Abstract
Parkinson’s disease (PD) is a neurodegenerative disease with a heavy burden on patients, families, and society. Deep brain stimulation (DBS) can improve the symptoms of PD patients for whom medication is insufficient. However, current open-loop uninterrupted conventional DBS (cDBS) has inherent limitations, such as adverse effects, rapid battery consumption, and a need for frequent parameter adjustment. To overcome these shortcomings, adaptive DBS (aDBS) was proposed to provide responsive optimized stimulation for PD. This topic has attracted scientific interest, and a growing body of preclinical and clinical evidence has shown its benefits. However, both achievements and challenges have emerged in this novel field. To date, only limited reviews comprehensively analyzed the full framework and procedures for aDBS implementation. Herein, we review current preclinical and clinical data on aDBS for PD to discuss the full procedures for its achievement and to provide future perspectives on this treatment.
INTRODUCTION
Parkinson’s disease (PD) is the second most common neurodegenerative disease [1, 2]. In 2019, the global PD prevalence was estimated at over 8.5 million individuals, and 106.3 per 100,000 people had been affected with PD [3]. Moreover, the incidence and prevalence of the disease as well as disability and death due to PD rise even more rapidly than for other neurological disorders, which places a heavy burden on patients, families, and society [3, 4]. Based on its main neuropathology, which is the loss of dopaminergic neurons and depletion of dopamine in the nigrostriatal pathway [5, 6], PD is initially managed with dopaminergic medications [7]. However, as the disease progresses, many PD patients have a poor quality of life because of fluctuations in medication response, levodopa-unresponsive tremor, and medication-related complications (such as levodopa-induced dyskinesia) [7, 8]. For these patients, deep brain stimulation (DBS) can provide better improvement in function and quality of life [9].
As a minimally invasive procedure, DBS shows high effectiveness and safety for several neurological diseases [10, 11]. Concerning PD, multiple clinical studies have demonstrated that DBS was more effective than best medical therapy for moderate-to-severe PD [9] even after a 10-year follow-up and was accompanied by limited adverse events [12], suggesting its acceptable long-term benefits and safety. Most previous studies have applied open-loop conventional DBS (cDBS) for PD, which delivers continuous, uninterrupted stimulations with constant amplitude, frequency, and pulse width and shape, regardless of the patient’s functional status or needs [13, 14]. However, the design of cDBS has inherent limitations, including various stimulation-induced adverse effects, such as dysarthria and imbalance [12, 16], increased battery consumption [13, 14], and frequent parameter adjustments during follow-up to address symptom changes [17].
To overcome these shortcomings of cDBS, the strategy of closed-loop adaptive DBS (aDBS) has been proposed [18–23]; scientific interest in this strategy has increased in recent years [11, 25]. For this design, aDBS recognizes input signals such as brain or motor signals that correlate to a patient’s changed symptoms to provide responsive optimized stimulation in real time [13, 14]. In theory, this on-demand aDBS design can improve therapeutic efficacy and generate fewer side effects with increased battery longevity [13, 26]. Encouragingly, several animal models [21, 27] and clinical studies of aDBS have shown its benefits [28–31]. This topic has attracted scientific interest while both achievements and challenges appear [32]. Although several previous reviews discussed different aspects of aDBS for various neurological disorders [11, 33–40], few of them analyzed the full framework to achieve aDBS for PD [14, 38]; the literature still needs a comprehensive, up-to-date summary of advances in the detailed procedures for aDBS. Herein, we review current preclinical and clinical data on aDBS for PD to discuss a full framework for its implementation in terms of its optimal phenotypes, targets, and patients; its input signals; and its adaptive strategy and stimulation parameters; we also aim to provide a comprehensive review of its implementation procedures, directions of development, and future perspectives and ultimately to promote its future development for the practical benefit of PD patients.
PROCEDURES TO ACHIEVE aDBS FOR PD
Overall, to achieve aDBS for PD, the first step is to identify optimal symptoms, targets, and PD patients. After that, the aDBS is designed with three main modules for sensing, control, and stimulation with associated functions of detecting input signals that reflect symptoms, determining adaptive strategy and adjusting parameters according to the input signal, and providing stimulation, respectively [13]. These procedures are discussed in the following sections, and a diagrammatic sketch of the implementation is shown in Fig. 1.
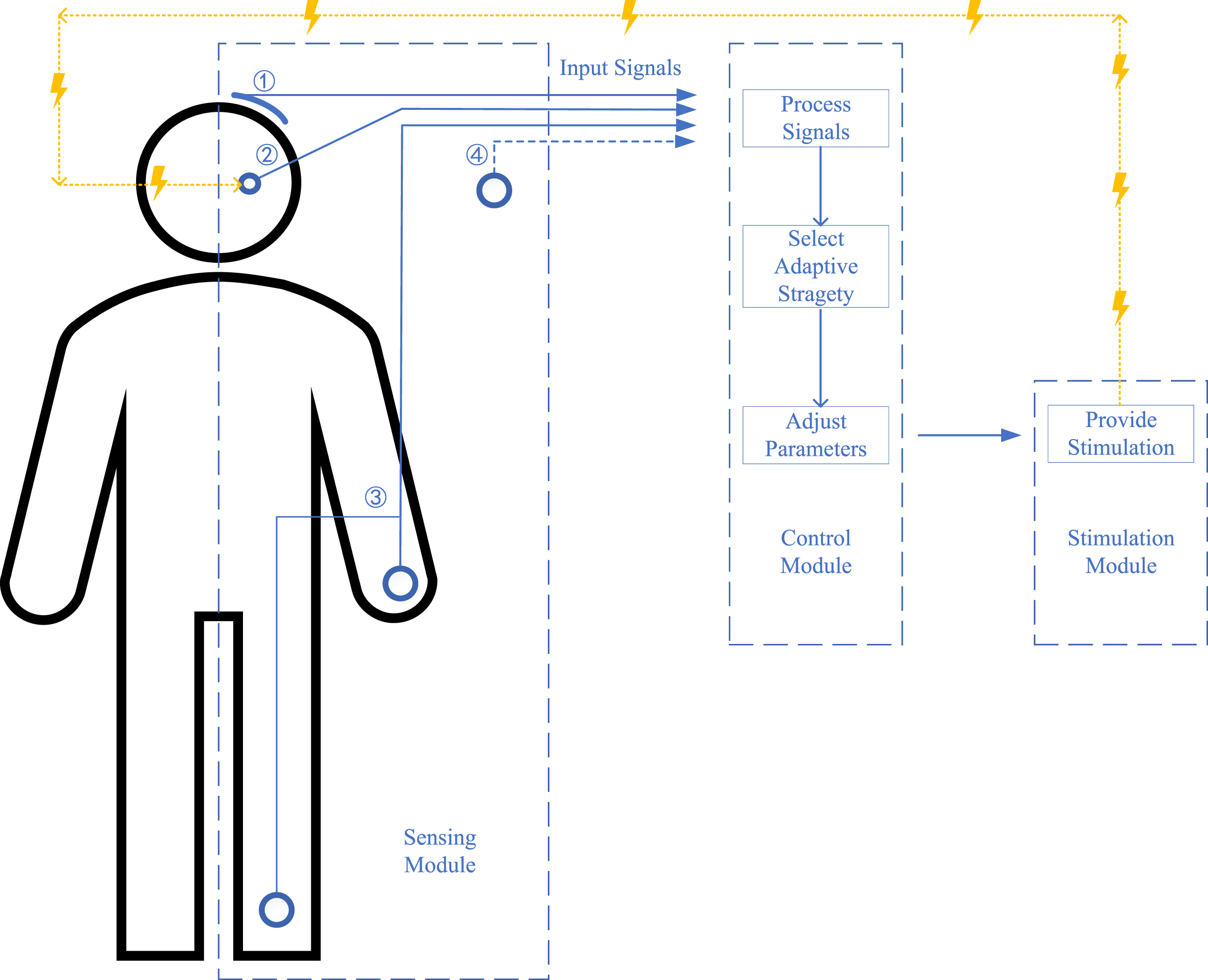
A diagrammatic sketch of the current adaptive deep brain stimulation (aDBS) design for Parkinson’s disease (PD). No. 1: input signals as cortical signals; No. 2: input signals as subcortical signals; No. 3: input signals as surface electromyography (sEMG) and other wearable devices; No. 4: input signals as future potential signals; lines in blue show the signals line and lines in yellow show the stimulation line; the figure shows three main modules for sensing, control, and stimulation with the function of detecting input signals reflecting symptoms, processing signals and selecting adaptive strategy and adjusting parameters, and providing stimulation, respectively.
Optimal symptoms, targets, and PD patients
Essentially, aDBS only changes the way it delivers stimulation to make it more appropriately correlated with the clinical condition of the patient [13], which means its main indications do not overstep the boundaries of cDBS [41]. Thus, the general eligibility principle for aDBS candidates should be consistent with cDBS in theory [42]. However, available studies and devices on aDBS for PD are still limited, especially on its input signals for specific symptoms, which means aDBS only covers several optimal phenotypes, targets, and groups of patients under the current design [14]. A table summarizing current animal models and clinical studies on aDBS in PD is presented as Table 1 to provide more information for readers.
Current animal models and clinical studies of adaptive deep brain stimulation (aDBS) in Parkinson’s disease (PD)
MPTP, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (primate model of Parkinson’s disease); DBS, deep brain stimulation; aDBS, adaptive DBS; cDBS, conventional DBS; GPi, globus pallidus internus; M1, primary motor cortex; PD, Parkinson’s disease; STN, subthalamic nucleus; LFP, local field potentials; n.s, not significant; FoG, freezing of gait. *This patient accepted bilateral cDBS initially and transferred to unilateral aDBS; **Patients were implanted bilaterally but under aDBS unilaterally; ***Patients applied bilateral aDBS on PPN and bilateral cDBS on STN.
Predominant Parkinsonian symptoms mainly include bradykinesia/rigidity, tremor, freezing of gait (FoG)/imbalance, dyskinesia, and nonmotor symptoms [1, 43]. Bradykinesia-rigidity PD is the most studied phenotype for aDBS [13, 14]. Both subcortical and cortical signals show an ability to represent bradykinesia and rigidity [44] and can be input signals for aDBS [22, 45]. Most previous studies on aDBS also focused on bradykinesia-rigidity PD [29, 47] or reported significant improvement in akinesia and rigidity fluctuations [21, 49], suggesting an optimal phenotype for aDBS. In tremor-dominant PD, wearable devices show advantages in detecting and predicting tremor [50–52], while the correlation of subcortical and cortical signals with tremor remains controversial [53–57]. Not only did the study of Malekmohammadi et al. achieve aDBS for tremor-dominant PD based on wearable device signals [58], but some aDBS efforts for other PD phenotypes based on subcortical and cortical signals also provided improvement in tremor [28, 49]. In addition, Cernera et al. [59], He et al. [60], and Opri et al. [61] applied closed-loop aDBS for essential tremor (ET) based on wearable-sensor-driven signals, subcortical (thalamic) LFPs, and cortical (M1) activity. These studies in ET also revealed the potential of different biomarkers for tremor-dominant PD. However, only a limited number of studies with small sample sizes have focused on FoG/imbalance [62, 63], dyskinesia [64], and speech side effects [65] of aDBS for PD, and its benefits are still debated, requiring more studies. Moreover, aDBS targeting nonmotor symptoms of PD is still lacking. For PD combined with psychiatric symptoms, the progress of aDBS for psychiatric disorders [66, 67] can be a paradigm to modulate both motor and psychiatric pathological brain circuit activities together [25, 68]. Future development of aDBS for PD requires further understanding of the pathophysiological mechanisms of different symptoms to cover more symptoms [25].
Another concern relates to the stimulation target. Commonly used targets for cDBS of PD include the subthalamic nucleus (STN) and the globus pallidus internus (GPi) [69] while these two targets are believed to have similar motor benefits [70, 71]. STN cDBS may allow a greater reduction of medication, while GPi cDBS exerts a direct anti-dyskinetic effect [70, 71]. In addition to these common targets, recent cDBS studies have suggested that stimulation of the ventral intermediate nucleus (Vim) has long-term benefit for tremor control and that the pedunculopontine nucleus (PPN) cDBS improves axial motor deficits, particularly FoG and falls [71, 72]. As stimulation of the Vim and PPN addressed other motor features of PD insufficiently, they only applied for patients with particular severe symptoms [71, 72]. Almost all previous studies on aDBS of PD were applied for the STN and showed favorable efficacy [23, 65]. The STN featured subcortical signal (local field potentials, LFP) correlates with several clinical symptoms [73, 74], making it a sufficient input signal during delivery stimulation [14]. Only a clinical study on PPN (targets PD patients with FoG) based on PPN-LFP [62] and a nonhuman study on GPi based on Gpi and primary motor cortex (M1) spikes [27] performed aDBS on other targets. Furthermore, limited by devices and algorithms, most prior clinical studies only applied unilateral aDBS [23, 64] (or bilaterally implanted but unilaterally turned on aDBS [46, 49]), while bilateral aDBS is becoming available in recent aDBS studies for PD [28, 65]. Because bilateral stimulation shows greater improvement in motor symptoms and gait [75], unilateral stimulation is now only proposed for asymmetric PD patients [71]. Thus, aDBS should be studied more on bilateral targets. Synchronous stimulation on multiple targets may provide additional efficacy for wider symptom coverage [72, 76]. However, a bilateral GPi cDBS plus bilateral PPN aDBS study for PD patients with medication-refractory FoG did not reveal more benefit but reported more adverse events [62]. Studies exploring the efficacy and safety of bilateral aDBS on different targets for PD are still needed, especially comparisons of different targets.
In general, current studies have provided initial experience with optimal symptoms, targets, and PD patients receiving aDBS. Available data suggest that phenotypes such as bradykinesia/rigidity and tremor and targets such as bilateral STN could be adequate choices, while further studies on FoG/imbalance, dyskinesia, nonmotor symptoms, and side effects based on bilateral aDBS of different targets need more exploration. Furthermore, since it involves a novel brain signal process and neuromodulate strategy, data safety and ethics concerns can also make acceptance challenging for potential PD patients on aDBS, which means that more detailed preoperative communication with patients is needed.
Input signals of aDBS for PD
A sensing module that measures the symptoms and status of PD patients is the first step in developing a valid aDBS system [14]. Thus, it is necessary to determine robust input signals of aDBS for PD [25]. To date, an increasing number of studies on electrophysiological and brain circuits have provided several biomarkers correlating to PD patients’ symptoms and status, which are considered promising input signals of aDBS [73]. These input signals mainly included subcortical signals, cortical signals, signals from surface electromyographic (sEMG) and other wearable devices, and future potential signals [77]. In the following, we review each of these input signals.
Subcortical signals
Concerning the input signal of aDBS for PD, most of the current subcortical signals refer to the recoding of the basal ganglia LFP, which reflects the sum of the extracellular electrical activity of a group of neurons in the vicinity of the recording electrode [73]. LFP can be captured through microelectrodes during DBS implantation [78], through cables connecting DBS leads during the temporary lead externalization period (3–4 days after implantation and before the final connection to the implanted pulse generator) [79], and through new generation chronic bidirectional sensing and stimulation electrodes [24, 80]. In this aspect, STN-LFP is tested the most as a biomarker, and it has been tested with clinical relevance and stable consistency [81]. Numerous studies have demonstrated that the beta band power (13–30 Hz) of STN-LFP is abnormally high in PD patients [73, 82–85]. During movement preparation and execution, this abnormal high beta activity decreased and then rebounded after movement, suggesting a correlation between beta activity of STN-LFP and movement status and symptoms [86–88]. Dopaminergic intake and/or DBS can suppress this beta activity, and the degree of suppression of oscillatory power correlates with improved Parkinsonian symptoms such as bradykinesia, rigidity, and akinesia [22, 89]. In addition, tremor [90, 91] and FoG/imbalance [92, 93] symptoms as well as sleep stage [94, 95], speaking behavior [96–98], and emotional symptoms [99] can also be detected by different frequency bands of STN-LFP. A number of proof-of-concept clinical studies have applied STN beta activity as an input signal of aDBS [23, 65]. These studies suggested that beta-LFP-based STN aDBS provided greater or at least comparable motor improvement [23, 65] with less energy delivered [23, 64] than cDBS for PD. A recent meta-analysis comparing aDBS with cDBS (mainly in the beta band) for PD suggested that aDBS might outperform cDBS in overall motor improvement with a reduction in energy delivery (55% less than cDBS), while rigidity-bradykinesia, levodopa-induced dyskinesia, and speech intelligibility in aDBS and cDBS were comparable, and beta-based aDBS might not be as efficient as cDBS for tremor control [100]. However, most of the current studies were acute trials with limited sample sizes; thus, there is still a need for future long-term class I evidence such as that which will result from the ongoing randomized multicenter clinical trial NCT04547712 [101]. It is also important to note a recent study suggesting beta-driven aDBS can compromise human motor behavior in PD, especially impairing movement termination [102], which merits further research and attention. As mentioned before, LFP can be recoded chronically through new generation electrodes, making it available for safe long-term recording without additional devices [79, 80]. Another advantage is that the correlation between the beta power of STN-LFP and motor symptom severity shows stable consistency for PD [103] even after long-term STN-DBS [104]. These findings suggest that STN-LFP may be an available input and feedback signal for aDBS that can stably reflect Parkinsonian symptoms [24, 30].
In addition to beta activity of STN-LFP, other STN-LFP-based electrophysiological biomarkers may also be suitable as input signals for aDBS targeting different symptoms, such as low-frequency band (< 12 Hz) [105], gamma band (31–100 Hz) [106], high-frequency band (> 200 Hz) [107] activity and single-unit activity [108]. However, only limited studies on these biomarkers and their correlations to different symptoms are still under further exploration. Furthermore, an animal model using LFP of Gpi [21] and a clinically used LFP of PPN [62] also showed that LFP of other targets also had the potential to become input signals of aDBS. Another promising source of input signals for aDBS could be evoked potentials elicited by applied stimuli (recorded from local stimulation sites such as the STN and GPi as subcortical signals or from remote areas of the cortex as cortical signals), which reveal information about neural connectivity and function [109–111]. A recent study by Sanabria et al. suggested that evoked potentials generated in the brain by GPi-DBS could be recorded to suppress or amplify frequency-specific (16–22 Hz, resonating in the beta band) neural activity in a PD patient implanted with a directional DBS lead [112]. These studies describe encouraging attempts, but their effectiveness and applicability still need to be confirmed.
Although subcortical signals may be regarded as the most studied and potentially suitable input signals of aDBS for PD [24], they still have some limitations. First, movement artifacts and stimulation artifacts make the detection and recognition of LFP a challenge [113, 114], and some patients may not easily be recorded with all bands of LFP [45]. Progress with more sensitive sensors and real-time noise removal algorithms can be helpful [113, 114]. Second, the ability of subcortical signals to capture all different symptoms and statuses remains unclear [53–57]. For example, the abilities of LFP to reflect tremor, FoG/imbalance and nonmotor symptoms of PD are still under debate [28, 62]; machine learning applied to multiple frequency bands and the addition of other input signals may be required [55, 115].
Cortical signals
Cortical signals are recorded through noninvasive scalp electroencephalography (EEG) [116] or through invasive subdural electrocorticography (ECoG), which requires an implanted grid with contacts [117]. However, disturbance of the scalp makes the stability and consistency of EEG unable to fit the sufficiency requirement of input signal [118]. Thus, ECoG oscillations instead of EEG are utilized as input cortical signals of aDBS for PD [118, 119]. ECoG studies on PD patients revealed that high beta (20–30 Hz) power and gamma power are increased in M1 during movement tasks [120]. These abnormal oscillations in sensorimotor cortices can be suppressed and normalized by DBS [121, 122]. A recent study suggested that ECoG might be superior to LFP for movement decoding in PD [123]. Moreover, ECoG can also be recorded to monitor dyskinesia [124] and to recognize speech [125]. Recent studies on phase-amplitude coupling (PAC), whereby the amplitude of one frequency signal is coupled to the phase of another frequency signal (such as beta-phase/broadband-gamma amplitude coupling) either within an area or between areas [126], also show its role in reflecting Parkinsonian symptoms. M1-beta-gamma-PAC is exaggerated in PD patients with dystonia compared to humans without movement disorders, and peaks in M1 gamma amplitude are coupled to, and precede, the STN beta-trough, suggesting a model of the basal ganglia-cortical circuit in PD [126]. This M1-beta-gamma-PAC decreases after STN-DBS or GPi-DBS, and the change is parallel to a decrease in bradykinesia-rigidity [126–128], which can be observed even after chronic stimulation [126, 128]. In contrast, M1-beta-gamma-PAC decreases during resting tremor [53]. Our team also revealed that the intensity of M1-beta-gamma-PAC predicted FoG severity and that STN-DBS alleviated FOG by reducing cortical PAC and increasing cortical resilience [129]. In addition, several studies have confirmed that ECoG technology produces chronic recordings safely [126, 128]. These findings provide a further foundation for ECoG as an appropriate input signal in aDBS for PD.
In developing cortical signals as input signals of aDBS for PD, a monkey 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) PD model used Gpi and cortical ECoG oscillatory burst as input signals for GPi aDBS and suggested that aDBS could better suppress pallidal discharge rate and oscillations and better ameliorate akinesia than cDBS [21]. Swann et al. applied cortical signals as input signals of aDBS for two PD patients [31]. They adjusted the stimulation voltage according to ECoG narrowband gamma (60–90 Hz) oscillations and suggested similar bradykinesia and dyskinesia scores for aDBS and cDBS, while aDBS had substantial energy savings (38% –45%) [31]. A recent long-term study applied STN-LFP and cortical ECoG as input signals of STN aDBS for 5 PD patients with rigidity and bradykinesia and revealed an increased time without dyskinesia during aDBS compared with clinically optimized cDBS [30]. These studies showed the ability to use cortical signals as input signals, but their limitations on sample size, follow-up periods, and efficacy assessments still indicate the need for more studies.
The use of cortical signals as the input signal of aDBS for PD also has some challenges. A major concern is that the recording of ECoG requires an invasively implanted subdural grid. Although this grid can be placed through the same burr hole as DBS electrodes, the additional implant may still increase the risk of complications, such as infection and hemorrhage [57]. Long-term studies with large sample sizes on the safety of the chronically implanted ECoG grid should be conducted. Another question is whether cortical oscillations have some limitations as an input signal of aDBS for PD. During movement preparation and execution [128] and stimulation [130], cortical beta-PAC may be altered, which has implications for feedback signal analysis. Additionally, the ability of cortical signals to represent all motor and nonmotor symptoms is still unclear [44, 130]. Generally, the potential abilities of reflecting and predicting Parkinsonian symptoms (especially on FoG) and decoding movement and behavior make cortical signals a promising input signal of aDBS for PD. Further steps to be taken are developing cortical signal algorithms for different motor and nonmotor symptoms and integrating other potential signals for the whole PD spectrum.
sEMG and other wearable devices
Noninvasive sEMG and other wearable devices (gyroscopes, accelerometers, or magnetometers) provide useful information for movement status and symptoms, especially for tremor detection and prediction [51, 131–134]. Thus, for tremor-dominant PD, these devices could be considered as a potential input signal of aDBS. Studies based on sEMG and other wearable devices as input signals of aDBS for ET revealed sufficient efficacy [135]. Malekmohammadi et al. innovatively used 4–8 Hz tremor power measured by a wearable smartwatch as an input signal for STN aDBS in 8 tremor-dominant PD patients [58]. The researchers observed a significantly lower (36.6%) resting tremor and a lower (76.35%) voltage used than cDBS [58]. This study expands insight regarding these wearable devices as a source of input signals for tremor-dominant PD. In addition, several studies have demonstrated the potential of wearable devices for monitoring postural instability and gait in PD [136]; such devices include inertial measurement units (IMUs) [137–139]. Although these wearable technologies provide an opportunity to monitor PD symptoms chronically [140], research is still needed to standardize them and improve their resolution [136].
However, evidence on the use of sEMG and other wearable devices to detect bradykinesia-rigidity symptoms is limited [132, 142]. This makes it necessary for these devices to combine other input signals for akinetic-rigid PD patients and patients with multiple symptoms. Another concern is the consistency. Patients are required to wear and manage these devices by themselves, which may lead to position shifting and data quality loss [132]. Subcutaneous implantation of sensors may increase stability. However, an implanted device may also face complications. These shortcomings make sEMG and other wearable devices more suitable for combining other stable signals to cover more symptoms or to identify symptoms and verify efficacy than providing a single input signal of aDBS for PD [30, 61].
Future potential signals
Patient-dependent mobile health using telemonitoring or video monitoring is proposed for PD patients’ care at home [143]. This design allows patients to adjust or choose parameters and even turn ON/OFF according to their individual needs [144]. This flexible method may make the simulation more suitable for the needs of patients [144]. Our team also revealed that simple and flexible voltage-based programming can be effective for remote self-administration [145]. However, when the patient’s symptoms or adverse effects cannot be resolved by self-control programing, a comprehensive evaluation and adjustment is still needed at the hospital [145]. Furthermore, the development of smartphones and remote sensors increases the ability to capture symptom fluctuation when patients are at home [146, 147]. These remote information technologies provide complementary support for input signals of aDBS [146, 147], especially when aDBS is applied in chronic daily use at home.
Mechanism-based neurochemical recordings of dopamine may be a future input signal for aDBS [148]. An important step is to develop real-time measurement of neurochemical substances using biocompatible devices and to verify its applicability for aDBS in PD [149]. Recently, optogenetic studies have uncovered unique functionality in stimulating specific groups of neurons to control the basal ganglia circuitry [150]. This novel optogenetic technology may be helpful in locating individual circuits [150] and new targets [151]. For instance, Zhang et al. suggested that targeting parafascicular thalamic circuits may be helpful for motor and nonmotor deficits in PD [151]. Fougère et al. identified the glutamatergic neurons of the cuneiform nucleus as a potential stimulation group to improve locomotor activity in PD [150]. These optogenetic works show great potential in the future development of aDBS for PD.
Although the development of aDBS based on these signals is in an early phase, these studies have proposed new paths or complementary supports and provided insights that may enhance methods in aDBS for PD. However, it is still a long way to develop, verify, put in use, and realize “from bench to bedside.”
Adaptive strategy and stimulation parameters
After determining and sensing input signals, a sensing module can be realized. The next step is to establish a control and stimulation module. Together, these three technological components can establish an available aDBS for PD [13]. In the following sections, we discuss the current progress and remaining challenges regarding adaptive strategies and stimulation parameters.
First of all, neural recordings of brain signals may encode various clinical states at different time scales, requiring fast or slow adaptation strategies according to different temporal dynamics [152]. A common example is the fast and slow modulation of beta-LFP-based aDBS. Previous studies on PD revealed that beta-LFP is not consistently elevated but rather comes in bursts [23, 153]; and these beta bursts have prolonged duration (pathological 200–1,000 ms vs. physiological 100–200 ms) and increased amplitudes when Parkinsonian motor impairment is present [153–155]. Thus, a fast adaptive strategy is required to capture these fast-changing dynamics in the range of milliseconds; to date, timescales of less than 400 ms [23, 153] and 800 ms [29] have been achieved. Another slow adaptive strategy is to process beta activity in the range of minutes without detecting beta bursts but considering medication-related OFF/ON Parkinsonian symptom fluctuations; this strategy has been tested with a 50 s smoothing time [46, 64]. Moreover, gamma-ECoG-based aDBS applies a design with 30 s evaluating averaged power followed by a 600 ms decision, which is another example of a slow adaptive strategy [31]. Methods to capture and integrate distinct biomarkers under different temporal dynamics still need to be explored [152]. Readers are encouraged to review the study of Tinkhauser et al. regarding adaptive strategies with different temporal dynamics [152].
Next, amplitude (either of voltage or of current) serves as a major automatically adjusted parameter to improve symptoms for PD patients in existing treatments [24]. Regarding amplitude modulation approaches, three adaptive strategies are proposed: the ON/OFF design, the gradual design, and the continuous design [14]. The ON/OFF design decides a fixed setting on amplitude, frequency, pulse width, and stimulation period before the adaptive mode, and a certain threshold (for example, exceeding a given beta amplitude of the LFP) is determined [27]. After the adaptive mode of aDBS is turned on, the input signal is continuously recorded. When the input signal reaches and exceeds a certain threshold, stimulation is turned on instantly and is turned off after the signal returns to the given range [27]. This design has been widely used in aDBS using beta-LFP as an input signal for PD patients [23, 65]. The ON/OFF design is a simple and stable adaptive strategy for aDBS. However, some details should be considered. First, to reduce paresthesia, the amplitude should ramp up gradually from zero to the predefined setting [23, 153]. Next, stimulation can be locked to specific pathological neurophysiological phases in PD as a phase-dependent stimulation strategy [23, 153], which may be useful in enhancing long-lasting therapeutic efficacy and minimizing stimulation-related side effects [156]. Cagnan et al. designed the phase-dependent strategy aDBS for ET with locked tremor phase, which reduced tremor severity while also mitigating energy consumption and potentially the risk of tolerance and rebound [157, 158]. Their latest results further supported phase specificity in achieving postural and kinetic tremor control and suggested that ET patients with more stable tremor characteristics might benefit more from this strategy [159]. These findings suggest potential insights for phase-dependent strategy of aDBS for PD. Moreover, to reduce influences on patients’ daily activities, machine-learning algorithms may be applied to predict the occurrence of symptoms and turn on stimulation before the symptoms [160]. However, the ON/OFF design does not sufficiently correspond to the input signal, which makes it necessary to propose gradual and continuous designs [14]. Gradual design applies multiple thresholds and increases or decreases the amplitude stepwise when the input signal is above or below these thresholds, respectively [29, 30]. For instance, Velisar et al. applied a dual threshold algorithm on beta-LFP of STN aDBS for PD based on patients’ specific therapeutic voltage windows [29]. Continuous design links the amplitude to input signals, which makes the output amplitude incline toward a line parallel to input signals [42, 64]. Gradual and continuous designs are believed to have better consistency with input signals as well as symptom severity; in theory, they may have better efficacy and be associated with fewer adverse effects [24]. However, the increased stimulation time introduces more battery consumption [79], and a stable feedback signal is required [42]. Additionally, no studies have compared these different designs of amplitude adaptive strategies; they deserve further research, especially for PD patients with different phenotypes and aDBS with different input signals. Concerning other stimulation parameters, such as frequency and pulse width, most current aDBSs for PD apply a preset fixed setting during stimulation [29, 42]. However, reprogramming these parameters is also an important step toward realizing the best compromise between therapeutic response and adverse effects [17]. In particular, high (130 Hz), low (60–80 Hz), and varied frequencies may have different efficacies in Parkinsonian symptoms and adverse events [17, 161]. The ultimate goal is to develop reliable semiautomated [162] or even fully automated [163] approaches to optimize multiple parameters of aDBS for PD; this will require the building of machine learning models [164].
In summary, the adaptive strategy and stimulation parameters are also important for the development of an available aDBS for PD. Most of the current studies only adjusted the amplitude with several designs. Future research directions should include comparison of different adaptive designs and development of a reliable automated module to adjust multiple parameters and to realize better efficacy and fewer side effects with less battery consumption.
FUTURE PERSPECTIVES OF aDBS FOR PD
From unimodal to multimodal inputs
As mentioned above, a single input signal has inherent limitations. For example, subcortical signals are featured in reflecting bradykinesia/rigidity [22, 89] but are debated in tremor and FoG/imbalance [55, 115]. Cortical signals and sEMG and wearable devices show advantages in detecting FoG/imbalance [127, 165] and tremor [58], respectively. Patient-dependent mobile health provides complementary support for input signals of aDBS [146, 147] when chronically used at home. Thus, combinations of different input signals may be more helpful to cover different symptoms. The FDA-approved Activa PC C + S and Summit RC C + S platforms for experimental platforms have chronic neural sensing from both subcortical and cortical signals and can wirelessly stream neural recordings, providing a perspective on multimodal inputs [24, 30]. With these devices, Arlotti et al. used Activa PC C + S in aDBS mode for 24 hours, and Golron et al. applied Summit RC C + S during normal daily activities for 14 months, showing its great potential in chronic applicability [30]. Additionally, an automatic and validated Parkinsonian symptom identification system using these multimodal inputs can be developed to choose individual-specific inputs and provide symptom-guided stimulation. Furthermore, multimodal inputs have advantages in identifying activity status (walking, resting, sleeping, etc.) [86–88, 123], which makes on-demand stimulation possible. These state-based on-demand aDBSs are particularly useful for the future application of chronically implanted aDBSs [24]. For instance, tremor-dominant PD patients can reduce stimulation during sleeping and increase stimulation after awaking. Despite these insights, more energy consumption because of multiple signals and complex algorithm calculations as well as increased risk of adverse events because of increased implants should be considered for multimodal input design.
On nonmotor symptoms and side effects
Apart from motor symptoms, a wide variety of nonmotor complications, including psychiatric symptoms, cognitive impairment, and somatosensory changes, are usually observed in PD patients [2, 43]. Previous studies suggested discordant or even conflicting results in mood effects [166] and cognitive outcomes [167] of cDBS for PD. A meta-analysis suggested that DBS improved depression and anxiety symptoms, which might be associated with the improvement of motor symptoms [166]. In general, DBS has no severe impact on cognitive functions in PD patients [167]. However, some patients experienced cognitive decline [167, 168], which might be related to exceptional brain network connectivity and could be relieved by stimulation reprogramming [168]. However, the current study on cDBS targeting motor and nonmotor symptoms together is still lacking. The advantage of aDBS from its ability to detect neurophysiological signals and circuit activities makes it available to modulate motor and nonmotor circuits together. The progress of identifying state-dependent responses and applying circuit-based aDBS for psychiatric disorders such as depression [66, 67] and brain stimulation for cognitive defect diseases such as Alzheimer’s disease by tackling circuit dysfunction [169] could provide helpful foundations. Future aDBS can combine these available circuits with motor circuits to develop treatments for PD patients with both motor and nonmotor symptoms. Furthermore, continuous cDBS could induce side effects such as dysarthria and imbalance, which might be related to excessive stimulation and exceptional circuits [12, 16]. Research on aDBS for PD should be conducted to identify these side effects and provide sufficient stimulation to reduce their occurrence.
A future next-generation fully adaptive aDBS
In summary, a future next generation aDBS for PD should be “fully adaptive” with the following features. First, the sensing module identifies the patient’s symptoms, phenotype, circuit, and biomarkers to choose the best combination of stimulation targets and multimodal input signals for symptom detection. Next, the control module processes these input signals, analyzes the optimal adaptive strategy and determines the stimulation parameters. After that, the stimulation module provides the best optimized therapeutic effects. The sensing module continuously monitors efficacy, side effects, activity status, and multimodal inputs as feedback signals and then sends them to the control module for further adaptive strategy and parameter adjustments. After patients return home for chronic use, remote mobile health provides patients with some self-control parameter choices, and patients can make adjustments according to their individual needs. This remote mobile health system also monitors patients’ symptoms and needs and sends them to clinicians for guidance. When problems cannot be resolved remotely, the monitoring system alerts a hospital revisit.
Clearly, more efforts are still needed to achieve this fully adaptive “one-time implant for long-term use” aDBS and achieve better outcomes for PD patients. With the development of new devices and technologies, the proceedings can be accelerated, predictably. A diagrammatic sketch of this future next-generation fully adaptive aDBS and future key perspectives is shown in Fig. 2.
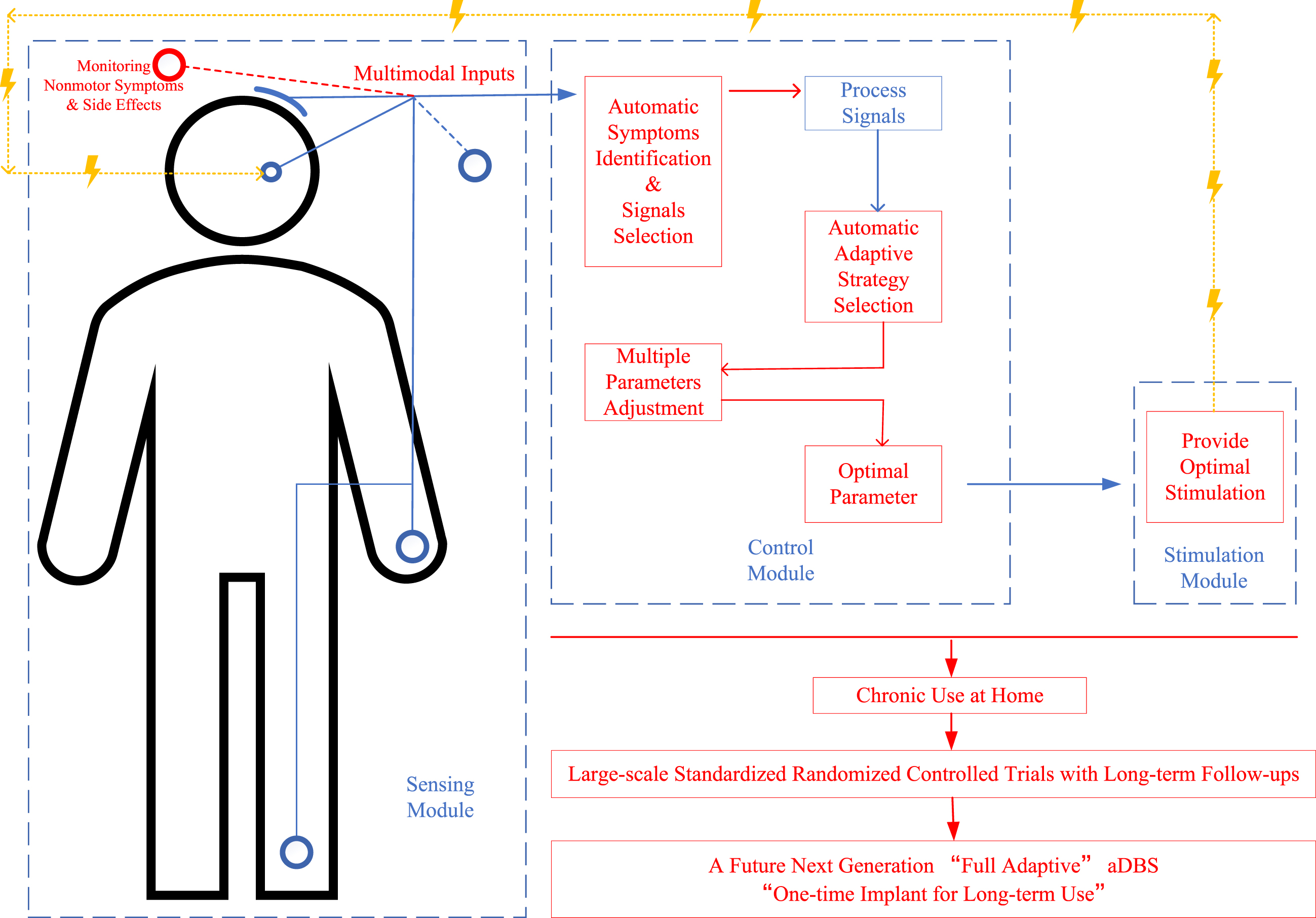
A diagrammatic sketch of a future next-generation “fully adaptive” deep brain stimulation (aDBS) design for Parkinson’s disease (PD) and its future key perspectives. Words and lines in red show the improvement perspectives, lines in blue show the signals line, and lines in yellow show the stimulation line; the figure shows the main procedures in monitoring multimodal inputs for identifying motor and nonmotor symptoms, analyzing optimal adaptive strategy and stimulation parameters, providing best optimal stimulation according to the input and feedback signals, and monitoring efficacy and side effects for further adaptation.
Toward standardized clinical trials
Performing well-designed standardized clinical trials is an important step to verify the efficacy and safety of aDBS for PD. Several papers have reported the application of aDBS in PD patients, and a summary is shown in Table 1. Although previous studies have provided beneficial explorations, most of these studies are proof-of-concept studies with limited sample sizes [28, 80]. To date, the largest cohort was the study of Arlotti et al., which enrolled 11 PD patients with aDBS in 2018 [46]. A sufficient sample size is needed to meet the requirement of test power for providing convincing results. Another concern is about the stimulation time and follow-up period. Prior studies tested aDBS during the lead externalization period, which lasted only several minutes during the perioperative window period [23, 65]. These studies only tested the availability of aDBS design, but the “stun” or “microlesion” effect might make it not accurately represent the real efficacy of aDBS and restrict its long-term use [14]. The development of new chronically implanted devices provides opportunities to explore their long-term efficacy and applicability for home use [30, 42], regarding which further studies are required to analyze benefits and safety. Furthermore, most previous study designs only applied self-control (switched from cDBS to aDBS [23, 65] or only tested aDBS with baseline [29, 46]), while few studies directly compared cDBS and aDBS in separate groups [64]. Additionally, comparisons of aDBS with different designs in input signals and adaptive strategies are lacking. In general, the crucial next step for upcoming aDBS for PD trials will be to formally compare the clinical benefits and safety of aDBS with cDBS using different designs for future clinical application. Well-designed standardized clinical trials with sufficient sample sizes, adequate follow-up and stimulation periods, and appropriate analytical procedures are recommended.
CONCLUSIONS
Growing evidence has suggested that aDBS, as an emerging field, may have potential to outstrip traditional cDBS as a therapeutic choice for PD. Important steps in successful attempts to achieve aDBS include identifying optimal phenotypes, targets, patients, input signals, and adaptive strategy and stimulation parameters. However, further studies exploring multimodal inputs covering more Parkinsonian symptoms and targeting nonmotor symptoms and side effects are needed to realize a future next-generation fully adaptive “one-time implant for long-term use” aDBS. Additionally, large-scale standardized clinical trials with long-term follow-ups and high quality are required to better verify the clinical efficacy and limitations of aDBS. Together, these findings will shed light on the future clinical use of aDBS for PD.
Footnotes
ACKNOWLEDGMENTS
This work was supported by the National Natural Science Foundation of China (81830033).
CONFLICT OF INTEREST
The authors have no conflicts of interest to report.
DATA AVAILABILITY
All data generated or analyzed during this study are included in this published article.
