Abstract
PURPOSE:
Patients with neurogenic bladder (NB) often perform clean intermittent catheterization (CIC) and are predisposed to bladder colonization. Antibiotics are not routinely indicated in those with asymptomatic bacteriuria (ASB). The original purpose of this study was to compare patients that received antibiotics for ASB and those that did not. However, because the non-antibiotic group was very small, the final analysis evaluated treatment patterns of ASB in children with NB.
METHODS:
A retrospective chart review was completed, including patients who presented with urinary tract infection (UTI) and NB managed by CIC. Patients with symptoms of UTI were excluded. Basic demographics, urinalysis, culture results, and antibiotic prescriptions were collected.
RESULTS:
The sample included 272 patient encounters for 109 unique patients. Of these, 50.7% were female, and the median age was 10.25 years. More than half the urine cultures (56.2%) grew gram-negative organisms, and 31.3% contained 2 or more organisms. Nearly all encounters received treatment with antibiotics. Twenty-three encounters with no culture performed or the culture resulted in no growth received antibiotic therapy.
CONCLUSIONS:
Antibiotic resistance and antibiotic stewardship are primary concerns in healthcare today. This organization’s current practice pattern shows high antibiotic use for ASB in patients with NB. Future studies are required to identify outcomes associated with treatment versus non-treatment in these patients.
Keywords
Introduction
Neurogenic bladder (NB) or Neuropathic bladder is a dysfunctional bladder that results in inefficient and ineffective emptying due to impaired nerve signals to the bladder. A number of neurologic disorders can cause NB, including, but not limited to, spinal cord injury, Parkinson’s, Multiple Sclerosis, and Spina Bifida [1, 2]. Management of NB often involves clean intermittent catheterization (CIC) in order to adequately empty the bladder and achieve continence.
Patients with NB managed by CIC are predisposed to colonization which is the presence of bacteria in the bladder that does not cause a host response; therefore, there is no immune or clinical reaction [3]. Poor emptying and daily instrumentation of the bladder results in bacterial colonization of the lower urinary tract in 50-60% of patients with NB [2]. The presence of bacteria in the urine without signs and symptoms of infection is called asymptomatic bacteriuria (ASB) [4]. Colonization causes interpretation of various tests for urinary tract infections to be difficult because it reports bacteria in urine similar to that in the setting of a urinary tract infection (UTI) [5, 6]. Patients participating in CIC programs are more likely to develop ASB and are at increased risk of developing UTIs [5, 7]. For clinicians, distinguishing ASB from a true urinary tract infection can often be difficult, given that urine-based laboratory investigations can yield identical results in either condition [1, 4].
Patients performing CIC are considered to have a UTI when symptoms of fever greater than 101, hematuria, change in continence, or pain are present [8]. The standard treatment for symptomatic UTI is antibiotic therapy for patients not on CIC programs. Many of the same patients with NB have recurrent UTIs and will likely receive multiple courses of antibiotic therapy [5]. Treating colonization in the setting of ASB with antibiotics has been controversial for the past 2 decades due to the significant consequences to the patient, healthcare system, and community antibiotic resistance rates [7, 9]. Antibiotics are considered a resource that can lose effectiveness over time, with overuse leading to resistance and the subsequent emergence of multi-drug resistant organisms. Those organisms pose a serious health threat of sepsis and even death [7].
The objectives of this study were to evaluate the practice pattern in treating asymptomatic bacteriuria in children with neurogenic bladder and identify outcomes associated with different treatment methods.
Methods
This study was a single-center retrospective chart review of patient encounters between 1/1/2015 and 4/30/2018. Institutional Review Board approval was obtained (IRB #00000300). The electronic medical records for patients with a past medical history diagnosis code for spina bifida (Q05.9) and/or neurogenic bladder (N31.9), and who received a diagnosis code of UTI (N39.0) for the encounter visit. Patients who had been evaluated in the emergency department, urgent care, rehabilitation, and urology clinics were included. Patient encounters from other clinics or inpatient units were excluded.
Patients were excluded from the study if they were already on antibiotic treatment at the time of encounter, not on a CIC regimen, or if they presented with symptoms of UTI. These symptoms included fever greater than 38 degrees Celsius, gross hematuria, change in continence pattern, or pain. This definition of UTI was based on the Centers of Disease Control (CDC) National Healthcare Safety Network (NHSN) definition of UTI from 2018, and the 2009 and 2019 Infectious Diseases Society of America (IDSA) guidelines [4].
Demographic, urinalysis, and urine culture data, along with treatment variables were extracted. Demographic data included age at the time of the encounter, sex, and race/ethnicity. Circumcision status in boys was not routinely documented and was not considered in this analysis. Patients’ urinalyses and culture data included detection of nitrites, leukocyte esterase, white blood cell (WBC) count, red blood cell (RBC) count, along with microorganism identification, and concentration of colony forming units (CFUs). In cultures with 2 organism strains, the predominant organism was considered for the CFU data. Cultures were defined as mixed when 2 different microorganism strains were present. Definitions of skin flora, gram positive, gram negative, and yeast were based on microorganism speciation. The type of antibiotic(s) prescribed for treatment was recorded. Data analysis was performed using IBM® SPSS® v23.
Results
A total of 272 encounters from 109 unique patients met all inclusion criteria. Nearly half in the sample were seen one time (n = 53, 48.6%). The median number of encounters per patient was 2 (range 1 to 10). The median age at the time of each encounter was 10 years (range 4 months to 21 years). Table 1 summarizes the patient level demographic information.
Sample characteristics
Sample characteristics
Urinalysis and microscopy were available for 263 encounters. The complete results are displayed in Table 2. More than half (53.5%) had urine positive for nitrites, and 87.3% had more than trace leukocyte esterase present in their urine. In addition, 69.4% had 16 or more WBCs per high-powered field (hpf). Eighty-eight (33%) contained > 100 WBC/hpf, and 2 were reported as too numerous to count (TNTC). For RBCs, 41.8% had 5 or more RBCs/hpf, and 44% had 1-4 RBCs/hpf. Additionally, just under 40% of encounters were noted to have single organism growth with > 100 CFU, along with > 15 WBC/hpf.
Urinalysis and urine microscopy results
Of the 272 patient encounters included in this study, no culture was obtained in 11 encounters, and the culture results were negative in another 12 encounters. Figure 1 depicts the distribution of cultures for all 272 encounters. Of the remaining 249 cultures that showed organism growth, more than half (56.2%) grew gram negative organisms. Nearly one-third (31.3%) of the cultures contained 2 or 3 different organisms. Gram-negative and gram-positive organisms accounted for 68% of all positive cultures. Table 3 displays the distribution of organism sub-types with the gram-negative and gram-positive culture groups.
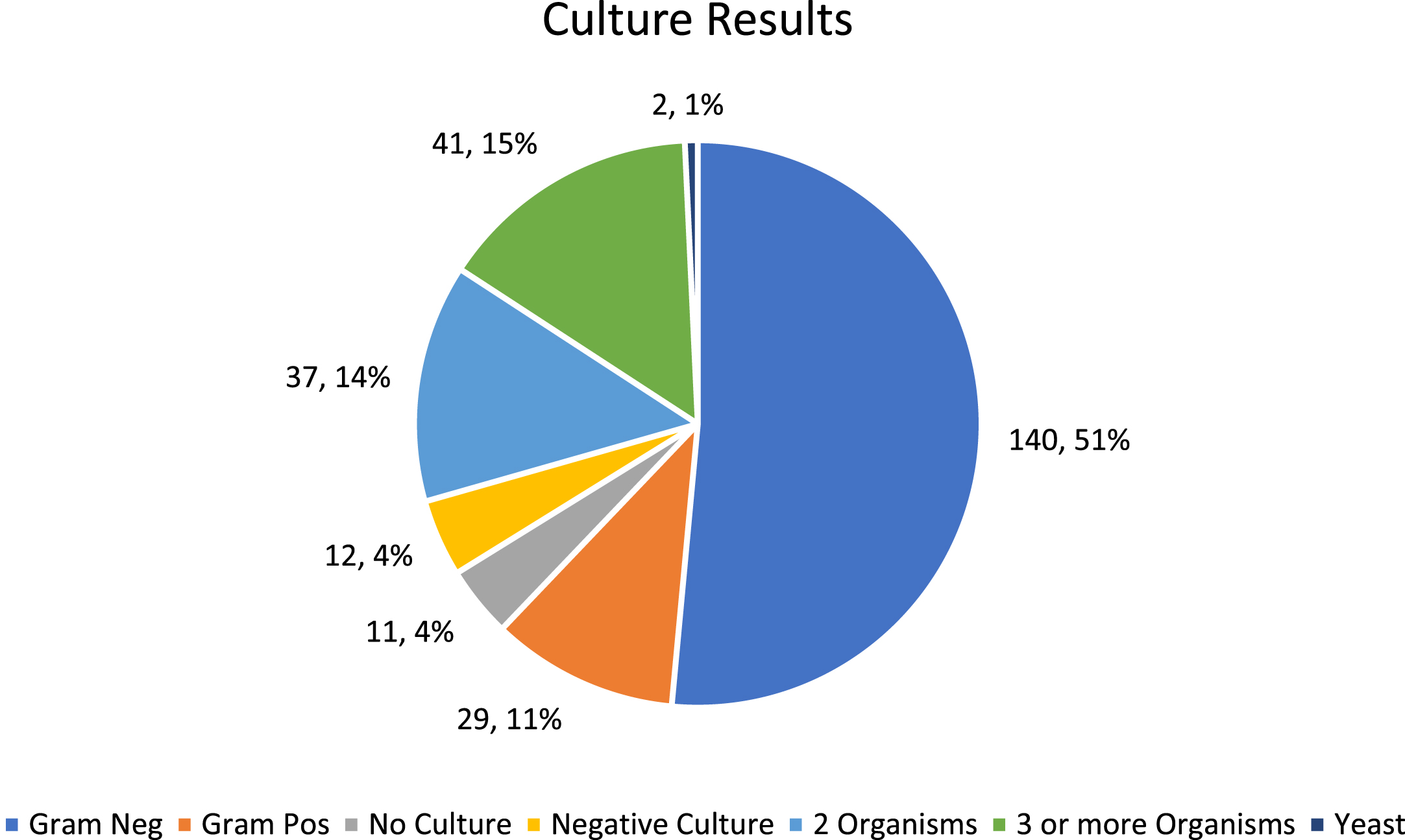
Distribution of cultures for all patient encounters (n = 272).
Gram-negative and Gram-positive Sub-types (n = 169)
Antibiotics were prescribed in all but 4 of the 272 encounters, and in 13 encounters, more than one antibiotic was prescribed. Of the 23 encounters where either no culture was obtained, or the culture resulted in no growth, patients received antibiotic therapy. In two of those encounters, the patient received 2 antibiotics. Table 4 outlines the distribution of drug classes prescribed across the 272 encounters.
Antimicrobial regimens (n = 272)
Clean intermittent catheterization is the leading treatment for NB and patients with spina bifida [10]. Pediatric patients with NB, managed with CIC, are frequently found to have bladder colonization of bacteria resulting in asymptomatic bacteriuria. Given the difficulty in interpreting routine urinalyses and urine cultures, there is a risk of unwarranted treatment with antibiotics that can lead to significant morbidity for the patient [7]. Antibiotics can also result in various adverse-drug events, yielding approximately 70,000 pediatric emergency department visits annually in the United States, with children≤2 years being at a four-fold risk [11].
In 2019, the IDSA provided new guidelines to specifically address ASB to help guide clinicians’ management of it in patients with NB. The IDSA’s guideline recommends against treating bacteriuria in the absence of UTI symptoms and acknowledges that signs and symptoms of a UTI in the NB population may differ from patients without NB [4]. This guideline does not specify signs and symptoms of UTI, but other studies have provided recommendations on symptoms to be considered indicative of UTI in this patient population [2, 12]. These studies included symptoms of suprapubic pain/tenderness, new onset of flank pain/costovertebral-angle tenderness, new or worse incontinence, or— in the absence of another identifiable cause: altered mental status, fever, rigors, malaise, lethargy. The most common symptoms of UTI, including dysuria, frequency, and urgency, are rarely present in a patient with NB, thus, additional symptoms to consider are increased spasticity, autonomic dysreflexia, or a sense of unease. Pyuria is not diagnostic of ASB or UTI and malodorous or cloudy urine are not symptoms that distinguish UTI from ASB [13].
Given the difficulty of interpretation of various tests differentiating between UTI and ASB, practice patterns of antibiotic stewardship and the symptoms used to distinguish ASB from UTI likely vary among clinicians. A survey-based research study from 2018 provides evidence for variability in these practice patterns by showing significant heterogeneity between different specialties when defining which symptoms distinguish a UTI from ASB [14]. The study aimed to determine the treatment pattern of ASB in patients with NB who are on CIC. Despite the latest IDSA guidelines and publications, which further emphasize the existing recommendation against antibiotic treatment of ASB in this population, these findings demonstrate that antibiotic treatment of ASB continues. In fact, this study was originally developed with the notion that two naturally occurring ASB groups would develop: those that received antibiotics and those that did not. Had that been the case, group outcomes would have been investigated as intended in the study aim. However, 98% of the encounters of ASB were managed with antibiotics, and therefore group comparisons were impossible.
Madden-Fuentes and colleagues recommended standardizing the definition of UTI in Spina Bifida patients for the purposes of comparing research findings. They suggested that a urine culture with greater than 100 CFU/ml of a single organism and greater than 10 WBC/HPF via urine microscopy be used as a uniform definition [15]. These laboratory results would lead to a diagnosis of UTI in the presence of at least 2 symptoms such as fever, pain, or change in continence. In this study, 39.7% of the encounters demonstrated the lab parameters but did not meet the definition of UTI because additional symptoms were not present.
The results of this investigation highlighted the need for robust antibiotic stewardship programs and to change the prescribing culture for ASB at this organization. Antibiotic stewardship should be emphasized to decrease inappropriate antibiotic use, thereby decreasing rates of antibiotic resistance and additionally reduce antibiotic adverse events. In this population of young patients with many years ahead of them and likely numerous UTIs in their future, the early development of antibiotic resistance could be detrimental. It is in the patients’ best interest to avoid antibiotic exposure outside of true clinical infection by promoting proper antibiotic stewardship. Providing care for the pediatric NB population is complex, and existing literature suggests that antibiotic treatment for ASB can be due to multiple factors such as the complexity of care, the inability of urine-based laboratory data to distinguish ASB from UTI, and perceived patient/family expectations [16]. Irrespective of the underlying motivations, the data highlight the scope of ASB overtreatment, which likely leads to increased morbidity for these patients and rising antibiotic resistance rates. Over a period of 40 months, patients received 268 courses of antibiotic therapy out of 272 encounters. It is possible that antibiotic treatment was prescribed more frequently since this study only included encounters in the emergency department, urgent care, along with urology, and rehabilitation clinics. It excluded encounters from other clinics or inpatient units where these patients are also commonly treated.
A prominent limitation of this study is the retrospective nature and reliance on the data that were captured at the time of the encounter. There was limited insight into the clinical decision-making process regarding what prompted urinalysis and urine culture orders and why antibiotic treatment was started by the treating clinician. Another limitation is that the number of encounters that may have detected ASB but did not diagnose it as a UTI is unknown. In addition, because this research used a single ICD-9 code for Spina Bifida to identify subjects, future work should expand inclusion to other types of patients with neurogenic bladder to broaden the understanding of treatment patterns in diverse patients with neurogenic bladder. Finally, the data were collected from one central United States pediatric hospital. Prescribing patterns may differ widely in other regions and organizations, so these results may not necessarily be generalized to other healthcare institutions.
Conclusion
In this study, 98% of NB patients on CIC with ASB received antibiotic treatment despite recommendations against it. The practice of overtreatment can increase morbidity and healthcare costs for this patient population and is not in alignment with current guidelines. Attention to this difficult clinical scenario, along with the development of clear guidelines and multidisciplinary collaboration, are needed to provide high-quality care for these complex patients.
Footnotes
Acknowledgments
This study was internally funded by Children’s Mercy-Kansas City.
Conflict of interest
The authors have no conflicts of interest to declare related to this work.
