Abstract
PURPOSE:
Report urologic outcomes among newborns with spinal dysraphism managed within an expectant clean intermittent catheterization (CIC) program.
METHODS:
Newborns were followed clinically and with serial ultrasound (US). Urodynamics (UD) and dimercaptosuccinic acid (DMSA) renal scan were obtained at 3–6 months, 1 year, 3 years, then as needed. Patients with initial evaluation after 6 months were excluded.
RESULTS:
Median follow-up was 3.2 years. 11/102 began catheterization for continence (median
4.0 years) and 47/102 did not start CIC. Of these, 2/58 developed a DMSA abnormality.
44/102 began CIC early, often for elevated storage pressures and febrile urinary tract
infection (UTI). Of these, 20/44 developed a DMSA abnormality including 9 who had
abnormality detected prior to starting CIC. Being on CIC or starting immediately upon
recognition of new hydronephrosis, reflux, elevated filling pressures, or febrile UTI
was associated with lower chance of DMSA abnormalities (4/17, 24%) compared to delaying
CIC (16/27, 60%) (
CONCLUSIONS:
CIC can be deferred until continence in select infants with a low risk of significant DMSA abnormality. However, immediate initiation of CIC upon recognition of risk factors is recommended as this was associated with fewer DMSA abnormalities than delaying CIC. Recommendations for expectantly-managed patients include close follow-up, serial US and UD, and prompt initiation of CIC upon recognition of new hydronephrosis, reflux, elevated storage pressures, or febrile UTIs.
Keywords
Introduction
Patients with spinal dysraphism (SD) merit urologic care from birth to minimize renal functional loss and prevent chronic kidney disease (CKD) later in life [1, 2]. Some urologists favor proactive clean intermittent catheterization (CIC) in a majority of infants [3, 4, 5]. Others manage expectantly and intervene selectively based on clinical, radiographic, and/or urodynamic (UD) risk assessment [6, 7, 8].
The optimal management approach remains unclear [9, 10]. Proactive CIC in infants has been associated with low rates of upper urinary tract deterioration [3, 4, 11], infrequent renal functional loss [3, 12, 13], and reduced rates of augmentation cystoplasty [14, 15]. Other studies suggest that proactive CIC might prevent anatomic [16] and functional [17] bladder deterioration. However, CIC adds significantly to care burden [18], and proactive intervention may result in treatment of many patients whose kidneys are not jeopardized [19]. Recent studies suggest that CIC may not protect against urinary tract infection (UTI) [20], or prevent renal parenchymal loss [21]. In 2016, a Centers for Disease Control (CDC) working group issued a protocol which aims to standardize newborn management and allow more rigorous outcomes analysis moving forward [9].
The purpose of the current article is to report outcomes among newborns with SD managed with expectant CIC. Some patients began CIC “early,” based on problems with UTI, or in response to radiographic or UD findings. Others began later if the family sought to achieve continence for the child. It is hypothesized that (1) many children can be managed without CIC until efforts are made to achieve continence, and that if properly selected, (2) these patients will also have a low risk of renal parenchymal injury or functional loss on technetium-99m dimercaptosuccinic acid (DMSA) renal scan. It is also suspected that (3) should new hydronephrosis (HN), vesicoureteral reflux (VUR), elevated storage pressures, or interim febrile UTI be recognized during follow-up, patients who begin CIC immediately will be less likely to develop DMSA scan abnormalities compared to those in whom intervention is delayed.
Methods
Following institutional review board approval, infant SD patients who had initial evaluation at our center between January 2009 and October 2016 were identified via institutional registry. Retrospective review of the electronic medical record was performed. Only patients who underwent initial evaluation prior to six months of age were included. Newborn patients were managed initially without CIC. The clinical follow-up plan included a renal ultrasound (US) every 3 months in year 1, every 6 months in year 2, then annually. UD evaluation and DMSA scan were obtained at 3–6 months, 1 year, 3 years, then as needed. Informed families participated in escalation-of-care decisions.
Demographic, clinical, radiographic and UD data were extracted from the
electronic health record, and organized by clinical encounter in a REDCap (research
electronic data capture system) [22] to create a
longitudinal dataset. HN grading was done based on the Society for Fetal Urology (SFU)
system [23]. Early CIC is defined as initiation
of catheterization prior to efforts to achieve urinary continence with CIC. Febrile UTI is
defined as a febrile illness (temperature over 38.5 degrees Celsius) with positive urine
culture (greater than 50,000 colony forming units of pathogenic bacteria) from a
catheterized specimen. Elevated storage pressure is defined as end fill pressure (EFP)
Results are described using total proportion with outcome and survival curve analysis with cumulative incidence. Proportions are compared by Fisher’s exact test. Log rank test and cox proportional hazard analysis is used to compare longitudinal data between groups.
Characteristics of cohort
Characteristics of cohort
One hundred and two patients were followed for a median duration of 3.2 years (interquartile range [IQR] 1.9–5.0 yrs). Patient characteristics are shown in Table 1. Spina bifida was the most common dysraphism (91%) and lumbar lesions (75%) were most common overall. The majority of the spina bifida and lipomengocele patients (78/96, 81%) had a ventriculoperitoneal shunt. Forty-nine patients were female.
CIC outcomes
Early CIC was started in 44/102 patients at a median age of 1.2 years (IQR
0.5–3.0 years) and characteristics by group is shown in Table 1. The only significant difference between those started on
early CIC and those not started on early CIC was number on CIC since birth
(
Reasons for starting CIC
Reasons for starting CIC
Note: many patients had more than one reason.
58/102 patients did not start early CIC. In 11/58, CIC was started for the singular purpose of continence at a median age of 4.0 years (range 3.4–4.8 years). The remaining 47 patients did not start CIC within 2.2 year median follow-up (IQR 1.2–3.6 years).
DMSA abnormalities were found in 22/102 patients (22%) at a median age of
1.0 years (IQR 0.6–2.0 years). Abnormalities were detected on initial DMSA scan in 8
patients, 2
Clinical progress in patients who may have benefitted from earlier CIC.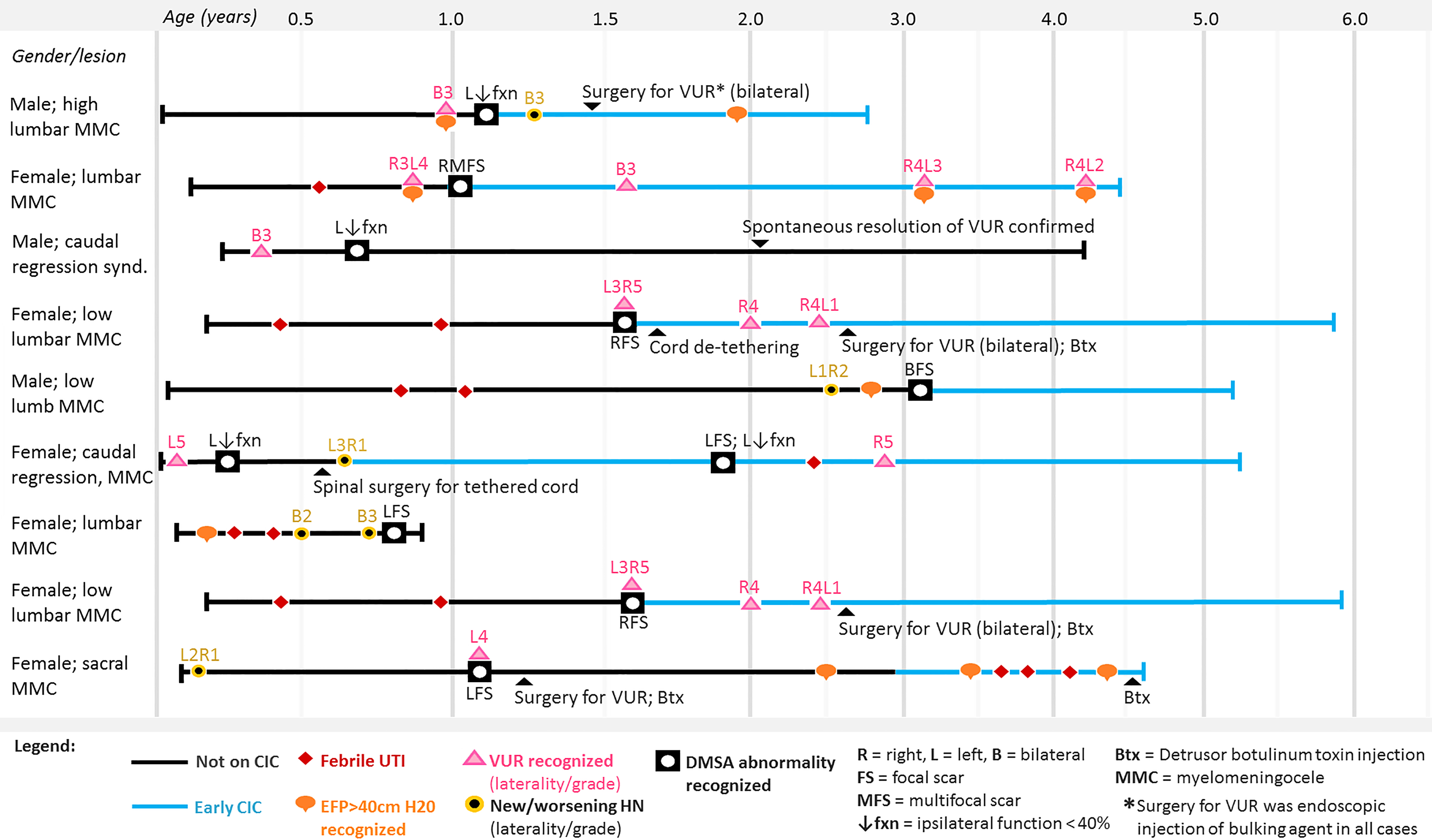
Among 44 patients in whom early CIC was initiated, 20 (45%) were found to have a DMSA abnormality. In 9/20, the DMSA abnormality was recognized prior to initiation of early CIC (Fig. 1) at a median age of 8.9 months (range 1.5–37.9 months). In all 9 of these patients, at least one of the following had been documented prior to abnormal scan: VUR (4/9), febrile UTI (4/9), and/or elevated storage pressure (4/9). In 11/20, DMSA abnormality was recognized after initiation of early CIC.
Among patients who (a) either had yet to start CIC at study completion (N
All 44 patients in whom early CIC was initiated were recognized to have
one or more of the following: new HN, VUR, elevated storage pressures, or interim febrile
UTI. Seventeen of these 44 patients were either already catheterizing or began immediately
upon initial recognition of one or more of the above findings at a median age of 1.0 years
(IQR 0.3–1.4 years). 27/44 patients did not start CIC immediately when one or more of
these findings was recognized. In these patients, a median delay of 1.2 years (IQR 0.3–2.6
years) was observed from recognition said findings to CIC start at a median age of 2.3
years (IQR 0.6–3.2). Four of the 17 patients (24%) who began CIC prior to or promptly upon
initial recognition of new HN, VUR, elevated storage pressure or interim febrile UTI, were
found to have a DMSA abnormality compared to 16/27 patients (60%) in whom CIC initiation
was delayed (
Total proportion and estimated cumulative incidence of outcomes of interest
Total proportion and estimated cumulative incidence of outcomes of interest
UTI
Table 3 lists total proportion and estimated cumulative incidences of outcomes of interest (febrile UTI, elevated storage pressures, DMSA abnormality, early CIC). The failure curves are shown in Fig. 2. An estimated 29% (95% CI: 19–43%) of patients will develop a DMSA abnormality by 5 years of age and 49% (95% CI: 38–61%) of patients will start early CIC.
Failure curves for outcomes of interest.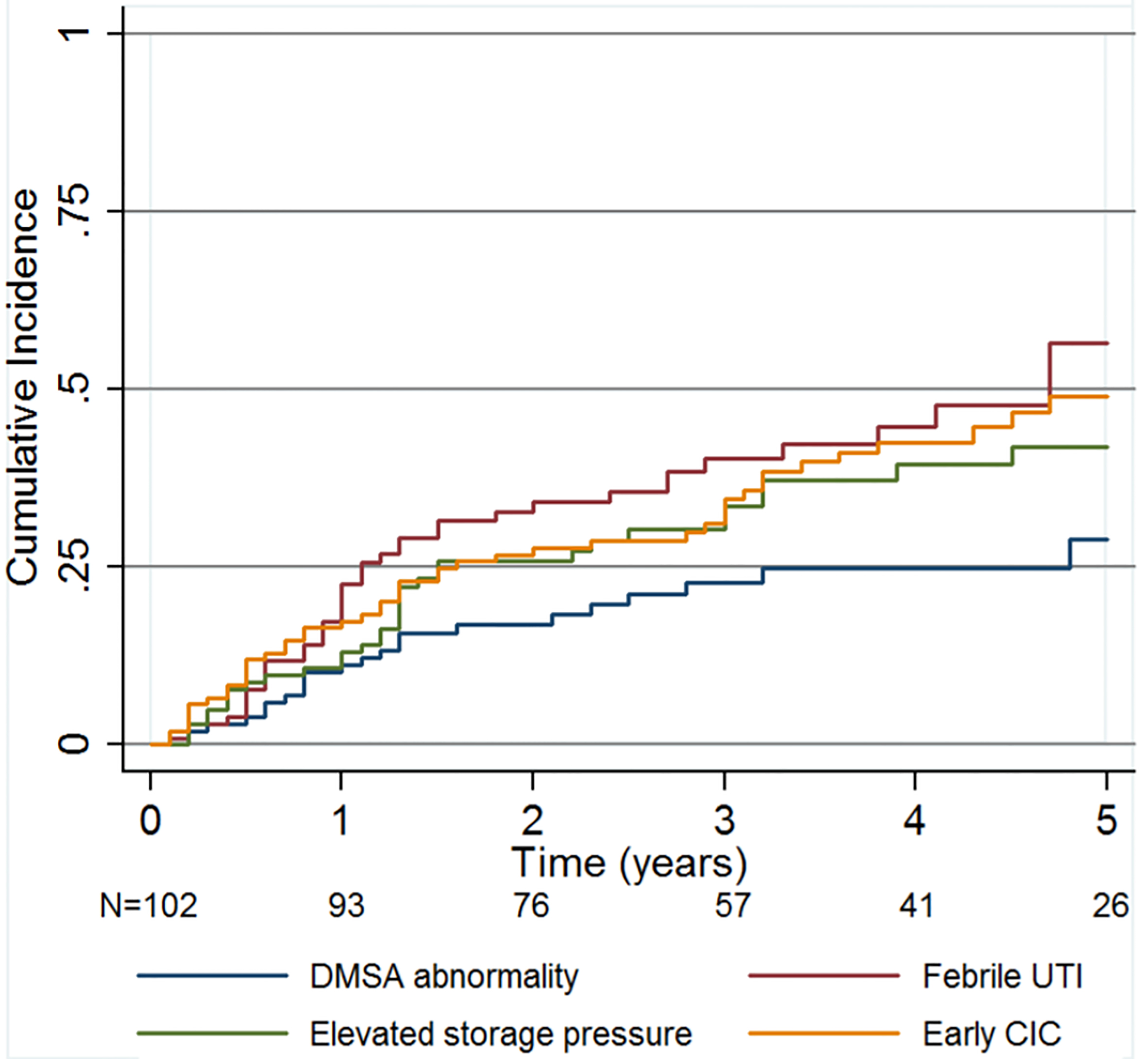
This study reports outcomes among children with SD who were managed within an expectant CIC program. It was hypothesized that in select patients, CIC might be deferred until efforts to achieve continence with low risk of DMSA abnormality. Indeed, it was observed that around 60% of children were managed without early CIC, and only two of these patients had an abnormal DMSA (one had a pelvic kidney with no focal defect and the other expired from a non-urologic cause before CIC was initiated). Therefore, these findings are congruent with previous studies demonstrating safety of expectant management in select newborns [6, 7, 8], and stratification of low-risk patients appears to have been accurate within this program. These findings are important because CIC is the only medical factor associated with parenting stress in mothers of children with spina bifida [18].
Early CIC was instituted in a minority of patients (43%) due to identification of risk factors. As expected, these children had a significantly-higher rate of abnormal DMSA findings (45%) which is also congruent with previous work [7]. In 9 patients – all with previously-recognized VUR, febrile UTI or elevated storage pressures – a DMSA abnormality was recognized before early CIC was initiated. Therefore, about 10% of patients in this cohort may have potentially benefitted from earlier initiation of CIC, which may have prevented DMSA abnormalities in some.
Some patients in this cohort began CIC immediately upon recognition of new HN,
VUR, elevated storage pressures, or interim febrile UTI during follow-up. In others,
intervention was delayed (likely secondary to provider practice variation, family
preferences, etc.), allowing serendipitous comparison. Already being on CIC or beginning
immediately upon recognition of new HN, VUR, elevated storage pressures, or interim febrile
UTI was associated with lower chance of DMSA abnormalities (4/17, 24%) compared to delaying
initiation of CIC (16/27, 60%) (
Renal outcomes
Direct comparison of renal outcomes to the existing literature is
difficult due to variability among protocols and inconsistency with respect to how renal
outcome measures are defined [10]. Woo et al.,
in a cohort of 83 expectantly-managed patients (N
Urodynamic findings
Detrusor storage pressure over 40 cm H
Limitations
This study is limited by retrospective nature and lack of a control group.
While the schedule for follow-up and diagnostic evaluation outlined in the methods section
was generally followed, not all patients obtained imaging at the planned time. Our
description of renal outcomes included only DMSA results, a study with inherent
limitations including technical variability and subjective interpretation. Difficulty in
distinguishing congenital dysplasia and transient pyelonephritic lesions may have
contributed to overestimation of the incidence of acquired renal injury in this series.
However, there were no patients who had an abnormal DMSA who then had a subsequent normal
DMSA. While there were no patients with abnormal serum Cr values in this cohort, more
precise measures of glomerular filtration rate were not routinely performed. Another
limitation was that CIC was not started in all patients who had an indication, as can be
gleaned from the text. Additionally, a discrete cutoff for elevated filling pressures
(
Conclusions
In a select group of newborn patients with SD, CIC can be deferred until efforts to achieve continence with a low risk of developing a significant DMSA abnormality. Conversely, identifying and protecting patients at high risk for renal injury is important but remains challenging. Many severely-affected patients developed new DMSA abnormalities and/or underwent vesicostomy despite CIC. Upon recognition of new HN, VUR, elevated storage pressures, or febrile UTIs, immediate compared to delayed initiation of CIC may decrease chance of subsequent DMSA abnormality. About 10% of patients in this program could have potentially benefitted had CIC been started immediately upon recognition of these factors. Recommendations for expectantly-managed patients include close clinical follow-up, serial renal US and UD evaluation, and prompt initiation of CIC upon recognition of new HN, VUR, elevated storage pressures or febrile UTI.
Footnotes
Conflict of interest
The authors have no conflict of interest to report.
Abbreviationss
SD – spinal dysraphism
CKD – chronic kidney disease
CIC – clean intermittent catheterization
UD – urodynamic
HN – hydronephrosis
US – ultrasound
VUR – vesicoureteral reflux
DMSA – (technetium-99m) dimercaptosuccinic acid
UTI – urinary tract infection
CDC – Centers for Disease Control
