Abstract
BACKGROUND:
The role of isoforms of prostate specific antigen (PSA) and other kallikrein-related markers in early detection of biochemical recurrence (BCR) after radical prostatectomy (RP) is not well known and serum PSA is currently used in preoperative risk nomograms.
OBJECTIVE:
The aim of this research was to study pre- and early postoperative levels of important PSA isoforms and human kallikrein-2 to determine their ability to predict BCR and identify disease persistence (DP).
METHODS:
This study included 128 consecutive patients who underwent RP for clinically localized prostate cancer. PSA, fPSA, %fPSA, [–2]proPSA, PHI and hK2 were measured preoperatively, at 1 and 3 months after RP. We determined the ability of these markers to predict BCR and identify DP.
RESULTS:
The DP and BCR rate were 11.7%and 20.3%respectively and the median follow up was 64 months (range 3–76 months). Preoperatively, the independent predictors of BCR were PSA (
CONCLUSIONS:
[–2]proPSA and PHI are preoperative predictors of BCR and DP that outperform the currently used serum PSA. At the early postoperative period there is no additional benefit of the other markers tested.
Keywords
Introduction
Prostate cancer (PC) affects approximately 12 percent of men during their lifetime [1] and is associated with a 3 percent risk of PC death [2]. Prostate-specific antigen (PSA), also known as human kallikrein-3 (hK3) is a biomarker secreted by the epithelial cells of the prostate gland that belongs to the family of serine proteases. Initially used in monitoring disease progression of PC patients, its use in screening, diagnosis, preoperative risk nomograms and follow up after primary therapy has become the current standard. After radical prostatectomy (RP), patients are surveilled by PSA measurements at three, six and twelve months, then every six months until three years, and then annually [3]. The level of PSA should be undetectable at 4–6 weeks postoperatively [4, 5]. A persistently elevated value after RP signals residual disease or micrometastases, while a fastly or slowly rising PSA during surveillance suggests metastases or local recurrence, respectively. The current definition of biochemical recurrence (BCR) after RP includes 2 consecutive PSA rises > 0.2 ng/ml and occurs in approximately 20–40%of patients [6, 7].
While the focus has been on developing new diagnostic markers for PC in the preoperative setting, that could either replace or enhance the performance of PSA, no alternatives have been approved in the postoperative period. Early prediction of BCR would be of paramount importance in planning adjuvant or salvage forms of therapy. The importance of identifying early patients at risk for BCR resides in the fact that the earlier radiation therapy (RT) is delivered postoperatively (either in adjuvant or salvage setting), the better seem to be the outcomes [8].
Although ultrasensitive PSA (uPSA) assays are able to detect minimal postoperative PSA levels of 0.001 ng/ml [9], the advantage of their use in clinical practice remains unclear.
New markers which have been extensively studied in the diagnostic setting of PC are recently being investigated as predictors of BCR in the preoperative setting. Among the studied markers are [–2]proPSA - a free-PSA (fPSA) isoform -and the derived formula Prostate Health index (PHI) [–2]proPSA/fPSA * √
Studies on the utility of PHI in the preoperative context confirmed its value as a predictor of BCR [13, 14], tumor volume [15] and pathologic outcomes [16, 17].
Another approved tool in PC diagnosis is the four kallikrein (4K) score test that includes the four parameters free PSA (fPSA), intact PSA, total PSA and human kallikrein-2 (hK2) and has shown excellent results in selecting patients with significant disease for prostate biopsy [18]. hK2 in particular belongs to the protein kallikrein family (such as PSA) and previous studies refer to its use in early detection of PC as an adjunct to PSA and fPSA [19], its value in BCR-prediction [20, 21] and the potential value of hK2/fPSA and hK2/%fPSA in detecting minimal PC disease [22].
To our knowledge, besides one study comparing the trends of [–2]proPSA and uPSA in the first year after RP in a high-risk population [23], and a second study [24] comparing the kinetics of [–2]proPSA and PSA in the first 3 years post-RP, no recent studies exist on the potential use of markers such as fPSA, %fPSA, hK2 and PHI to predict BCR and identify disease persistence (DP) in the postoperative setting.
The aim of our study was to investigate the use of these markers in both preoperative and early postoperative settings and examine their predictive power.
Materials and methods
Study population
A cohort of 128 consecutive patients underwent open or laparoscopic radical prostatectomy (RP) for clinically localized prostate cancer at the author’s institution between January 2013 and January 2014. Among them, 24 patients (18.8%) underwent laparoscopic RP and 104 patients (81.3%) underwent an open procedure. The median (range) follow up period was 64 months (3–76 months). When patients were classified as having either BCR or DP their follow up in this study was ended. Extended pelvic lymph node dissection was planned according to the MSKCC nomogram [25]. Pathological staging was assessed according to the TNM Classification of Malignant Tumours seventh edition [26] and histological grading according to the Gleason grading system [27]. Forty-eight patients (37.5%) had adverse pathological features such as extracapsular extension, seminal vesicle invasion or positive surgical margins. No patient received preoperative or postoperative hormonal androgen deprivation therapy or adjuvant radiotherapy (RT). Blood samples were collected before surgery and the follow up included complete clinical assessment including digital rectal examination and blood sample collection at 1 and 3 months after RP. Samples were collected before any manipulations that could transiently increase the marker levels.
All blood samples were centrifuged for 15 minutes at 3000 revolutions per minute, the serum pipetted into 1,8ml aliquots (Nunc Cryotube Vials, Thermo Fisher scientific, Roskilde, Denmark) and stored at –76°C. The markers evaluated were PSA, fPSA, [–2]proPSA, PHI and hk2. BCR was defined as 2 consecutive rises of PSA > 0.2 ng/ml, while DP was defined as PSA≥0.1|ng/ml at 6 weeks after RP.
The study complied with The Code of Ethics of the World Medical Association (Declaration of Helsinki), printed in the British Medical Journal (18 July 1964). Institutional Review Board approval (reference: EK-377/13) was obtained for the analysis of the patients and all samples were collected and analysed after a written informed consent signed by each of the participants.
Marker measurement
PSA, fPSA and [–2]proPSA serum samples were analysed by Access Hybritech Immuno-assay on Access2 (Beckman Coulter USA). The calibration procedures were performed by a 6-point recombinant curve for PSA and fPSA (both 0–20 ng/ml) and a 7-point recombinant [–2]proPSA curve (0–5000 pg/ml). Quality control was performed daily using quality controls provided by Beckman Coulter. FreePSA/PSA and PHI were calculated by Access2 system.
The Human kallikrein 2 (hK-2) ELISA kit –Cusabio Biotech Co., LTD., Houston, USA was used for the analysis of hK-2. The detection range of the kit was 62.5–4000 pg/ml. Frozen samples were incubated at room temperature two hours before the assay. Samples were mixed several times and transparent supernatants were used for the assay. An added protocol for each kit was followed. Standardisation of measurement was assured by the creation of a calibration curve for each assay. A four-parameter logistic calibration curve was used for the calculation of the hK2 concentration. All analyses were performed in the same laboratory by the same board-certified biochemist.
Statistical analysis
Variables of our interest were compared between studied groups of patients and the recurrence-free group using a Mann-Whitney Wilcoxon U-test in case of numerical variables and using a Fisher Exact test in case of categorical ones. Multivariable models were constructed using a logistic regression. To evaluate a predictive value of the variables a ROC analysis including an AUC evaluation was adopted.
Results
Baseline characteristics
The main patient characteristics are listed on Table 1.
Main patient characteristics. All the continuous variables are expressed as median (range) and categorical estimates as number (percentage)
Main patient characteristics. All the continuous variables are expressed as median (range) and categorical estimates as number (percentage)
During the median follow up period of 64 months, 87 patients (68%) were free from recurrence, a total of 26 patients developed BCR (20.3%) and 15 patients (11.7%) had disease persistence manifested by a persistently high PSA. The statistically significant differences between the BCR-free, BCR and DP groups were the pre- and postoperative tumour grading, clinical (cT) and pathological stages (pT) and nodal status (pN). Parameters such as patient age or prostate mass were not significantly different between the patient groups. The mean time to BCR was 19 months (range 3–49 months) while the 1-year post-RP BCR rate was 8.6%(11 patients).
The marker levels measured at diagnosis, preoperatively, at 1 month and 3 months after RP are shown in Table 2.
Marker levels at diagnosis, preoperative, 1-month and 3-month time period. All the results are expressed as a median (range). P -values were calculated using the Wilcoxon test
Marker levels at diagnosis, preoperative, 1-month and 3-month time period. All the results are expressed as a median (range).
Besides the markers shown on Table 2, the predictive ability of hk2/fPSA and hK2/%fPSA was also tested. Both ratios did not significantly correlate to either BCR or DP at both preoperative and postoperative periods (
Figures 1–3 depict the ROC curves obtained for BCR at the different time points using the different markers.

ROC curves for all the markers at preoperative period and their relation to BCR. Red PSA (AUC 0.64), Blue [–2]proPSA (AUC 0.70), Green PHI (AUC 0.74), Brown hK2 (AUC 0.60), Violet fPSA (AUC 0.59), Black fPSA/PSA (AUC 0.60).
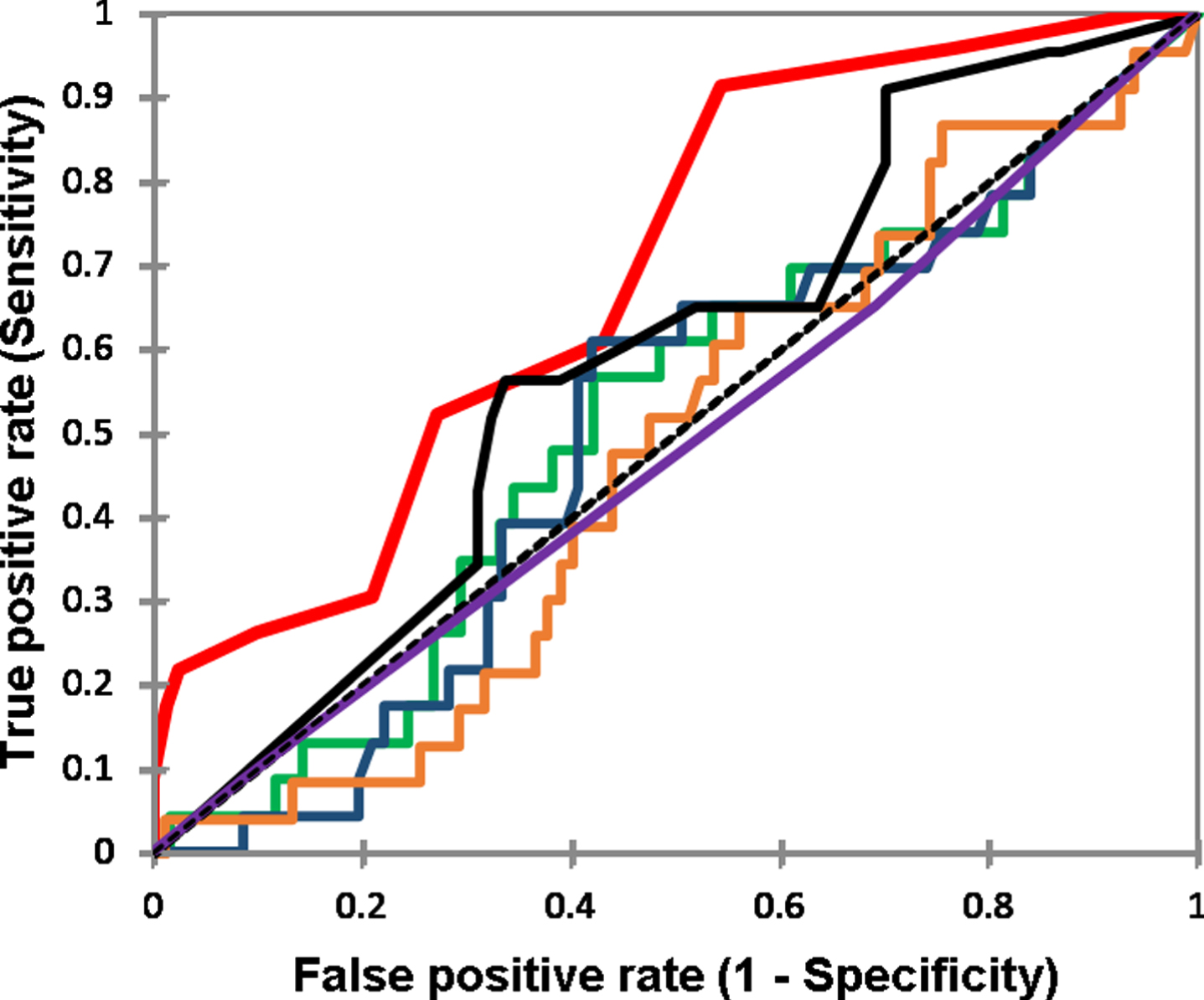
ROC curves for all the markers at 1 month period and their relation to BCR. Red PSA (AUC 0.69), Blue [–2]proPSA (AUC 0.50), Green PHI (AUC 0.52), Brown hK2 (AUC 0.48), Violet fPSA (AUC 0.48), Black fPSA/PSA (AUC 0.59).
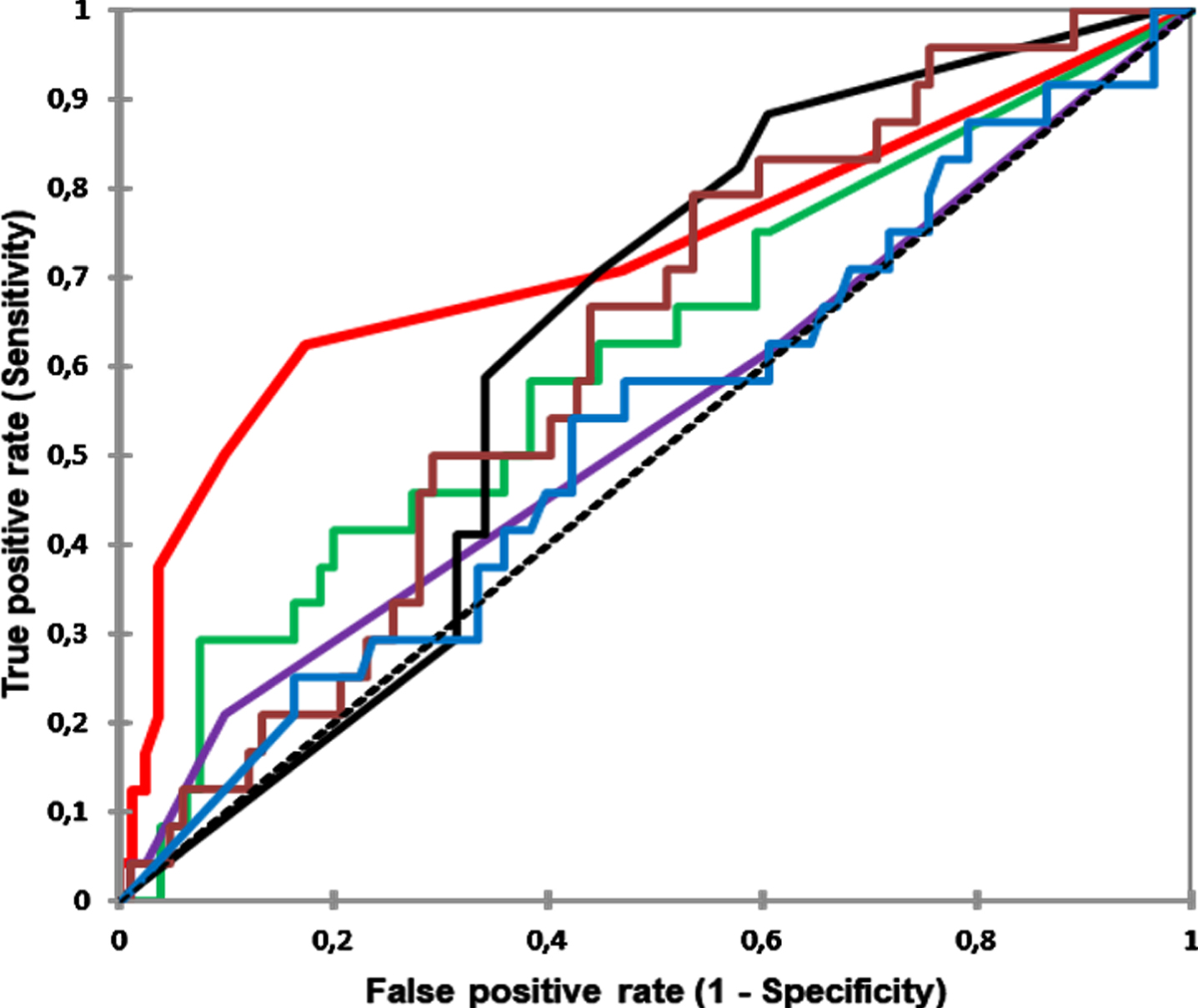
ROC curves for all the markers at 3 months period and their relation to BCR. Red PSA (AUC 0.72), Blue [–2]proPSA (AUC 0.53), Green PHI (AUC 0.61), Brown hK2 (AUC 0.62), Violet fPSA (AUC 0.54), Black fPSA/PSA (AUC 0.62).
In the preoperative setting, the predictors of BCR were preoperative PSA (AUC 0.64;
PSA (AUC 0.68;
Multivariate models to predict BCR using preoperative data including clinical stage (cT1 and cT2-T3), preoperative Gleason score (GS 6 and GS≥7) and the different studied markers were created (Table 3).
Multivariate models using preoperative data and selected markers
Multivariate models using preoperative data and selected markers
At multivariate analysis, PSA, [–2]proPSA and PHI were all independent predictors of BCR. Among these, PHI was the most important marker predicting BCR both when used alone (Model C) and when combined to PSA (Model E). Both models attained areas under the curve of 0.86 (
So far, not much is known about the ideal follow up of patients after RP. Not only no other marker with the exception of PSA has been approved for disease surveillance and BCR identification, but also the schedule of PSA testing is a matter of debate, with some experts advocating a risk-adapted follow up versus the current ‘one-size-fits-all’ standard schedule of PSA measurements at 3,6,12 months after RP then semiannually until 3 years, and then annually [3].
After surgery, the level of PSA should drop to undetectable levels within 21–30 days, considering that the half-life of PSA is approximately 3.2 days [28]. Therefore, there is a window of opportunity for the early use of markers of disease recurrence and persistence.
An early and more accurate prediction of BCR and DP is of the utmost importance. It would have an impact on patient counseling, selecting patients for nuclear imaging techniques such as 68GaPSMA-11 Positron Emission Tomography and risk-adapting surveillance schedules. It could select a group of patients who would benefit of a stricter follow up protocol and would benefit from adjuvant or early-salvage therapy. Although adjuvant therapy is considered in patients with adverse pathological features (extracapsular extension, seminal vesicle invasion and positive surgical margins), an overtreatment risk of approximately 50%[29] should not be neglected. Early prediction of BCR would thus spare these patients from unnecessary morbidity.
Despite recent advances in PC diagnosis brought by PSA isoforms such as [–2]proPSA and PHI, their use in BCR prediction and in surveillance is virtually unexplored. Preoperative risk nomograms such as the MSKCC nomogram [25] which guides the surgeon in decision making concerning pelvic lymph node dissection and nerve-sparing procedures based on the probability of extraprostatic disease, lymph node and seminal vesicle invasion and risk of BCR still rely, among other parameters, on serum PSA.
The present work aimed to investigate the predictive ability of PSA isoforms to detect BCR and DP at three distinct time points: preoperatively and in the early post-operative period (one month and 3 months after RP).
With the present work we showed that PHI (AUC 0.73;
This finding is in accordance with previous results by Lughezzani et al. [14] and Maxeiner et al. [13] who tested preoperative PHI values in populations of 313 and 437 patients respectively and confirmed the value of PHI as an independent biomarker of BCR prediction.
There are not many studies on the use of PSA isoforms in the surveillance period. We found that only PSA was able to correlate with BCR at the first (AUC 0.64;
As far as we are aware, the present work is the first recent study testing the remaining PSA isoforms in the postoperative period and their relation to the occurrence of BCR.
Disease persistence is defined in most studies as a detectable level of PSA > 0.1 ng/ml within 4–8 weeks after RP and occurs in 5–20%of patients [30]. This may be the result of residual benign or malignant prostate tissue or the presence of micrometastases. In our cohort of 128 patients, 15 patients (11.7%) developed DP. We tested the aforementioned markers and concluded that preoperatively both [–2]proPSA (AUC 0.73;
Besides recent nuclear imaging studies on the detection of DP [31] there are currently no studies on potential serum markers signaling DP, with the exception of studies using PSA and uPSA.
The significance of our work resides in the finding that both [–2]proPSA and PHI are stronger predictors of BCR and DP than the current serum PSA at the preoperative setting, so their use in preoperative nomograms and validation by larger scale studies would be a logical next step.
Conversely, the use of any other tested marker besides PSA brings no benefit in the period post-RP.
Although our study is innovative in testing PSA isoforms both at the preoperative and postoperative periods, analysed in the same laboratory, by the same operator, in a homogenous population, it is not devoid of limitations. The relatively small number of patients enrolled, the low rate of BCR and DP, and the use of stored serum samples albeit the study’s prospective design are the main drawbacks of our study.
We believe that cross-validation of models including [–2]proPSA and especially PHI in a preoperative nomogram, on a larger independent population, in a prospective and multicentre setting would be of a great value.
Conclusions
proPSA and PHI outperform serum PSA as predictors of BCR and DP preoperatively. PHI is the strongest independent predictor of BCR. At the early surveillance period only PSA correlated to BCR, and there is no additional benefit of using its isoforms in detecting DP. Further research on the use of [–2]proPSA and PHI in BCR prediction is needed to prove its clinical usefulness.
Footnotes
Acknowledgments
Project supported by the Ministry of Health of the Czech Republic under Grant 00064203.
Author contributions
CONCEPTION: Stepan Vesely, Joana Do Carmo Silva
DATA CURATION: Joana Do Carmo Silva
ANALYSIS OF DATA: Joana Do Carmo Silva, Hana Luksanova
PREPARATION OF THE MANUSCRIPT: Joana Do Carmo Silva
REVISION FOR IMPORTANT INTELLECTUAL CONTENT: Stepan Vesely
SUPERVISION: Richard Prucha, Marko Babjuk
Conflict of interest
Joana Do Carmo Silva, Stepan Vesely, Hana Luksanova, Richard Prucha and Marko Babjuk declare they have no conflicting interests.
