Abstract
Background:
Non-motor symptoms are common in Parkinson’s disease (PD) and some, including hyposmia, constipation, and REM sleep behavior disorder, often precede the clinical diagnosis.
Objective:
To assess the relation between combinations of non-motor features and presence of PD among women.
Methods:
A nested case-control study was conducted among women in the Nurses’ Health Study. Women were eligible if they responded to screening questions for constipation and probable REM sleep behavior disorder (pRBD) on a 2012 questionnaire and were under age 85 on January 1, 2012. 87 women with confirmed PD and 14,170 women without PD agreed to participate and completed in 2015 the Brief Smell Identification Test to assess hyposmia, as well as a questionnaire to assess parkinsonism and other non-motor PD features, including depressive symptoms, excessive daytime sleepiness, impaired color vision, and body pain.
Results:
In age-adjusted logistic models, each non-motor feature was significantly associated with PD, and the odds of PD increased exponentially with the number of features. Women with constipation, pRBD, and hyposmia had an age-adjusted OR for PD of 211 (95% CI 84.2–529) compared to women with none of these features. The odds of having PD rose further with the presence of additional non-motor signs. Comparing women with at least 6 of the 7 features assessed in this study to women with one or none, the age-adjusted OR for PD was 356 (95% CI 113–1126).
Conclusion:
Results suggest that these non-motor features could be useful in discriminating PD patients from controls in women, and since they often appear during the prodromal period of PD, their combinations may prove useful for identifying populations at high risk of developing PD.
INTRODUCTION
Numerous sex differences have been identified in the epidemiology of Parkinson’s disease (PD). Compared to men, women are less susceptible to PD, have a later age of onset, and may have a more benign phenotype [1, 2]. In addition, the association between certain non-motor symptoms and PD may be weaker among women than men [3]. Non-motor symptoms are common in PD and some, including hyposmia, constipation, and REM sleep behavior disorder, often precede the clinical diagnosis [4]. We have recently reported that the combination of probable REM sleep behavior disorder (pRBD), hyposmia, and constipation is rare among apparently healthy men and could be useful for identifying those with a high probability of developing PD [5]. However, it is unclear whether these initial findings would generalize to women.
We therefore conducted a large-nested case-control study among participants of the Nurses’ Health Study (NHS) to assess cross-sectional associations between non-motor symptoms and prevalent PD among women.
METHODS
Source population
The NHS was established in 1976 when 121,700 female registered nurses aged 30 to 55 years completed a mailed questionnaire regarding their medical histories and health-related behaviors; follow-up questionnaires have been sent every two years [6]. Selection procedures for the current study are similar to those of our previous study among men in the Health Professionals Follow-up Study [5]. Briefly, women were eligible for the current study if they completed the 2012 questionnaire and responded to questions regarding constipation and pRBD (because the pRBD question required the response of the participant’s sleep partner, 15,860 otherwise eligible women were excluded because they reported that they did not have a sleep partner), were under age 85 on January 1, 2012, and were alive and eligible to receive mailings in May 2015.
Identification of PD cases and controls
PD case documentation procedures have been described previously [7]. Briefly, PD cases are first identified through responses to the biennial questionnaires. Then, with the participants’ consent, their medical records are requested and then reviewed by a neurologist specializing in movement disorders. Cases were considered confirmed if the medical record included a final diagnosis of PD by a neurologist, or if the medical record demonstrated evidence of at least 2 of the 3 cardinal signs of PD (rest tremor, rigidity, bradykinesia) in the absence of features suggesting other diagnoses. 110 confirmed PD cases met the above eligibility criteria and were invited to participate in the current study.
To increase efficiency, we selected from over 35,000 eligible controls—comprising those women at risk of PD who would have been included as cases if they had been diagnosed—all women who screened positive for constipation or pRBD (N = 13,892), as well as 4,869 women without constipation or pRBD. We accounted for the bias caused by undersampling women without constipation or pRBD using inverse probability weighting [8]. 71 PD cases and 14,100 controls completed mailings sent in 2015 and were included in analyses. An additional 16 PD cases and 70 controls had in 2011 completed the Brief Smell Identification Test (B-SIT) and questionnaire as part of a pilot case-control study. The procedures and measures were essentially identical to those used in the 2015 study, and therefore data from the pilot study are included in this analysis. A flow chart depicting the study design can be found in Fig. 1. This study was approved by the Human Research Committees at the Brigham and Women’s Hospital.
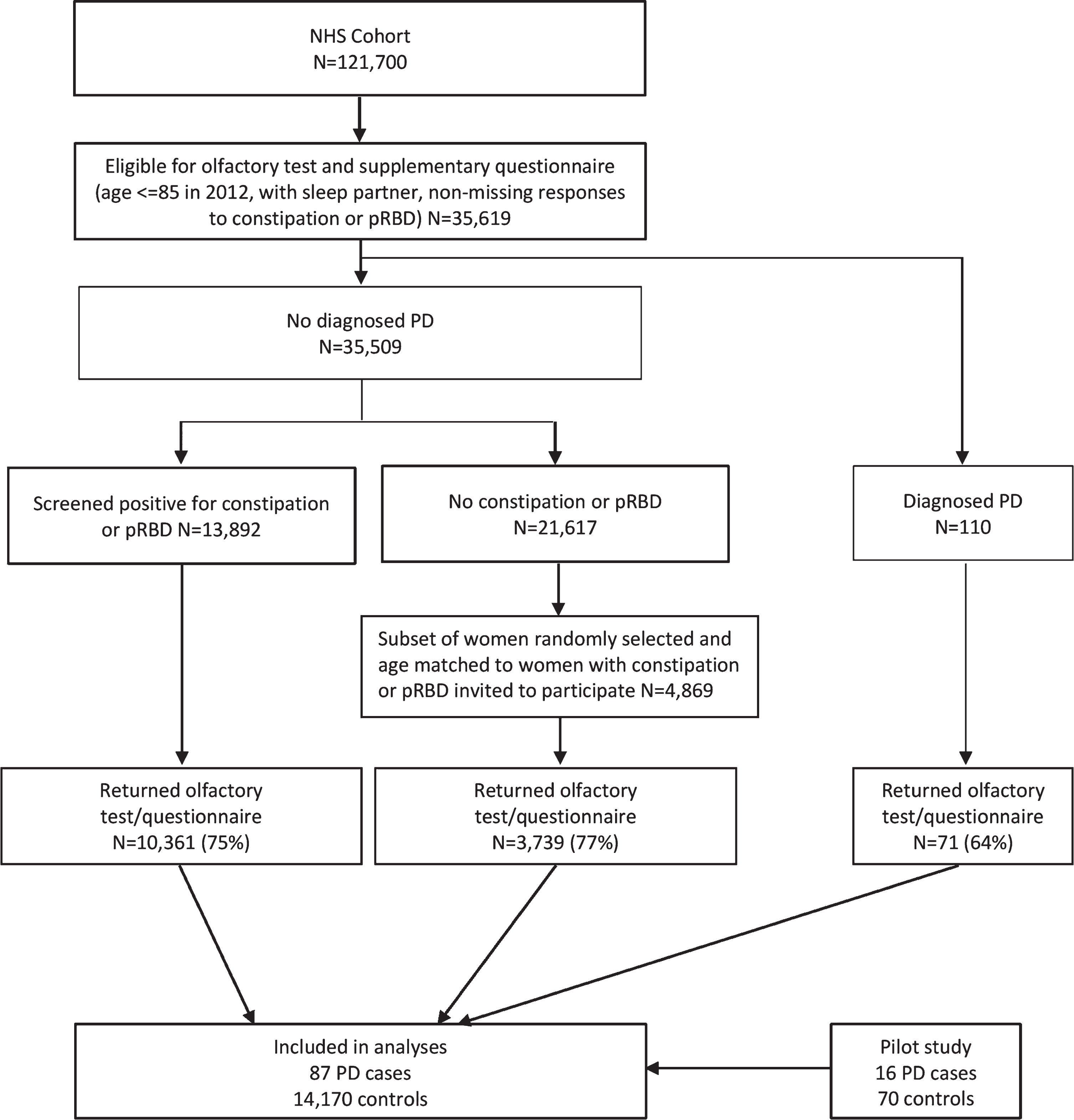
Flow chart of the study design.
Assessments
Study assessments have been described previously [5]. Participants reported bowel movement and laxative frequencies on the 2012 questionnaire; women were considered constipated if they reported a bowel movement frequency of every other day or less, and/or laxative use at least twice a week. An RBD screening question adapted from the validated Mayo Sleep questionnaire was used to define pRBD (“Has your spouse [or sleep partner] told you that you appear to “act out your dreams” while sleeping [punched or flailed arms in the air, shouted or screamed], which has occurred at least three times?”) [9]. The original question, which did not include the specification of dream enactment having occurred at least three times, had a sensitivity of 100% and a specificity of 95% for the diagnosis of polysomnography-confirmed RBD in a community-based sample [10]. We assessed hyposmia using the B-SIT, which consists of a booklet containing 12 odorants. Participants are asked to identify each odorant from a list of four alternatives [11]. Olfactory scores were calculated by summing the number of correctly identified odors, and hyposmia was defined as a score in the bottom 10% of women without pRBD or constipation (score≤8). Excessive daytime sleepiness was measured using the Epworth Sleepiness Scale [12]; excessive daytime sleepiness was defined as a score of at least 10. Color discrimination was assessed using a mailed version of the Roth color discrimination test, which is an abridged version of the Farnsworth-Munsell Test [13]. In a validation study among 28 participants from the NHS and Health Professionals Follow-up Study, the Spearman correlation coefficient between the mailed version of the Roth color discrimination test and the Farnsworth-Munsell 100 hue test was 0.73 (p < 0.0001) [5]. Depressive symptoms were assessed using the Mental Health Inventory (MHI) [14], a 5-item subscale of the Short-Form Health Survey (SF36), and body pain presence and severity were also assessed using questions from the SF36. Impaired color vision, depressive symptoms, and body pain were all defined as scores in the bottom 10% of women without pRBD or constipation. Parkinsonian symptoms were measured using a nine-item screening questionnaire that assessed self-reported changes in motor function [15]; the number of changes in motor function were summed to calculate a motor score. Among 51 HPFS and NHS participants assessed in person by neurologists specializing in movement disorders, the Spearman correlation coefficient between this score and the MDS-UPDRS part 2 (“Motor experiences of daily living”) was 0.82 (p < 0.0001), and with part 3 (“Motor examination”) was 0.66 (p < 0.0001)[5]. Parkinsonism was defined as reporting at least 4 changes in motor function, which has been shown to have a sensitivity of 90% and a specificity of 94% for neurologist-confirmed parkinsonism [16].
Statistical analysis
In women without PD, we used logistic regression to calculate age-adjusted odds ratios (ORs) between individual prodromal features, and between prodromal features and probable parkinsonism. Logistic regression was also used to calculate age-adjusted ORs for confirmed PD according to presence of prodromal features. We used inverse probability weighting to account our sampling strategy in which only a subset of women without pRBD or constipation were invited to participate in follow-up screening. Women were weighted by the inverse of their probability of being selected into the study, conditional on pRBD and constipation status. We plotted receiver operating characteristic (ROC) curves and estimated the area under the curve (AUC) for number of prodromal signs for classifying PD [17]. We considered two possible screening strategies for ROC analyses—first, screening for pRBD, constipation, and hyposmia only (0–3 signs), and second, additionally considering impaired color vision, body pain, excessive daytime sleepiness, and depressive symptoms (0–≥6 signs). Finally, we calculated positive predictive values (PPV) for PD at different cutoff points of number of prodromal signs for both of these screening strategies, assuming a prevalence of PD between 1–3%. All analyses were performed using SAS statistical software (SAS Institute, Inc., Cary, NC) or R version 3.4.0.
Data availability
Anonymized data will be shared by request from qualified investigators.
RESULTS
The prevalence of each non-motor feature in cases and controls are presented in Table 1. Each non-motor feature was significantly associated with PD in age-adjusted analyses, with ORs ranging from 2.64 for body pain to 13.15 for pRBD (Table 1). The stronger association for self-reported hyposmia as compared to the results of the olfactory test is consistent with the low sensitivity of self-reported hyposmia—only individuals with severe olfactory impairment and thus higher PD risk are aware of their deficit [18]. Among women without PD, non-motor features also tended to be positively associated with each other; the only exceptions were that impaired color vision was not associated with pRBD or body pain (Table 2).
Prevalence of non-motor features among PD cases and controls
Results are weighted using inverse-probability weights.
*Mean (SD). Value is not weighted.
Age-adjusted ORs (95% CIs) between individual non-motor features
752 women had missing data for impaired color vision data, 25 had missing data for excessive daytime sleepiness, 176 had missing data for body pain, and 95 had missing data for depressive symptoms.
Constipation, pRBD, and hyposmia
The odds of PD increased exponentially with additional prodromal features (Fig. 2A). Women with constipation, pRBD, and hyposmia had an age-adjusted OR of 211 (95% CI 84.2–529) for PD compared to women with none of these three features. These three signs were present in 30.5% of PD cases, but only 0.9% of controls. We plotted ROC curves (Fig. 3A) based on a score representing the number of prodromal features (pRBD, constipation, and hyposmia). Using this score, the AUC for PD was 0.839 (95% CI 0.785–0.888). An alternative score, where hyposmia measured using B-SIT was replaced with self-reported hyposmia, yielded an AUC for PD of 0.828 (95% CI 0.772–0.876). A different model that included variables for constipation, pRBD, and the olfactory score modeled continuously produced an AUC of 0.869 (95% CI 0.817–0.914). Figure 4A shows the relationship between number of prodromal signs and PPV for PD as a function of the prevalence of PD in a hypothetical screened population of women. Assuming a prevalence of PD of 2%, we estimate a PPV for PD of 40% using these three prodromal features.
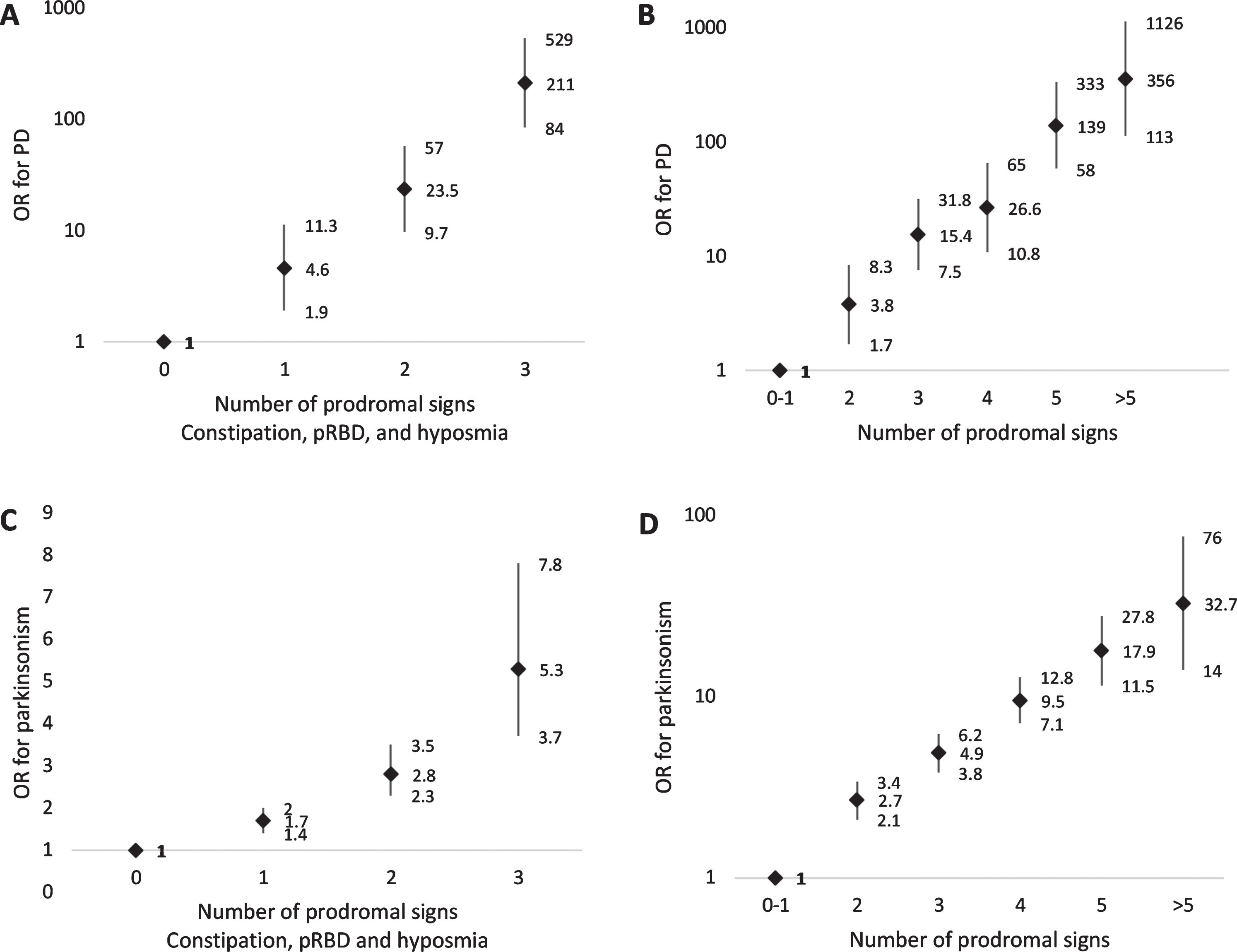
A) Age-adjusted associations between number of prodromal signs (including constipation, pRBD, and hyposmia) and PD. B) Age-adjusted associations between number of prodromal signs (including constipation, pRBD, hyposmia, impaired color vision, depressive symptoms, excessive daytime sleepiness, and body pain) and PD. C) Age-adjusted associations between number of prodromal signs (including constipation, pRBD, and hyposmia) and parkinsonism. D) Age-adjusted associations between number of prodromal signs (including constipation, pRBD, hyposmia, impaired color vision, depressive symptoms, excessive daytime sleepiness, and body pain) and parkinsonism.
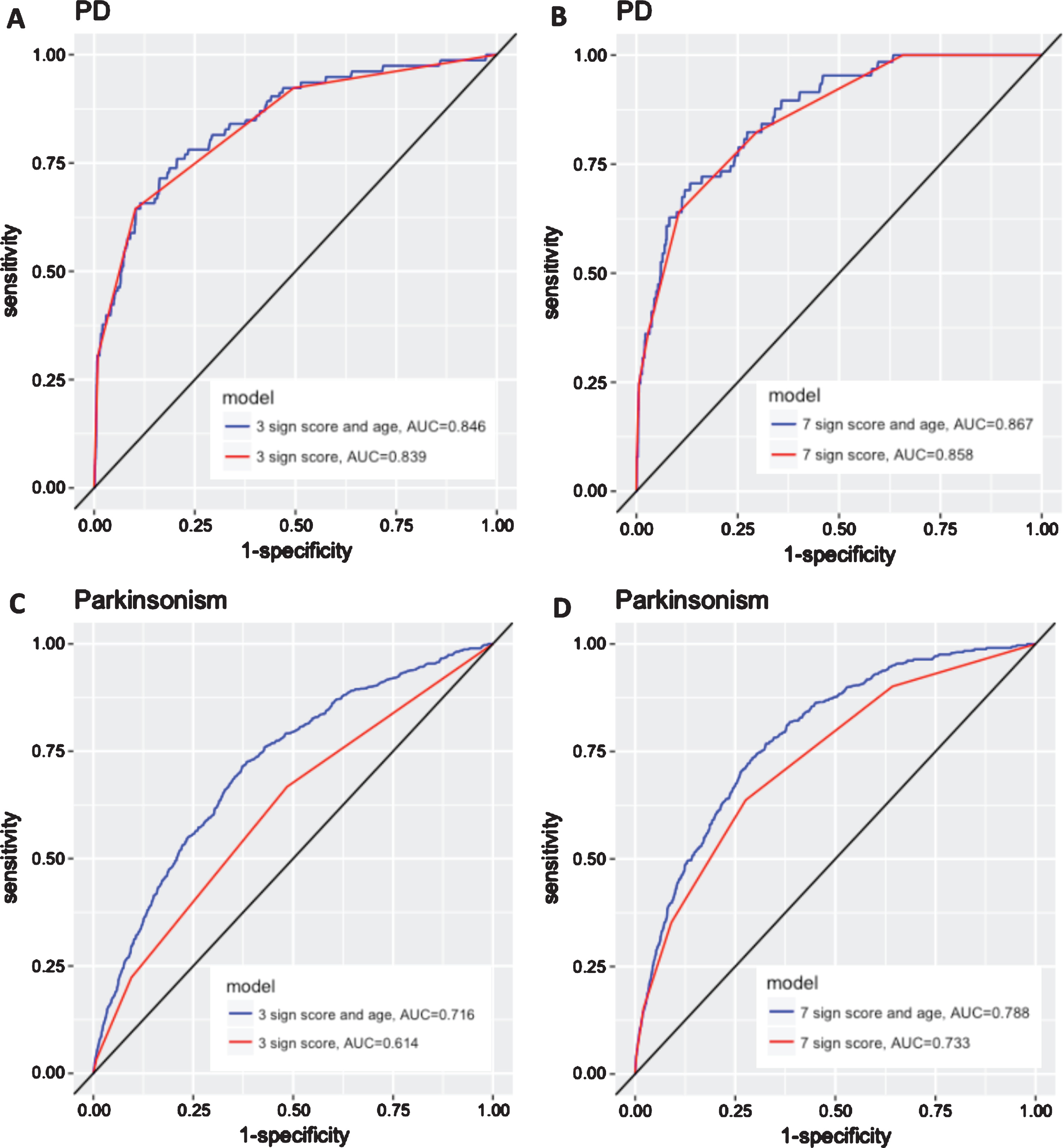
A) ROC curve for PD using model with variable for number of prodromal signs, including hyposmia, pRBD and constipation (0 to 3). B) ROC curve for PD using model with variable for number of prodromal signs, including hyposmia, pRBD, constipation, excessive daytime sleepiness, impaired color vision, body pain, and depressive symptoms (0 to ≥6). C) ROC curve for parkinsonism using model with variable for number of prodromal signs, including hyposmia, pRBD and constipation (0 to 3). D) ROC curve for parkinsonism using model with variable for number of prodromal signs, including hyposmia, pRBD, constipation, excessive daytime sleepiness, impaired color vision, body pain, and depressive symptoms (0 to ≥6).
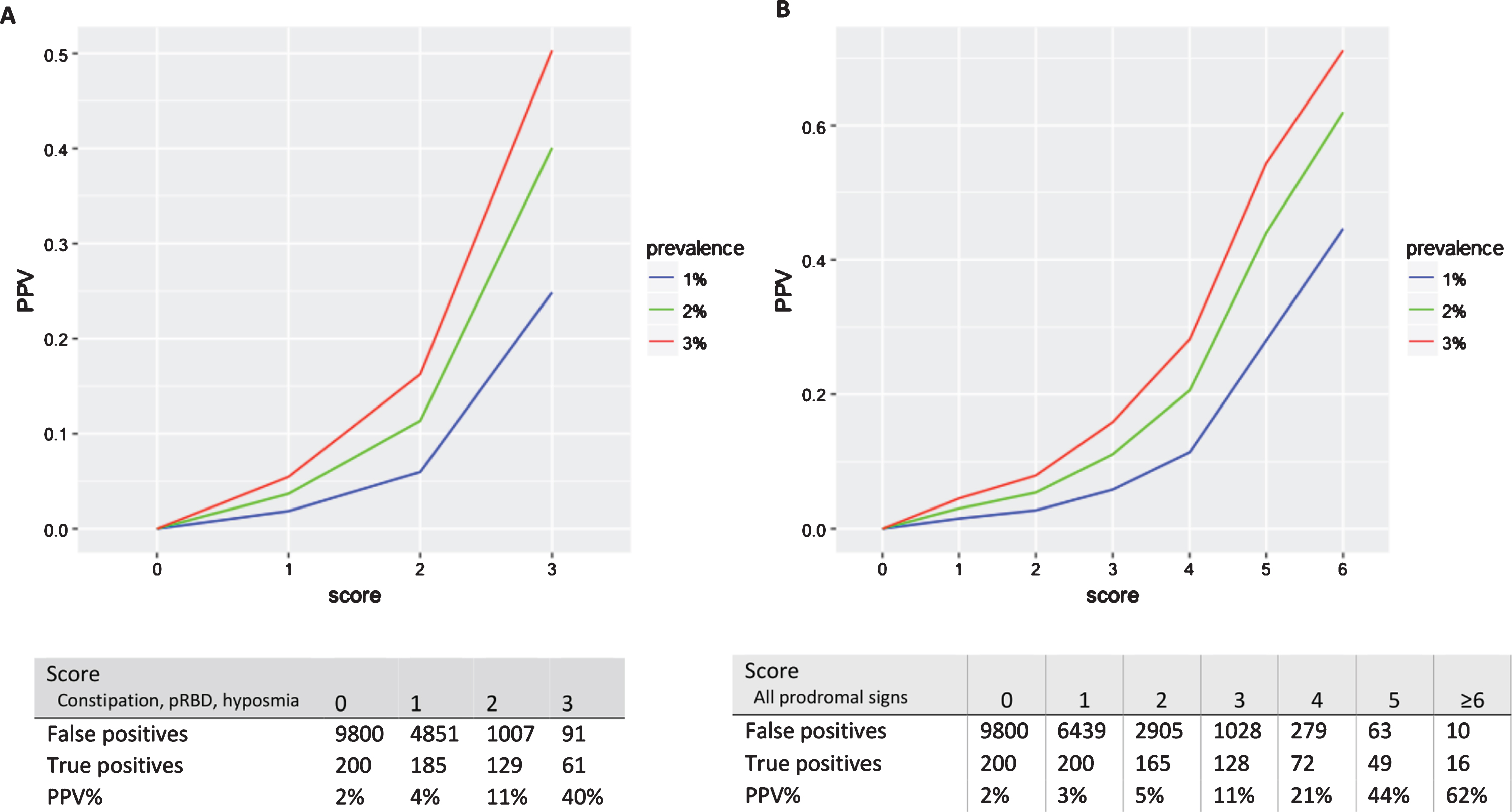
Relationship between PPV and score, assuming prevalence of PD of 1%, 2%, or 3%. A) Using strategy of screening for constipation and pRBD, followed by hyposmia testing. Table shows number of false positives, true positives, and PPV% using this screening strategy with a given cutoff point in a hypothetical cohort of 10,000 older women, assuming a prevalence of 2% (i.e., 200 true cases); the corresponding PPV is shown by the green line in graph above. B) Using strategy of screening for constipation and pRBD, followed by hyposmia, impaired color vision, excessive daytime sleepiness, depressive symptoms, and body pain screening. Table shows number of false positives, true positives, and PPV% using this screening strategy with a given cutoff point in a hypothetical cohort of 10,000 older women, assuming a prevalence of 2%; the corresponding PPV is shown by the green line in graph above.
Combined prodromal features
Other prodromal signs
The odds of PD rose further with the presence of additional prodromal features. Comparing women with at least 6 prodromal signs to women with one or no signs, the age-adjusted OR for PD was 356 (95% CI 113–1126) (Fig. 2B). At least 6 prodromal signs were present in 7.8% of PD cases, compared with only 0.1% of controls. ROC curves for PD based on number of prodromal signs (0 through≥6) are shown in Fig. 3B. AUC for PD was 0.858 (95% CI 0.817–0.895). When hyposmia measured using B-SIT was replaced with self-reported hyposmia, the AUC for PD was 0.840 (95% CI 0.793–0.884). A different model that included variables for constipation, pRBD, and the scores modeled continuously for the other five features yielded an AUC of 0.896 (95% CI 0.861–0.935). The positive predictive value for PD increased with increasing number of prodromal signs, to a maximum of 62% for women with at least 6 prodromal signs assuming a prevalence of PD of 2%. This PPV is more than 20% higher than that for the screening strategy described above only using 3 prodromal signs, but at the cost of identifying less than a third the number of true cases (Fig. 4B).
Associations between non-motor features and parkinsonism
1,042 (7.4%) women without diagnosed PD were classified as having parkinsonism. All of the non-motor features under study were significantly associated with parkinsonism, with ORs ranging from 1.71 to 4.23 (Table 3). We also examined associations between number of non-motor signs and having parkinsonism; the odds of parkinsonism increased with the number of non-motor features, but the increase was not as pronounced as that described for PD. The OR for parkinsonism for women with constipation, pRBD, and hyposmia was 5.35 (95% CI 3.67–7.80) compared to women with none of these three features. When we assessed additional non-motor signs, the OR for parkinsonism comparing women with at least 6 signs to those with one or none was 32.7 (95% CI 14.01–76.18) (Fig. 2C, D). The AUC for a model based on pRBD, constipation and hyposmia was 0.614 (95% CI 0.591–0.637) and increased to 0.733 (95% CI 0.711–0.759) when we included additional features (Fig. 3C, D).
Associations of non-motor features with screening positive for parkinsonism
DISCUSSION
In this large study of women, we found that concurrent hyposmia, pRBD, and constipation were strongly associated with having PD. The combination of these three features had high accuracy in classifying women as PD cases or controls (AUC 0.839); classification was modestly improved by adding additional non-motor features of PD (AUC 0.858). Assuming a prevalence of 2%, the PPV for PD for women with at least 6 non-motor signs was 62%. These results suggest that these features could be useful in discriminating PD patients from controls, and since these features often appear during the prodromal period of PD, their combinations may also prove useful for identifying populations at high risk of developing PD or in the earliest stages of neurodegeneration. Although the same non-motor features were also associated with parkinsonism, these associations were weaker than those for PD, a finding consistent with the heterogeneity of parkinsonism, which could be drug-induced or caused by vascular disease.
Overall, results of this study closely mirror previously reported results in a large cohort of men, the Health Professionals Follow-up Study [5]. The associations between combined pRBD, hyposmia, and constipation with PD were similar between men and women—OR of 160 (95% CI 72.8–353) in men compared to 211 (95% CI 84.2–528) in women for having concurrent pRBD, hyposmia, and constipation versus none of these three features. The presence of additional non-motor signs was strongly associated with PD in both cohorts—OR for ≥6 versus 0 signs of 1,325 (95% CI 333–5,279) in men, compared to OR for ≥6 versus 0-1 signs of 356 in women (95% CI 113–1126). Both the model featuring pRBD, constipation, and hyposmia and the model featuring additional non-motor signs performed modestly better among men than women at classifying PD cases from controls (AUC of 0.850 in men versus 0.839 in women for 3 sign score; AUC of 0.879 in men versus 0.858 in women for score including additional signs). For individual non-motor features, although the confidence intervals overlap, the association between hyposmia and PD appeared to be weaker in women—OR of 8.84 (95% CI 5.58–13.99) versus 19.03 (95% CI 11.80–30.68) in men, as was the association between impaired color vision and PD—2.69 (95% CI 1.59–4.55) versus 5.80 (95% CI 3.86–8.71) in men, whereas the association between pRBD and PD was stronger—OR of 13.15 (95% CI 8.56–20.21) in women versus 7.98 (95% CI 5.52–11.53) in men. Otherwise, associations were similar between men and women. The similarities of our results in two separate cohorts, both in men and women, lend further support to the results and suggest that they are likely to generalize to other similar populations of older individuals.
Sex differences in non-motor features of PD have been reported previously. In the Parkinson’s Progression Markers Initiative (PPMI) Study, a large multicenter study designed to identify PD progression biomarkers, male PD patients performed worse than females on odor identification, while women experienced higher trait anxiety [19] and depressive symptoms [20]. Male and female patients also differed on certain cognitive domains, with women performing better on global cognition and memory, and men performing better on visuospatial tasks. In another study of US farmers and their spouses, there was a suggestion that the associations of hyposmia and infrequent bowel movement with PD were both modified by sex [3]. In addition, while present in both sexes, the dose-response relationship between number of non-motor symptoms and PD was stronger among men than women.
Other studies that assessed combinations of non-motor features in relation to PD were restricted to men [21] or did not report results separately in men and women [22–25]. In the Honolulu-Asia Aging Study, a cohort comprising only men, PD incidence increased from 16 cases per 10,000 person-years for participants without non-motor features to 215 cases per 10,000 person-years for participants with 3 or more features (out of impaired olfaction, constipation, slow reaction time, and excessive daytime sleepiness) [21]. In the Prospective evaluation of Risk factors for Idiopathic Parkinson’s Syndrome (PRIPS) study, a population-based cohort of 1,847 PD-free individuals age 50 or older at baseline, a PPV for PD over three years of follow-up of 25% was found by combining multiple risk markers (hyposmia, UPDRS motor score, and hypoerechogenicity of the substantia nigra). However, sensitivity using this approach was low—only 2 out of 11 individuals who developed PD exhibited these three markers at baseline [22]. In the Parkinson Associated Risk Study (PARS), individuals with and without a family history of PD were recruited to complete the University of Pennsylvania Smell Identification Test (UPSIT), and those with hyposmia plus a normosmic control group were subsequently evaluated with dopamine transporter (DAT) imaging. Out of 21 participants with both hyposmia and a DAT deficit at baseline, 14 (67%) converted to PD at 4 years follow-up [23]. In the Parkinson’s Progression Marker Initiative (PPMI) study, a model using UPSIT score, a genetic risk score, family history of PD, sex, and age achieved an AUC of 0.923 discriminating participants with PD from healthy controls [24]. Finally, a recent analysis from the Health, Aging, and Body Composition Study found that the hazard ratio (95% CI) for PD among participants with both a self-reported poor sense of smell and an objectively measured poor sense of smell, compared to those who reported and tested normal, was 7.8 (3.2, 19.4). In comparison, the hazard ratios were 2.2 (1.0, 4.6) for self-reported poor smell only and 3.6 (1.9, 6.9) for objectively measured poor smell only [25].
Our study is unique in that we assessed several non-motor features of PD among a large, well established cohort of women. The measures used were well-validated, simple, and inexpensive, and could thus be scaled up for use on a larger population at minimal cost. One limitation of our study is that the Nurses’ Health Study is a relatively homogeneous population of nurses; it is possible that the assessments used in this study would not perform as well in other populations. In addition, some self-reported measures are inevitably measured with error, which could result in under estimation of the associations. Another important limitation is that the results reported are cross-sectional, and therefore it cannot be determined from our data when these non-motor features onset during the course of PD progression. For this reason, prospective studies will be necessary to better determine the sensitivity, specificity, and PPV of these non-motor features.
Footnotes
ACKNOWLEDGMENTS
This study was supported by Department of Defense grant W81XWH-14-0131. The NHS cohort is funded by NIH grant UM1 CA186107.
CONFLICT OF INTEREST
The authors have nothing to report.
Dr. Hughes is an employee of Optum.
X. Gao has served on a committee of the Parkinson Study Group and received funding from the NIH/National Institute of Neurological Disorders and Stroke.
Dr. Baker reports no disclosures.
Dr. Stephen received honoraria from serving on the scientific advisory board of Xenon Pharmaceuticals. Dr. Stephen has received research support (7% effort) from Sanofi-Genzyme for a study of video oculography in late-onset GM2 gangliosidosis. Dr. Stephen has received financial support from Pfizer, Sanofi-Genzyme, Bristol-Myers Squibb, Biogen and Biohaven for the conduct of clinical trials.
Dr. Kim reports no disclosures.
Dr. Valeri reports no disclosures.
Dr. Schwarzschild is an employee of Massachusetts General Hospital and a member of the faculty of Harvard Medical School, and is supported by research funding (from the National Institutes of Health, Michael J Fox Foundation, Parkinson Foundation, Target ALS, Maximilian E. & Marion O. Hoffman Foundation, Farmer Family Foundation, Biotie/Acorda and the US Department of Defense), clinical practice compensation, an endowment and University funds. He serves on the scientific advisory boards of the Michael J Fox Foundation, CorticoBasal Degeneration Solutions, Collaborative Center for the X-linked Dystonia Parkinsonism, and Cure Parkinson’s Trust. He has served as an advisory board member/consultant for Denali Therapeutics, Eli Lilly & Company, nQ Medical, and Prevail Therapeutics. He has received royalties from MGH for licensing of adenosine A2A receptor knockout mice.
Dr. Ascherio serves on the editorial board of the Journal of Parkinson disease, is funded by Department of Defense research grants W81XWH1410131 and W81XWH1810341, and by NIH research grants R01NS089619 and R01NS103091. He has received research support from the National MS Association and the ALS Association.
