Abstract
As a member of helix–loop–helix protein family, transcription factor 12 functions as either an oncogene or a tumor suppressor in various human cancers. However, there are no reports on its involvement in prostate cancer. To investigate clinical relevance of transcription factor 12 in prostate cancer and to evaluate its roles in malignant phenotypes of this cancer in vitro and in vivo, we here examined expression patterns of transcription factor 12 protein in 50 prostate cancer tissue specimens by immunohistochemistry. Then, associations of transcription factor 12 expression with various clinicopathological characteristics and patients’ prognosis of prostate cancer were evaluated. Its involvements in cancer cell proliferation, migration, invasion, and tumor growth were determined by in vitro and in vivo experiments. As a result, the positive immunostaining of transcription factor 12 protein was localized in cytoplasm and/or nucleus of prostate cancer cells. Its expression levels were decreased with prostate cancer Gleason score increased. Statistically, the decreased expression of transcription factor 12 protein more frequently occurred in prostate cancer patients with high Gleason score, positive metastasis, prostate-specific antigen failure, and short biochemical recurrence–free survival (all p < 0.05). Importantly, multivariate analysis showed that the status of transcription factor 12 expression was an independent predictor of biochemical recurrence–free survival in prostate cancer. Functionally, enforced expression of transcription factor 12 suppressed cell proliferation, migration, and invasion in vitro and inhibited tumor growth in vivo. In conclusion, transcription factor 12 protein may be a novel molecule which plays a critical role in prostate cancer progression and patients’ prognosis, suggesting it might be a promising therapeutic target for prostate cancer therapy.
Keywords
Introduction
Prostate cancer (PCa), one of the most common noncutaneous cancer among males, represents the second leading cause of cancer-related deaths worldwide, particularly in the Western countries. 1 Although there have been great advances in early diagnosis and prevention, the incidence and mortality of PCa are increasing constantly. The patients hospitalized were frequently diagnosed as having advanced diseases characterized by metastasis during which cancer cells may be moved from the point of origin within the prostate gland to multiple distant organ sites throughout the body. 2 Various clinicopathological characteristics, such as serum levels of prostate-specific antigen (PSA), tumor–node–metastasis (TNM) stage, and Gleason score, have been extensively used to divide PCa patients into different risk groups. 3 However, these parameters all have limitations and cannot be used as a sole criterion. 4 Therefore, it is necessary to identify more accurate biomarkers for PCa diagnosis and prognosis.
Transcription factor 12 (TCF12, also known as HEB or HTF4), as a member of helix–loop–helix (HLH) protein family, has the direct DNA (E-box) binding ability. 5 TCF12 protein is found to be extensively expressed in many tissues and can form homodimers or heterodimers. 6 Functionally, TCF12 is responsible for cell development and differentiation. 7 It has been reported that TCF12 can regulate the differentiation of lymphocytes or the development of neural or mesenchymal tissues. 8 The mutations in this gene or the translocation fusion of its fragment with other molecules may result in craniosynostosis or mesenchymal malignancies. 9 Growing evidence shows that TCF12 may function as either an oncogene or a tumor suppressor in various human cancers. For example, He et al. 10 found that TCF12 expression was elevated in gallbladder cancer tissues and correlated with poorer overall survival in patients. Tang et al. 11 indicated that TCF12 was obviously elevated in cancer-associated fibroblasts, leading to extracellular matrix remodeling and triggering the invasion and metastasis of breast cancer cells both in vitro and in vivo. Lee et al. 12 determined that TCF12 may act as a transcriptional repressor of E-cadherin, and its overexpression may be closely correlated with the occurrence of colorectal cancer metastasis. In contrast, Chen et al. 13 identified TCF12 as a direct target of miR-211 in oral squamous cell carcinoma and confirmed that it might function to suppress the oncogenicity of this malignancy. However, there are no reports on the involvement of TCF12 in PCa.
As a member of HLH protein family, TCF12 functions as either an oncogene or a tumor suppressor in various human cancers.10–13 However, there are no reports on its involvement in PCa. To investigate clinical relevance of TCF12 in PCa and to evaluate its roles in malignant phenotypes of this cancer, we here examined the expression patterns of TCF12 protein in PCa and benign prostate tissue specimens by immunohistochemistry. Then, associations of TCF12 expression with various clinicopathological characteristics and patients’ prognosis of PCa were evaluated. Moreover, its involvements in cancer cell proliferation, migration, invasion, and tumor growth were determined by in vitro and in vivo experiments.
Materials and methods
Ethic statement
This study was approved by the human study ethics committees at the Ministry of Public Health of China. All specimens were handled and made anonymous according to the ethical and legal standards.
All animal experiments in this study were performed in compliance with the guidelines of the Institute for Laboratory Animal Research at Guangzhou Medical University, Guangzhou, China.
Patients and tissue samples
For the immunohistochemistry analysis, a tissue microarray (TMA) of 50 primary PCa tissues were purchased from Xi’an Ailina Biotechnology Co, Ltd (China; cat. no. PR807a). Patients received chemotherapy or radiotherapy before surgery were excluded from the study.
For the evaluation of the clinical relevance of TCF12, a publicly available dataset named the Taylor dataset including 150 primary PCa tissues and 29 adjacent non-cancerous prostate tissues with messenger RNA (mRNA) microarray expression data was collected. 14 The overall survival was defined as the time interval from the date of surgery to the day of the last follow-up or death. Biochemical recurrence (BCR) was defined as PSA ⩾0.2 ng/mL on two consecutive measurements after radical prostatectomy. The BCR-free survival was determined from the initial surgery to the day of BCR. The clinicopathological characteristics of the above two datasets were summarized in Table 1.
Associations of TCF12 protein and mRNA expression with various clinicopathological characteristics of patients with PCa.
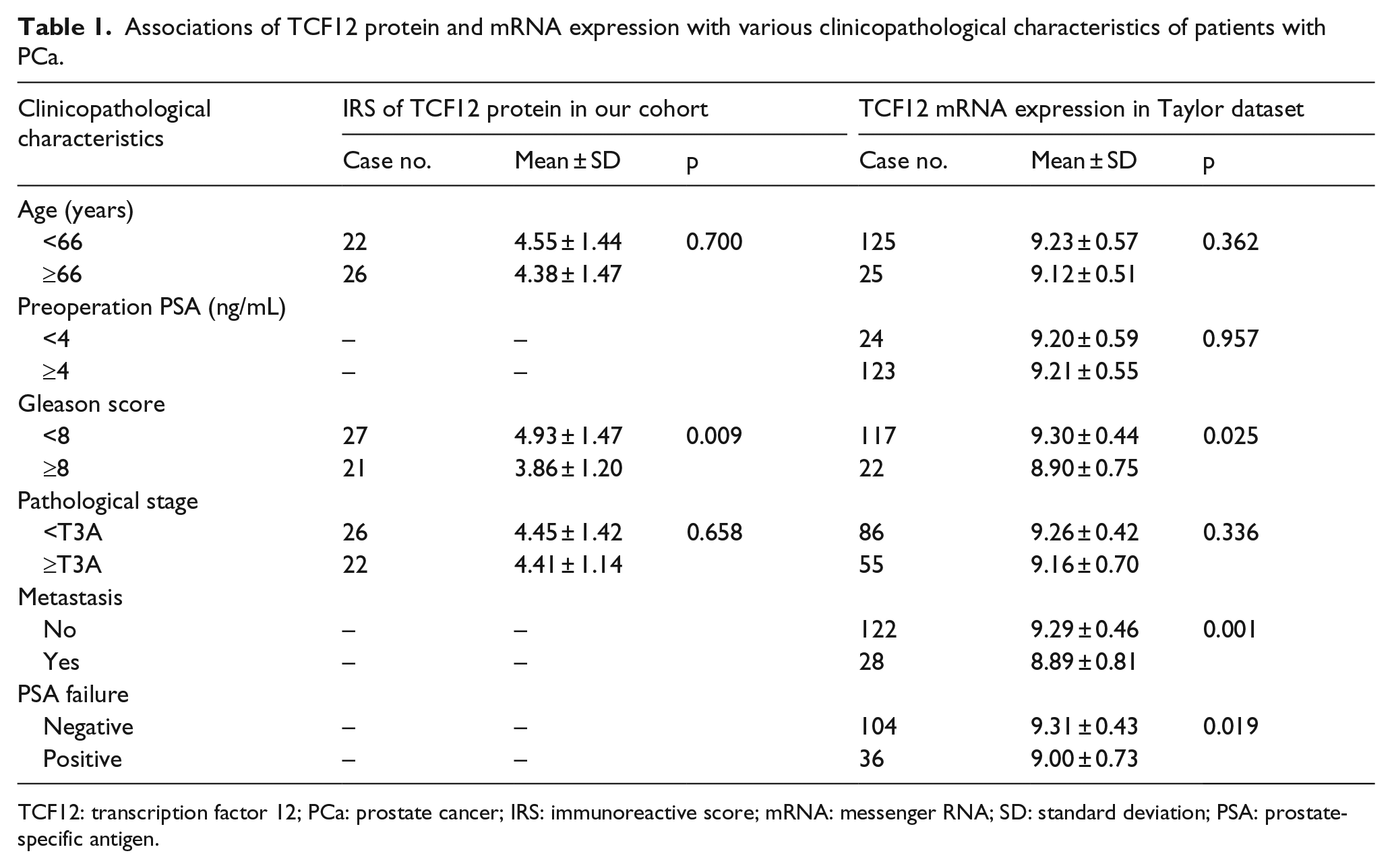
TCF12: transcription factor 12; PCa: prostate cancer; IRS: immunoreactive score; mRNA: messenger RNA; SD: standard deviation; PSA: prostate-specific antigen.
Cell culture and transfection
Two human PCa cell lines (DU145 and LNCaP) were purchased from American Type Culture Collection (ATCC, USA) and cultured in Dulbecco’s Modified Eagle’s Medium (DMEM; cat. no. SH30022.01; HyClone, USA) supplemented with 10% fetal bovine serum (HyClone, USA; cat. no. SH30087.01), 100 U/mL penicillin, and 100 µg/mL streptomycin. DU145 and LNCaP cells were placed in 37°C and 5% CO2 humidity incubator and were subcultured for passage every 3–4 days. The stable cell line of overexpression of TCF12 and control were made by HYYMed Company based on lentivirus with the open reading frames (ORF) sequence of TCF12 (NM_207036.1) and transfected into DU145 and LNCaP cells.
Immunohistochemistry
Subcellular localization and expression levels of TCF12 protein in PCa and benign prostate tissue specimens were examined by immunohistochemistry according to the protocol of our previous reports.15–18 The primary antibody against TCF12 (bs-11237R; Bioss, China) was used at a dilution of 1:200.
Results of immunohistochemistry were scored by two independent experienced pathologists who were blinded to the clinical data. Discrepancy of the scores was resolved through re-examination of the staining to reach to consensus. Five high-power fields were randomly selected to observe cell staining in each section using Leica fluorescence microscope. The density and proportion of the positively stained cells were determined. The immunoreactive score (IRS) was calculated based on the staining intensity and the positive percentage according to the protocol of our previous reports.15–18
Western blot analysis
Expression levels of TCF12 protein in DU145 and LNCaP cells with or without the transfection of TCF12-overexpression plasmid were detected by western blot analysis according to the protocol of our previous reports.15–18 The following antibodies were used: anti-TCF12 (bs-11237R; Bioss) and anti-β-actin antibody (BM0627; Boster, China). The results were visualized with the SuperSignal West PICO Chemiluminescent Detection System (Thermo Fisher Scientific Inc., Rockford, USA).
Cell Counting Kit-8, wound-healing assay, and Transwell assay
Effects of TCF12 in the cell proliferation, migration, and invasion of DU145 and LNCaP cells with or without the transfection of TCF12-overexpression plasmid were detected by Cell Counting Kit-8 (CCK-8) assay according to the protocols of our previous reports.15–18
Generation of the in vivo xenograft model
Effect of TCF12 protein in tumor growth was evaluated using in vivo xenograft model according to the protocols of our previous reports.15–18
Statistical analysis
The version 19.0 SPSS for Windows (SPSS Inc., USA) were used for all statistical analyses in this study. Continuous variables were expressed as mean ± standard deviation (SD). Differences among groups were assessed using Wilcoxon signed-rank test. Associations between TCF12 expression and various clinicopathological characteristics were evaluated by Fisher’s exact test for any 2 × 2 tables and Pearson χ2 test for non-2 × 2 tables. Kaplan–Meier method was used for the survival analysis and Cox regression analysis was used for the univariate and multivariate analyses. Differences were considered statistically significant when the p value was less than 0.05.
Results
Decreased expression of TCF12 protein associates with aggressive clinicopathological characteristics of patients with PCa
Positive immunostaining of TCF12 protein was localized in cytoplasm and/or nucleus of PCa cells (Figure 1). In addition, PCa patients with high Gleason score more often had decreased expression of TCF12 protein than those with low Gleason score (p = 0.009, Figure 1 and Table 1). We also evaluated the associations between TCF12 mRNA expression and various clinicopathological characteristics of PCa patients based on the Taylor dataset. Similarly, the expression levels of TCF12 mRNA in PCa patients with high Gleason score (p = 0.025), positive metastasis (p = 0.001), and PSA failure (p = 0.019) were dramatically lower than those with low Gleason score, negative metastasis, and PSA failure (Table 1).

Decreased expression of TCF12 protein associates with aggressive progression of patients with PCa. (a) Representative photographs of TCF12 protein immunostaining in PCa tissues with Gleason Score: 2 + 3 = 5. (b) Representative photographs of TCF12 protein immunostaining in PCa tissues with Gleason Score: 4 + 5 = 9.
Decreased expression of TCF12 protein predicts BCR of patients with PCa
Next, we determined whether the expression level of TCF12 mRNA correlated with the prognosis of PCa patients after radical prostatectomy. Kaplan–Meier analysis showed that PCa patients with low TCF12 mRNA expression had a significantly shorter BCR-free survival than those with high TCF12 mRNA expression (log-rank test, p = 0.004, Figure 2(a)). However, there was no significant correlation between TCF12 mRNA expression and overall survival of patients with PCa (Figure 2(b)). As a result of univariate analysis shown in Table 2, advanced pathological tumor stage (p = 0.0001), high Gleason score (p < 0.001), and low TCF12 mRNA expression (p = 0.006) were significantly correlated with poor BCR-free survival in patients with PCa (p < 0.05). Further multivariate analysis showed that pathological tumor stage, Gleason score, and TCF12 mRNA expression were all independent prognostic markers for BCR-free survival of PCa patients (Table 2).

Kaplan–Meier curves of (a) biochemical recurrence–free survival and (b) overall survival of patients with PCa based on TCF12 mRNA expression.
Associations of various clinicopathological characteristics with biochemical recurrence–free survival of patients with PCa.

PCa: prostate cancer; PSA: prostate-specific antigen; HR: hazard ratio; CI: confidence interval.
Enforced expression of TCF12 suppresses cell proliferation, invasion, and migration of PCa cells
Since TCF12 was lowly expressed in PCa tissues compared with benign prostate tissues, and its associations with aggressive progression and BCR, we hypothesized that TCF12 might function as a tumor suppressor in this cancer. To validate this hypothesis, we transfected the TCF12-overexpression plasmid into two PCa cell lines—LNCaP and DU145. Western blot analysis showed that the expression levels of TCF12 protein in two cell lines were both significantly increased after the transfection (p < 0.05, Figure 3(a)).

Enforced expression of TCF12 suppresses cell proliferation, invasion, and migration of PCa cells. (a) Expression levels of TCF12 protein in LNCaP and DU145 cells transfected with TCF12-overexpression or control plasmid detected by western blot analysis. (b) Cell viabilities of LNCaP and DU145 cells transfected with TCF12-overexpression or control plasmid detected by CCK-8 assays. (c) Cell invasion abilities of LNCaP and DU145 cells transfected with TCF12-overexpression or control plasmid detected by Transwell assay. (d) Cell migration abilities of LNCaP and DU145 cells transfected with TCF12-overexpression or control plasmid detected by wound-healing assay. The data were from three independent experiments and presented as mean ± SD (*p < 0.05 and **p < 0.01, comparison to the control group).
In addition, CCK-8 assays were performed to evaluate the impact of TCF12 overexpression on proliferation of PCa cells, and the results revealed that the enforced expression of TCF12 dramatically suppressed cell proliferation compared with the negative control in both LNCaP and DU145 cells (p < 0.05, Figure 3(b)). Moreover, we also explored the effect of TCF12 overexpression on PCa cell invasion by Transwell assay. Our data indicated that the invasion ability of TCF12-transfected PCa cells was reduced compared to the control group (p < 0.001, Figure 3(c)).
Wound-healing assay further revealed that the overexpression of TCF12 efficiently inhibited cell migration of two PCa cell lines (p < 0.001, Figure 3(d)).
Enforced expression of TCF12 suppresses tumor growth in vivo
To further determine the roles of TCF12 in tumor growth in vivo, LNCaP cells stably expressing TCF12 or control vector were subcutaneously implanted into nude mice. LNCaP cells stably expressing TCF12 formed significantly smaller tumor nodules (Figure 4(a)) and remarkably slowed down tumor xenografts’ growth compared with the controls (Figure 4(b)).

Enforced expression of TCF12 suppresses tumor growth in vivo. (a) LNCaP cells stably expressing TCF12 or control vector were subcutaneously implanted into nude mice. LNCaP cells stably expressing TCF12 formed significantly smaller tumor nodules. (b) TCF12 overexpression remarkably slowed down tumor xenografts growth compared with the controls. **p < 0.01 compared with control.
Discussion
Improved therapeutics of PCa patients are highly dependent on efficient biomarkers for early diagnosis and prognosis. There is an urgent need to identify new and reliable molecular markers of PCa development and progression. The aberrant expression of TCF12 has been recently reported to be associated with cancer aggressiveness. However, the role of TCF12 in prostate carcinogenesis is unknown. On the basis of immunohistochemistry analysis and the Taylor dataset, we determined the negative correlations of TCF12 protein and mRNA expression with advanced cancer progression and patients’ BCR. Using in vitro and in vivo studies, we provide the first evidence of tumor-suppressive roles of TCF12 in PCa behavior. These findings suggest that TCF12 may serve as a novel biomarker of PCa progression and outcome.
TCF12 is involved into T-cell development and regulation and myogenesis. Its upregulation also functioned in the proliferation of neural stem and progenitor cells. 19 Growing evidence shows the crucial roles of TCF12 in various human cancers. Functionally, TCF12 acts as an oncogene in breast cancer, gallbladder cancer, and colorectal cancer, but as a tumor suppressor in oral squamous cell carcinoma.10–13 Thorsen et al. 20 in 2011 indicated that TCF12 protein expression was upregulated in colorectal adenoma and cancer samples as well as was correlated with progression-free survival. Lee et al. 12 in 2012 demonstrated that TCF12 was tightly correlated with cellular expression of Bmi1 and EZH2 and was co-immunoprecipitable with Bmi1 and EZH2, suggesting that TCF12 transcriptionally suppressed E-cadherin expression via polycomb group-repressive complexes. They also found that TCF12 mRNA overexpression was correlated with E-cadherin mRNA downregulation in colorectal cancer tissues and the occurrence of cancer metastasis. He et al. 10 identified TCF12 as a potential prognostic maker and a molecular target for inhibiting invasion and metastasis in gallbladder cancer and also revealed that HDAC1 promoted migration and invasion binding with TCF12 by promoting epithelial-to-mesenchymal transition progress in this malignancy. Tang et al. 11 found that TCF12 was obviously elevated in cancer-associated fibroblasts, and the upregulation of fibronectin and lysyl oxidase indirectly through TCF12 contributed to extracellular matrix remodeling, triggering the invasion and metastasis of breast cancer cells both in vitro and in vivo. Chen et al. 13 verified TCF12 as a target of miR-211 which may enhance the oncogenicity of carcinogen-induced oral carcinoma by repressing TCF12. In this study, our analysis identifies the decreased expression of TCF12 protein and mRNA as a risk factor for aggressive progression of PCa. The patient group with low TCF12 expression indeed exhibited a poorer BCR-free survival when compared with patients with high TCF12 expression in PCa tissues. However, there was no significant association between TCF12 expression and overall survival of PCa patients based on the Taylor dataset. Since these results were obtained from the existing database, further validation based on a large clinical cohort should be performed. Cancer cell migration and invasion have been indicated to be an initial and essential step for the development of metastasis and controlled by a series of complex cellular and molecular changes. Moreover, we inducted the TCF12-overexpression plasmid into PCa cell lines. The in vitro experiments demonstrated that the enforced expression of TCF12 inhibited PCa cell proliferation, migration, and invasion. Further in vivo experiments also confirmed its suppressive effects on tumor growth in a xenograft mouse model.
In conclusion, our data suggest that TCF12 protein may be a novel molecule which plays a critical role in PCa progression and patients’ prognosis, suggesting it might be a promising therapeutic target for PCa therapy. The molecular mechanisms underlying TCF12 affecting PCa required further investigation.
Footnotes
Acknowledgements
W.-d.Z. and H.-c.H. helped in conception and design of this study; Q.-b.C., Y.-k.L., M.-y.J., Z.-d.H., Y.-x.L., Y.-p.W., and J.Y. participated in the development of methodology; Y.-x.L. and Y.-q.Z. helped in acquisition of data and analyzed and interpreted the data; and Q.-b.C., Y.-k.L., and Y.-q.Z. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from National Natural Science Foundation of China (81571427, 81470983, and 81641102), Science and Technology Project of Guangdong Province (2016A020215018 2013B021800055, and 2014A020212471), and Guangzhou Municipal Science and Technology Project (2014J4100072 and 201607010398).
