Abstract
Background:
Emerging evidence suggests that gut dysbiosis contributes to Parkinson’s disease (PD) by signaling through microbial metabolites. Hippuric acid (HA), indole derivatives, and secondary bile acids are among the most common gut metabolites.
Objective:
To examine the relationship of systemic concentrations of these microbial metabolites associated with changes of gut microbiota, PD status, and severity of PD.
Methods:
We enrolled 56 patients with PD and 43 age- and sex-matched healthy participants. Motor and cognitive severity were assessed with Movement Disorder Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) part III motor score and the Mini-Mental State Examination (MMSE), respectively. Plasma concentrations of targeted gut metabolites were measured with liquid chromatography-tandem mass spectrometry. Gut microbiota was analyzed with shotgun metagenomic sequencing.
Results:
Compared with controls, PD patients had significantly higher plasma levels of HA, indole-3-propionic acid (IPA), deoxycholic acid (DCA), and glycodeoxycholic acid (GDCA). After adjustment for age and sex in a multivariate logistic regression analysis, plasma levels of HA (odds ratio [OR] 3.21, p < 0.001), IPA (OR 2.59, p = 0.031), and GDCA (OR 2.82, p = 0.036) were associated with positive PD status. Concentrations of these gut metabolites did not correlate with MDS-UPDRS part III score or MMSE after adjustment for confounders. Microbial metabolite levels were associated with the relative abundance of pro-inflammatory gut bacteria.
Conclusion:
Aberrant gut microbial metabolites of HA, indole derivatives and secondary bile acids associated with specific gut microbiota changes were observed in patients with PD.
Keywords
INTRODUCTION
Emerging evidence suggests that the pathological hallmarks of Parkinson’s disease (PD), intraneuronal aggregation of α-synuclein forming Lewy bodies, may originate in the gut enteric nervous system and then ascend through the vagus nerve to the brainstem through cell–cell transmission [1, 2]. These findings are consistent with clinical observations that gastrointestinal dysfunction is one of the most common preceding symptoms, possibly appearing up to decades before the onset of characteristic motor symptoms [3]. In addition, PD patients have an altered gut microbiota composition compared with unaffected controls, and changes in the gut microbiota correlate with distinct disease phenotypes and motor symptom severity in patients with PD [4, 5]. These observations have prompted the hypothesis that PD is caused by an altered gut environmental trigger leading to increased gut permeability. This permeability in turn allows for leakage of bacteria and their potential metabolites into the systemic circulation and promotes central dopaminergic neurodegeneration through the gut–brain axis [6].
Microbial metabolites are small molecules synthesized from dietary compounds and produced by gut microbiota to interact with hosts [7]. As the main communicating signals between gut microbiota and hosts, gut microbial metabolites and their pivotal impacts on PD pathogenesis are being gradually unraveled [7]. Alterations in gut microbial metabolites, including trimethyl-N-oxide, short-chain fatty acids, and catechol sulfate, have been observed in PD patients and are reported to be associated with disease severity and progression [7, 8]. Of note, in a mouse model of PD, short-chain fatty acids have been implicated in promoting α-synuclein pathology and neurodegeneration by activating neuroinflammation [9]. Additional metabolites are the lipophilic microbial metabolites that could cross the blood–brain barrier. These include tryptophan-derived indole derivatives, secondary bile acids from bacterial conversion of bile acids in the colon, and hippuric acid (HA), which is produced by microbial degradation of dietary aromatic compounds in the intestine. Because of their lipophilicity, these metabolites have the potential to serve as signaling molecules in regulating inflammatory responses and metabolic homeostasis [10–12]. Increased cerebrospinal fluid or plasma levels of HA and the indole derivative indoleacetic acid (IAA) have been reported in patients with PD compared with controls [13, 14], although conflicting results existed [15, 16]. Additionally, a correlation between cerebrospinal fluid IAA levels and the progression of PD has been described [14], but IAA also paradoxically has been shown to reduce pro-inflammatory cytokine production by lipopolysaccharide-stimulated macrophages [17]. Clarifying these inconsistent findings requires an independent cohort study with a large number of enrolled participants to elucidate these candidate gut metabolic pathways associated with disease risk and progression in PD.
We previously have established a long-term follow-up cohort of PD patients and age, sex-, and diet-matched controls as a way to identify several gut-oriented biosignatures for PD progression, including plasma levels of trimethyl-N-oxide and short-chain fatty acids [18–20]. Hence, in the current study, we aim to analyze and compare plasma levels of HA, indole derivatives, and secondary bile acids in PD patients and unaffected controls to delineate the interrelations of these metabolite levels and their potential links to PD status and severity and gut microbiota changes.
METHODS
Participants
A total of 56 PD patients and 43 control parti-cipants were recruited from National Taiwan University Hospital. PD was diagnosed according to the UK PD Society Brain Bank Clinical Diagnostic Criteria [17]. Controls were neurologically unaffected spouses, family members, or accompanying friends of the PD patients. Participants were excluded if they were vegetarians, had taken antibiotics or probiotics within 3 months of enrollment, or had irritable bowel syndrome, inflammatory bowel disease (IBD), colon cancer, or colitis. The institutional ethics board committees of National Taiwan University Hospital approved the study. The experiments and assays were in accord with the Helsinki Declaration of 1975. Informed consent was obtained from all participants. All PD patients and controls were matched for diet habits using the Food Frequency Questionnaire and had undergone 16S ribosomal RNA sequencing, as previously published [18].
Clinical evaluation
We assessed clinical motor severity with Movement Disorder Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) part III motor scores [19] and with Hoehn and Yahr (H-Y) staging for patients with PD [20]. At “off” phase, patients were defined as early-stage PD when the H-Y stage was < 3 and as advanced-stage PD when H-Y severity was≥3. We evaluated cognitive function with the Mini-Mental State Examination (MMSE) during the “on” phase [21]. Patients who had PD with dementia (PDD) were diagnosed according to the criteria proposed by the Movement Disorder Society [22]. We applied MMSE scores of 25 or less as the cut-off value for significant cognitive impairment, combined with any impairment in the eight instrumental activities for the diagnosis of PDD [23]. Levodopa equivalent daily dose (LEDD) was calculated for all participants.
Measurement of plasma levels of gut microbial metabolites
For blood samples, 10 mL of venous blood after fasting for over 8 h was drawn from each participant in the morning. Plasma concentrations of microbial metabolites including HA, tryptophan, and tryptophan-derived indole derivatives (indolelactic acid (ILA), indole-3-acetic acid (IAA), and indole-3-propionic acid (IPA)), and secondary bile acids (cholic acid (CA), glycocholic acid (GCA), chenodeoxycholic acid (CDCA), glycochenodeoxycholic acid (GCDCA), deoxycholic acid (DCA), glycodeoxycholic acid (GDCA), ursodeoxycholic acid (UDCA), and glycoursodeoxycholic acid (GUDCA)) were measured with liquid chromatography (LC)-tandem mass spectrometry (MS/MS). Each analyte was measured twice in each sample, and the mean value of the two measurements was used for analysis.
LC-MS/MS was performed as previously des-cribed [21]. In brief, 60μL of plasma was extracted with 420μL of methanol using a Geno/Grinder 2010 (SPEX SamplePrep, Metuchen, NJ, USA). We performed derivatization of the supernatant with 30μL of 0.2 M 3-nitrophenylhydrazine and 30μL of 0.12 M 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (40°C, 20 min). After derivatization, isotopically labeled internal standards were added before LC-MS/MS analysis was performed. Metabolites and corresponding internal standards were analyzed using an Agilent 6470 triple quadrupole mass spectrometer (Agilent Technologies, Santa Clara, CA, USA). The separation was performed on a ZORBAX Eclipse Plus C18 column (2.1 mm×100 mm, 1.8μm) with a mobile phase consisting of 0.1% formic acid in water (solvent A) and 0.1% formic acid in 2-propanol:ACN (3:1, v:v) (solvent B) at a flow rate of 0.35 mL min–1. The LC gradient profile was as follows: 0–0.5 min, 0% B; 0.5–1.7 min, 0% –15% B; 1.7–3.5 min, 15% B; 3.5–9.0 min, 15% –30% B; 9.0–9.1 min, 30% –40% B; 9.1–16.0 min, 40% –72% B; 16.0–16.1 min, 72% –80% B; 16.1–16.2 min, 80% –100% B; 16.2–18.2 min, 100% B; and 18.2–20.0 min, 0% B. The injection volume was 5μL. The negative electrospray ionization mode was used with the following parameters: 325°C for drying gas temperature, 7 L min–1 for drying gas flow, 45 psi for nebulizer pressure, 325°C for sheath gas temperature, 11 L min–1 for sheath gas flow rate, and 2500 V for capillary voltage. Nozzle voltage was set at 1500 V. Concentrations of each analyte in the samples were determined from calibration curves using the peak area ratio of the analyte to its corresponding isotope internal standard.
Gut microbiota analyses with shotgun metagenomic sequencing
Fresh feces from each participant were collected into stool specimen collection tubes containing DNA stabilizer (Sarstedt), which were immediately flash-frozen on dry ice and stored at –80°C before analyses. The feces were collected within one week after venous blood sampling. Total fecal DNA was extracted using a QIAamp DNA Stool Mini Kit (Qiagen, Hilden, Germany) as previously described.[18] The metagenomic Illumina paired-end reads for both case and control samples were trimmed using Trimmomatic v0.39 48 with parameters “PE -phred33 ILLUMINACLIP:TruSeq3-PE.fa:2:30:10 LEADING:12 TRAILING:12 SLIDINGWINDOW:4:15 MINLEN:36” [22]. We then searched the trimmed paired-end reads of the samples for their prokaryotic species profiles using Kraken2 v2.0.6-beta 49 [23]. To maximize the search efficiency, we used the maxikraken2 database (total size 140 GB; constructed by Daniel Fischer at Natural Resources Institute Finland, available at https://lomanlab.github.io/mockcommunity/mc_databases.html) instead of the default kraken2 database. The resulting microbial species proportion for all case and control samples was collected and analyzed using Student’s t-tests to identify microbial species that were significantly different (p < 0.05) between case and control samples. Only species with a mean abundance of at least 0.1% in either case or control (but not necessarily both) were included. We performed all analyses with R software 3.4.1 [24].
Statistical analysis
Continuous variables are expressed as mean±standard deviation and categorical factors as number (percentage). The homogeneity of variances was tested by using Levene’s test. Variables that followed a Gaussian distribution were compared with two-tailed t-tests or analysis of variance (ANOVA). For variables that violated the assumptions of normality or homoscedasticity, the groups were compared with the non-parametric Mann–Whitney U test or the Fisher’s exact test. Plasma concentrations of microbial metabolites were square root-transformed to achieve normal distribution before further analyses. We applied logistic regression models to examine the associations between microbial metabolites and PD status with adjustment for age and sex. Area under the receiver operating characteristic (ROC) curve (AUC) was performed to quantify the model’s diagnostic performance for exploring the ability of microbial metabolites to distinguish PD patients from healthy controls. To examine the associations between microbial metabolites and disease severity as measured by MDS-UPDRS part III motor score at “off” phase or MMSE score, we applied multivariate linear regression models with adjustment for age, sex, disease duration, and LEDD.
RESULTS
We enrolled 99 participants, including 56 PD patients (67.3±11.9 years old, 61% male) and 43 healthy controls (66.2±8.4 years old, 63% male). The clinical characteristics and plasma microbial metabolite concentrations of all participants were listed in Table 1. There were no significant differences between groups in demographic distribution, medical comorbidities, or body mass index. Among PD patients, 6 (10.7%) patients were clinically diagnosed with PDD.
Clinical characteristics and plasma concentrations of microbial metabolites in all participants
Variables are expressed as mean±standard deviation or number (percentage). IBD, inflammatory bowel disease; IBS, irritable bowel syndrome; LEDD, levodopa equivalent daily dose; MDS-UPDRS, Movement Disorder Society Unified PD Rating Scale; MMSE, Mini-Mental State Examination; N.A., not available. *p < 0.05; **p < 0.01.
Plasma levels of gut microbial metabolites in PD patients and controls
Patients with PD had significantly higher levels of HA than control participants (4.28±5.11 vs. 1.70±1.88μM, p < 0.001; Fig. 1A). For tryptophan and tryptophan-derived indole derivatives, PD patients had increased levels of IPA (1.26±1.43 vs. 0.83±1.02μM, p = 0.013) compared with controls, whereas plasma concentrations of tryptophan, ILA, and IAA were comparable between groups (Fig. 1B). For secondary bile acids, plasma levels of DCA (0.39±0.52 vs. 0.27±0.38μM, p = 0.037) and GDCA (0.69±0.84 vs. 0.46±0.72μM, p = 0.034) were elevated in PD patients compared with controls (Fig. 1C). We also examined whether age had any influence on plasma microbial metabolite levels. With the exception of a modest correlation of plasma HA with age (ρ= 0.269, p = 0.007), age did not correlate with either measure for the individual metabolites.
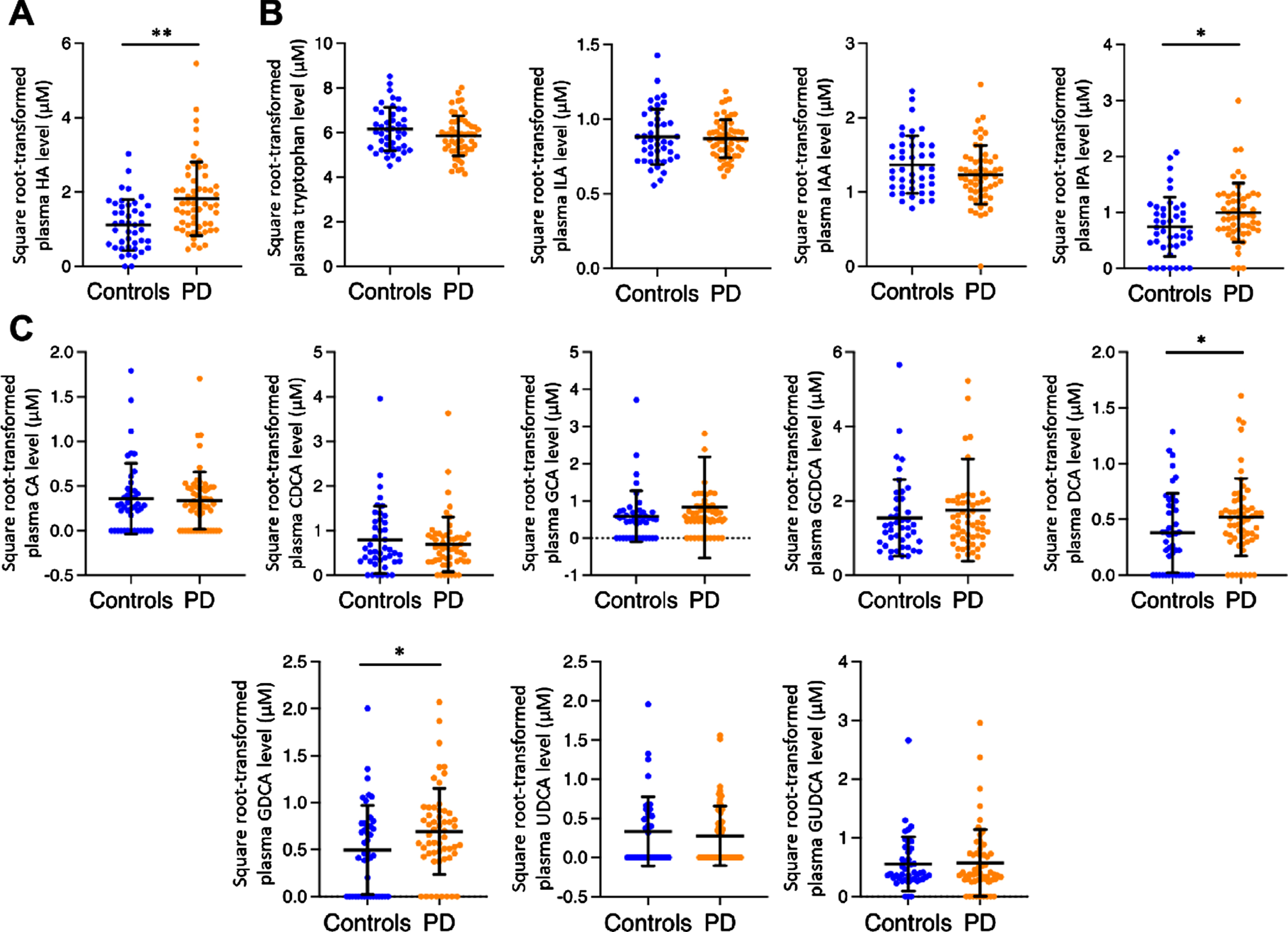
Plasma levels of microbial metabolites in PD patients and healthy controls. The scatterplots display data density and means with standard deviations of plasma levels of hippuric acid (A), tryptophan-related indole derivatives (B), and secondary bile acids (C) in PD patients and healthy controls. Plasma levels of targeted gut metabolites are square-root transformed. *p < 0.05, **p < 0.01. HA, hippuric acid; ILA, indolelactic acid; IAA, indole-3-acetic acid; IPA, indole-3-propionic acid; CA, cholic acid; GCA, glycocholic acid; CDCA, chenodeoxycholic acid; GCDCA, glycochenodeoxycholic acid; DCA, deoxycholic acid; GDCA, glycodeoxycholic acid; UDCA, ursodeoxycholic acid; GUDCA, glycoursodeoxycholic acid.
We next investigated whether microbial metabolites in systemic circulation could distinguish PD patients from controls. After adjustment for age and sex, multivariate logistic regression analyses showed an association between PD status and higher plasma concentrations of HA (odds ratio [OR] 3.21, 95% confidence interval [CI] 1.75–6.58, p < 0.001; Table 2), IPA (OR 2.59, 95% CI 1.14–6.53, p = 0.031), and GDCA (OR 2.82, 95% CI 1.12–7.87, p = 0.036). Elevated plasma concentrations of DCA (OR 3.45, 95% CI 1.05–12.89, p = 0.051; Table 2) showed a trend to associate with PD status.
Associations between plasma microbial metabolites and PD status in all participants after adjusting age and sex
Multivariate logistic regression models were applied to examine the associations between microbial metabolites and PD status. In the models, presence of PD was set as dichotomous endpoint. The covariates included age, sex, and square root-transformed microbial metabolites. CI, confidence interval; OR, Odds ratio. *p < 0.05; **p < 0.01.
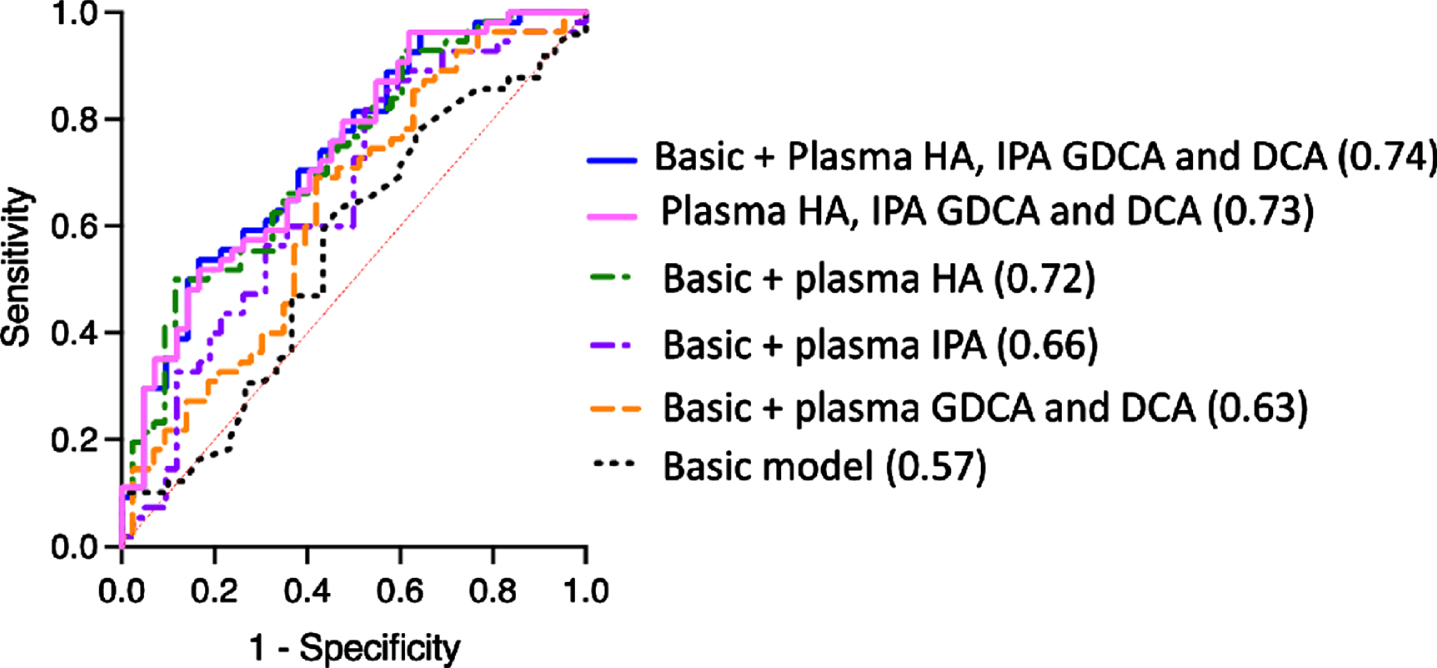
Based on the results of the aforementioned multiple logistic regression model, we performed ROC curve analysis to predict PD status. The prediction accuracy, as expressed by AUC, improved from 0.57 in the basic model containing age and sex only (p = 0.273) to 0.63 with the addition to the model of plasma levels of the targeted secondary bile acids GDCA and DCA (p = 0.033) (Fig. 2 and Table 3); to 0.66 with the addition of plasma IPA levels (p = 0.009); and to 0.72 after incorporation of plasma HA levels (p < 0.001). Notably, the prediction accuracy was 0.73 in the model only containing plasma levels of HA, IPA, GDCA, and DCA (p < 0.001). The AUC further increased to 0.74 in the full model containing age, sex, and plasma concentrations of HA, IPA, DCA, and GDCA (p < 0.001) (Fig. 2).
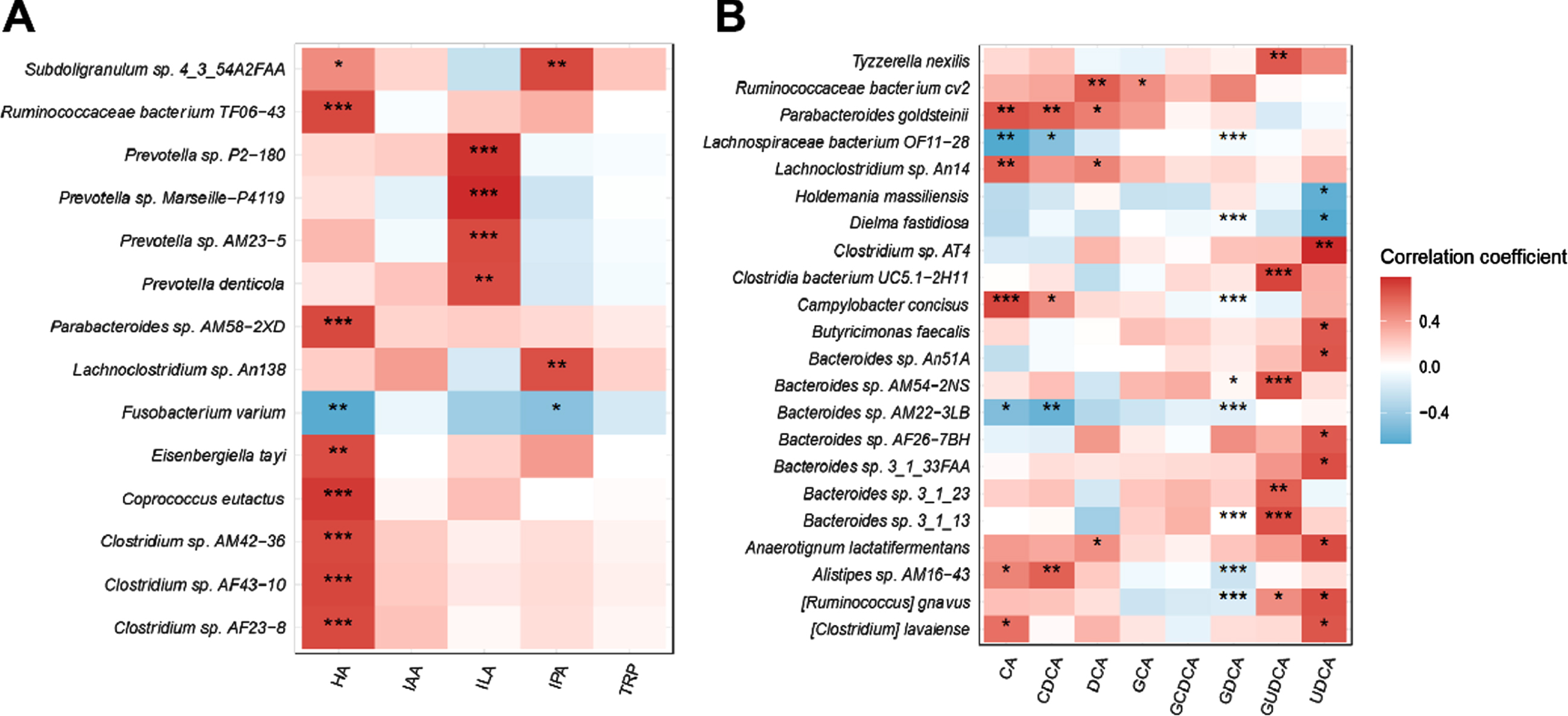
The correlations between plasma levels of targeted microbial metabolites in patients with PD. Heat maps representing the Spearman correlation of the relative abundance of different bacteria and the plasma levels of HA, tryptophan-related indole derivatives (A), and secondary bile acids (B). *p < 0.05, **p < 0.01. PD, Parkinson’s disease; HA, hippuric acid; IAA, indole-3-acetic acid; ILA, indolelactic acid; IPA, indole-3-propionic acid; CA, cholic acid; CDCA, chenodeoxycholic acid; DCA, deoxycholic acid; GCA, glycocholic acid; GCDCA, glycochenodeoxycholic acid; GDCA, glycodeoxycholic acid; GUDCA, glycoursodeoxycholic acid; UDCA, ursodeoxycholic acid.
Receiver operating characteristic curve analysis for differentiating PD patients from healthy controls
Variables in the full model were square root-transformed plasma levels of HA, IPA, GDCA, and DCA in addition to age and sex. AUC, area under the curve; CI, confidence interval; DCA, deoxycholic acid; GDCA, glycodeoxycholic acid; HA, hippuric acid; IPA, indole-3-propionic acid. *p < 0.05; **p < 0.01.
Plasma levels of microbial metabolites and disease severity in PD
For motor symptom severity, plasma levels of ILA were modestly increased in PD patients in the advanced-stage group compared with those who had early-stage PD (0.73±0.21 vs. 0.85±0.25μM, p = 0.041), but plasma levels of other gut metabolites did not differ between the groups. Furthermore, we found that plasma levels of all targeted microbial metabolites did not correlate with motor symptom severity. In multivariate linear regression models, after correction for age, sex, disease duration, and LEDD, plasma concentrations of HA, tryptophan, indole derivatives, and secondary bile acids were not associated with MDS UPDRS part III motor scores (Supplementary Table 1).
For cognitive symptom severity, we found that plasma concentrations of all targeted microbial metabolites were comparable between PD patients with normal cognition and patients with PDD. Using multivariate linear regression analyses adjusting for age, sex, disease duration, and LEDD, we found that plasma levels of HA, tryptophan, indole derivatives, and secondary bile acids did not correlate with MMSE scores (Supplementary Table 1).
Correlation of gut microbiota species with plasma levels of microbial metabolites
We next explored correlations between the relative abundance of gut microbiota and plasma levels of targeted microbial metabolites, including HA, IPA, DCA, and GDCA, which showed differences between PD and control groups. The heat map representing Spearman correlations for the relative abundance of differential bacteria and plasma concentrations of gut metabolites revealed that plasma levels of HA positively correlated with the relative abundance of Ruminococcaceae bacterium TF06–43, Parabacteroides sp. AM58–2XD, Eisenbergiella tayi of family Lachnospiraceae, Coprococcus eutactus, and Clostridium sp. AM42–36, AF43–10, and AF23–8 of family Clostridiaceae (Fig. 3A). Plasma IPA levels correlated with the relative abundance of Subdoligranulum sp. 4_3_54A2FAA and Lachnoclostridium sp. An138 of family Lachnospiraceae. For the secondary bile acids, plasma levels of DCA correlated with the relative abundance of Ruminococcaceae bacterium cv2 of family Ruminococcaceae, Lachnoclostridium sp. An14 and Anaerotignum lactatifermentans of family Lachnospiraceae, and Parabacteroides goldsteinii. Plasma GDCA concentrations were inversely associated with Ruminococcus gnavus and Lachnospiraceae bacterium OF11-28 of family Lachnospiraceae, and Alistipes sp. AM16-43 and Bacteroides sp. AM22-3LB of order Bacteroidales (Fig. 3B).
ROC curves for distinguishing PD patients from controls. The ROC curve indicating the accuracy of discriminating PD using age and sex alone (AUC = 0.57, p = 0.273) improved with the addition of plasma levels of GDCA and DCA (AUC = 0.63, p = 0.033), IPA (AUC = 0.66, p = 0.009), or HA (AUC = 0.72, p < 0.001). The prediction accuracy was 0.73 in the model only containing plasma levels of HA, IPA, GDCA, and DCA (AUC = 0.73, p < 0.001). The accuracy of estimating PD development further improved in a full model incorporating plasma levels of targeted microbial metabolites, which showed differences between PD and controls (AUC = 0.74, p < 0.001). PD, Parkinson’s disease; HA, hippuric acid; IAA, indole-3-acetic acid; ILA, indolelactic acid; IPA, indole-3-propionic acid; CA, cholic acid; CDCA, chenodeoxycholic acid; DCA, deoxycholic acid; GCA, glycocholic acid; GCDCA, glycochenodeoxycholic acid; GDCA, glycodeoxycholic acid; GUDCA, glycoursodeoxycholic acid; UDCA, ursodeoxycholic acid.
DISCUSSION
Our study demonstrated that the microbial metabolites HA, IPA, and secondary bile acids, including DCA and GDCA, are increased in the systemic circulation of PD patients compared with unaffected controls. After adjustment for cofounders, the increased plasma levels of these four microbial metabolites were associated with positive PD status and with changes in specific gut microbiota, albeit without correlating with symptoms severity in PD.
HA is one of the major host–microbiota co-metabolites [12], mainly produced in liver and kidney by glycine conjugation of benzoic acid formed by gut microbiota from dietary polyphenolic compounds [12]. In our study, plasma concentrations of HA were significantly higher in PD patients, which is in line with a previous study showing increased HA concentrations in the serum of PD patients [25]. However, one recent serum metabolomic study of PD patients with PRKN mutations revealed reduced concentrations of benzoate-related metabolites, including HA [26]. In addition, gut microbiota components and metabolomic profiling differ between patients with hereditary PD and those with idiopathic PD [27, 28]. Loss-of-function mutation in PRKN gene leads to dysregulated mitochondrial function and impaired mitophagy, which may result in impaired glycine conjugation of benzoic acid, leading to reduced HA production in those PD patients with PRKN mutations [29, 30]. In support of the findings in the current study, another serum metabolomic study in eight drug-naïve patients with idiopathic PD also showed higher plasma HA concentrations in the patients compared with controls [31]. Although the physiological function of HA remains largely elusive, it has been linked to metabolic syndrome, depression, anxiety, and IBD [12]. IBD is associated with an increased risk for PD with age [32], and PD is hypothesized to be caused by an altered gut microenvironmental trigger leading to gut hyperpermeability and allowing leakage of bacteria and their metabolites into the systemic circulation to promote neurodegeneration [6]. Further in vitro and in vivo research is needed to explore the molecular mechanisms of HA in neuronal function.
Indole derivatives are degradation products of tryptophan, which is absorbed from a protein-based diet mainly by commensal bacteria in the colon [11]. In our population, plasma levels of IPA were increased in PD patients compared with controls, which is consistent with a recent European study showing elevated plasma IPA concentrations in drug-naïve PD patients [31]. IPA is a ligand of the aryl hydrogen receptor and pregnane X receptor, which regulates intestinal barrier integrity, neutralizes hydroxyl radicals, and modulates gut immunity and systemic inflammation [33, 34]. Although studies have shown that IPA has anti-inflammatory and anti-oxidative effects [11, 36], emerging in vitro and in vivo studies have shown that high-dose IPA could induce mitochondrial dysfunction with suppressed mitochondrial respiration and prolonged stimulation of the aryl hydrogen receptor, which was related to oncogenic potential [37, 38]. In addition to IPA, previous studies have shown reduced plasma IAA and ILA in PD patients compared with controls [15, 39], which we did not observe in our work. We also found that plasma concentrations of tryptophan, the precursor of indole derivatives, were comparable between PD patients and controls. These results highlight the importance of changes in specific gut microbiota resulting in different patterns of changes in individual subtypes of indole-derivatives. Further studies are warranted to investigate the physiological functions of individual subtypes of indole-derivatives.
Bild acids are divided into primary and secondary forms. Primary bile acids are produced in the hepatocytes, whereas secondary bile acids are formed by modification of the primary bile acids in the intestinal lumen [10]. In the intestine, primary bile acids are deconjugated by bacterial flora to form secondary bile acids, and then are re-conjugated again with glycine or taurine during recycling [10]. In this study, we observed that PD patients had higher plasma levels of the secondary bile acids DCA and GDCA, whereas plasma concentrations of primary bile acids were similar to controls. Studies on comparing fecal levels of secondary bile acids in PD patients and unaffected controls are lacking. One recent study analyzing the abundance of bile acids in ileum and appendix showed comparable production of primary bile acids between PD patients and controls but increased appendix concentrations of DCA in PD patients [40]. In support of our findings, a recent large cohort study also showed increased levels of DCA and GDCA in the systemic circulation of PD patients [41]. Furthermore, our results demonstrated that plasma levels of DCA correlated with the relative abundance of Lachnoclostridium sp. An14 and Anaerotignum lactatifermentans of family Lachnospiraceae, which are species enriched in the appendix of patients with PD [40]. We also observed an association of plasma GDCA concentrations with the pro-inflammatory bacteria genera Alistipes and Bacteroides, which have been described as increased in PD patients compared with controls [42]. Participation of bile acids in modulation of gut immunity and neuroinflammation has been reported, and they also are considered to be pro-inflammatory and cytotoxic because of their detergent-like features [43, 44]. Secondary bile acids could cross and increase the permeability of the blood–brain barrier, inducing cell membrane disruption and production of oxidative stress and mitochondrial dysfunction, leading to apoptosis and neurodegeneration [43–45]. Our results indicate that dysregulated secondary bile acid metabolism may link to aggravated neurodegeneration in PD. Further research is needed to examine the impacts of DCA and GDCA in neuronal degeneration.
We also found that plasma microbial metabolites were associated with changes in the relative abundance of specific gut microbiota linked to PD. Consistent with previous findings that plasma HA is a marker of gut microbiota diversity and correlates positively with the Ruminococcaceae and Clostridiaceae families [46], in our cohort, plasma HA levels were associated with the relative abundance of Subdoligranulum sp. 4_3_54A2FAA, unclassified Clostridium (Clostridium sp. AM42–36, AF43–10, and AF23–8), and Parabacteroides. Previous studies have shown increases in these species in PD patients and an association with constipation and inflammation [47–50]. In addition, increased circulating levels of IPA in PD were associated with the pro-inflammatory bacteria Subdoligranulum and Lachnoclostridium, which also are enriched in colorectal neoplasm [11, 52]. Of note, the gut microbiota identified here as associated with increased plasma levels of targeted microbial metabolites in PD patients are mostly members of Clostridiales. These species are abundant in the appendix of PD patients and modulate inflammatory responses through interaction with regulatory T cells and pro-inflammatory cytokines [53]. Taken together, our results provide further evidence that dysbiosis of pro-inflammatory gut microbiota may modulate PD process through promoting levels of the microbial metabolites HA, IPA, DCA, and GDCA in the systemic circulation.
Our study has several strengths. First, the enrolled participant number that has a concomitant data of gut metabolites and fecal metagenomic sequencing information is substantial compared to previous studies. Second, the control participants were age, gender and diet-habit matched to the patients with PD. However, our study still has several limitations. First, the cross-sectional design precludes any inferences of a causal relationship between the microbial metabolites HA, IPA, DCA, and GDCA and the risk and disease procession in PD. Further large cohort longitudinal studies are needed. Second, constipation and use of laxatives and anti-cholinergic agents or cholinesterase inhibitors may influence gut microbiota and gut metabolites. Future studies should take these potential confounders into account. Third, we did not measure other benzoic acid–related metabolites associated with HA formation and degradation. Untargeted microbial metabolomic studies are needed to further clarify the role of the altered HA metabolism pathway in PD. Forth, we did not measure microbial metabolites in feces and other tissues, including liver and kidney. Aromatic compounds, including tryptophan, involves the combined activity of gut microbial metabolism and liver and kidney functions [12]. Furthermore, a disrupted intestinal barrier with intestinal hyperpermeability has been observed in patients with PD [54], an integrated study examining both plasma and fecal and levels of HA, indoles-derivatives and secondary bile acids is needed. Fifth, the influence of anti-PD medications on gut metabolites and drugs-induced changes in metabolism should be considered. Although previous studies have shown anti-parkinsonism medications may not significantly affect the profiling of metabolism, with the exception of the dopamine pathway [55], further studies enrolling drug naïve PD patients are needed. Furthermore, we only assessed cognitive function using MMSE, a screening measurement for global cognitive function. Further detailed neuropsychological tests are warranted for assessing the correlation of cognitive function and change of plasma gut metabolites. Finally, all the enrolled participants in this study were Asians who have different diet habits compared with western populations. People from different geographic locations may have diverse diet habits and may influence plasma levels of gut metabolites. Further studies in different geographic regions with comprehensive diet assessments are needed to confirm our findings.
In conclusion, our results showed that the microbial metabolites HA, IPA, DCA, and GDCA were elevated in the systemic circulation of PD patients and associated with changes in individual gut microbiota. These findings suggest that dysregulation in the metabolism of HA, tryptophan-derived indoles, and secondary bile acids contributes to the disease process in PD. Further experimental research exploring the exact mechanisms of these metabolic pathways in neurons would help to elucidate gut–brain interactions involved in the development of PD.
Footnotes
ACKNOWLEDGMENTS
The authors are grateful to all study participants. We thank the staff of the Second and Eighth Core Lab, Department of Medical Research, National Taiwan University Hospital, for technical support during the study. We also thank the Ramaciotti Centre for Genomics, University of New South Wales, for the shotgun sequencing in this study. This work was financially supported by grants from National Taiwan University (NTU-110-A-CC-5400-64841), National Taiwan University Hospital (NTUH 109-T18, 110-EDN01), National Taiwan University Hospital Bei-Hu Branch (11005), and National Health Research Institutes (NHRI-EX111-11136NI).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
