Abstract
Parkinson's disease symptoms, including gait disturbances and speech impairment, substantially impact quality of life. This n-of-1 study examined the effects of deep brain stimulation (DBS) frequency on a 54-year-old man with Parkinson's disease. The participant conceived and led the study, embedding patient insight within the project design and execution. The impacts of three DBS frequencies were assessed using markerless motion capture for gait, voice recording for speech analysis, and Kinarm robotic tasks of upper limb coordination and cognitive assessment. The study found that DBS frequency has a clinically significant impact on multiple symptoms, including gait, rigidity, speech and cognitive-motor ability.
Plain language summary
This study explored how different deep brain stimulation (DBS) settings impact walking and speech difficulties in a person with Parkinson's disease. The research focused on the effects of three different DBS pulse frequencies on gait, speech and upper limb coordination. This work is significant for two reasons: The effect of DBS pulse frequency (number of pulses per second) is less well understood compared to other parameters, such as DBS pulse strength (current) and pulse width (time). The participant in this study proposed and led this project, enabling a much greater degree of patient insight and engagement than typically occurs.
Dear Editor,
Parkinson's disease (PD) is the second-most prevalent neurodegenerative disorder, presenting with combinations of bradykinesia, tremor, rigidity, along with gait and speech impairments. 1 Deep brain stimulation (DBS) is one therapeutic option. 2 DBS electrode contact selection, frequency, current and pulse width collectively define DBS dose. 3 PD gait impairment can severely impact quality of life, 4 yet DBS parameters to address gait impairment are not well understood. 5 Prior studies have indicated DBS targeting both the sub-thalamic nucleus (STN) and substantia nigra pars reticulata (SNr) may alleviate PD-related gait disturbances.6,7
This n-of-1 study is remarkable as the participant, a 54-year-old right-handed man diagnosed with PD at 40 years of age, conceived and led the project. At age 50 years, his worsening PD symptoms (right-side-dominant) led to the decision to implant DBS (Vercise Gevia, Cartesia directional leads; Boston Scientific, Valencia, CA, USA) in the STN. His speech, fine motor control and rigidity were controlled with stimulation of the four anterior mid-tier electrode contacts, each at 60 Hz, −2.3 mA, and 60 µs; however, his gait was severely impaired. 8 Electrode localization suggested the participant's most ventral DBS electrode contacts might directly influence SNr neurons, 9 and stimulation with the left-most ventral electrode (179 Hz, −2.8 mA, 60 µs) alleviated his right-sided gait difficulties. Three years later, following gait deterioration, the right-most ventral electrode contact was activated and stimulation intensity increased across the other five active contacts by ∼10% with beneficial impact on his gait.
The present study was conducted to evaluate the effects of DBS frequency on PD symptoms. The participant's medication remained unchanged throughout the study: pramipexole extended-release 3 mg daily and amantadine 100 mg twice daily. He had not taken levodopa for >3 years prior. The study's DBS parameters were selected by the participant, based upon his personal notes from DBS programming sessions in the six-month period post-DBS surgery. The proposed DBS programs were reviewed by the participant's treating neurologist in Australia, deemed safe, then uploaded to the patient controller. A neurologist in Canada with PD-DBS expertise assessed the participant at study commencement (MDS-UPDRS-III of 20).
Five 40-min trial sessions, each with different DBS programs, were conducted during a single day using a randomized design, with the participant and project team blinded until initial analyses were complete. Each session incorporated assessment of gait, speech, cognitive-motor performance, and cortical activity. All five DBS programs used the pulse generator as the anode and electrode segments as cathodes, with pulse width 60 µs; the anterior mid-tier contacts of the left- and right-STN electrodes were activated and remained at 60 Hz and −2.5 mA throughout the study. For the most-ventral electrode contacts (C1/C9), Session #1 used the participant's baseline setting (179 Hz, −3.1/−2.9 mA, not blinded). In random order, the four subsequent sessions tested two alternative frequencies for contacts C1/C9, each with two current strengths (Session #2: 104 Hz, −3.1/−2.9 mA; #3: 104 Hz, −3.3/−3.2 mA; #4: 149 Hz, −3.1/−2.9 mA; #5: 149 Hz, −3.4/−3.2 mA). To minimize frequency changes, Sessions #3 and #5 were paired with the same frequency as Sessions #2 and #4, respectively. Fifteen- and 60-min rest periods were held between current changes and frequency changes, respectively.
Gait was assessed for 5 min per session using a markerless motion capture system (Theia3D, Theia Markerless Inc., Kingston, ON, Canada), 10 while the participant walked on a treadmill at speed 1.7 m/s. Arm swing, reflecting rigidity and indicating gait quality, was measured per stride as the wrist-to-pelvis distance in the anterior-posterior plane. From the arm swing assessment, there was greater rigidity of the right arm (right < left arm swing median per session) under all conditions (Figure 1:A1–2). Notably, the participant held the support bars for 51% and 45% of the time during Sessions #2 and #3, respectively, indicating substantial gait difficulty under those settings (Figure 1:A4). During Sessions #4 and #5, relative to baseline, greater left-right arm swing asymmetry (measured by left / right arm swing median) and greater variation in arm swing (measured by interquartile range) indicated more gait difficulty during those sessions. 11 Torso lean, indicating postural instability, was calculated per stride as the thorax angle relative to the perpendicular from treadmill base, and did not vary substantially across conditions (Figure 1:A3).
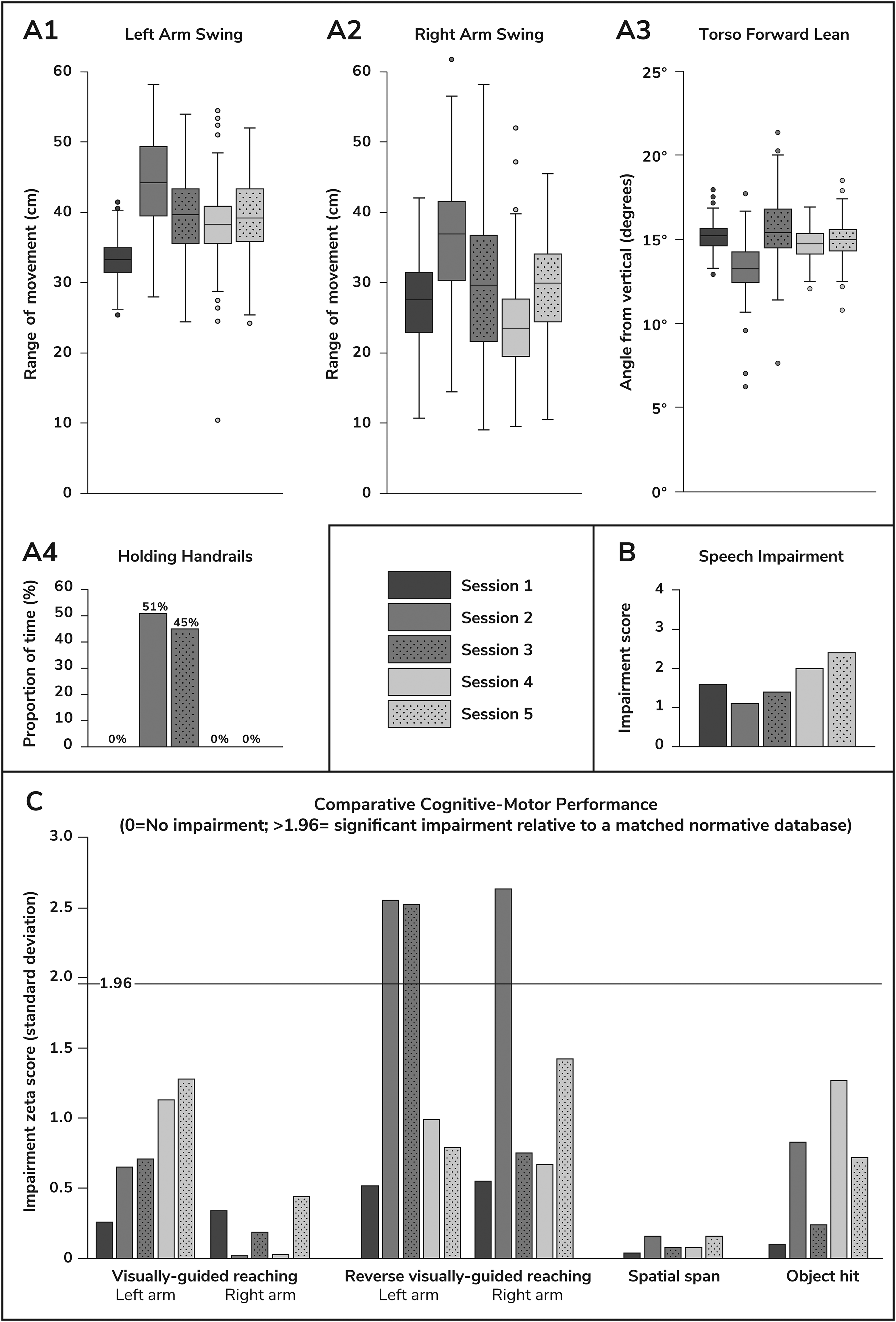
Selected motion capture, speech and Kinarm results, by study session. DBS settings for Session #1 were: contacts C1/C9 179 Hz, −3.1/−2.9 mA; C2, C5, C10, C13 each 60 Hz, −2.5 mA; all contacts 60 µs; the other sessions had contacts C1/C9 varied to #2: 104 Hz, −3.1/−2.9 mA; #3: 104 Hz, −3.3/−3.2 mA; #4: 149 Hz, −3.1/−2.9 mA; #5: 149 Hz, −3.4/−3.2 mA. (A) Markerless motion capture while walking on a treadmill. (B) Speech. (C) Kinarm. (A1–3) Boxes denote interquartile range with line at median, and whiskers denote 1.5 x interquartile range with data outliers plotted. The right arm experienced a greater variation in its range of motion, compared with the left arm, on all DBS settings: interquartile range (cm) for left arm swing 4, 10, 7, 5, 7 and right arm swing 8, 12, 15, 9, 10, for Sessions #1 to #5, respectively. Note: Data are presented only from periods when the participant was not holding the treadmill handrails. (A4) Proportion of time the participant was holding the treadmill handrails during each session. (B) Speech impairment scale, 0 = none, 1 = mild, 2 = moderate, 3 = severe, 4 = profound. (C) Zeta scores (lower score = less impairment) are age-, sex- and dominant-hand-matched to the Kinarm database, where scores >1.96 indicate statistically significant impairment.
Speech was recorded for 5 min per session using an iPhone. The participant responded to prompts designed to elicit extemporaneous speech. Recordings were analyzed by a speech-language pathologist experienced in PD, using a five-point impairment scale for voice quality, resonance, articulatory precision, and rhythm. 12 Speech impairment in the recorded samples ranged from mild (Session #2) to moderate-severe (Session #5) (Figure 1:B). The participant reported that moderate-severe speech impairment would require him to take early retirement.
The participant performed four Kinarm tasks (https://kinarm.com/download/kinarm-standard-tests-tech-sheet/) using the Kinarm Exoskeleton Lab (Kinarm, Kingston, ON, Canada). 13 Tasks included visually-guided reaching (motor), reverse visually-guided reaching (cognitive-motor), spatial span (memory), and object hit (bimanual coordination). The first two tasks were conducted with each arm, for a total of six Kinarm tests. Kinarm measurements of cognitive motor function revealed significant impairments under Sessions #2 and #3, relative to matched controls in the Kinarm normative database. The spatial span task is a measurement of short-term memory, and the participant was unimpaired with this task for all the sessions. The baseline session (Session #1) was the least impaired in five of the six Kinarm tests (Figure 1:C).
Functional near infrared spectroscopy (fNIRS; NIRx Medical Technologies LLC, New York) was used to measure cortical activity at rest, and during gait and speech measurements. The fNIRS results will be reported separately.
These findings underscore the need to reconsider DBS programming protocols, as the frequency parameter is often overlooked in favor of adjustments to current strength and choice of electrode contacts. 14 The DBS frequency-induced changes in symptoms were not compensated by adjustments to stimulation current. The use of the Total Electrical Energy Delivered (TEED) is considered by some as a relevant metric in such scenarios. 15 Sessions #1 and #5 had the same TEED but had a different impact on PD-symptoms.
In conclusion, the participant's firsthand experiences helped identify clinically relevant insights that might have been overlooked in traditional research paradigms, including the clinical impact of DBS frequency. While the n-of-1 design limits the generalizability of the findings, it provides proof of concept for future clinical trials.
Footnotes
Acknowledgments
The authors would like to thank Doctors Richard Blazé (Australia) and Jennifer Sharma (Canada), neurologists who cared for the participant, Matthew Jacobs, Sophie DeCoste and Kerlas Samaan, for their contributions to the initial stages of the project, and Associate Professor Andreas Horn, for advice on multiple occasions.
ORCID iDs
Ethical considerations
This study was approved by the Queen's University's Health Sciences Research Ethics Board (approval no. 6041027) on May 10, 2024.
Consent to participate
Informed consent for participation was obtained on the day of study, before study procedures commenced.
Consent for publication
Informed consent for information published in this article was not separately obtained because the first author is the sole participant in the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was conducted as part of the Capstone Project Course of the Neurotech Microcredential Program, for which SB received funding from the Province of Ontario. SB receives funding from the Connected Minds Program, supported by Canada First Research Excellence Fund, Grant #CFREF-2022-00010.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy considerations.
