Abstract
Background:
Although sleep disturbances are highly prevalent in patients with Parkinson’s disease, sleep macroarchitecture metrics show only minor changes.
Objective:
To assess alterations of the cyclic alternating pattern (CAP) as a critical feature of sleep microarchitecture in patients with prodromal, recent, and established Parkinson’s disease.
Methods:
We evaluated overnight polysomnography for classic sleep macroarchitecture and CAP metrics in 68 patients at various disease stages and compared results to 22 age- and sex-matched controls.
Results:
Already at the prodromal stage, patients showed a significantly reduced CAP rate as a central characteristic of sleep microarchitecture. Temporal characteristics of CAP showed a gradual change over disease stages and correlated with motor performance. In contrast, the sleep macroarchitecture metrics did not differ between groups.
Conclusion:
Data suggest that alterations of sleep microarchitecture are an early and more sensitive characteristic of Parkinson’s disease than changes in sleep macroarchitecture.
Keywords
INTRODUCTION
Dysfunctional sleep is a hallmark of Parkinson’s disease (PD), and sleep-wake problems are among the most commonly reported non-motor symptoms in PD patients [1]. The interaction of sleep and neurodegeneration is assumed to be bi-directional: while healthy sleep can attenuate neurodegeneration, ongoing neurodegeneration disturbs physiological sleep [2]. A deeper understanding of dysfunctional sleep in PD is pivotal for addressing this vicious cycle.
Overnight video-polysomnography (PSG) is the gold standard for studying sleep, but changes in sleep macrostructure in PD are subtle: it was estimated that PD patients have, on average, only 2% less non-rapid eye movement (NREM) stage 2 sleep and slow wave sleep (SWS) compared to healthy subjects [3]. Hence, a closer look beyond the analysis of sleep macrostructure could help to better understand the complex alterations of sleep dysregulation in PD neurodegeneration [4–7]. The cyclic alternating pattern (CAP) represents oscillations of cortical activity during NREM sleep independent of the classic sleep staging system [8] (Fig. 1 provides an example) that can be detected in the electroencephalographic (EEG) signal. CAP is considered a significant feature of sleep microarchitecture, and recently CAP alterations have been linked to earlier phenoconversion to PD in patients with isolated REM sleep behavior disorder (iRBD), a prodromal state of the disease [9]. Still, it remains unclear how CAP alterations relate to the progress of neurodegeneration in PD.
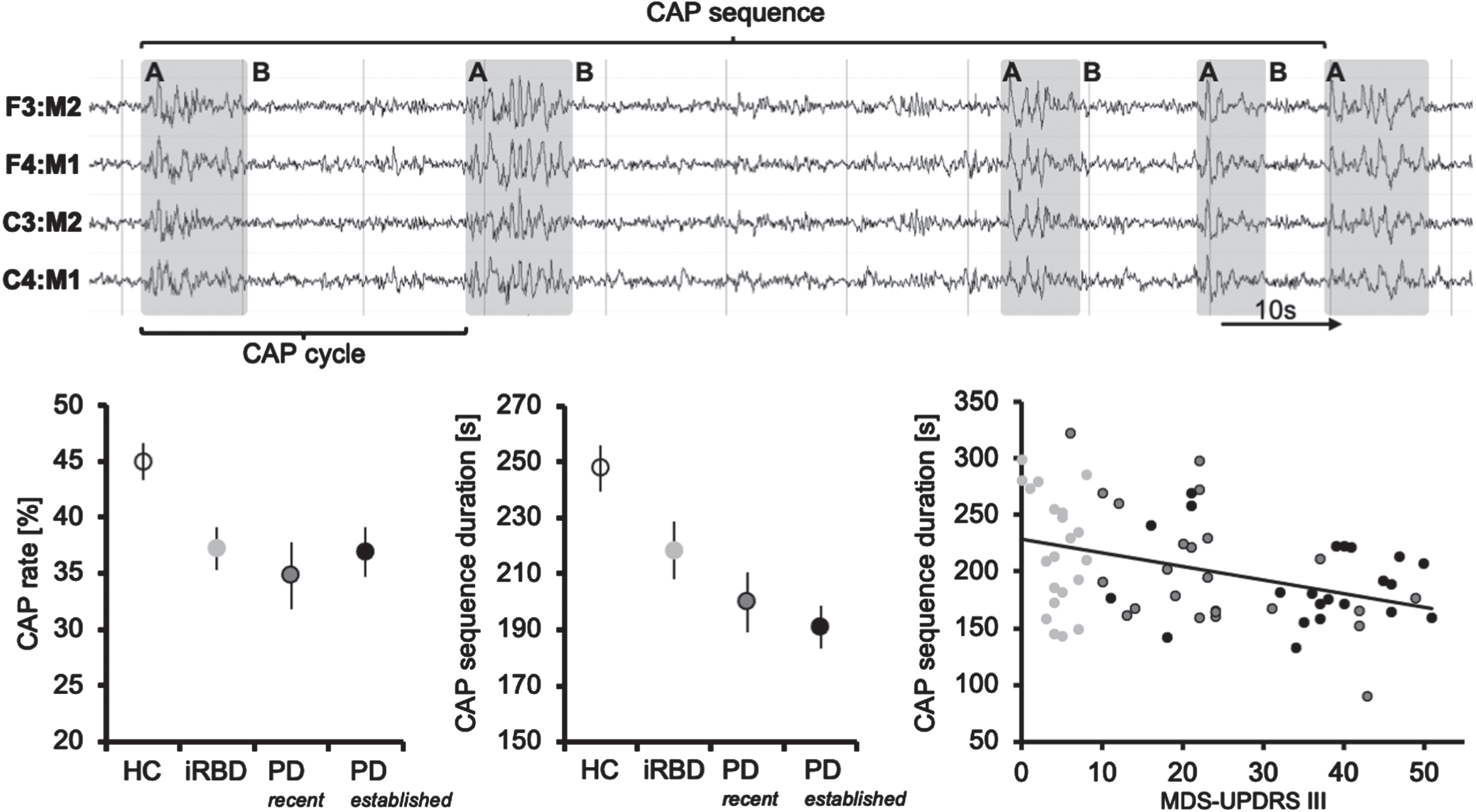
Features of the cyclic alternating pattern (CAP). Upper panel: Example of a CAP sequence composed of events with high amplitude (A phases, light grey) alternating with sections of electroencephalographic (EEG) background activity (B phases). One A and one B phase form a CAP cycle; multiple CAP cycles represent a CAP sequence if not interrupted by EEG background activity for more than 60 seconds (s). Classic sleep-EEG montage with references of scalp electrodes to the contralateral ear is provided. Lower panel: Left, CAP rate (amount of non-rapid eye movement (nREM) sleep comprising CAP) is already reduced in prodromal Parkinson’s disease (PD) (=patients with isolated REM sleep behavior disorder (iRBD)). Center, duration of CAP sequences gradually decreases with PD stages progressing from prodromal to recent and established PD. Right, CAP sequence duration negatively correlates with motor impairment evaluated with the Movement Disorders Society Unified Parkinson’s disease Rating Scale across stages of prodromal and overt PD (light grey = iRBD, dark grey = recent PD, black = established PD). Error bars indicate the standard error of the mean.
We analyzed CAP metrics in patients at various stages of PD (prodromal, recent, and established PD) compared to healthy controls (HC) to assess the relationship of altered sleep microstructure during the progression of PD and compared the results to classic sleep macroarchitecture analysis.
MATERIALS AND METHODS
Participants
We evaluated datasets from currently ongoing case-control studies [10, 11] and one previous study [5]. These studies were approved by the local ethical committees and all subjects provided informed written consent according to the Declaration of Helsinki.
To avoid a potential bias due to comorbidities, we excluded datasets from 3 HC, 5 iRBD, and 7 PD patients due to extraordinarily high apnea/hypopnea indices (AHI,>30/h) or periodic limb movement indices (PLMI,>100/h). The final sample consisted of 22 HC subjects, 22 iRBD, and 46 PD patients. All subjects were between 50–85 years old and non-demented (Montreal Cognitive Assessment score >22). iRBD patients were part of an ongoing study recruiting a local prodromal PD cohort in Cologne. iRBD patients were classified according to the current Movement Disorder Society (MDS) criteria on prodromal PD [12] (see Supplementary Table 1 for details). PD diagnosis was made according to the current consensus criteria [13]. Levodopa equivalent daily doses (LEDD) were estimated as previously recommended [14], disease severity was judged according to the Hoehn and Yahr stage [15], and motor symptoms were quantified with the MDS Unified Parkinson’s disease Rating Scale part III (MDS-UPDRS III) after 12 h of medication withdrawal. Depending on their motor symptom duration, PD patients were considered as patients with recent PD (<3 years) or established PD (>3 years).
Demographic and clinical characteristics and metrics of sleep macro- and microarchitecture
AHI, apnea-hypopnea index; BMI, body mass index; CAP, cyclic alternating pattern; f, female; h, hour; iRBD, isolated REM sleep behavior disorder; kg, kilogram; LEDD, levodopa equivalent dose; m, male or meter; MDS-UPDRS III, Movement Disorders Society Unified Parkinson’s disease Rating Scale part III; mg, milligram; min, minutes; N, non rapid eye movement sleep; PLMI, periodic limb movement index; RBD, REM sleep behavior disorder; REM, rapid eye movement sleep; s, second; SWS, slow wave sleep; y, years. Statistics: significant post-hoc tests are indicated to the corresponding group: HC, healthy controls; rPD, recent PD; ePD, established PD. Differences in RBD status were not calculated as HC are RBD-negative, and iRBD patients are RBD-positive by definition. RBD status did not differ between PD groups. The probability of prodromal PD was estimated according to Movement Disorder Society research criteria [12].
Polysomnographic data and CAP analysis
We used a mobile SOMNOscreenTM plus device for overnight PSG, including 10 EEG recordings (according to the international 10/20 system: F3, F4, C3, C4, O1, O2, M1, M2, Fpz as grounding, and Cz as reference) as previously described [5]. Visual PSG scoring was performed on 30-s epochs according to the AASM Manual for the Scoring of Sleep and Associated Events, Version 2.6 [16].
CAP scoring followed the rules of the consensus criteria published in 2001 [8]. In brief, episodes of phasic events with high amplitude (A phase) alternate with sections of EEG background activity (B phase). One A and one B phase together represent a CAP cycle, and both A and B phases must have a duration of between 2–60 s. To constitute a CAP sequence, at least three A and two B phases must occur (Fig. 1). Background activity longer than 60 s, REM sleep, or awake episodes end a CAP sequence. Scoring was performed using DOMINO software in an iterative process: first, a medical doctoral candidate (JS), who was trained on CAP scoring, and second, a board-certified sleep expert (MS) marked A phases. Both raters were blinded to the subjects’ clinical features. F3, F4, C3, C4, O1, and O2 EEG channels were presented referenced to the contralateral ear as well as bipolar in a longitudinal montage. The A phases were tagged directly onto the EEG data and exported with sleep stage scorings from DOMINO software into the American Standard Code for Information Interchange (ASCII). A custom-made Python script was used to identify NREM episodes, including CAP, by applying the consensus criteria [8]. The following CAP parameters were calculated: CAP rate (CAP time / total NREM time×100), duration of A and B phases, sequence duration, and CAP cycles per sequence.
Statistical analysis
We analyzed the data with Statistical Package for the Social Sciences (SPSS) version 28. Group data are presented as mean±standard deviation unless otherwise stated. The normal distribution of the data was assessed with the Shapiro-Wilk test, Q-Q plots, and box plots. Group comparisons were calculated using analysis of variance, Kruskal Wallis, and chi-square statistics with equivalent post-hoc tests as appropriate. Correlation analyses were calculated with Spearman’s rank coefficient and Pearson’s r. Significance was accepted at p < 0.05.
RESULTS
Demographic and clinical data
We included 22 HC subjects, 22 iRBD patients, 24 patients with recent, and 22 with established PD. Groups were age- and sex-matched (Table 1). All iRBD patients were naïve to dopaminergic treatment, as well as 11 recent and one established PD patient. Eighteen of 22 iRBD patients fulfilled the criteria for probable prodromal PD, defined as ≥80% probability [12].
We did not observe statistically significant differences in sleep macroarchitecture parameters between HC subjects and patient groups, i.e., sleep latency, sleep efficiency, sleep stages, and sleep cycles. AHI was comparable between groups, but PLMI was higher in iRBD patients than PD patients (Table 1).
CAP sleep microstructure analysis
CAP rate was significantly lower in patients than HC (p = 0.017, Table 1, Fig. 1 lower panel, left). Reduced CAP rate occurred in the context of shorter CAP sequences and reduced CAP cycles per sequence (p < 0.001, and p = 0.001, respectively, Table 1). The duration of A and B phases (the components forming the CAP cycles) did not differ between HC and patients (Table 1). CAP sequences progressively shortened from HC along PD stages (rho = –0.436, p < 0.001) (Fig. 1, lower panel, center). CAP sequence duration also negatively correlated with MDS-UPDRS III values in patients (r = –0.393, p < 0.001) (Fig. 1 lower panel, right). This correlation was independent of LEDD and RBD status in partial correlation analysis. RBD status in PD patients was not linked to CAP metrics. When investigating only the 18 iRBD patients fulfilling the MDS criteria on prodromal PD, results of group comparisons and correlation analyses did not change significantly (correlation with PD stages: rho = –0.425, p < 0.001; correlation with MDS-UPDRS III: r = –0.355, p = 0.004). Comparing both subsets of iRBD patients, iRBD patients not fulfilling the MDS criteria showed a numerically (albeit not statistically significant) higher CAP rate (39.9±9.2 vs. 36.6±9.1%, p = 0.529), longer sequence duration (253.7±23.4 vs. 210.5±50.0, p = 0.112), and more cycles per sequence (7.0±0.9 vs. 5.7±1.7, p = 0.156).
DISCUSSION
Our data on CAP across various PD stages indicate that microsleep alterations may be a sensitive and early event in PD evolution. Additionally, temporal characteristics of CAP showed a gradual decrease along PD stages and could be linked to the motor performance of patients. This finding aligns with and complements data from a previous report on reduced CAP rate in mid-stage PD patients [5].
For iRBD patients, Melpignano and colleagues also showed a lower CAP rate than in HC and besides that a reduction in CAP rate was linked to earlier phenoconversion [9]. As phenoconversion typically implies break-through of apparent motor symptoms, this finding fits well with the correlation between microsleep disturbances and the motor performance decline we observed. Notably, iRBD patients not fulfilling the MDS criteria for prodromal PD showed microsleep parameters similar to healthy controls which supports the notion of CAP being a sensitive marker of PD progression. PD motor deficits mainly arise due to dopaminergic deficiency, which points to a monoaminergic regulation of (micro)sleep homeostasis. In this connection, we recently reported a link between noradrenaline transporter density and CAP rate in PD [5].
Dopamine and noradrenaline act as wake-promoting neurotransmitters and are assumed to be silenced to promote sleep [17]. Beside their function in prompting arousals and awakenings during sleep, these monoamines also initiate high-amplitude slow oscillations during NREM sleep— a hallmark of the A phases of CAP [18]. Interestingly, silencing the locus coeruleus, the primary source of noradrenaline in the brain, during sleep is associated with alterations of these oscillations and eventually leads to memory dysfunction [19]. This is in line with evidence for a reduced CAP rate in patients with mild cognitive impairment who converted to dementia in the following two years [20]. Similarly to our data, the authors did not observe a difference in sleep-macroarchitecture metrics in these patients [20]. In a study with recent PD patients, changes in conventional sleep parameters could not be detected either [21]. Notably, a reduction of delta power could be attenuated by dopaminergic medication. Therefore, altered microsleep might be a highly sensitive marker of monoaminergic dysfunction and, hence, a potential marker of incipient PD pathology, even at a prodromal stage.
Recently, the concept of a brain- vs. body-first PD phenotype has been proposed [10, 22]. According to this concept, the occurrence and timing of RBD onset represent critical clinical features in discriminating between both phenotypes [10]. We did not observe a difference in CAP metrics between patients with recent PD with or without RBD. Therefore, alterations of microsleep might represent a potential marker of earliest PD pathology independent of the brain- vs. body-first concept. Altered microsleep could be of high value as prodromal features of brain-first PD remain scarce [22–24].
The major limitation of our study is the lack of longitudinal data to assess the predictive value of microsleep changes on an individual patient level. Nevertheless, we sought to carefully match groups concerning sex, age, and relevant sleep-specific co-morbidities (operationalized by elevated AHI and PLMI) to avoid potential bias on sleep microarchitecture [25, 26]. Still, iRBD patients showed higher PLMI than PD groups.
In conclusion, we observed that microsleep alterations as assessed by the cyclic alternating pattern are an early event in PD evolution and more sensitive than classic parameters of sleep macroarchitecture. Future studies are warranted to elucidate their association with the patients’ motor and non-motor symptom burden and to collect data on longitudinal changes of microsleep parameters.
Footnotes
ACKNOWLEDGMENTS
C. E. J. Doppler received grants from the Clinician Scientist Program (CCSP), funded by the German Research Foundation (DFG, FI 773/15-1).
M. Sommerauer received grants from the Else Kröner-Fresenius-Stiftung (grant number 2019_EKES.02), and the Koeln Fortune Program, Faculty of Medicine, University of Cologne (grant number 453/2018).
CONFLICT OF INTEREST
G. R. Fink serves as an editorial board member of Cortex, Neurological Research and Practice, NeuroImage: Clinical, Zeitschrift für Neuropsychologie, and DGNeurologie; receives royalties from the publication of the books Funktionelle MRT in Psychiatrie und Neurologie, Neurologische Differentialdiagnose, and SOP Neurologie; received honoraria for speaking engagements from Bayer, Desitin, Ergo DKV, Forum für medizinische Fortbildung FomF GmbH, GSK, Medica Academy Messe Düsseldorf, Medicbrain Healthcare, Novartis, Pfizer, and Sportärztebund NRW.
The other authors have no conflict of interest to report.
