Abstract
Acute flaccid myelitis (AFM) is an acute condition occurring mostly in children. Although affected patients can show a broad spectrum of symptoms, it is mainly characterized by the progressive onset of focal limb weakness, presenting over a period of hours to a week, with or without pain, and spinal cord gray matter involvement on magnetic resonance imaging (MRI). There is compelling evidence to support the role of viruses, in particular enteroviruses. Studies describing electrodiagnostic (EDX) abnormalities and long-term outcomes in AFM are limited. In this report, the clinical presentation, EDX and MRI findings, and long-term outcomes in three children with AFM who presented with progressive and asymmetric weakness of the limbs are described. MRI showed a long segment cervicothoracic lesion with vertical expansion in all three cases. Patients do recover in time, but many continue to have permanent muscle weakness in the worst affected part(s), and complete recovery is rare. Follow-up of these three cases at 18 months demonstrated a partial recovery in the motor system.
Introduction
Acute flaccid myelitis (AFM) is an important cause of acute flaccid paralysis (AFP) in children and is associated with spinal cord gray matter abnormality. It is usually manifested as a sudden onset of motor dysfunction which may be associated with pain, cranial nerve involvement, and bulbar and respiratory weakness [1–3].
Although AFM is not very common, it is a serious neurologic condition that can be life-threatening for a small number of children, particularly those whose respiratory system is affected [1, 2]. The incidence of AFM was reported to be 1.46 per 100,000 people [4]. There is an association between viral agents and AFM including enterovirus D68 and A71, West Nile virus, wild-type poliovirus, flavivirus, adenoviruses, and other types of enteroviruses. While historically, poliovirus was recognized as the leading cause of AFM, it is uncommon nowadays due to vaccination programs. However, in communities in which vaccination rates are low, this should be considered a cause of AFM. There is a causal relationship between these viral agents and AFM, but it is still unclear whether this association is related to direct viral infection of anterior horn cells or an autoimmune reaction [2–8].
AFM can be difficult to be distinguished from other neurological disorders because it shares many of the same symptoms as other conditions like acute transverse myelitis (ATM), Guillain-Barre syndrome (GBS), acute disseminated encephalomyelitis (ADEM), and spine compressive lesions.
Assessing children to establish a diagnosis is challenging. While children may be able to report the nature of symptoms, they may not be able to report the timing and duration of their problems. They are often not cooperative with neurologic examinations. Additionally, many key signs or symptoms typical of AFM may not be readily evident on initial presentation [2, 3].
Long-term neurological sequelae of AFM require better surveillance and investigations.
This report exhibits the clinical presentations, imaging and EDX findings, and outcomes of three patients who were admitted to the center.
Case report
The clinical characteristics of the three cases in this study are summarized in Table 1. All these three children were under 10 years old (one male and two females) and were initially admitted to the pediatric hospital of Tabriz University of Medical Sciences, Tabriz, Iran. All three cases have completed routine national vaccination programs, and there was no history of trauma, toxin exposure, tick bites, or recent travel. The study was approved by the local institutional review board, and a written informed consent form was obtained from all of the parents.
Clinical characteristics of the cases and results of the paraclinical data
Clinical characteristics of the cases and results of the paraclinical data
Abbreviations: yrs, years; GI, gastrointestinal symptom; MMT, manual muscle test; CSF, Cerebrospinal fluid; WBC, white blood cell; MRI, magnetic resonance imaging; UL, upper limb; LLL, left lower limb.
In this report, the Centers for Disease Control and Prevention (CDC), USA criteria was used to establish AFM diagnosis, including acute flaccid weakness with a spinal cord lesion on MRI restricted to the gray matter in one or more segments of the spinal cord with no identified etiology [1]. The diagnosis further was confirmed by EDX findings.
A 9-year-old female patient reported for the first time to the Children’s Hospital with a symptom of limb weakness. One day before admission, she developed progressive right upper limb (RUL) weakness and neck pain. Three days before the limb weakness, she had a history of mild fever, headache, and nausea.
On physical examination, there was a significant weakness in neck flexion and extension. Further examination revealed grade 1+ and 2+ deep tendon reflexes (DTRs) in upper limbs (UL) and lower limbs (LL), respectively. Elbow flexion/extension grade in manual muscle strength testing was 1/5. Shoulder abduction strength was 1/5 (trace activity), and forearm and intrinsic hand muscles strength in RUL was 3/5 (fair, full range of motion against gravity).
The left upper limb (LUL) exam showed a score of 3 to 4 out of 5 for muscle strength, while the LL were almost normal. The plantar reflex showed a flexor sign on both sides.
No bowel/bladder dysfunction or sensory or cranial nerve involvement was detected. During her stay, her symptoms progressed to the point that she had movement problems and difficulty walking due to severe limb weakness, difficulties with head control, and neck pain.
Case 2
A 5-year-old female patient reported for the first time with limb weakness and back pain. One day before admission, she developed LUL and left lower limb (LLL) weakness. Two days before the limb weakness, she had a history of flu-like symptoms. On admission, there was no sensory or sphincter problem, but the patient had ptosis, miosis, and anhidrosis (Horner’s Syndrome) on the left side.
On physical examination, manual muscle strength testing of distal LUL (wrist and hand) and LLL showed grades of 2/5 and 3/5, respectively. DTRs on the left side were 1+ at the time of the admission which gradually increased in the LLL in the first few days of her stay. Plantar responses were flexor bilaterally. The muscular strength, DTRs, and tone of the right half of the body were normal.
Case 3
A 5-year-old male patient was admitted for the first time with lethargy and neck stiffness. He was hospitalized due to suspected meningitis. Two days before admission, his parents reported a history of fever and nausea. Surprisingly, there was no report of any weakness at the time of admission, but he had a temperature of 38°C with a positive Kernig sign. Serial examinations on day two revealed complete remission of fever, but the patient developed an impaired gag reflex with increased pharyngeal secretions and mild respiratory distress. In further neurological exams, involvement of cranial nerve VII on the right side was spotted with no cognition deficit, but the patient was unable to hold his head in a steady upright position. Although the muscle strength and reflexes were normal in the LL, the UL exam showed reduced DTRs and a muscle strength grade of 2/5 in proximal (shoulder and elbow) and 3/5 in distal (wrist and hand) muscles.
Laboratory findings
Metabolic panel, urine analysis, liver function test, ESR, and complete blood count (CBC) were normal in all three cases.
Serologic tests for human immunodeficiency virus (HIV 1, 2), p24 antigen, Hepatitis B and C panel, and anti neuromyelitis optica (NMO; anti-aquaporin-4) antibody were negative.
Cerebrospinal fluid analysis (CSF) revealed a lymphocytic pleocytosis > 5 WBC/mm3 in two cases (1 & 3). The Oligoclonal band was negative in CSF, and CSF cultures were negative. Stool examination for poliovirus was negative in all cases.
Radiologic findings
Brain and spine MRI was performed for all cases, which showed significantly increased T2 signal intensity at cervicothoracic spinal cord gray matter in all cases. T2W images obtained from the posterior fossa showed brain stem lesions/ pontine tegmentum involvement in case 3 (Fig. 1).
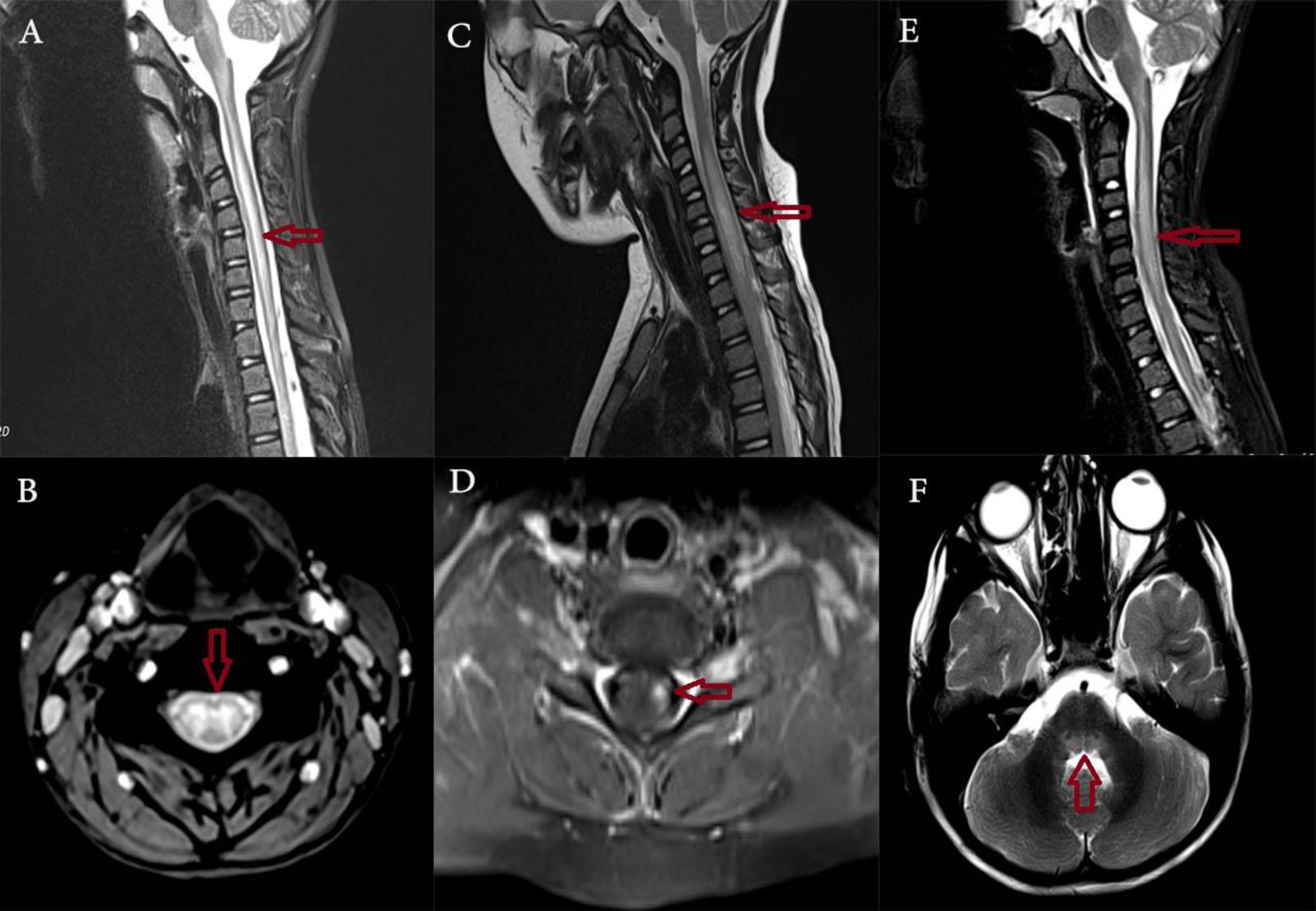
A & B. Sagittal and axial T2W Fat Sat images from a cervical region which shows long segment involvement of central portion of cord. Mentioned changes are ill-defined and involve the entire central gray matter (case 1); C. Sagittal T2W images from cervical region which shows long segment involvement of central portion of cord associated with mild expansion of cord at this area; D. Sagittal T1W Fat Sat images with contrast administration from cervical region which shows asymmetric enhancement in the left anterior horn of spinal cord (case 2); E. Sagittal T2W Fat Sat images from cervical region which shows long segment involvement of central portion of cord. Mentioned changes are ill-defined hyperintensity throughout the central spinal cord gray matter at this area; F. Axial T2W images from posterior fossa which shows brain stem lesions and mentioned changes involve pontine tegmentum (case 3) (shown by arrows).
EDX studies were performed by a pediatric electromyographer using a Medelec Synergy electromyography machine with temperature control. The EDX studies showed normal motor and sensory peripheral nerves conduction velocities (NCV) and amplitudes with unobtainable F waves in upper limbs (in case 2). There was a reduced recruitment pattern and no voluntary motor unit action potential (MUAP) in the proximal part of the upper limb, including biceps, triceps, and deltoid muscles (cases 1, 3) and distal muscles of case 2 including hand intrinsic and wrist extensors in the first week of symptoms onset. Sensory nerve action potentials (SNAPs) were normal.
Treatment
After administrating a course of intravenous immunoglobulin (IVIG) and/or intravenous methylprednisolone, cases 1 & 2 were able to walk during the first few weeks. However, the strength of the upper limbs did not change during this time. The patients were discharged with recommendations for physical therapy and follow-up.
Follow up
EDX evaluation during a 12-month follow-up showed normal sensory nerve conduction studies with absent or reduced Compound Muscle Action Potential (CMAP) amplitude in right axillary and musculocutaneous nerves in cases 1 & 3. There was reduced CMAP amplitude of median and ulnar nerves in case 2. These results are summarized in Table 2. Denervation potentials were also found mainly in proximal upper limb muscles (biceps, triceps, and deltoid) in two cases (1 & 3) and distal upper limb muscles (hand intrinsic muscles and wrist extensors) in case 2. There was no electrodiagnostic evidence of reinnervation in case 1 (Table 3).
Electrodiagnostic findings (NCS) of patients at 12 months follow-up
Electrodiagnostic findings (NCS) of patients at 12 months follow-up
Abbreviations: Amp, Amplitude; Lat, Latency; F.wave.; NCV, Nerve Conduction Velocity; Rt, Right; Lt, Left; CMAP, Compound Muscle Action Potential; DPN, Deep-Peroneal Nerve; SNAP, Sensory Nerve Action Potential; Mus cut, musculocutaneous; NP, Not performed; Abs, absent. *Abnormal parameters are in bold. Nerve conduction parameters were compared with unaffected side or aged-matched normal data reported by Parano et al. [22].
Electrodiagnostic findings (EMG) of patients at 12 months follow-up
Abbreviations: EMG, Electromyography; Rt, Right; Lt, Left; M, Muscle; MUAP, Motor-Unit Action Potential; EDC, Extensor Digitorum Communis; ADM, Abductor Digiti Minimi; FCR, flexor carpi radialis.
During an 18-month follow-up, case 1 had weakness of cervical muscles with severe RUL weakness (0/5 proximally at shoulder and elbow and 3/5 distally at wrist and hand) and scapular winging. In case 3, a moderate RUL weakness (3/5 proximally at shoulder and elbow and 4/5 distally at wrist and hand) with scapular winging was observed. In case 2, the muscle strength of the LUL was 3/5 and 4/5 for distal (wrist and hand) and proximal (shoulder and elbow) muscles, respectively.
AFM is characterized by acute progressive limb weakness which may be accompanied by pain. Weakness develops in a short period from hours to days, with an appearance of more severely involving proximal and upper limbs than distal and lower limbs. AFM usually occurs in healthy children with no previous medical history, but a recent febrile illness with a median of five days preceding onset of neurological symptoms can be found in most cases [2, 3]. Those reported here also followed the classic pattern of disease progression with neck or back pain and asymmetric acute weakness after a period of febrile illness. Asymmetrical weakness mainly was in proximal upper limbs in two cases (1&3), and distal upper and lower limb in case 2. Case 3 was hospitalized due to suspected meningitis, and weakness started while in the hospital. In cases 1&2, the onset of weakness was initially unilateral which progressed during hospitalization. Case 1 symptoms started with an asymmetrical weakness in the cervical area and UL.
Sensory involvement and sphincter dysfunction are not common in AFM. Patients may have autonomic dysfunction, respiratory distress, and need for mechanical ventilation [2, 9]. None of the cases in this report had these symptoms.
Pain is reported in 51–69% of patients and bulbar and cranial nerve involvement is reported in > 20% of patients [3]. In the present report, case 3 had cranial nerve VII involvement and bulbar symptoms. All cases had neck or back pain.
In practice, AFM should be considered for children presenting with acute neurological symptoms. A combination of clinical and imaging findings, repeated electrodiagnosis, close monitoring of the disease course, and response to treatment can also be helpful in this regard.
Clinical clues and EDX findings can help to differentiate AFM from other causes of AFP such as ATM and AIDP. Patients with AIDP have slowed NCVs reflecting segmental demyelination. ATM is characterized by paraplegia, sensory level (defined as lowest spinal cord level that still has normal sensation), sphincter dysfunction, and weakness which generally begins in the LL. Furthermore, MRI findings reveal white and gray matter involvement of the spinal cord. Oligoclonal bands can be identified in the CSF, nerve conduction study is usually normal, and somatosensory and motor evoked potentials are abnormal [2, 10].
In the absence of sensory and sphincter problems, the presentation of weakness, pain, and hyporeflexia can make it difficult to distinguish AFM from other acute neurological disorders, like GBS. This challenge was encountered in case number two where loss of DTRs and an unobtainable F wave response initially suggested GBS.
The diagnosis of AFM can be established based on the clinical picture, with acute flaccid limb weakness and MRI findings of spinal cord lesion in one or more segments is mainly restricted to gray matter [1].
The recommended work-up in patients with suspected AFM includes blood tests, stool and respiratory samples, CSF analysis, MRI of the brain and entire spinal cord, and EDX [2, 3].
CSF analysis reveals pleocytosis (> 5 WBC / mm3) in most cases with a mild increase in protein levels in half of the patients [2]. Viral agents can be rarely detected in the CSF [2, 11].
Brain and spinal cord MRI is important in the diagnosis of AFM and is often needed to differentiate it from other conditions. The involvement of more than one segment of the spinal cord is the most common finding of AFM in children. A T2 hyperintense signal in the central cord is known as a classic MRI finding and is the hallmark of AFM [8]. Diffuse spinal cord edema is usually seen at the initial stages along with the consequent T2 hyperintensity at anterior horns. Brainstem lesions are reported in 11–75% of cases [2, 12], and brain MRI may reveal hyperintensity in the dorsal pons and caudate nucleus. AFM diagnosis is definite when characteristic MRI findings and CSF pleocytosis are present [8]. However, MRI findings are not identified as prognostic factors for AFM [12, 13]. In this study, all three cases had long segment cervicothoracic lesions with vertical expansion restricted to the gray matter as well as negative IgG for neuromyelitis optica. Case 2 showed an atypical presentation (hemiparesis and no CSF pleocytosis) with characteristic MRI findings (Fig. 1). Based on the diagnostic criteria reported by Murphy et al. [8], AFM is probable when MRI findings are characteristic with no CSF pleocytosis. However, electrophysiologic findings and clinical course support AFM diagnosis in this patient.
Electrophysiologic tests can have a role in the AFM diagnosis in regions where MRI is not available [8]. EDX findings in AFM are compatible with anterior horn cell involvement (commonly observed in the C5 to C7 levels) and include decreased CMAP amplitude, normal motor conduction velocity, and normal SNAPs; unobtainable F waves are often observed as well. Needle examination shows a neurogenic pattern and denervation potentials. During the first week, EDX may be normal except for showing the reduced recruitment pattern and possible unobtainable F waves in the affected segments of limbs [8].
All three cases reported here had normal SNAPs, low amplitude CMAPs, denervation potentials, and neurogenic MUAPs in cervical roots-related muscles during the follow-up period. Changes in the electromyography of these patients have persisted even after several months, suggesting a pronounced anterior horn cell involvement (Tables 2 & 3).
AFM is an acute condition with a high potential for residual deficit. Complete recovery in AFM cases is rare; less than 10% of patients show complete recovery and most have residual deficits and oftentimes prolonged need for ventilation support, but cranial nerve recovery is a recognized feature [2, 14]. Aubart et al. reported 10 cases and followed them for one to three years. Incomplete recovery after 12 months was reported in eight cases. Motor recovery after four weeks of disease onset was introduced as a prognostic factor in their study [15]. None of the three cases in this study achieved complete motor recovery after an 18-month follow-up.
In a retrospective study by Patel et al., 15 patients were included; nerve transfer surgery was performed in six cases that had poor recovery. Active movements improved after surgery in four patients [16]. It is well understood that in those who will not finally reinnervate, surgery should be performed for the best outcome. Identifying patients who will not spontaneously recover is challenging, but any with muscle that has not reinnervated should be considered for nerve transfer surgery [17].
Electrodiagnostic findings at presentation and follow-up may be helpful for the prediction of prognosis and for management. Severe denervation potentials and neurogenic changes in the weeks after AFM onset are possible predictors of poor outcomes and long-term sequelae. Muscle groups with severe weakness at the neurological nadir are less likely to recover [2, 18]. Saltzman et al. reported the outcome of two AFM cases, where patients with profound weakness and denervation without any recovery on EMG were selected for nerve transfer. They demonstrated successful reinnervation and functional recovery in postoperative follow-up [17].
Although prognostic factors are not well defined, it seems that severe weakness at nadir and denervation potentials can predict poor outcomes in these cases. There is a correlation between EV-D68 and poor outcomes [2, 3]. In the present report, case one, who had a severe weakness (MMT 0/5) and severe denervation potentials on EMG, had very poor motor recovery. The two other cases with MMT 2/5 at neurological nadir had partial recovery during 18 months.
Currently, there is no effective and curative treatment for AFM, and it is difficult to distinguish whether the initial partial improvement is the result of early intervention or the natural course of the disease. IVIG, steroids, and plasmapheresis have been tried in the treatment of patients with this disease [2, 3]. Due to the beneficial effects of IVIG in animal studies, IVIG has been recommended in the acute phase of AFM combined with respiratory support, pain control, and physical therapy [19]. Steroids may increase viral loads and anterior horn cell damage; however, their anti-inflammatory effects may reduce spinal cord edema [20]. In addition, fluoxetine may inhibit viral replication, but there is no improvement in the function of those patients treated with this medicine [21]. The cases in this report received IVIG and steroids (as shown in Table 1).
Conclusion
AFM, as a challenging condition, is a devastating disease in children, which may have long-term sequelae and disability. Rapidly progressive asymmetric weakness, mainly in upper limbs (proximal > distal), with or without pain in a child with a recent febrile illness is the typical presentation of AFM. Severe motor impairment, denervation potentials, and neurogenic changes on EMG may also be associated with poor motor outcomes.
Further studies are needed to introduce optimal treatment and elucidate the best predictors of long-term outcomes in AFM.
Footnotes
Acknowledgments
The authors have no acknowledgments.
Author contributions
VT conceptualized and designed the report; performed electrodiagnostic studies; supervised data collection and revised the manuscript; and contributed to the submission process of the manuscript. MB was the main clinical manager of the patients and coordinated in data collection and reviewed the manuscript. SRK, AS, FJ and SK contributed to data collection and helped in drafting the manuscript. AJ reported MRI. All authors read and approved the final manuscript.
Conflict of interest
The author reports no conflicts of interest in this work.
Funding
There was no funding source for this study.
