Abstract
PURPOSE:
Acute flaccid myelitis (AFM), an uncommon but serious neurologic condition, primarily affects children, and can progress quickly to paralysis and respiratory failure. Data on long-term outcomes of patients with AFM are limited. This study reports on functional status through 12 months for AFM patients who became ill in 2018 in the United States.
METHODS:
Health departments collected information on outcomes at 6 and 12 months after onset of AFM using a standardized form that asked patients or their parents/guardians about functional status. Analyses were restricted to confirmed cases.
RESULTS:
Of the 238 confirmed AFM cases reported to CDC in 2018, 90 (38%) had assessments at 6 months, 82 (34%) at 12 months, and 49 (21%) at both 6 and 12 months. Among the 49 patients with data at both time points, the proportion of patients reporting significant or severe impairment at 6 months ranged from 2% to 59% depending on the outcome. Although proportions decreased by 12 months and ranged from 2% to 51%, most patients had some impairment at 12 months. No deaths were reported.
CONCLUSION:
Six- and 12-month outcomes in patients with onset of AFM in 2018 span a wide range of functionality, particularly of upper and lower extremities. Importantly, improvement appears to occur over time in some patients.
Introduction
Acute flaccid myelitis (AFM) is an uncommon but serious neurologic condition primarily affecting children [1–4]. During the acute phase of illness, patients often present with a respiratory or febrile illness followed by sudden onset of weakness in one or more limbs. AFM can progress quickly to paralysis, with ventilatory support needed in a quarter of patients [5]. Muscle weakness in AFM patients often affects the proximal muscles, such as the muscle groups in the neck, shoulder girdle, and hips. Consequently, proximal muscle weakness can significantly impact activities of daily living and self-care. Data on long-term outcomes of patients with AFM are limited by the small number of participants and only a few studies have described functional changes over time. These studies suggest that although weakness persists, patients continue to improve [6–8].
National surveillance for AFM started after the first large increase in cases in the United States occurred in 2014 [1, 9]. Subsequent national increases in cases occurred during 2016 and 2018, with the largest number reported in 2018 [10]. To better understand outcomes through 12 months among patients with confirmed AFM, those with illness onset in 2018 were contacted about their functional status at two time points after onset of their illness.
Methods
As part of AFM surveillance, health departments reported patients with acute onset of flaccid limb weakness (the clinical criterion for AFM) to the Centers for Disease Control and Prevention (CDC). A standardized form was used to collect demographic and clinical data, and biological specimens were submitted for enterovirus testing. For surveillance purposes, confirmed cases of AFM were defined as patients who met the clinical criterion as verified by CDC. In addition, they had magnetic resonance imaging showing spinal cord lesions largely restricted to gray matter in one or more vertebral segments [11]. Clinical and radiographic information from patients meeting the clinical criterion for AFM were reviewed and classified by a panel of neurologists for surveillance purposes and included in the CDC AFM surveillance data.
Beginning in 2018, surveillance processes for AFM were expanded to collect information on outcomes at 6 and 12 months after onset of limb weakness. Health departments were asked to follow up with patients with confirmed AFM who were included in the CDC AFM surveillance data using a standardized form that asked patients or their parents/guardians to categorize patient status on seven functional domains (overall impairment, physical condition, upper limb functions, lower limb functions, sensory components, excretory functions, and support factors). The form was adapted from a previously validated assessment tool “PULSES” that assessed impairment and disability [12] (Appendix). Each question was rated on a 4-point scale from 1 to 4 (normal or independent to severely disabled or dependent). Assessments were conducted within a range of time after onset of limb weakness; ranges were 5–9 months and 11–15 months for the 6- and 12-month assessments, respectively.
Descriptive analyses were restricted to confirmed cases with onset in 2018 who had available outcomes data at 6 months, 12 months, or both. Outcomes were examined by severity of the acute illness using the following definitions of severity: 1) need for mechanical ventilation; 2) all four limbs affected; and 3) admission to intensive care unit (ICU). Outcomes were also examined by pathogen detected; patients who were enterovirus D68 (EV-D68) positive were compared with patients who were EV-A71 positive or not positive for EV-D68 or EV-A71 because data have suggested that certain enterovirus types lead to more severe presentations of AFM [13]. For the severity and pathogen analysis, each of the outcome questions was collapsed to dichotomous categories of no/mild dependence versus moderate/severe dependence and compared using the chi-square test. A separate descriptive analysis was conducted to examine change in each outcome measure among cases with outcomes data at both the 6- and 12-month time points.
The functional outcomes assessment was not included as part of AFM surveillance until 2018. However, an attempt was made to collect functional outcomes data at a single time point greater than 12 months after onset of illness from AFM cases with onsets before 2018 (onset of illness from 2014 to 2017) and who are part of the CDC AFM surveillance data. Patients were contacted during summer 2021 by their respective health department or by the CDC through a contract research organization funded to assist with this activity. Those who were successfully located and agreed to participate were administered the same standardized form used for the 2018 cohort; descriptive analyses were also performed for these data.
Data were analyzed using SAS software (version 9.4; SAS Institute) and p-values< 0.05, calculated using the chi-square test, were considered statistically significant. Data on outcomes for patients with confirmed AFM and onsets from 2014 through 2018 were collected as part of standardized public health surveillance and determined by the CDC not to be research involving human subjects.
Results
Of the 238 confirmed AFM cases reported to the CDC in 2018, 120 patients from 26 of the 42 states with AFM cases reported responded to at least one assessment; 90 (38%) had assessments at 6 months, 82 (34%) at 12 months, and a total of 49 (21%) had assessment data at both 6 and 12 months. Characteristics of patients during their acute illness by assessment time point are presented in Table 1. Characteristics of non-participants at either 6 or 12 months were not statistically significantly different from participants (data not shown).
Demographic and clinical characteristics of confirmed acute flaccid myelitis cases at time of acute illness (onset of illness in 2018) who participated in functional outcome assessments at six- and/or 12-months*
Demographic and clinical characteristics of confirmed acute flaccid myelitis cases at time of acute illness (onset of illness in 2018) who participated in functional outcome assessments at six- and/or 12-months*
*Data for some characteristics may be missing; the denominator is provided when data are missing. †Percentages may not add up to 100%, as they are rounded to the nearest percent. §One patient who participated in the six-month assessment did not have number of limbs affected indicated. ¶Both upper and lower limbs affected is defined as having three limbs affected.
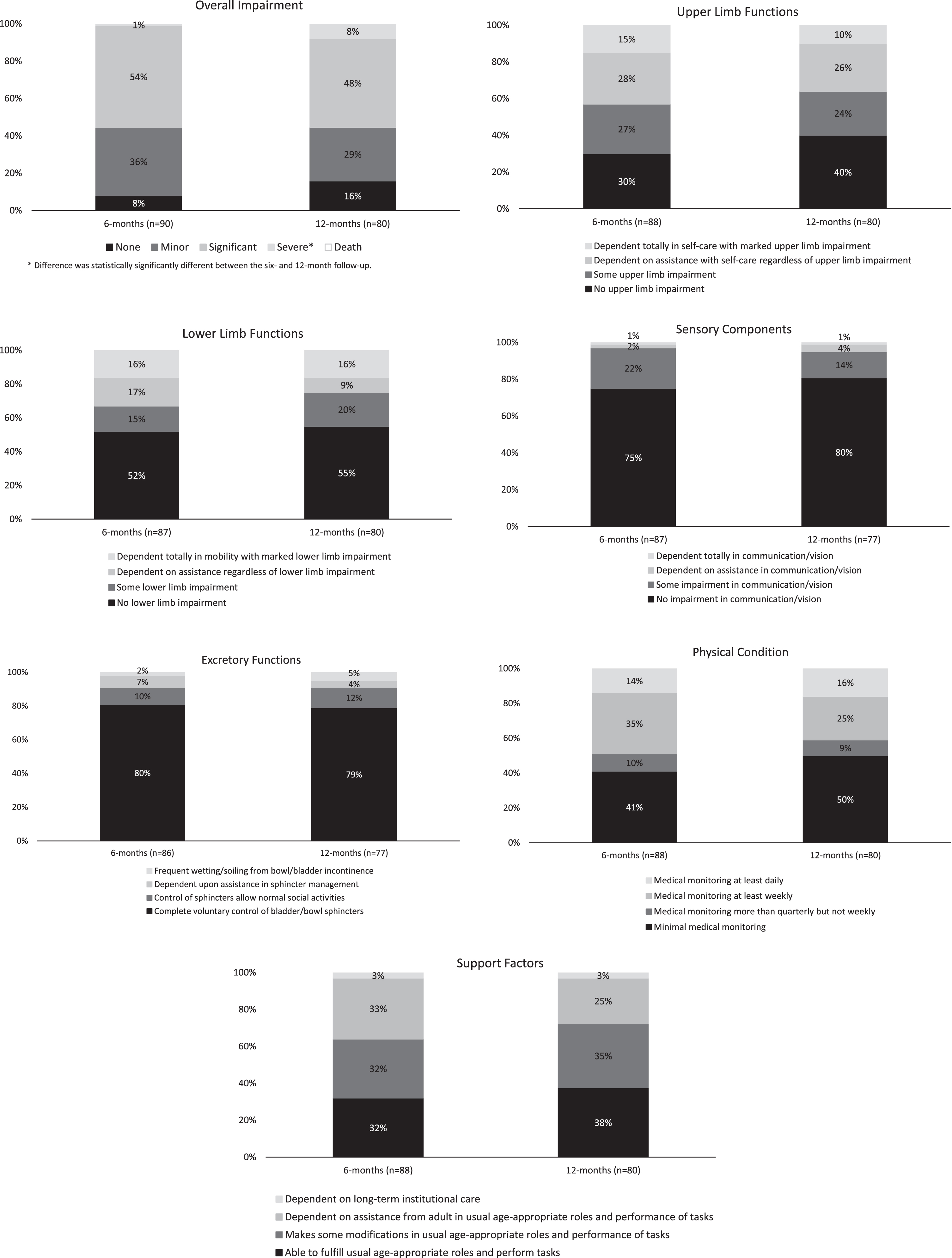
Results of outcomes at the 6- and 12-month assessments are presented in Table 2. At 6 months, one patient (1/90, 1%) responded as having severe overall impairment, defined as involvement of at least 3 extremities and respiratory impairment; most patients (54%, 49/90) responded as having significant overall impairment, defined as major involvement of at least 2 extremities; 36% (32/90) responded as having minor overall impairment, defined as any minor involvement; and 8% (7/90) responded as having no overall impairment. Most patients (70%, 62/88) responded that they had upper limb impairment, including 38 (43%) who were dependent on some assistance for activities of self-care and 24 (27%) who had some impairment but had age-appropriate independence in these activities; 30% (26/88) responded as having no upper limb impairment. Almost half of the patients (42/87, 48%) responded as having lower limb impairment, including 29 (33%) who were dependent on some assistance for mobility and 13 (15%) who responded as having independent mobility with some impairment. Sensory (i.e., ability to communicate through speech, hearing, and vision) and excretory function impairments were less common; 75% (65/87) responded as having independence in communication and vision with no impairment, and 80% (69/86) had complete voluntary control of bladder/bowel sphincters. For physical condition, which describes how frequently the patient requires medical monitoring to assess functioning of cardiovascular, gastrointestinal, urologic, and endocrine, as well as neurologic systems after AFM diagnosis, 41% (36/88) responded as needing minimal medical monitoring, 35% (31/88) as having medical problems that required at least weekly medical attention, and 14% (12/88) as requiring at least daily medical attention. Overall, 32% (28/88) were able to fulfill age-appropriate roles and perform tasks without assistance, 65% (57/88) required at least some adult assistance, and 3% (3/88) reported being dependent on long-term institutional care. No deaths were reported among patients at either the 6- or 12-month assessment period among this cohort.
Number and proportion of confirmed acute flaccid myelitis cases with onset in 2018 with functional outcome measures on seven domains using the adapted “PULSES” assessment tool (Appendix) at the six- and/or 12-month assessment period*, United States
*Some patients may not have responded to all of the questions about specific functional outcomes in the assessment at the time of interview.
For most outcome measures, the proportion of patients responding with no or minimal dependence was slightly higher among participants at 12 months compared with participants at 6 months, but these differences were not statistically significant (Fig. 1). Most patients also continued to have no sensory and excretory function impairment. For the overall impairment and physical condition categories, some patients responded as having more severe impairment or needing more monitoring at 12 months than at 6 months. The difference in severity was only statistically significant for overall impairment. A total of 49 patients had outcomes data available at both 6 and 12 months to examine whether changes in impairment occurred over time at the individual level. The proportion of patients reporting significant or severe impairment at 6 months ranged from 2% to 59% depending on the outcome. Although the proportions decreased slightly by 12 months and ranged from 2% to 51%, most patients still had some impairment at 12 months. Additionally, the number of patients reporting no impairment at 6 months for the different functional outcomes increased or remained the same for most outcomes by 12 months. Of the 29 patients reporting significant overall impairment at 6 months, 4 reported less and 3 reported greater impairment at 12 months (Fig. 2).
Comparison of functional outcome measures among confirmed acute flaccid myelitis cases with onset in 2018 who had data for the six- and/or 12-month assessments, United States.
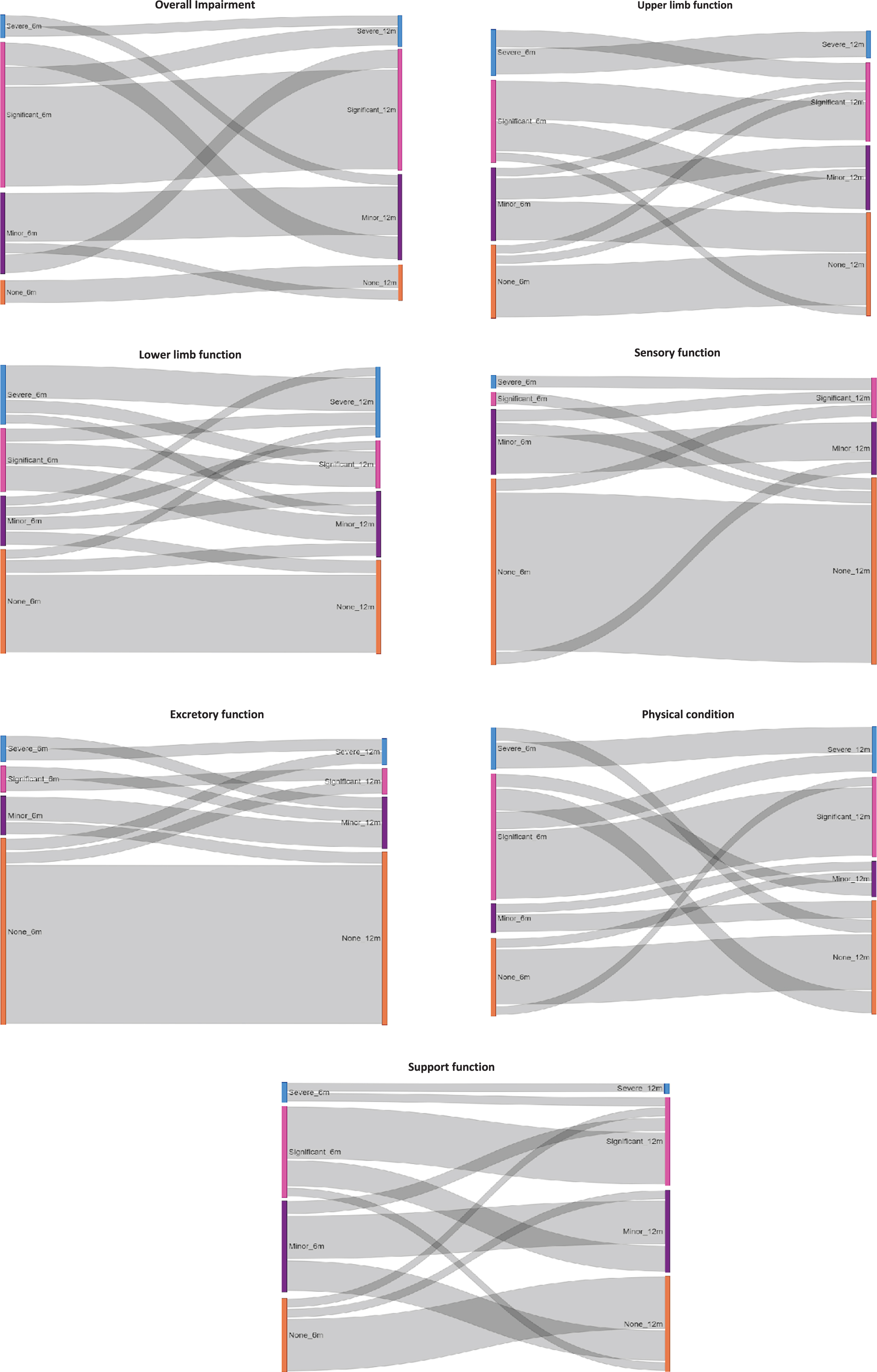
Outcomes of patients with confirmed acute flaccid myelitis with onset in 2018 and follow-up data at both the six- and 12-month assessments to show individual progression over time (N = 49).
Analysis of outcomes by severity of the acute illness differed based on the definition of severity used. For patients requiring mechanical ventilation during acute illness (severity definition 1; n = 26), none of the outcome variables were statistically significantly different from patients not requiring ventilation during acute illness at 6 months. However, at 12 months, patients who required mechanical ventilation during acute illness were more likely to require support (i.e., dependence on assistance in age-appropriate roles and performance of tasks or dependence on long-term institutional care) than patients who were not ventilated during acute illness (p = 0.01). Patients with all four limbs affected during acute illness (severity definition 2; n = 25) were more likely to have moderate to severe lower limb involvement (p = 0.002) and require more support (p = 0.006) at 6 months, but none of the outcome variables were statistically significantly different at 12 months. Patients requiring ICU admission during acute illness (severity definition 3; n = 47) were more likely to have moderate to severe overall (p = 0.02), upper limb (p = 0.02) and lower limb impairment (p = 0.03) and require support (p = 0.01) at 6 months compared with patients not admitted to the ICU. At 12 months, these patients were more likely to require support (p < 0.001). When comparing patients by pathogen, those with a specimen positive for EV-D68 (n = 16) were more likely to have moderate to severe overall (p = 0.001) and upper limb impairment (p = 0.03) and require support (p = 0.01) at 6 months compared with patients who had a specimen positive for EV-A71 (n = 4). Results were similar when comparing patients with an EV-D68 positive specimen to those with a specimen not positive for EV-D68 or EV-A71 (n = 68; data not shown). At 12 months, in addition to having more moderate to severe overall (p < 0.001) and upper limb impairment (p = 0.05) and requiring support (p = 0.04), EV-D68 patients (n = 13) were also more likely to have moderate to severe physical involvement (p = 0.04) compared with patients who had a specimen positive for EV-A71 (n = 7). Compared with patients not positive for EV-D68 or EV-A71 (n = 60), EV-D68 patients (n = 13) were more likely to have sensory impairments (p = 0.02) and require support (p = 0.05) at 12 months.
Of the 333 patients with confirmed AFM and onset during 2014 through 2017 who were part of the CDC AFM surveillance data, 49 (15%) were successfully located and agreed to participate. The median length of follow-up for these patients was 4.6 years (range, 3.3–6.9 years). Similar to results from patients with onset in 2018 at the 12-month assessment (Table 2), most had no sensory (81%) or excretory involvement (65%) and reported fewer severe outcomes for upper (82%) and lower limb function (68%), physical condition (60%), and support factors (81%); however, a greater proportion responded as having severe overall impairment unlike the patients with onset in 2018 (24% vs 8%, respectively). One patient (2%) had died by the time of contact for follow-up; details were not available about the timing or cause of death.
Overall, 38% and 34% of patients with onset of AFM in 2018 provided responses at the 6- or 12-month assessments, respectively, and 21% provided responses at both time points, making this the largest and most representative study of AFM long-term functional outcomes to date. Most patients had minimal involvement of sensory and excretory components at both 6 and 12 months but were more likely to be dependent on help for upper and lower limb and support factors at 6 months; the proportion that was dependent decreased slightly by 12 months. Up to 22% and 35% of patients with assessments at both 6 and 12 months reported at least some impairment of limbs and support functions at 6 months, respectively; by 12 months, the proportion of patients responding with no dependence increased by a range of 0% – 35% depending on the outcome. AFM can be a devastating illness that can result in lifelong consequences. Importantly, mortality associated with the acute and convalescent phases is uncommon.
Reliably measuring and summarizing AFM outcomes in a standardized way is challenging because there is no single tool currently available for this purpose. Several different tools have been used in various studies [6, 7, 14] and all included a clinical evaluation by a physician. The assessment tool used in the surveillance activity in this study included a survey that relied only on self-report via interview, the validity of which can be limited by subjectivity. Although the tool that was used has been validated for functional impairments [15], the development of a standardized tool for evaluating functional outcomes among patients with AFM across institutions will provide more comparable results.
Consistent with other studies examining long-term outcomes among patients with AFM [8, 16, 17], the assessments found that few patients fully recovered, and a majority still reported having significant overall impairment at the 12-month follow-up period. However, when comparing dependence at 6 and 12 months after onset of weakness, some patients were less likely to report complete dependence at 12 months, suggesting possible improvement or adaptation over time. Children may learn to adapt when their physical strength has not improved and that could be interpreted as progress or improvement. Alternatively, because responses were based on self-report, perceptions of improvement or decline at the time of the assessment could have impacted the responses. Varying perceptions may have therefore resulted in reporting that indicated slight improvement or worsening at 12 months compared to 6 months for some of the patients. This may also have been true for the cohort with onset of AFM from 2014–2017 who reported overall impairment as more severe after a longer follow-up period than patients from the 2018 cohort. Overall, patients with AFM followed longer than 12 months have shown continued minor improvements in motor recovery and function most often in a distal to proximal pattern [6–8, 18]. Rehabilitation therapy, especially when provided early but even when provided months to years after onset of illness, has been shown to help lessen long-term consequences of AFM [14, 19] and is a critical component of the clinical management and treatment of patients with AFM.
Although there was no data on initial impairment for comparison, certain indicators of clinical severity in patients with AFM during the acute phase of illness that might be associated with worse outcomes were identified. In the assessment, patients who were admitted to the ICU were more likely to have more severe overall, upper and lower limb impairment and require more support factors at 6 months and continued support at 12 months. Similarly, patients who had all four limbs affected were more likely to have more severe lower limb impairment and require more support factors at 6 months. Patients who were positive for EV-D68 were more likely to have more severe overall and upper limb impairment and require more support factors at 6 and 12 months when compared with patients who were positive for EV-A71. Similar results were found when EV-D68 positive patients were compared with those who were not positive for either EV-D68 or EV-A71 (results not shown), indicating that EV-D68 might be associated with worse outcomes than other enteroviral causes of AFM. Other studies examining outcomes of patients with EV-D68 positive AFM have shown that outcomes are severe, with disability lasting long-term, especially in the most affected muscle groups [7, 8, 13]. Clinical and neuroimaging data suggest that EV-D68, like poliovirus, has an affinity for motor neurons in the spinal cord. Damage, either due to direct neuronal injury or the inflammatory response related to infection, can result in long-term deficits [16, 20]. The findings support previous studies and suggest that severity during the acute illness, along with etiology, can help guide long-term prognosis.
This assessment is subject to several limitations. Only about a third of the total number of patients with confirmed AFM in 2018 participated. The same ones did not always participate in both the 6- and 12-month assessments, which limited the ability to assess longitudinal changes. Those who did respond may not be representative of AFM patients overall, as respondents with more severe outcomes may have been more likely to participate than those with minimal involvement or full recovery. However, when comparing the baseline characteristics of acute illness in participants and non-participants, no differences were found. Additionally, to minimize the burden on families when completing these assessments, the follow-up period was limited to 12 months post onset and details on therapies or treatments received during hospitalization or the follow-up periods were not collected. Thus, determining whether improvements continued to occur after 12 months or exploring possible links between treatments and outcomes was not possible. However, collecting data from an earlier cohort of patients provided additional information on functional outcomes at a time point greater than 12 months after onset of illness. Furthermore, phone interviews made it difficult to assess whether improvements in outcomes were related to muscle strength or functional adaptations to compensate for weakness. The availability of better, standardized follow-up and functional outcome measures will be important for understanding long-term outcomes of AFM.
Understanding outcomes among patients with AFM is important for targeting treatment and management plans to provide optimal care. Based on data from this cohort of patients with AFM with onset during the last peak year in 2018 – the largest long-term follow-up cohort to date – patients reported continued dependence on care, particularly for function of upper and lower extremities, up to 12 months after initial onset of weakness. Importantly, improvement, which may include adaptation to allow more independence in activities in daily living, does appear to occur over time in some patients. Studies have suggested that continued rehabilitation therapy and targeted treatments could result in further improvements. Longer periods of systematic, clinic-based follow-up that includes collection of data on acute and ongoing treatments and services received will be important for examining and understanding factors that influence longer-term outcomes among these patients.
Footnotes
Acknowledgments
We thank the local and state health departments for their work on AFM surveillance and collecting the long-term outcomes data for the AFM cases; the clinical research organization, General Dynamics Information Technology, and McKing Consulting Corporation for interviewing many of the families to collect the data for the longer-term outcomes activity from 2014–2017; Dr. James Sejvar for suggesting the tool used to assess outcomes; and Dr. Teri Schreiner from Children’s Hospital Colorado for her expertise and consultation.
Conflict of interest
The authors have no conflict of interest to report.
Disclaimer
The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention, US Department of Health and Human Services.
Ethical Considerations
This study, as part of standardized public health surveillance, is exempt from Institutional Review Board approval.
Funding
The authors have no funding to report.
Appendix
34. Date of follow-up: __ __/__ __/__ __ __ __ (mm/dd/yyyy)
35. Impairment: □ None □ Minor (any minor involvement) □ Significant (≤2 extremities, major involvement)
□ Severe (≥3 extremities and respiratory involvement) □ Death □ Unknown
35a. Date of death: __ __/__ __/__ __ __ __ (mm/dd/yyyy)
36.
i. Medical problems sufficiently stable that medical or nursing monitoring is not required more often than 3-month intervals
ii. Medical or nurse monitoring is needed more often than 3-month intervals but not each week.
iii. Medical problems are sufficiently unstable as to require medical and/or nursing attention at least weekly.
iv. Medical problems require intensive medical and/or nursing attention at least daily (excluding personal care assistance)
37.
i. Age-appropriate independence in self-care without impairment of upper limbs
ii. Age-appropriate independence in self-care with some impairment of upper limbs
iii. Dependent upon assistance in self-care with or without impairment of upper limbs.
iv. Dependent totally in self-care with marked impairment of upper limbs.
38.
i. Independent in mobility without impairment of lower limbs
ii. Independent of mobility with some impairment of lower limbs, such as needing ambulatory aids, a brace or prosthesis
iii. Dependent upon assistance or supervision in mobility with or without impairment of lower limbs.
iv. Dependent totally in mobility with marked impairment of lower limbs.
39.
i. Age-appropriate independence in communication and vision without impairment
ii. Age-appropriate independence in communication and vision with some impairment such as mild dysarthria, mild aphasia or need for eyeglasses or hearing aid.
iii. Dependent upon assistance, an interpreter, or supervision in communication or vision
iv. Dependent totally in communication or vision
40.
i. Complete voluntary control of bladder and bowel sphincters
ii. Control of sphincters allows normal social activities despite urgency or need for catheter, appliance, suppositories, etc.
iii. Dependent upon assistance in sphincter management
iv. Frequent wetting or soiling from bowel or bladder incontinence
41.
i. Able to fulfil usual age-appropriate roles and perform customary tasks
ii. Must make some modifications in usual age-appropriate roles and performance of customary tasks
iii. Dependent upon assistance, supervision, and encouragement from an adult due to any of the above considerations
iv. Dependent upon long-term institutional care (chronic hospitalization, residential rehabilitation, etc. Excluding time-limited hospitalization for specific evaluation or treatment)
