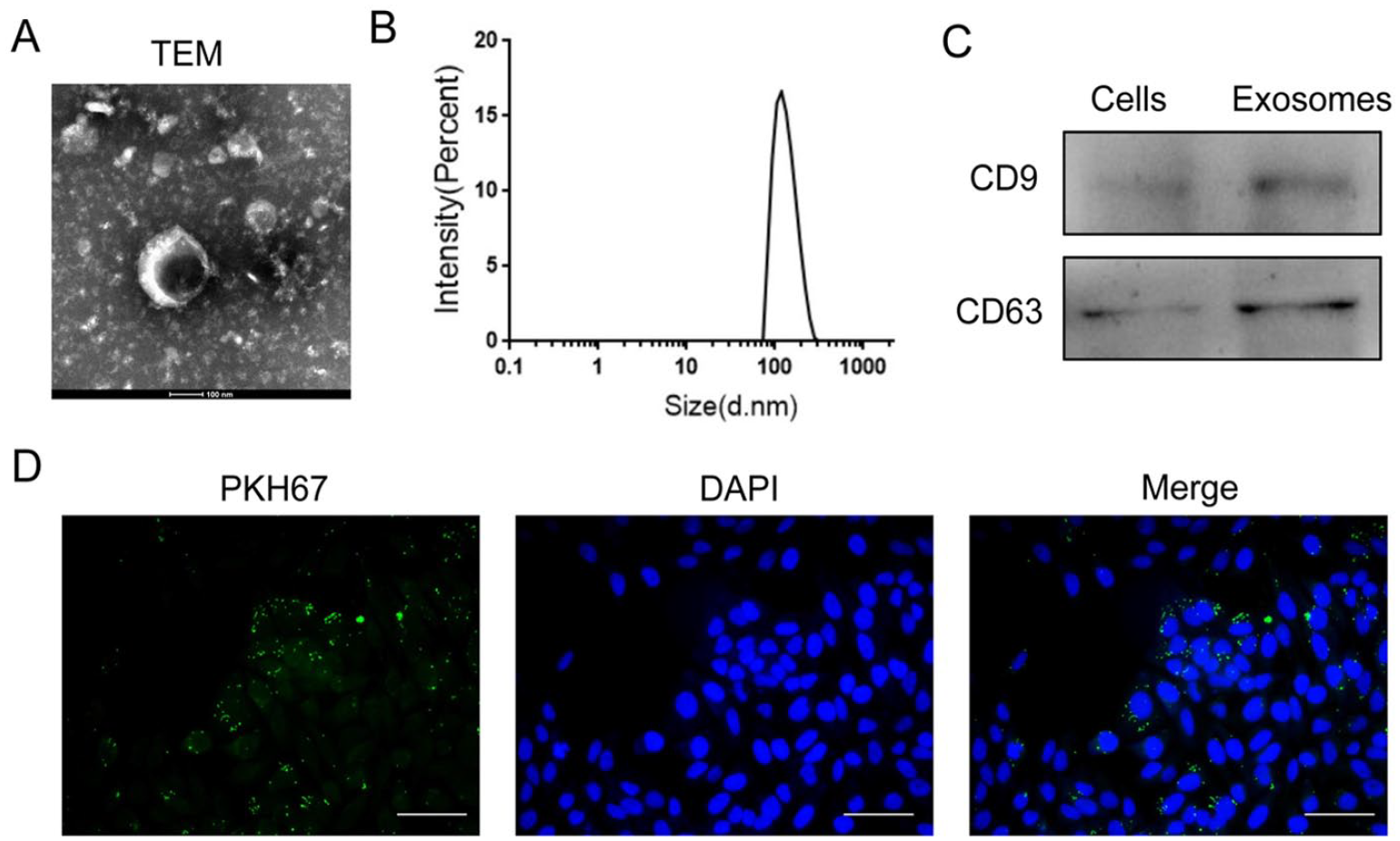
Characterization of isolated exosomes (Exos) from ADSCs. (A) The morphology of Exos was detected by TEM analysis. (B) Size distribution of Exos was indicated by intensity. (C) The expression of exosome markers was analyzed by Western blot. (D) The uptake of Exos by HESCs was accessed by immunofluorescence staining. ADSCs: adipose tissue–derived stem cells; HESCs: human endometrial stromal cells; TEM: transmission electron microscopy; DAPI:4’,6-diamidino-2-phenylindole.
Exos Inhibit LPS-Induced Inflammation in HESCs
To detect the function of Exos in CE, an in vitro CE model was established by treating HESCs with LPS. The results showed that treatment with LPS reduced the proliferation of HESCs (Supplemental Fig. S1A) and induced the expression of inflammatory factors, such as IL-1β, IL-6, and TNF-α (Supplemental Fig. S1B and C). Interestingly, administration of Exos restored the viability of HESCs inhibited by LPS (Fig. 2A). Moreover, while LPS increased the apoptosis of HESCs, treatment with Exos blocked this effect (Fig. 2B, C). In addition, cocultured with Exos decreased the mRNA and protein levels of IL-1β, IL-6, and TNF-α that were induced by LPS in HESCs (Fig. 2D–G). Together, these results indicate that Exos suppress LPS-mediated inflammation and increase HESC cell proliferation.

Exos inhibit the LPS-induced inflammatory response in HESCs. (A) HESC proliferation was detected by EdU staining following treatment with LPS alone or together with Exos. (B) Apoptotic rate of HESCs was determined by flow cytometry following treatment with LPS alone or together with Exos. (C) The expression of apoptosis-related proteins in HESCs after treatment with LPS alone or together with Exos. (D–F) The mRNA expressions of IL-1β (D), IL-6 (E), and TNF-α (F) were detected by qRT-PCR in HESCs following treatment with LPS alone or together with Exos. (G) The protein expression of TNF-α, IL-6, and IL-1β was detected by Western blot in HESCs following treatment with LPS alone or together with Exos. LPS: lipopolysaccharide; HESCs: human endometrial stromal cells; EdU: 5-ethynyl-2′-deoxyuridine; IL-1β: interleukin-1β; IL-6: interleukin-6; TNF-α: tumor necrosis factor-alpha; qRT-PCR: quantitative real-time polymerase chain reaction; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; DAPI:4’,6-diamidino-2-phenylindole.
Exos Suppress LPS-Stimulated Inflammation via MiR-21
miR-21 has been reported to be involved in LPS-induced inflammatory response in endometrium 17 . Therefore, we investigated whether miRNA-21 contributed to the inhibitory effect of Exos on inflammation in our model. Quantitative real-time polymerase chain reaction analysis showed that while LPS reduced the expression of miR-21 in HESCs, treatment with Exos restored the level of miR-21 (Fig. 3A). To confirm the function of miR-21 in HESCs, EdU staining assay was carried out. The result displayed that the number of EdU-positive cells was markedly decreased after treatment with LPS, and this effect was counteracted by miR-21 (Fig. 3B). Lipopolysaccharide increased BAX expression and decreased BCl-2 expression in HESCs, but these effects were reversed by miR-21 (Fig. 3C), indicating that miR-21 could repress LPS-induced HESC apoptosis. Moreover, overexpression of miR-21 decreased the levels of inflammatory cytokines, such as TNF-α, IL-6, and IL-1β, that were elevated by LPS exposure (Fig. 3D, E). Overall, these data indicate that Exos suppress LPS-stimulated inflammation response and enhance HESC cell proliferation via miR-21.

Exos suppress LPS-stimulated inflammatory response via miR-21. (A) The expression of miR-21 in HESCs was detected by qRT-PCR after treatment with LPS alone or together with Exos. (B) The proliferation of HESCs was determined by EdU staining after treatment with LPS alone or together with miR-21. (C) The expression of apoptosis-associated proteins Bcl-2 and Bax was determined by Western blot after treatment with LPS alone or together with miR-21. (D) The mRNA expression of TNF-α, IL-6 and IL-1β was determined by qRT-PCR after treatment with LPS alone or together with miR-21. (E) The mRNA expression of TNF-α, IL-6, and IL-1β was determined by Western blot after treatment with LPS alone or together with miR-21. LPS: lipopolysaccharide; HESCs: human endometrial stromal cells; qRT-PCR: quantitative real-time polymerase chain reaction; EdU: 5-ethynyl-2′-deoxyuridine; TNF-α: tumor necrosis factor-alpha; IL-6: interleukin-6; IL-1β: interleukin-1β; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; DAPI:4’,6-diamidino-2-phenylindole; NC:negative control.
Exos Repress LPS-Induced Inflammation via miR-21/TLR4/NF-κB Pathway
It is well known that TLR4 plays a key role in inflammation. TargetScan analysis revealed that there was a miR-21-binding site in TLR4 3′-UTR (Fig. 4A). The luciferase reporter assay further confirmed that TLR4 was a direct target of miR-21 in HESCs (Fig. 4B), which was consistent with previous studies in other cell types18–20. Moreover, overexpression of miR-21 could effectively reduce TLR4 expression (Supplemental Fig. S2). As shown in Fig. 4C, LPS increased TLR4 expression in HESCs, and this effect was counteracted by Exo treatment. In agreement with this result, administration of Exos decreased the phosphorylation of p65, a downstream effector of TLR4, but not the total protein of p65 (Fig. 4C). In addition, while Exos reduced LPS-induced production of TNF-α, IL-6, and IL-1β, overexpression of TLR4 abolished this effect (Fig. 4D, E). Therefore, our data suggest that Exos suppress LPS-induced inflammation via miR-21/TLR4/NF-κB signaling in HESCs.

Exos inhibit LPS-induced inflammatory response via miR-21/TLR4/NF-κB signaling. (A) The potential binding site of miR-21 in TLR-4 3′-UTR was detected by TargetScan. (B) The binding of miR-21 and TLR-4 3′-UTR was examined by dual-luciferase reporter assay. (C) The expression levels of TLR4, p65, and p-p65 were examined by Western blot after administration of LPS alone or together with Exos. (D) Overexpression of TLR4 reversed the effect of Exos on LPS-induced mRNA expression of TNF-α, IL-6, and IL-1β in HESC. (E) Overexpression of TLR4 reversed the effect of Exos on LPS-induced protein expression of TNF-α, IL-6, and IL-1β in HESCs. LPS: lipopolysaccharide; TNF-α: tumor necrosis factor-alpha; IL-6: interleukin-6; IL-1β: interleukin-1β; HESCs: human endometrial stromal cells; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; TLR4:Toll-like receptor4; NF-KB:Nuclear factor kappa beta; UTR:Untranslated Region; WT:wild type; MU:mutant; OE:overexpression.
Exos Suppress LPS-Mediated Inflammation In Vivo
To verify the effect of Exos on inflammatory activity, a CE mouse model was utilized. Immunohistochemistry staining showed that the content of TNF-α, IL-6, and IL-1β was significantly elevated in endometrial tissues of LPS-treated mice compared with the control mice. However, co-administration of Exos reduced the production of these inflammatory factors that were stimulated by LPS in endometrial tissues (Fig. 5A). Consistently, qRT-PCR and Western blot validated that LPS induction markedly increased the mRNA and protein levels of TNF-α, IL-6, and IL-1β, which was blocked by Exos in mouse endometrial tissues (Fig. 5B, C). Collectively, these results indicate that Exos have a protective activity against endometrial inflammation in vivo.

Exos repress LPS-triggered inflammatory response in vivo. (A) The protein levels of TNF-α, IL-6, and IL-1β in mouse endometrial tissues were detected by immunohistochemical staining. (B) The mRNA levels of TNF-α, IL-6, and IL-1β in mouse endometrial tissues were detected by qRT-PCR. (C) The protein levels of TNF-α, IL-6, and IL-1β in mouse endometrial tissues were detected by Western blot. LPS: lipopolysaccharide; TNF-α: tumor necrosis factor-alpha; IL-6: interleukin-6; IL-1β: interleukin-1β; qRT-PCR: quantitative real-time polymerase chain reaction; GAPDH: glyceraldehyde-3-phosphate dehydrogenase.
Discussion
Chronic endometritis is highly related to infertility in women. Hence, improving the treatment of CE is of significant importance. In this study, we reported for the first time that Exos derived from ADSCs had a suppressive effect on LPS-induced inflammation in endometrial cells and in a mouse model.
Exos are small vesicle secreted by different cell types, including ADSCs 9 . ADSC-Exos have attracted more and more attention as potential therapeutic agent 21 . ADSC-Exos have been reported to possess an anti-inflammatory activity. For example, Zhao et al. 14 report that Exos from ADSCs inhibit inflammation in white adipose tissue (WAT) of diet-induced obese mice via activation of M2 macrophage polarization. Feng et al. 22 show that treatment with ADSC-Exos decreases the production of inflammatory factors in microglia cells stimulated with LPS and thus prevent neuroinflammation. Chronic endometritis is an inflammation disease in the inner layer of uterine mucosa 23 , which is mainly caused by microbial infection in the uterine cavity 24 . Lipopolysaccharide is one of the important causes of endometritis 25 . Therefore, an in vitro endometritis model was established by treating HESCs with LPS. Using this model, we found that treatment with ADSC-Exos significantly enhanced the proliferation of HESCs and inhibited HESC apoptosis. More importantly, we found ADSC-Exos decreased the expression levels of IL-1β, TNF-α, and IL-6 stimulated by LPS, indicating that ADSC-Exos had a protective activity against LPS-induced inflammation. Moreover, the anti-inflammatory activity of ADSC-Exos was also verified by a mouse model. Therefore, our data indicate that ADSC-Exos have a protective activity against LPS-induced inflammation in endometrial cells both in vitro and in vivo.
It has been demonstrated that Exos can deliver different biomolecules to target cells to exert their function6–8. Among these cargo molecules, miRNAs have attracted great attention due to their extensive regulation in gene expression 26 . The function of miR-21 in inflammation modulation has been documented previously27–29. It has been shown that in equine endometrium, the expression of miR-21 is markedly decreased after LPS challenge, indicating an involvement of miR-21 in inflammatory response in endometrium 17 . Consistently, our work showed that LPS exposure reduced the level of miR-21 in HESCs, whereas administration of ADSC-Exos restored the expression of miR-21. Moreover, overexpression of miR-21 caused downregulation of inflammatory cytokines, including IL-1β, TNF-α, and IL-6, and increased HESC proliferation. Therefore, our results suggest that miR-21 plays an important role in ADSC-Exo-mediated suppression of inflammation response.
Toll-like receptor (TLR) is critical for the primary innate immune response to pathogenic challenge 30 . Studies from Ju et al. 31 reveal that TLR4 plays a critical role in the pathogenesis of human CE. They found that the expression of TLR4, as well as the levels of its corresponding downstream signaling factors, such as p-p65, TNF-α, and IL-1β, is upregulated in CE tissues compared with normal endometrial tissues 31 . miR-21 has been shown to regulate TLR4 expression18–20. It has been reported that overexpression of miR-21 inactivates TLR4/NF-kB pathway and thus suppresses myocardial apoptosis 18 . In our experiments, we found that TLR4 expression was downregulated by miR-21 and luciferase reporter assay confirmed that TLR4 was a direct target of miR-21 in endometrial cells. In addition, treatment with Exos decreased the expression of TLR4, as well as the phosphorylation of p65, a downstream factor of TLR4. In addition, administration of ADSC-Exos reduced the secretion of LPS-induced IL-1β, TNF-α, and IL-6, but this effect was blocked by ectopic expression of TLR4. Taken together, our results indicate the ADSC-Exos attenuates LPS-induced inflammation via miR-21/TLR4/Nf-κB signaling. In summary, this study reveals that ADSC-derived Exos have an inhibitory effect on LPS-stimulated inflammation in endometrial cells both in vitro and in vivo, which is mediated by the regulation of miR-21/TLR4/Nf-κB signaling pathway. Our results suggest that Exos-based therapy may be a promising strategy for the treatment of inflammation-associated diseases, including CE.
Supplemental Material
sj-jpg-1-cll-10.1177_09636897231173736 – Supplemental material for Exosomes From Adipose-Derived Stem Cells Suppress the Progression of Chronic Endometritis
Supplemental material, sj-jpg-1-cll-10.1177_09636897231173736 for Exosomes From Adipose-Derived Stem Cells Suppress the Progression of Chronic Endometritis by Bin Wang, Li Li and Ruizhu Yu in Cell Transplantation
Supplemental Material
sj-jpg-2-cll-10.1177_09636897231173736 – Supplemental material for Exosomes From Adipose-Derived Stem Cells Suppress the Progression of Chronic Endometritis
Supplemental material, sj-jpg-2-cll-10.1177_09636897231173736 for Exosomes From Adipose-Derived Stem Cells Suppress the Progression of Chronic Endometritis by Bin Wang, Li Li and Ruizhu Yu in Cell Transplantation
Footnotes
Availability of Data and Materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Ethical Approval
The study was approved by the Institutional Animal Care and Use Committee of Dongguan People’s Hospital.
Statement of Human and animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Guangdong Medical Science and Technology Research Fund (No. A2022273).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
