Abstract
GATA2 is a pioneering transcription factor governing androgen receptor expression and signaling in prostate cells. To understand the prognostic potential of GATA2 assessment in prostate cancer, we analyzed nuclear GATA2 expression on an annotated tissue microarray with 12,427 prostate cancer samples. Normal prostate glands were negative to weakly positive. GATA2 staining was found in almost all prostate cancers (95%). Strong GATA2 staining was linked to advanced tumor stage, high classical and quantitative Gleason grade (p < 0.0001 each), positive nodal stage (p = 0.0116), and early biochemical recurrence (p < 0.0001). GATA2 was linked to
Keywords
Introduction
In Western societies, prostate cancer is the most prevalent cancer in men. 1 A bulk of prostate cancers is indolent. Only a small subset is highly aggressive.2,3 Gleason grade and tumor extent on biopsies, prostate-specific antigen (PSA), and clinical stage are proven preoperative prognostic parameters. These data are statistically powerful for the cohort, but often insufficient for individual treatment decisions. Thus, it is hoped that a better understanding of disease biology will lead to novel clinically applicable molecular markers that enable a reliable prediction of prostate cancer aggressiveness.
The transcription factor GATA2 is a member of a family of six proteins that have been named after their ability to bind specific DNA consensus sequences characterized by the A/T-GATA-A/G sequence. 4 GATA proteins function as pioneering factors that open chromatin to initiate tissue-specific expression programs.5,6 GATA2 was initially identified as a fundamental factor in early steps of hematopoiesis 7 because inactivating mutations of GATA2 were linked to myelodysplastic disorders and leukemia.8,9 However, later it became evident that GATA2 is also a critical factor for urogenital development 10 and specifically for the prostate gland. 11 GATA2 is a key transcription factor for the androgen receptor (AR) and is, in turn, transcriptionally regulated by AR and androgens in a negative feedback loop. 12 Furthermore, GATA2 modulates the transcriptional activity of androgen-responsive genes as it co-localizes on their DNA enhancer elements with AR and Fork head box protein A1.12–15 Thus, it was not surprising to find that experimental modulation of GATA2 levels in prostate cells had a strong impact on androgen-dependent transcription and cell growth. 13 Accordingly, studies investigating cohorts of 109 and 383 prostate cancers reported immunohistochemical GATA2 expression to be linked to aggressive tumor features such as invasive and metastatic cancers,12,16–18 high Gleason grade, and advanced tumor stage.12,16,17
As such data identify GATA2 as a promising candidate for a clinically valuable prognostic marker, we studied the expression and prognostic impact of GATA2 in our large annotated prostate cancer prognosis tissue microarray (TMA), comprising more than 12,000 prostate cancer specimens.
Materials and methods
Patients
Radical prostatectomy specimens were available from 12,427 patients, who had surgery between 1992 and 2012. Median follow-up was 48 months (range: 1–280 months; Table 1). PSA recurrence was defined as the time point when postoperative PSA levels were at least 0.2 ng/mL and increasing at subsequent measurements. The TMA was annotated with tumor stage, Gleason grade, nodal stage, and resection margin status. The specimens were analyzed according to a standard procedure.
19
In addition to the classical Gleason categories, “quantitative” Gleason grading was performed as previously described.
20
In brief, every cancer demonstrating a Gleason 4 (and 5) pattern was subclassified according to its percentage of Gleason 4. For practical use, we subdivided the 3 + 4 and 4 + 3 cancers into seven subgroups: 3 + 4, ≤5% Gleason 4; 3 + 4, 6%–10%; 3 + 4, 11%–20%; 3 + 4, 21%–30%; 3 + 4, 31%–49%; 4 + 3, 50%–60%; and 4 + 3, 61%–100%. In addition, separate groups were defined by the presence of a tertiary Gleason 5 pattern, including 3 + 4 Tertiary 5 and 4 + 3 Tertiary 5. The molecular database attached to this TMA contained results on ERG expression,
Pathological and clinical data of the arrayed prostate cancers.
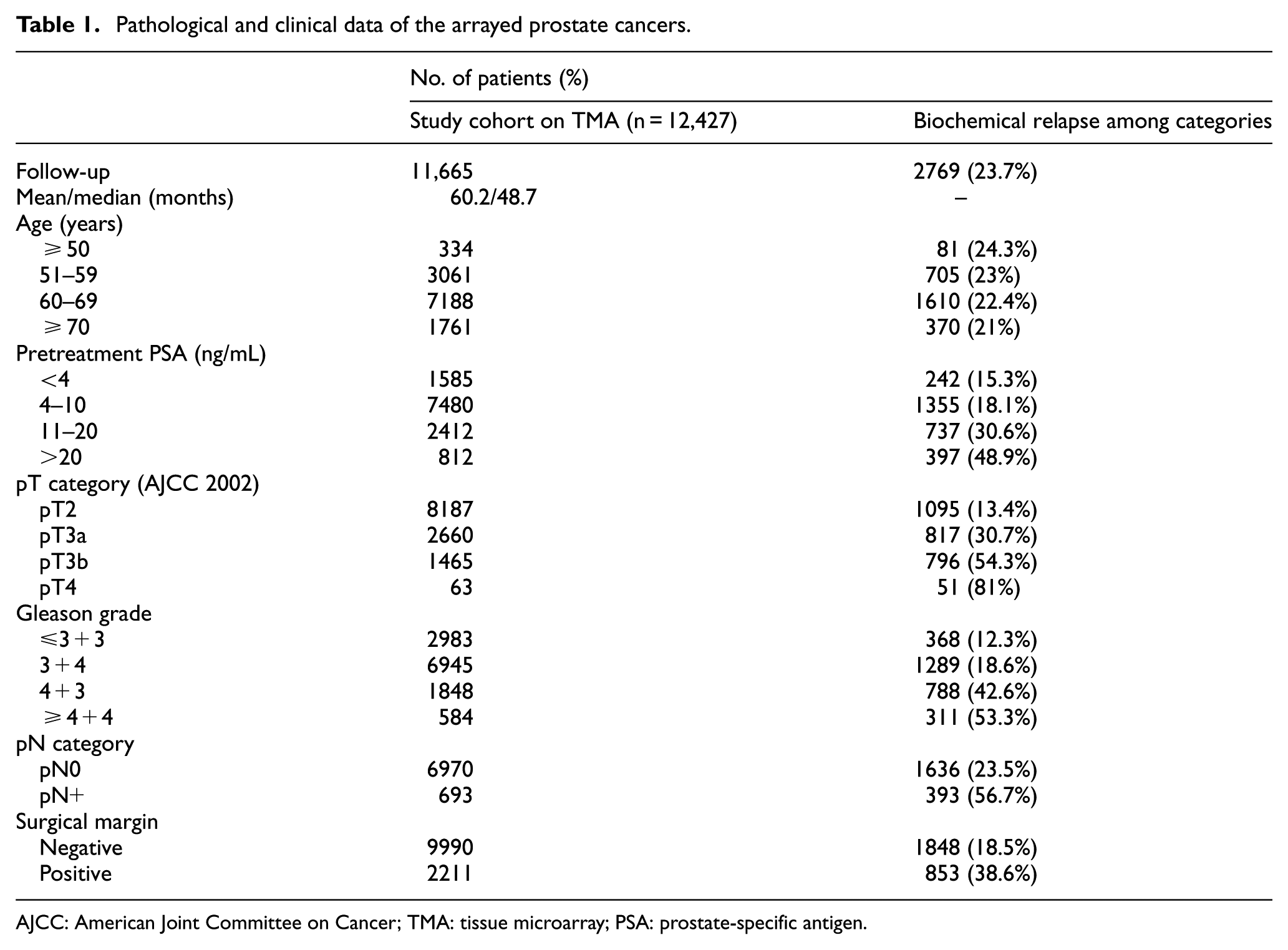
AJCC: American Joint Committee on Cancer; TMA: tissue microarray; PSA: prostate-specific antigen.
Immunohistochemistry
Freshly cut TMA sections were all stained on the same day in a single run. Slides were deparaffinized, rehydrated, washed in DAKO buffer (K8002, Agilent, Santa Clara, CA) and transferred to a DAKO Link 48 autostainer device. GATA2 staining was performed with the rabbit polyclonal anti-GATA2 antibody (Cat. # NBP1-82581; Novus Biologicals Europe, Abingdon, UK) at 1:1350 dilutions. Heat-induced antigen retrieval was performed at 98°C in DAKO at pH 6. GATA2 staining was typically nuclear, sometimes with slight cytoplasmic co-staining. The staining intensity (0, 1+, 2+, and 3+) and the fraction of positive tumor cells were separately recorded for each tissue spot. A final score was then built from these two parameters according to the following system as previously described. 24 Negative cases had complete absence of staining, weak cases had staining intensity of 1+ in ≤70% of tumor cells or staining intensity of 2+ in ≤30% of tumor cells; moderate cases had staining intensity of 1+ in >70% of tumor cells, staining intensity of 2+ in >30% but in ≤70% of tumor cells or staining intensity of 3+ in ≤30% of tumor cells; strong cases had staining intensity of 2+ in >70% of tumor cells or staining intensity of 3+ in >30% of tumor cells.
Statistics
Contingency tables and the chi-square test were performed to search for associations between molecular parameters and tumor phenotype. Analysis of variance (ANOVA) test was applied to search for differences of the Ki67LI. Survival curves were calculated according to Kaplan–Meier. The log-rank test was applied to detect significant differences between groups. Cox proportional hazards regression analysis was performed to test the statistical independence and significance between pathological, molecular, and clinical variables. Separate analyses were performed using different sets of parameters available either before or after prostatectomy. Calculations were done with JMP 12 (SAS Institute Inc., Cary, NC, USA).
Results
A total of 9769 (78.6%) of tumor samples were interpretable in our TMA analysis. Non-informative cases (2658, 21.4%) lacked tissue samples or unequivocal cancer tissue in the TMA spot. Normal prostate tissue showed negative to weakly positive nuclear staining. In cancers, nuclear GATA2 staining was seen in 9321 of 9769 (95.4%) samples and was considered weak in 12.2%, moderate in 43.5%, and strong in 39.7% (Table 2). Representative images of GATA2 staining are shown in Figure 1.
Association between GATA2 staining results and prostate cancer phenotype.
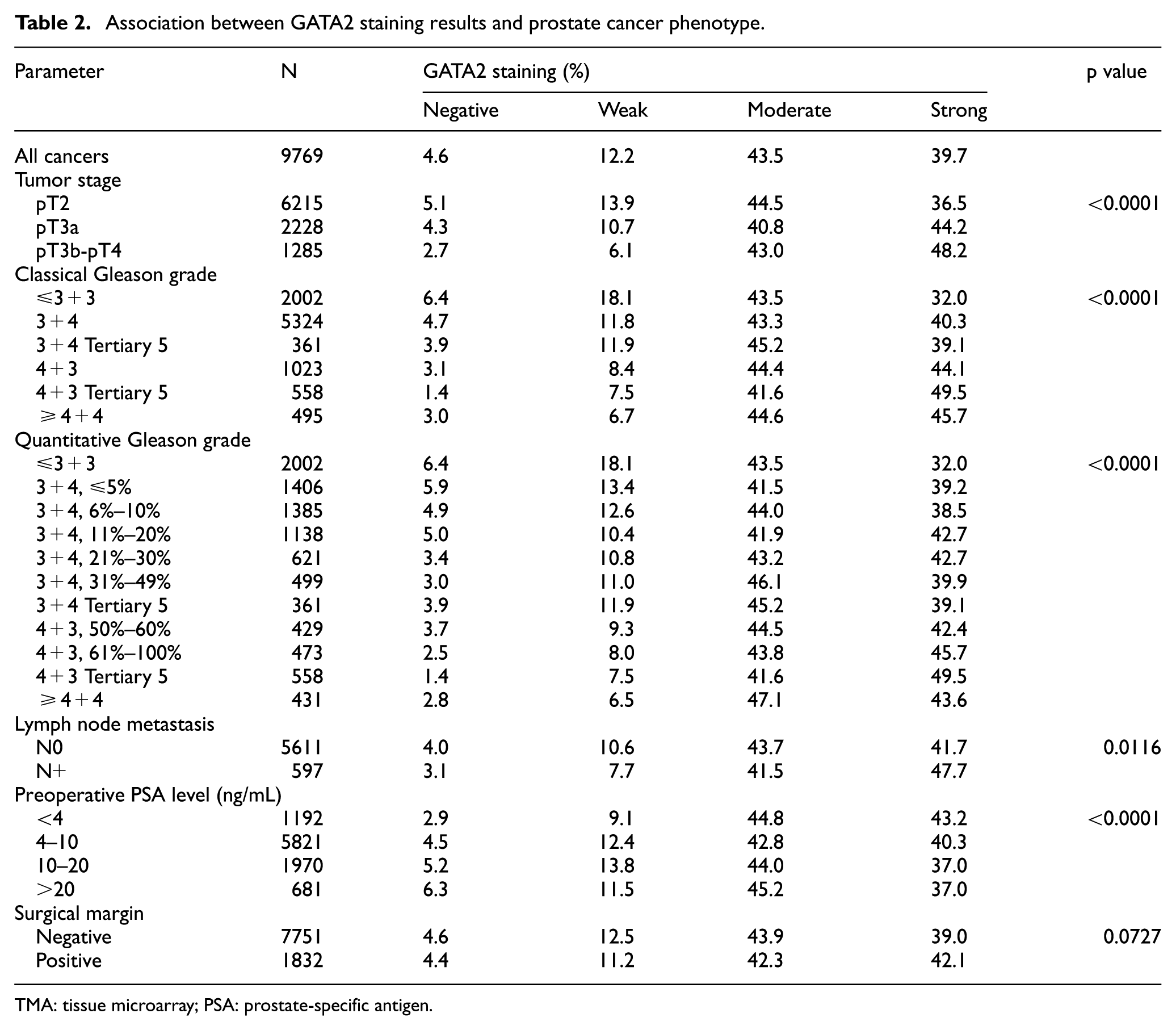
TMA: tissue microarray; PSA: prostate-specific antigen.
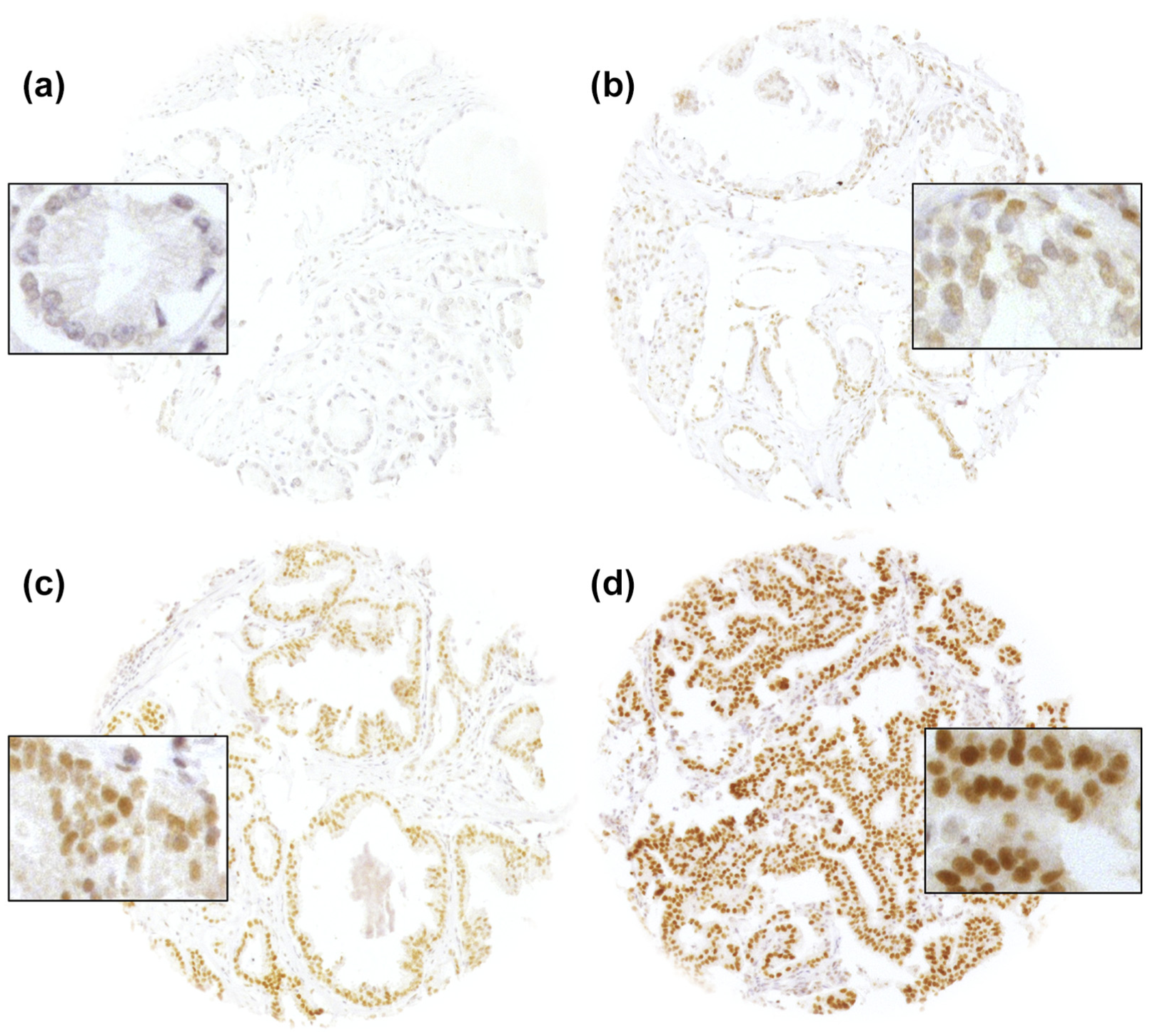
Representative pictures of GATA2 immunostaining in prostate cancer with (a) negative, (b) weak, (c) moderate, and (d) strong staining.
Association with TMPRSS2:ERG fusion status and ERG protein expression
To evaluate whether GATA2 expression is associated with ERG status in prostate cancers, we used data from previous studies (expanded from Minner et al.
21
). Data on
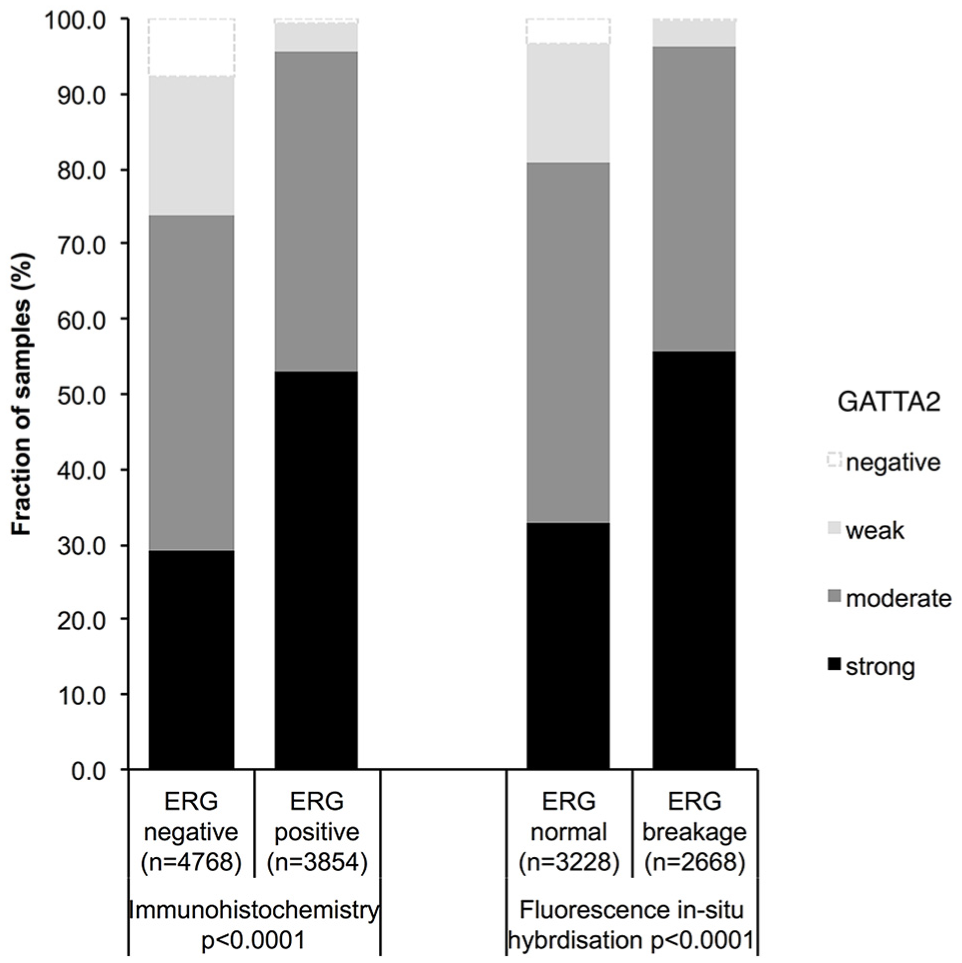
Association between positive GATA2 staining and ERG status (IHC/FISH) in all cancers.
Associations with tumor phenotype
Increased GATA2 expression was significantly linked to advanced tumor stage (p < 0.0001), classical and quantitative Gleason grade (p < 0.0001 each), positive nodal stage (p = 0.0116), and low preoperative PSA level (p < 0.0001; Table 2). These associations also held true in subset analyses of ERG-negative and ERG-positive cancers for ERG-positive cancers (Supplementary Tables 1 and 2).
Association with AR
GATA2 is a known transcriptional regulator of AR expression. 13 We utilized IHC data on AR expression from a previous study 23 to compare AR and GATA2 expression levels. Data on both GATA2 and AR were available for 6448 cancers. There was a strong positive association between AR and GATA2 expression in all cancers, including both the ERG-negative and ERG-positive subsets (p > 0.0001 each; Figure 3).

Association between positive GATA2 and AR expression levels in all cancers and subsets of ERG-negative and ERG-positive cancers.
Association with tumor cell proliferation (Ki67LI)
GATA2 was significantly linked to increased cell proliferation in all cancers, as measured by Ki67LI (p < 0.0001). The average Ki67LI increased from 0.68 ± 0.15 in cancers lacking GATA2 expression to 1.79 ± 0.10 (weak), 2.82 ± 0.05 (moderate), and 3.37 ± 0.06 in cancers with strong GATA2 expression (p < 0.0001). This association held true in all tumor subsets with identical Gleason score (≤3 + 3: p < 0.0001, 3 + 4: p < 0.0001, 4 + 3: p < 0.0001, ≥4 + 4: p = 0.0007; Table 3). This relationship was more prominent in ERG-negative than in ERG-positive cancers (Supplementary Tables 3 and 4).
Association between GATA2 staining and Ki67 labeling index in all cancers and in subsets of cancers with identical Gleason grade.
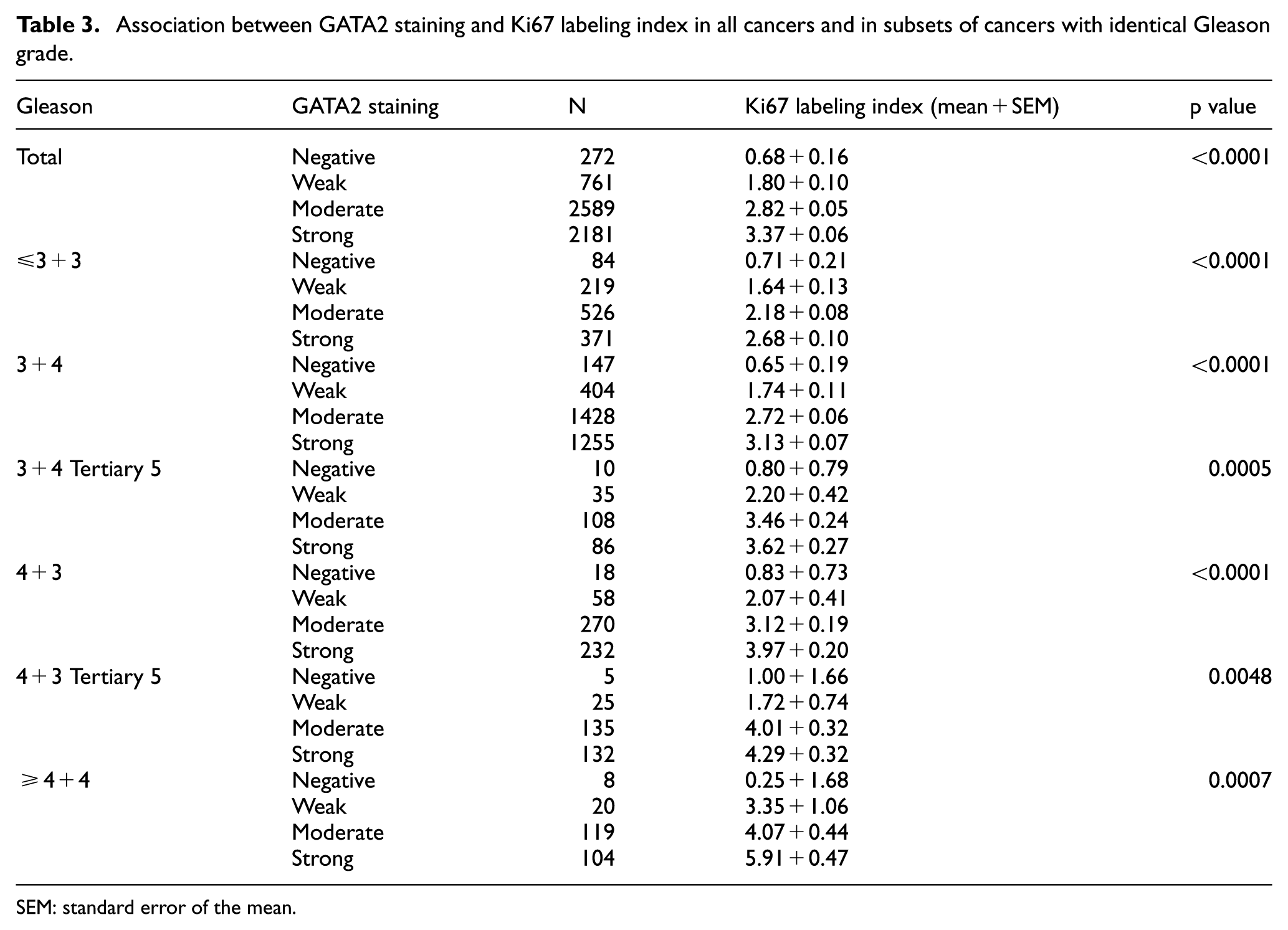
SEM: standard error of the mean.
Association with PSA recurrence
Follow-up data were available for 8787 patients with interpretable GATA2 staining. Highly significant associations were found between early PSA recurrence and GATA2 expression in all tumors (Figure 4(a), p < 0.0001) as well as in the subgroup of

Association between GATA2 expression and biochemical recurrence in (a) all cancers, (b)

Kaplan–Meier plot of prostate-specific antigen (PSA) recurrence-free survival after radical prostatectomy and low (negative/weak) versus high (moderate/strong) staining of GATA2 in subsets of ERG-negative cancers defined by identical (a) classical Gleason score and (b–h) quantitative Gleason score categories.
Multivariate analysis
Four different types of multivariate analyses were performed evaluating the clinical relevance of GATA2 expression in different scenarios (Table 4). Scenario 1 evaluated all postoperatively available parameters including pathological tumor stage, pathological lymph node status (pN), surgical margin status, preoperative PSA value, and pathological Gleason grade obtained after the morphological evaluation of the entire resected prostate. In scenario 2, all postoperatively available parameters with exception of nodal status were included. The rationale for this approach is that the indication and extent of lymph node dissection is not standardized in the surgical treatment of prostate cancer and that excluding pN in multivariate analysis can markedly increase case numbers. Two additional scenarios had the purpose of modeling the preoperative situation as much as possible. Scenario 3 included GATA2 expression, preoperative PSA, clinical tumor stage (cT stage), and Gleason grade obtained on the prostatectomy specimen. Since postoperative determination of a tumor’s Gleason grade is “better” than the preoperatively determined Gleason grade (subjected to sampling errors and consequently under-grading in more than one-third of cases 25 ), another multivariate analysis was added. In scenario 4, the preoperative Gleason grade obtained on the original biopsy was combined with preoperative PSA, cT stage, and GATA2 expression. These analyses revealed that GATA2 provided independent prognostic information in ERG-negative scenarios (hazard ratio 1.26–1.48, Supplementary Table 5), especially if the lymph node stage was not included. An independent prognostic value of GATA2 was lacking in ERG-positive cancers.
Multivariate analyses including GATA2 expression in all cancers, ERG-negative cancers, and ERG-positive cancers.

PSA: prostate-specific antigen.
Discussion
The results of our study demonstrate that overexpression of GATA2 is linked to adverse tumor features and early biochemical recurrence in the subset of prostate cancers lacking
Detectable nuclear GATA2 staining was found in virtually all-normal and cancerous prostate samples in our study. However, unequivocal upregulation in cancers as compared to normal glands, that is, moderate to strong staining was seen in more than 83.2% of our 9769 interpretable cancers. The total fraction of 95% cancers with detectable GATA2 staining in our study is slightly higher than in earlier studies on 109 and 383 prostate cancers, who reported positive staining in 77%, 82%, 84%, and >90% of cancers.12,16–18 These differences are remarkably small given that various antibodies (i.e. a mouse monoclonal antibody from Sigma, rabbit polyclonal antibodies from Cell Signaling Technology, and Novus) and laboratory protocols were used. It is possible that the Novus antibody used in our study resulted in a particularly sensitive detection of the ubiquitously expressed GATA2 protein. Chiang et al. 18 reported >90% positivity in their TMA study on 358 tumors with the same antibody. He et al. 12 reported increased staining intensity of cancer cells as compared to normal epithelium. Together, these findings demonstrate that upregulation of GATA2 expression typically accompanies malignant transformation of prostate cells.
A strong association with tumor phenotype and clinical outcome was found in this study. This is in concordance with earlier work reporting associations between increased GATA2 expression and one or more adverse tumor features including advanced stage, high Gleason grade, metastatic growths, and early biochemical recurrence in univariate analysis.12,16,17 A higher cancer aggressiveness in case of GATA2 overexpression fits well with the known roles of GATA2 as an inducer of AR expression and an enhancer of transcription of androgen-responsive genes.12,17 As summarized in a recent review by Rodriguez-Bravo et al., 26 GATA2 acts through three modes of action on a genome-wide level: GATA2 binds to upstream AR regulatory elements to enhance AR expression, creates open chromatin in AR enhancers by recruiting the p300 histone acetyltransferase that activates chromatin via acetylation of H3K27, and forms and maintains regulatory chromatin loops between AR-bound distal enhancers and AR target gene promoters via recruitment of the mediator co-regulatory complex subunit MED1. The central role of the AR for prostate cancer is well known from studies showing that the administration of androgen results in accelerated tumor growth in vitro, 27 and in vivo. 28
The availability of a molecular database attached to this prostate cancer TMA enables us to investigate the interaction with genes of interest including the AR and
The data of this study suggest that GATA2 expression may represent a clinically useful marker in ERG-negative cancer. In this subgroup, GATA2 expression had a prognostic impact that was independent of established prognostic parameters, especially if only parameters were included that are available at the moment in time when decisions on therapy are taken. Earlier studies of 109 and 383 prostate cancers had also demonstrated a statistically significant prognostic impact of GATA2 overexpression, but could not demonstrate an independent prognostic effect of GATA2 overexpression12,16 and were unable to separately analyze molecularly defined subgroups. This highlights the importance of large tumor sets that allow for statistically meaningful analyses. It is of note that several parameters that are usually included in multivariate analyses are statistically strong but suffer from significant shortcomings in clinical practice. pT stage and nodal status are lacking during the preoperative therapeutic decision-making. The Gleason grade, the most powerful preoperatively available prognostic marker, suffers from very substantial interobserver variability reaching beyond 40% in individual biopsies. 38 That GATA2 expression had a prognostic impact in Gleason 3 + 4 = 7 cancers illustrates that this subgroup contains highly diverse tumors ranging from Gleason 3 + 4 = 7 with 5% Gleason 4 and behaving very similar to Gleason 3 + 3 = 6 up to Gleason 3 + 4 = 7 with 40% Gleason 4 and aggressive tumor behavior more comparable to Gleason 4 + 3 = 7. 20 Accordingly, GATA2 expression lacked prognostic impact in cancers defined by identical quantitative Gleason grade. This demonstrates the power of the quantitative Gleason grading system, which is, however, not universally applied and does not solve all issues of interobserver variability in prostate cancer grading. GATA2 expression measurement may thus have potential to become an element in a future multiparametric prognostic test for ERG-negative prostate cancer.
Recent studies suggest that GATA2 may also serve as a predictive biomarker particularly in advanced and castration-resistant prostate cancers. For example, Vidal et al. 39 demonstrated that GATA2 overexpression is linked to chemotherapy resistance in vitro, and that GATA2 knockdown can reduce tumor growth in patient-derived, castration-resistant, taxane-resistant xenografts. Although direct inhibitors of GATA2 are currently not available, indirect targeting may be achieved through inhibitors of upstream or downstream factors such as NOTCH, AKT or Hedgehog signaling, histone acetyltransferases, and deacetylases, where inhibitory compounds are under development (reviewed in Rodriguez-Bravo et al. 26 ). The high fraction of about 80% of cancers with GATA2 upregulation suggests that many prostate cancer patients may benefit from such therapies should they become available.
In summary, our study demonstrates that GATA2 upregulation is a frequent event in prostate cancer, which is linked to poor prognosis specifically in the subset of ERG-negative cancers. GATA2 expression analysis—especially in combination with other prognostic markers—may have the potential for clinical application in prostate cancer.
Supplemental Material
Supplementary_information – Supplemental material for The independent prognostic impact of the GATA2 pioneering factor is restricted to ERG-negative prostate cancer
Supplemental material, Supplementary_information for The independent prognostic impact of the GATA2 pioneering factor is restricted to ERG-negative prostate cancer by Franziska Büscheck, Maciej Zub, Asmus Heumann, Claudia Hube-Magg, Ronald Simon, Dagmar S Lang, Doris Höflmayer, Emily Neubauer, Frank Jacobsen, Andrea Hinsch, Andreas M Luebke, Maria Christina Tsourlakis, Guido Sauter, Hartwig Huland, Markus Graefen, Alexander Haese, Hans Heinzer, Torsten Schlomm, Till S Clauditz, Eike Burandt, Waldemar Wilczak, Stefan Steurer and Sarah Minner in Tumor Biology
Footnotes
Acknowledgements
The authors thank W Fehrle for help in revision of the manuscript and are grateful to Janett Lütgens, Sünje Seekamp, and Inge Brandt for excellent technical assistance. The German Federal Ministry of Education and Research (BMBF) supported this work (grant number 01KU1505B). F.B. and M.Z. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
