Abstract
Parkinson's disease (PD) ranks the second place among neurodegenerative diseases in terms of its morbidity, which affects 1-2% people aged over 65 years. In addition to genetics, some environmental factors may exert vital parts in PD occurrence as well. At present, more and more studies are conducted to elucidate the association between gut microbial dysbiosis and the incidence of PD. Gut microbial dysbiosis has a certain effect on both the central nervous system (CNS) and the enteric nervous system (ENS), which indicates that there is a gut-microbiota-brain axis that induces CNS disorders. Some gut microbial strains are suggested to suppress or weaken the neuroinflammation- and gut-inflammation-immune responses, which suggests the protective and pathogenic effects of certain gut microbial species on PD progression. Therefore, gut microbiome may contain plenty of targets for preventing and managing PD. Faecal microbiota transplantation (FMT) may serve as a direct and useful treatment for PD in the future. Nonetheless, there is little available scientific research in this field. The present work reviewed the latest research to examine the association of gut microbiota with PD, and the future prospects of FMT treatment.
Keywords
Introduction
Parkinson's disease (PD), a kind of multifocal and progressive neurodegenerative disorder, is mostly caused by dopaminergic neuron loss within substantia nigra pars compacta. PD shows the major symptoms of rigidity, tremor, bradykinesia (Samii et al., 2004) and numerous non-motor symptoms (NMS). Typically, NMSs more adversely affect the life quality of PD cases than motor symptoms (Martinez-Martin, 2011). In general, such NMSs are neuropsychiatric diseases (anxiety, depression, dementia, psychosis and apathy), and problems related to dysfunction of the enteric nervous system (ENS) and autonomic nervous system (ANS) (orthostatic hypotension, urinary changes and constipation). PD is a complicated disorder that involves multiple systems, which is associated with diverse mechanisms. The PD pathogenic mechanisms are mainly related to protein (α-synuclein) aggregation, endoplasmic reticulum stress (ERS), calcium homeostasis and mitochondrial dysfunction (Goswami et al., 2017; Sun and Shen, 2018), but its precise etiology remains to be further investigated.
Gastrointestinal (GI) dysfunction has been identified as a prominent contributing factor to the PD pathogenesis, typically, gut possibly provides a route for pathogens to spread into the central nervous system (CNS). Recently, the bi-directional communications taking place along the brain-gut axis are suggested to modify the neurobiological processes (Mayer et al., 2014; Sun and Shen, 2018). The linkages or interactions between brain and gut are achieved in different ways. To be specific, these may be attained by the influences of central inputs on the gut functions (like stress response), the impacts of gut-derived activity on the CNS function (GI disorder-related changes in cognition and moods), and the comparable roles of gut and brain in identical diseases (like neuronal degeneration). In traditional practice, the interactions and communications between gut and brain are regarded to be mostly regulated by inflammatory mediators and neuro-hormonal factors. Typically, it is suggested in latest studies that, gut microbiota is also a GI factor that affects the functions of brain and gut.
Lately, gut microbiota is extensively reported to affect the gut-brain axis, which also plays a certain role in the CNS-associated disorders and neuropsychiatric diseases, like multiple sclerosis (Jangi et al., 2016), autism (Strati et al., 2017), depression (Foster and McVey Neufeld, 2013; Kang and Cai, 2017a) and schizophrenia (Caso et al., 2016). Given the effects of gut microbiota and the microbial metabolites on the interference with the host behavior, cognition, immunity and metabolism (Marques et al., 2010; Cryan and O'Mahony, 2011; Foster and McVey Neufeld, 2013; P. Li et al., 2021), great attention has been paid to their impacts on the pathogenic mechanism of PD, and certain phenotypic associations are illustrated in recent years (Unger et al., 2016; Hill-Burns et al., 2017; Sun and Shen, 2018 ). For instance, the changes in the abundances or compositions of gut microbiota along with the microbial metabolites can be detected among PD cases. The gut microbial changes exert a vital part in PD induction and progression. Nonetheless, related data and scientific work are lacking at present. The emerging technologies make it possible to systemically investigate the gut microbiota and provide the practical data regarding the compositions and pathological changes (Kang et al., 2019). The present work aimed to review the latest studies concerning the association of gut microbiota with PD, together with the future prospects of the faecal microbiota transplantation (FMT) therapy.
The Microbiota-Gut-Brain Axis
As a bidirectional communication between GI tract and the CNS, the microbiota-gut-brain axis is associated with immune, endocrine mechanisms and neural pathways (Cryan and O'Mahony, 2011; De Palma et al., 2014; Sun and Shen, 2018; Dogra et al., 2021 ). It modulates the digestion events (such as secretion and motility), immunologic function, and perception or emotional response to visceral stimuli under normal condition (Mayer et al., 2006). Its dysfunction is related to several disorders (Cryan and O'Mahony, 2011; Aziz et al., 2013; Bonaz and Bernstein, 2013; Davari et al., 2013; Felice et al., 2016; Grenham et al., 2011; Mayer, 2011; Parashar and Udayabanu, 2017), like depression, anxiety, hepatopathy (Iwaki et al., 2021), intestinal inflammation, GI motility disorder, and chronic abdominal pain syndrome (Cryan and Dinan, 2012; Strati et al., 2021). Besides, this axis is linked with stress behaviors and responses (Clarke et al., 2013; O'Mahony et al., 2015; Parashar and Udayabanu, 2017). At present, it is suggested that microbiota involved in bidirectional communication that takes place along the brain-gut axis may serve as the significant contributor (Rhee et al., 2009; Bercik, 2011; Grenham et al., 2011; Mayer, 2011; Collins et al., 2012; Felice et al., 2016; Parashar and Udayabanu, 2017). Apart from the neuroimmune and neuroendocrine systems, this axis also involves the CNS, together with the parasympathetic and sympathetic branches of ENS and ANS (Grenham et al., 2011; Parashar and Udayabanu, 2017). The afferent fibers that travel from gut to cortical centers (including anterior/posterior cingulate, cerebral, amygdalar and insular cortices), together with the effector fibers that project to gut smooth muscles, stand for the main routes of bidirectional communications along the microbiota-gut-brain axis (O'Mahony et al., 2011; Parashar and Udayabanu, 2017). The complicated network may possibly affect the GI and cerebral functions (Rhee et al., 2009; Mayer, 2011; Felice et al., 2016).
Roles of Gut Microbial Dysbiosis in PD
Increasing studies prove that PD is linked with gut microbial dysbiosis in both human beings and animals. At the same time, some mechanisms may be related to the effects of microbial alterations on the gut-brain interaction in PD. Figure 1 presents the mechanisms of gut microbial dysbiosis in affecting PD occurrence.
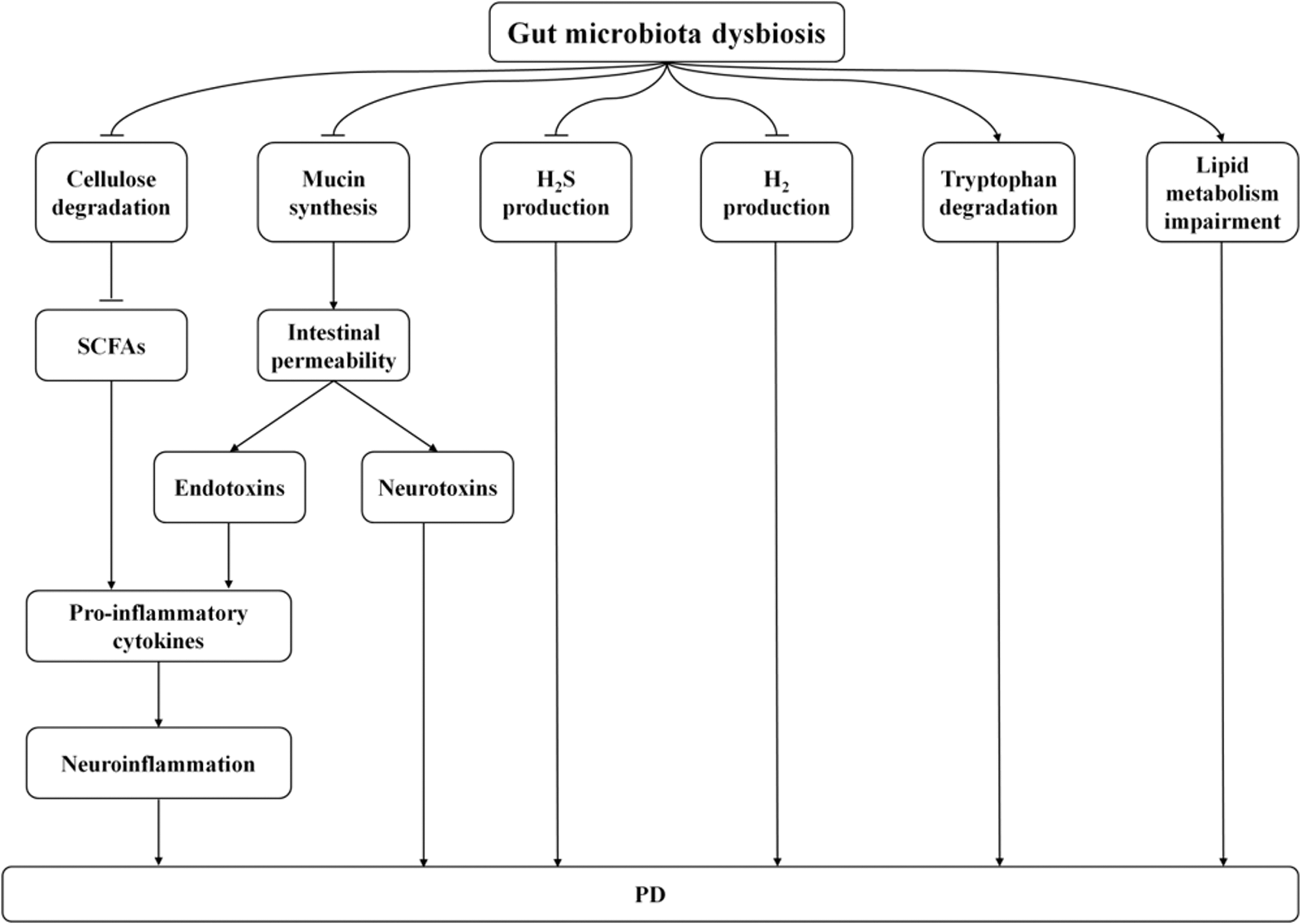
Possible Mechanisms Associated With the Relationship Between Intestinal Microbiota and PD. The ability of intestinal flora to degrade cellulose to synthesize SCFAs may reduce, leading to increased production of pro-inflammatory factors and ultimately inducing neuroinflammation. The gut microbiota may contribute to increasing intestinal permeability via decreasing the epithelial mucin proteins, which results in translocation of endotoxins and neurotoxins across the intestinal wall. In addition, endotoxins may induce production of pro-inflammatory cytokines, which eventually lead to neuroinflammation. Intestinal microbiota may reduce H2S production, H2 production, and increase tryptophan degradation. Gut microbiota may lead to the impairment of lipid metabolism. These mechanisms may lead to impaired CNS function, such as alterations in cognition, behavior and stress responses, finally causing or aggravating PD.
Experimental Studies
As suggested by animal studies, gut microbial dysbiosis can be detected in various animal models of PD. Most of these works indicate that, compared with normal controls, PD patients display changes in the relative abundances of potentially “harmful” and “beneficial” bacteria (Table 1). Research on germ-free (GF) mice first suggests that gut microbiota is related to PD. Sampson et al. (2016) found that specific pathogen-free (SPF)-Thy1-αSyn (alpha-synuclein-overexpressing [ASO]) animals suffered from progressively declined motor function, while such phenomenon was markedly postponed among the Germ-free (GF)-ASO animals. Besides, the reduced abundances of order Clostridiales and phylum Firmicutes, as well as the elevated abundances of orders Enterobacteriales and Turicibacterales and phylum Proteobacteria are recently detected in fecal samples collected from mice with PD induced by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)(Sun et al., 2018).
Changes in Microbiota Composition Associated With PD in Animals.

In line with the research, rotenone-induced PD mice also display fecal microbial dysbiosis, which is featured by the significantly changed microbial compositions (particularly the elevated Firmicutes-to-Bacteroidetes ratio) and the decreased bacterial diversity. Additionally, the rotenone-induced motor and GI impairments are suggested to markedly relate to the alterations of fecal microbial composition (Yang et al., 2017). Further, as discovered from Sprague-Dawley (SD) rats exposed to rotenone treatment the relative abundances of
Collectively, these findings obtained from diverse animal samples reveal the role of gut microbial dysbiosis in the pathogenic mechanism of PD. But, a lot of research is still needed to determine the specific strains of bacteria that can treat PD and the specific strains that can cause PD. Meanwhile, further study need to emphasize more on the metabolomic aspect and its role in pathophysiology and the nutritional aspect.
Clinical Studies
Increasing evidence has suggested that gut microbial dysbiosis is also found in PD patients (Table 2). Meanwhile, gut microbiota plays a certain role in the pathophysiology of PD. It is previously suggested that, bacterial overgrowth in the small intestine is prevalent among PD cases, which is related to poor motor function (Tan et al., 2014). Hopfner et al. (2017) found that the Beta-diversity was significantly different between PD cases and controls. Meanwhile,
Changes in Microbiota Composition Associated With PD in Humans.

In line with these results, Hasegawa et al. (2015) showed that the sum of fecal bacterial counts markedly decreased in PD patients relative to controls. Compared with normal controls, the abundance of
It is also discovered in another work that, the abundances of Bacteroidetes,
As suggested in more clinical studies, fecal samples from PD show high abundances of
All in all, these observations shed more lights on gut microbial dysbiosis among PD cases, which may be used to guide disease interventions through gut microbiota manipulation. However, how does the gut microbiota change during the course of PD remains unclear. Moreover, it remains unclear if different microbial compositions are related to different phenotypes of PD. Further research is needed to determine which bacteria and metabolites can serve as the biomarkers for accurately differentiating PD patients from the controls. In addition, PD-associated metagenomics studies should consider the family environmental factor.
Future Prospects of FMT Therapy for PD
The FMT therapy refers to the injection of fecal suspension obtained from one healthy subject to the GI tract of another subject for the treatment of a certain disorder. FMT is a direct and superior treatment for PD through the manipulation of human gut microbiota. The FMT therapy has been applied for more than 50 years. Typically, Ge Hong in China first described to use feces as a treatment for various disorders, like diarrhea, in the 14th century (Zhang et al., 2012). In 1958, Eiseman et al. used fecal enemas to treat pseudomembranous colitis (possibly induced by
Changes in Microbiota Composition Associated With PD and FMT Therapeutic Strategies.
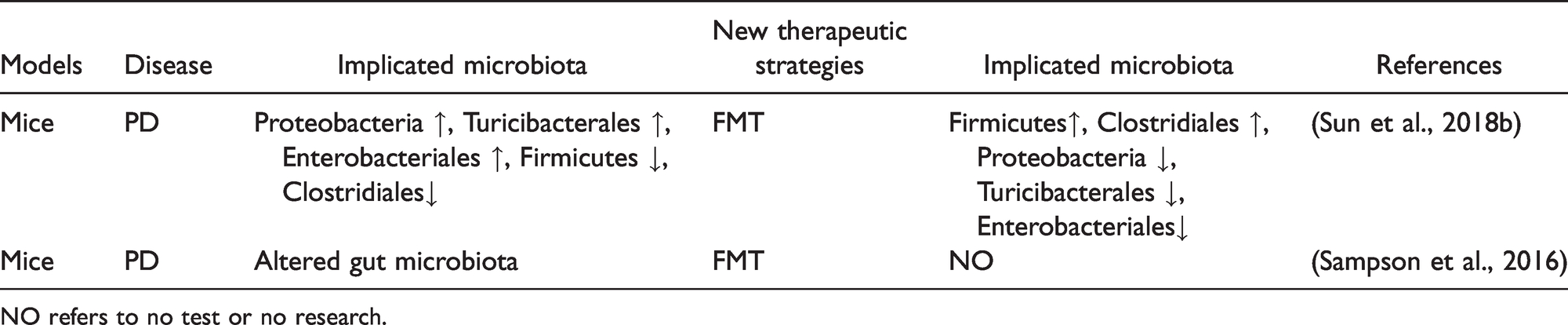
NO refers to no test or no research.
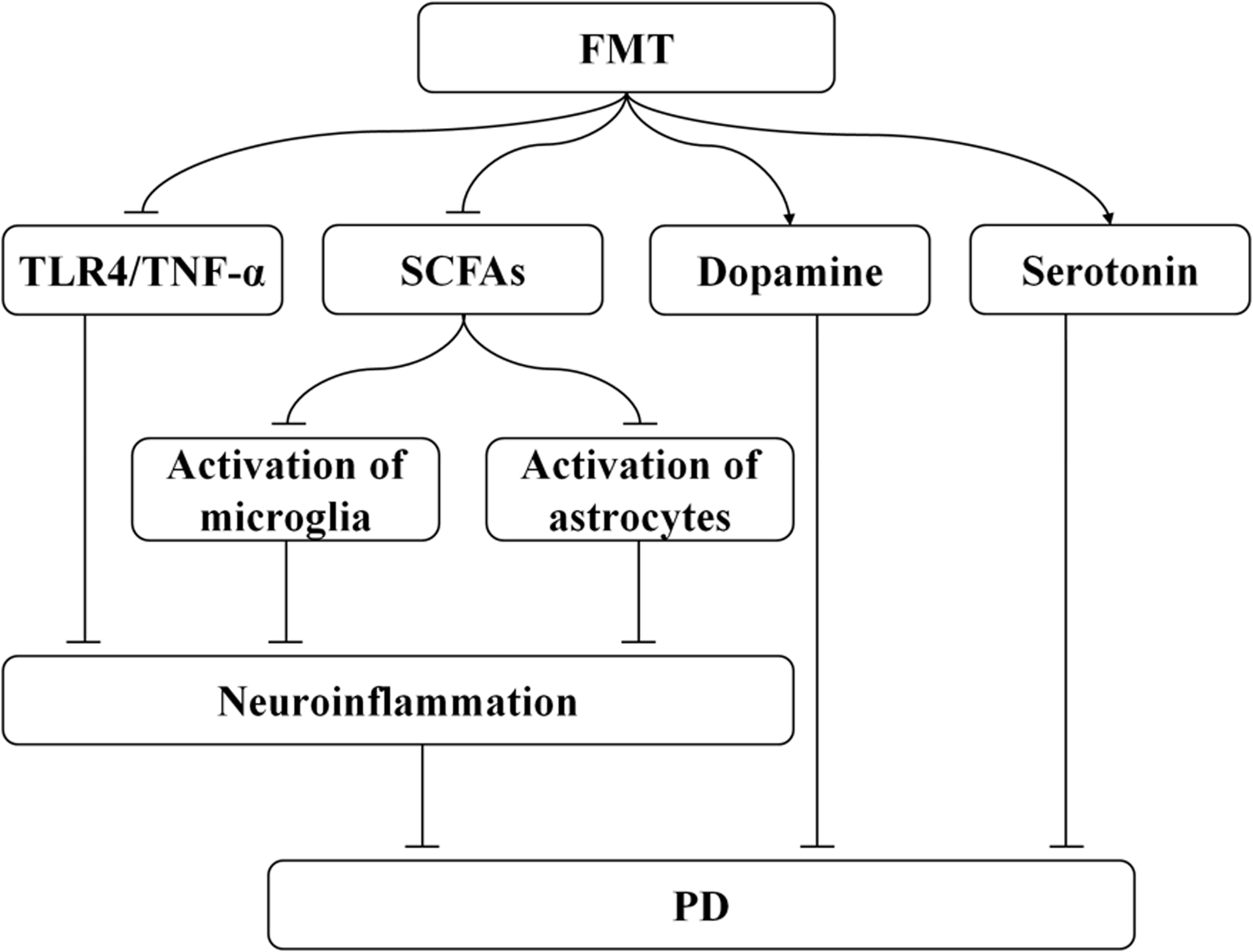
Possible Mechanisms Underlying the Relationship Between Fecal Microbiota Transplantation (FMT) and PD Are as Follows: FMT May Inhibit Neuroinflammation by Decreasing the TLR4/TNF-a Signal Transduction. FMT may decrease astrocytes and microglia activation by reducing production of SCFAs, and ultimately inhibits neuroinflammation. FMT may boost serotonin (5-HT) and dopamine (DA) levels.
Concluding Remarks and Perspectives
However, FMT therapy is a new and under-explored therapy for PD. The available scientific information in this field is lacking. Therefore, more efforts should be made to better understand the role of FMT in PD. First of all, a causal relation between alteration of gut microbiota and PD need to be proven. Application of fecal transplantation in PD disease may be regarded as an interesting research tool to identify the causally involved intestinal microbial species and/or non-microbial components as initiators of both gastrointestinal and systemic autoimmune reactions. Therefore, we should devote more effort to studying FMT therapy to PD. Secondly, it is worthwhile to find out how FMT re-establishes a balanced intestinal microbiota and results in impressive cure rates in patients with PD disease. Thirdly, more randomized controlled trials (RCTs) with reasonable design should be carried out to determine whether FMT is safe and effective on PD. Additional high quality data (e.g. longitudinal study) are also needed to analyze the potential adverse effects. Furthermore, it is important to analyze the fecal and small intestinal microbial compositions prior to and following FMT. The above studies shed more lights on the mechanism of FMT and help to discover the microbes together with the metabolites related to PD pathogenesis. Fourthly, the effects of microbial metabolites on PD should be further investigated. Particularly, it should be further investigated about whether SCFAs protect or promote PD. Last but not least, FMT is a simple treatment, which only requires a short operation time. Compared with repeated conventional treatments, the hospitalization cost of FMT is low. Thus, the standardization of FMT protocols is urgently established. For this reason, in addition to conventional approaches, FMT is promising as an alternative therapy for PD diseases in the future.
Footnotes
Author Contributions
Yongbo Kang collected and wrote most of the experiments with help from Xing Kang, Hongfang Zhang, Qingqing Liu, Hao Yang and Weiping Fan.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Science Research Start-up Fund for Doctor of Shanxi Medical University (Grant No.XD1807), Science Research Start-up Fund for Doctor of Shanxi Province (Grant No.SD1807), Scientific and Technological Innovation Programs of Higher Education Institutions in Shanxi (Grant No. 2019L0425) and Shanxi Province Science Foundation for Youths (Grant No. 201901D211314).
