Abstract
BACKGROUND:
OBJECTIVE:
The aim of this study is to reveal the underlying mechanism of
METHODS:
Ingredients of
RESULTS:
Eighteen active ingredients and 10 target genes were screened from
CONCLUSION:
The study showed that multi-components in
Introduction
Non-alcoholic fatty liver disease (NAFLD) is represented by excessive fat accumulation in the liver, and it causes non-alcoholic steatohepatitis, cirrhosis and hepatocellular carcinoma [1]. There are 1.8 billion people were attacked by NAFLD worldwide until 2015, and patients with NAFLD face a high risk of cardiovascular disease with a mortality up to 75% [2, 3]. Incidence of NAFLD keeps ever-rising rapidly, which leads to an enormous threat to human health. Unfortunately, there is presently no effective and satisfactory treatment for NAFLD [4]. In decades, traditional Chinese medicine (TCM) treating with major chronic diseases has attracted more and more attention due to its unique efficacy and relative safety [5, 6]. For example, previous studies revealed that Dachaihu decoction had a good effect in attenuating NAFLD [7].
The network pharmacology is built based on the interconnected molecular systems targeting multiple nodes, and it is in line with the mechanism of multiple compounds and multiple targets in TCM. Network pharmacology is now commonly used to reveal the complex mechanism of TCM though cheminformatics and bioinformatics technology [15]. In this study, network pharmacology and molecular docking are used to explore the mechanism of GF in the treatment of NAFLD. The current study is carried out to provide scientific support for the further research of GF and for new drug development in the treatment of NAFLD. The design of this research is shown in Fig. 1.
The workflow of the study to investigate the mechanism of GF in the treatment of NAFLD.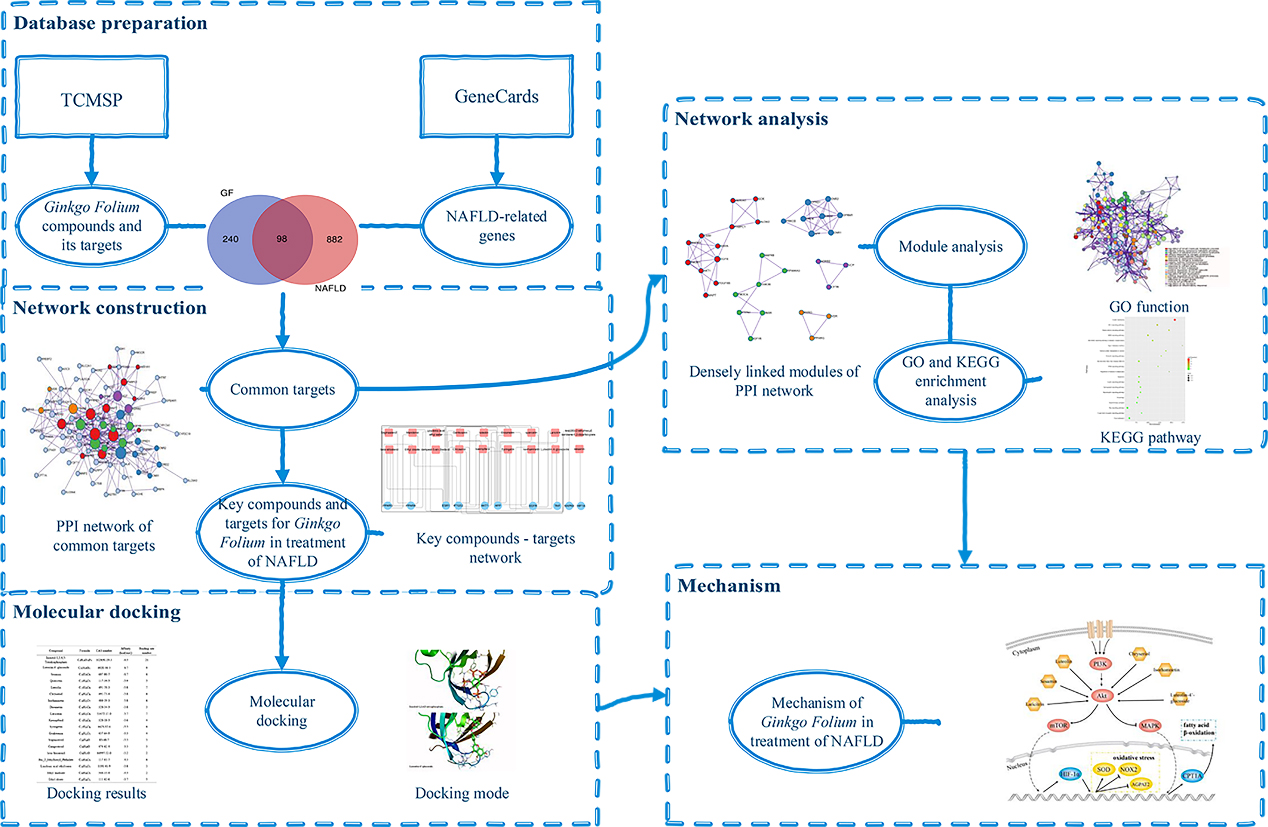
Ingredients and target-genes of GF
Ingredients of GF were collected from Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP) (
Potential genes of GF treating NAFLD
NAFLD-related genes were collected from GeneCards database (
Based on GF and NAFLD associated gene datasets, Venn diagram was constructed to identify common genes of NAFLD and GF.
Network construction and analysis
The common genes of NAFLD and GF were input into the Metascape [19], and protein-protein interaction (PPI) network and module analysis were carried out. A node with a higher degree value indicates that it is more important to the network. We considered genes as key genes if its degree value was greater than twice the median of all genes in PPI network [20]. Finally, key ingredients-genes network of GF treating NAFLD was established by Cytoscape 3.7.0 [21].
To cluster the biological functions of key genes, Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis were run through Metascape.
Molecular docking
The 3D structure of key ingredients and protein were acquired from PubChem database and The Research Collaboratory for Structural Bioinformatics Protein Data Bank (RCSB PDB;
The detail information of active compounds in Ginkgo Folium
The detail information of active compounds in
Identification of active components of GF
In the TCMSP database, Ginkgo Folium was searched, and a total of 307 compounds were found. There were 19 compounds satisfied the condition of OB
Identification of common genes of NAFLD and GF
338 GF-related genes and 980 NAFLD-related genes were retrieved from SwissTargetPrediction and Genecards database, respectively. After mapping Venn diagram based on GF and NAFLD associated gene dataset, 98 overlap genes of NAFLD and GF were extracted (Fig. 2).
Network construction and analysis
98 overlapping genes were inputted into the Metascape for PPI network and MCODE-based module analysis. Because CA1 had no interaction with the other genes and was excluded, 97 nodes and 608 edges were in PPI network, as shown in Fig. 3a. According to the criteria that degree of major hub was more than twice the median of all nodes in PPI network, 10 key target-genes were procured. They were AKT1, TNF, EGFR, PTGS2, MAPK8, PPAR
The detailed information of the target genes for GF in the treatment of NAFLD
The detailed information of the target genes for GF in the treatment of NAFLD
The Venn diagram of the common genes between GF and NAFLD (blue part marks the unique genes of GF, red part marks the unique genes of NAFLD, and the purple part is the common genes).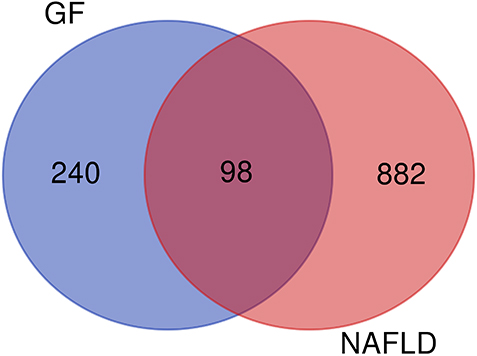
PPI network of potential target-genes of GF in the treatment of NAFLD (a is PPI network, b is densely linked modules of PPI network; line represents the interaction relationship, nodes represent the target-genes; node size is proportional to its degree in the network, and red belongs to the module of response to reactive oxygen species, blue belongs to the module of G protein coupled receptor transport regulation, green belongs to the module of cell response to insulin stimulation, purple belongs to the module of protein modification regulation, orange belongs to the module of nuclear receptor activity).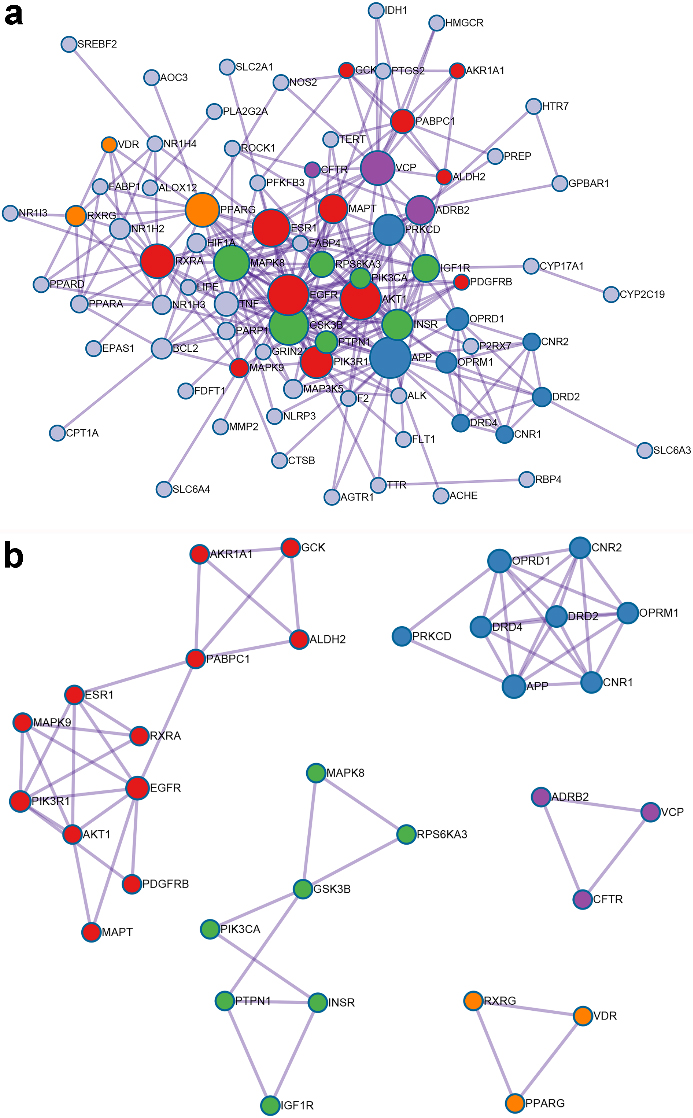
The key bioactive ingredients-genes network for GF in the treatment of NAFLD (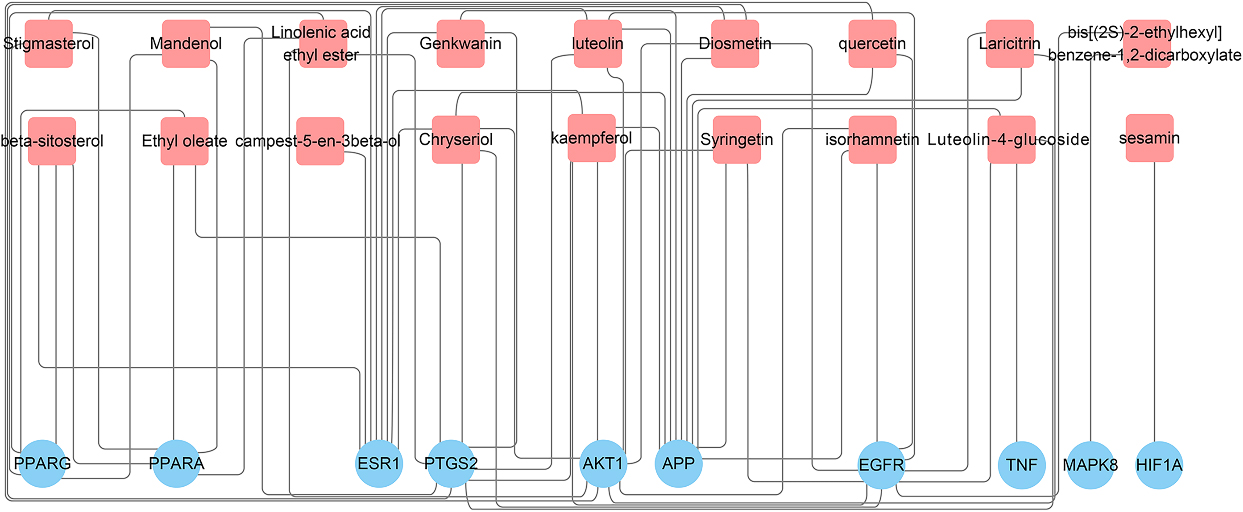
GO and KEGG analysis of common genes for GF in the treatment of NAFLD (nodes in a represent functions of common genes. node size is proportional to its counts, and node color is different with different function; b shows pathways of common genes. node color range from green to red represent -log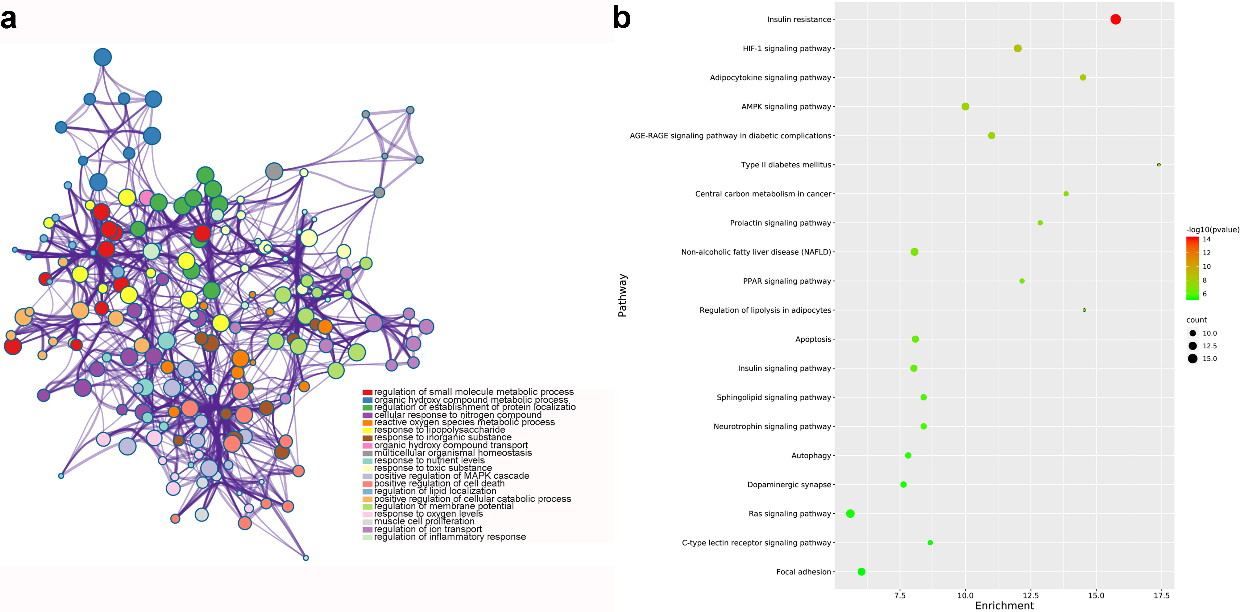
The PPI network was consisted with five densely linked modules, including response to reactive oxygen species, G protein coupled receptor transport regulation, cell response to insulin stimulation, protein modification regulation and nuclear receptor activity (Fig. 3b).
Flavoxanthin did not interact with any genes and was excluded. A total of 18 compounds in GF involved with 10 key target-genes. The data was entered into Cytoscape to construct a key ingredients-genes network, as shown in Fig. 4.
From GO analysis, results suggested that target genes were mainly enriched in regulation of small molecule metabolic process, nuclear receptor activity and RNA polymerase II transcription factor complex. Figure 5a highlighted the top 20 GO terms with smallest
Molecular docking
In order to clarify mechanism of GF in the treatment of NAFLD, molecular docking was used to present interaction between key compound and protein. We selected AKT1 with the highest degree value (PDB ID: 1h10) and 18 compounds of GF for molecular docking. The docking results of 18 compounds of GF were compared with endogenous ligand (inositol-1,3,4,5-Tetrakisphosphate) of AKT1. The affinity value (reversely represented the degree of docking coincidence of molecules) and number of hydrogen bonds are listed in Table 3. Results showed that the affinity values of luteolin-4’-glucoside, sesamin, luteolin, chryseriol, isorhamnetin and laricitrin with AKT1 were lower than
The docking results of 18 key bioactive compounds of GF to AKT1 protein, compared with endogenous ligand (inositol-1,3,4,5-Tetrakisphosphate) of AKT1
The docking results of 18 key bioactive compounds of GF to AKT1 protein, compared with endogenous ligand (inositol-1,3,4,5-Tetrakisphosphate) of AKT1
Docking model of endogenous ligand (inositol-1,3,4,5-tetraphosphate) (a) and luteolin-4’-glucoside (b) with AKT1 protein.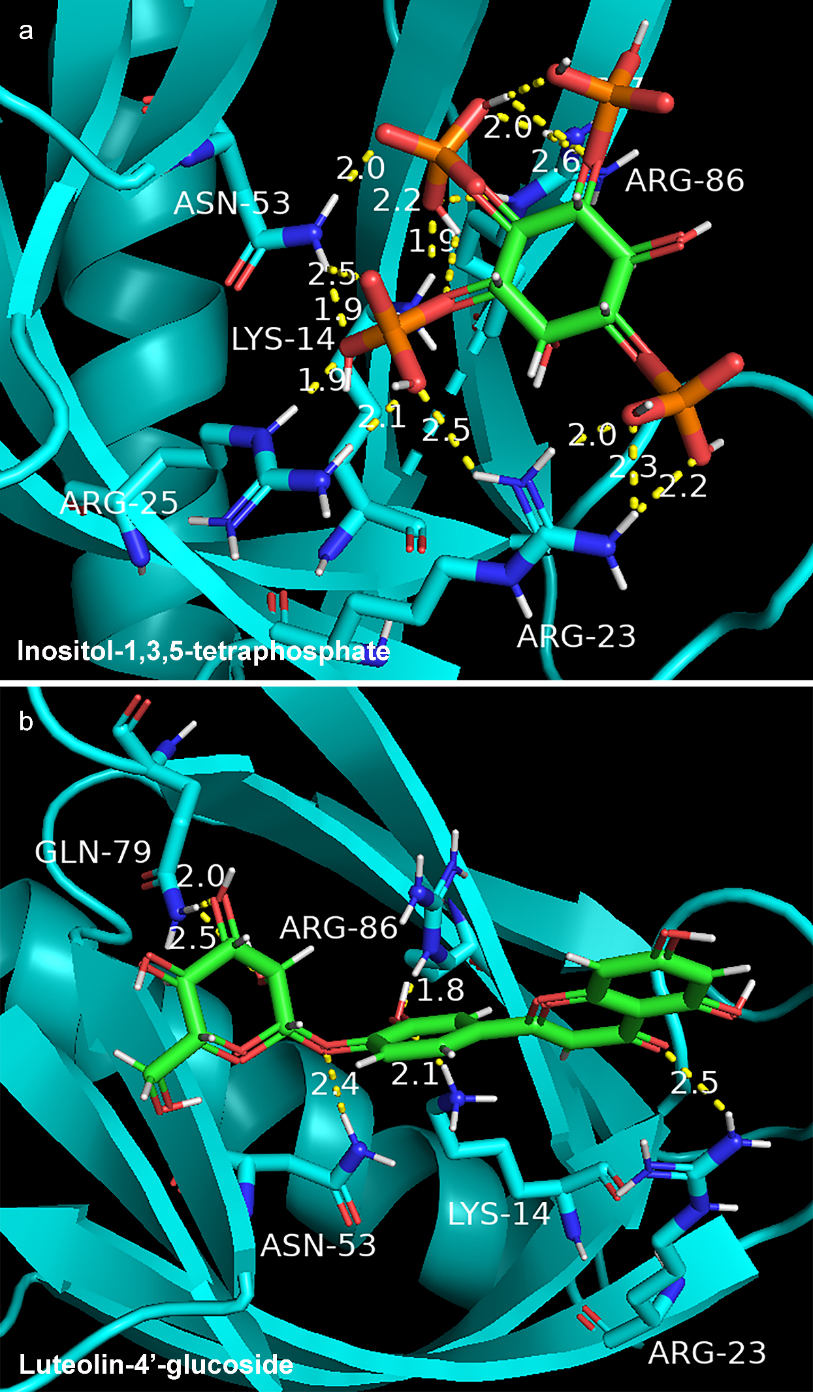
Schematic diagram of the molecular mechanism of GF in the treatment of NAFLD.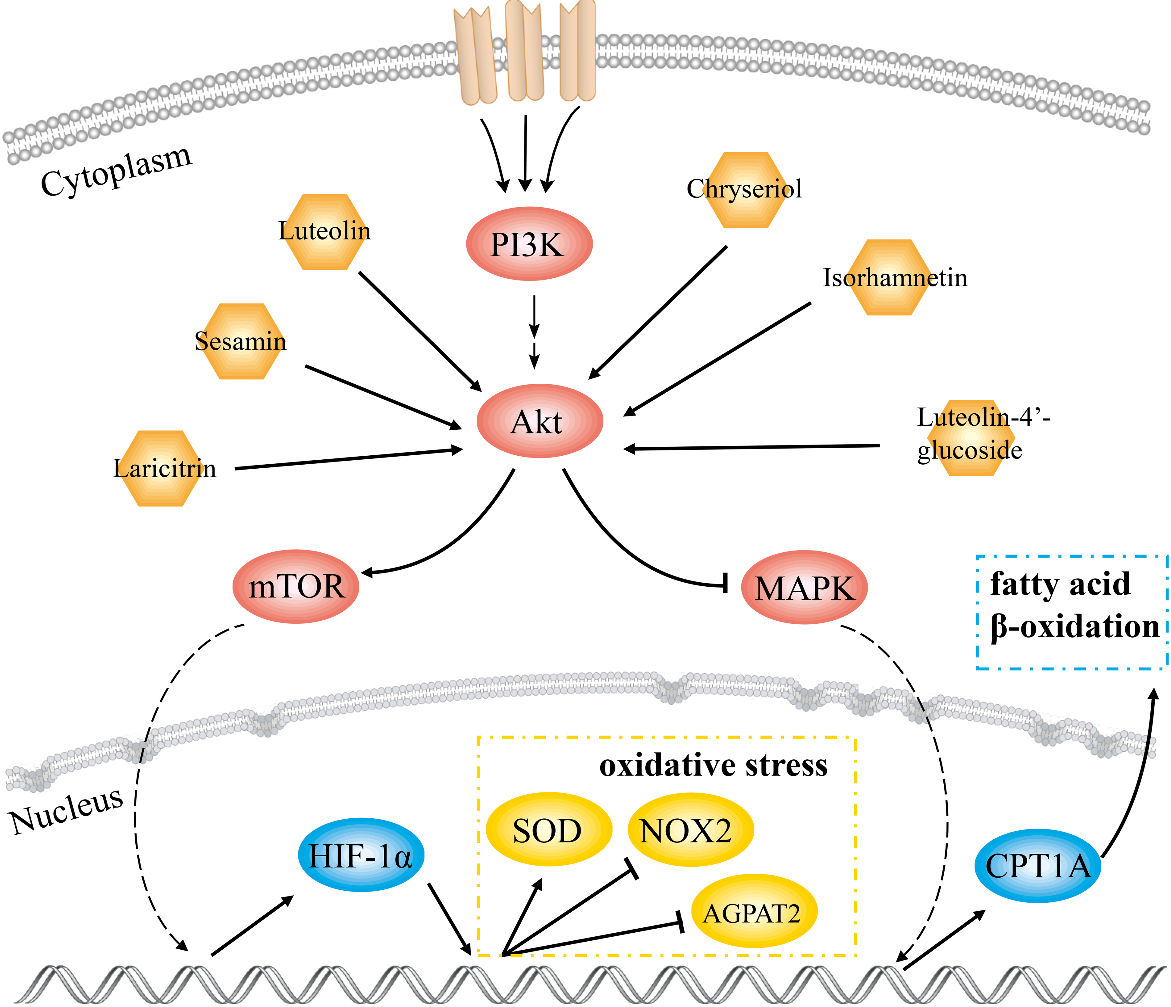
Globally, NAFLD is the most prevalent liver disease, which seriously threatens human health. Mechanisms for NAFLD progression and development are complex and multifactorial [24]. Studies have shown that NAFLD is influenced significantly by insulin resistance [25]. It increases production and secretion of adipokines and inflammatory cytokines [26]. Triglycerides accumulation in the liver causes lipo-toxicity and leads to oxidative stress and overloaded reactive oxygen species [27]. Additionally, altered gut flora causes increased fatty acid absorption and activated inflammatory [28]. Several factors contribute to the pathogenesis of NAFLD, including inflammation, lipo-toxicity and steatosis, as indicated by changes in serum biochemistry and histopathological features [29]. Previous studies indicated that GF has a favorable effect on regulating lipid metabolism [14], yet, its mechanism remains unclear.
In this study, 18 key compounds and 10 target genes of GF treating NAFLD were identified. AKT1 with highest degree value played a pivotal role in the PPI network. Results of GO and KEGG analysis showed that the mechanism of GF against NAFLD was associated with cell apoptosis, HIF-1, adipocytokine and AMPK signaling pathways. Molecular docking results displayed that luteolin-4’-glucoside, sesamin, luteolin, chryseriol, isorhamnetin and laricitrin had strong affinity with AKT1. This work indicated that these compounds were effective substances for GF in treatment of NAFLD.
Luteolin-4’-glucoside has an anti-dyslipidemia effect and inhibits expression of sterol regulatory element-binding protein-1 and
Overactivation of HIF-
Conclusion
The underlying mechanism of GF against NAFLD was investigated by network pharmacology combined with molecular docking. Our findings showed that GF alleviated NAFLD through interactions between effective ingredients and AKT1 and regulation of AKT-AMPK/HIF pathway. The present study revealed the molecular biological mechanism of GF against NAFLD and provided a basis to the clinical treatment of GF.
Footnotes
Acknowledgments
This research was supported by the Horizontal Research Project of Shanghai Jiao Tong University (Nos SA1700111, SA1700118), a project supported by the Department of Science and Technology of Guizhou (Grant no. [2018] 2831), and the Open Research Fund of NMPA Key Laboratory for Rapid Testing Technology of Drugs, Guangdong Institute for Drug Control (Nos KF2022002, KF2022006).
Conflict of interest
None to report.
