Abstract
Background:
Parkinson’s disease (PD) is a multi-system disorder consisting of not only classic motor symptoms but also a variety of non-motor symptoms including gastrointestinal (GI) dysfunction and mood disorders. The gut microbiota has been suggested to play a role in modulating PD motor and non-motor features, although the causality and mechanisms behind these proposed interactions remains largely understudied.
Objective:
In this study, we aimed to provide in-depth characterization of an established mouse model of PD (transgenic (TG)
Methods:
We profiled the PD-like phenotype of transgenic mice through a panel of motor, GI, and behavioral tests. We then investigated how antibiotic treatment or gut microbial community transfer (via cohousing with wild-type mice) impacted the PD-like phenotype.
Results:
We found that this mouse model demonstrated early (6 weeks of age) motor symptoms when compared to a wild-type control mouse strain. Transgenic mice also exhibited early GI dysfunction, as well as behavioral alterations, including reduced anxiety-like behavior, and increased depression-like and apathy-like behavior. Compared to wild-type mice, the transgenic fecal microbiota was less diverse and compositionally distinct. Interestingly, drastic alterations to the gut microbiota, through antibiotic treatment or cohousing with wild-type mice, had a minimal effect on the motor, GI, and behavioral phenotype of transgenic mice.
Conclusion:
We concluded that this mouse model effectively recapitulates motor and non-motor features of PD; however, the gut microbiota appears to exhibit a minor impact on the pathophysiology of this PD model.
INTRODUCTION
Parkinson’s disease (PD) is a progressive neurodegenerative disease that affects 1% of the population over 60 years of age [1]. It is characterized by the presence of motor symptoms such as bradykinesia (slowed movement), rigidity, and tremor; as well as the neuronal accumulation of the protein alpha-synuclein, forming Lewy bodies [2, 3]. Approximately 10% of PD cases can be directly attributed to genetic mutations [1] including copy number or point mutations in the gene encoding alpha-synuclein,
In addition to the classic motor symptoms of PD, patients typically experience a variety of non-motor symptoms [2]. Some of these, including constipation and sleep disturbances, often occur decades prior to the onset of motor symptoms. Gastrointestinal (GI) dysfunction and mood disorders, such as depression and anxiety, are particularly prevalent [5]. The multitude of non-motor symptoms greatly impacts patients quality of life [6] and demonstrates that PD is a multi-system disorder. GI symptoms can be particularly burdensome in PD and include difficulty swallowing, constipation, and difficulty with defecation [7]. The prevalence and early onset of GI dysfunction in PD has, in part, led to the hypothesis of a gut-first origin of pathogenesis [8] at least in a subset of PD patients [9].
The gut microbiota—the community of bacteria, archaea, fungi, and viruses in the gut—has recently been suggested as a potential mediator of environmental PD risk factors due to a close interaction with the enteric nervous system (ENS) and immune system [10]. Furthermore, gut microbiota composition is related to GI function [11] and shows links with depression and anxiety outside of the PD context [12]. Multiple clinical studies investigating differences in gut microbial composition between PD patients and non-PD control subjects (reviewed by [10]) have revealed common bacterial shifts, including an increased abundance of
Many mouse models of PD have been developed, ranging from toxin-induced to genetic models where alpha-synuclein is overexpressed or mutated [16, 17]. However, the progressive nature of the disease has been difficult to replicate in rodents. In addition, models that effectively recapitulate both motor and non-motor symptoms of PD are somewhat lacking [18]. Some animal studies have suggested a causal role for the gut microbiota in PD. Germ-free (GF) genetic or toxin-based PD rodent models show an attenuated disease phenotype compared to microbially colonized animals [19, 20]. Furthermore, fecal microbial transfer (FMT) from PD patients into a PD mouse model exacerbated PD pathophysiology [20]; and FMT from PD mice into C57BL/6J control mice induced motor deficits [21]. While recent work has examined the effect of microbial manipulation on PD motor symptoms and neurodegeneration, the role of the microbiota in PD non-motor symptoms remains grossly understudied. Moreover, reproducibility of the aforementioned studies is important in order to determine generalizability to PD as a whole.
A model of PD displaying early GI dysfunction followed by mild motor deficits was developed using mice expressing human alpha-synuclein with a common familial point mutation (A53T) on an artificial chromosome under native regulation [22]. The authors demonstrated that GI deficits—including increased transit time, reduced colonic motility, and reduced fecal water content—were apparent at 3 months of age with mild motor deficits becoming observable at 6 months of age.
Herein, we aimed to investigate the role the microbiota plays in PD motor and non-motor symptoms utilizing the
MATERIALS AND METHODS
Animals and treatment
FVB;129S6-
Mice were housed in the Modified Barrier Facility at the University of British Columbia on a 12 h light-dark cycle. Ventilated cages consisted of 2–5 mice and included wood chip bedding, nesting material, and a plastic hut for enrichment. Mice had access to food (PicoLab Rodent Diet 20 – 5053 (irradiated)) and water
Antibiotic treatment
A subset of transgenic (TG) and wild-type (WT) mice were treated with antibiotics via their drinking water. A cocktail of antibiotics consisting of ampicillin (1 g/L; GoldBio), vancomycin (0.5 g/L; GoldBio), neomycin (0.5 g/L; GoldBio), gentamycin (100 mg/L; GoldBio), and erythromycin (10 mg/L; Cayman Chemical Co.) was used as described previously [20]. Antibiotics were dissolved in drinking water, provided
Cohousing
Female TG and WT mice were cohoused from 3 weeks to 11 weeks of age (“cohouse”) after which motor, GI, and behavioral testing was performed. These mice were compared to mice of the same genotype that were not cohoused (“control”) and mice that were cohoused from 3 weeks to only 7 weeks of age and then regrouped by genotype (“split”). Fecal samples were collected prior to, 4 weeks into, and 8 weeks into the cohousing period for bacterial 16S rDNA sequencing.
Mouse testing
Motor, gastrointestinal, and behavioral tests were performed during the light phase. Cages were randomized to control for testing order. When possible, tests were performed on separate days to allow mice to rest and recover. All motor, gastrointestinal, and behavioral tests were performed in the same testing room. Where indicated, mouse movements were recorded via Go-PRO (HERO 6). Mice were tested at 6, 14, and 22 weeks of age for the model characterization and antibiotic treatment experiments. Mice were tested at 11–14 weeks of age for the cohousing experiment. Both female and male mice were used for experiments where possible. For tests where sex differences were observed, data from female and male mice were plotted separately, otherwise, data were pooled.
Motor tests
Rotarod
An IITC Rotarod was used to test gross motor function as described previously [22]. The rotarod apparatus was set to accelerate from 4 to 40 rpm over 5 min and then maintain a speed of 40 rpm for an additional 5 min for each trial. The time elapsed when mice fell off the rod onto the platform below (latency to fall) was recorded. Mice that remained on the rod until the end of the trial were given a latency of 600s for that trial. Mice underwent 4 trials per day for 3 consecutive days with 10–30 min of rest between trials. The machine was cleaned using alcohol between trials and between mice. For mice where the test was repeated at multiple time points, data at the later time points (14 and 22 weeks) is presented as the average of all trials for each mouse, as mice performed similarly over all three testing days when the test was repeated (data not shown).
Pole descent
The pole descent test was used to measure gross motor function [23] with minor modifications [20]. A 0.5 m long wooden pole (1 cm in diameter) with a circular base was placed upright in the home cages of mice, temporarily emptied of everything but bedding and nesting material. Testing consisted of two consecutive training days followed by a testing day. On the first training day, which consisted of three trials, mice were placed head-down 1/3 of the way up the pole, 2/3 of the way up the pole, and at the top of the pole and allowed to descend to the bottom each time. On the second training day, mice received another three trials each time being placed head-down from the top of the pole. On the testing day, mice were placed head-down at the top of the pole and the time from release to the placement of their forepaws on the bottom of the pole was recorded. The pole was cleaned with alcohol between mice and cages.
Hindlimb clasping
The hindlimb clasping test was used to assess striatal dysfunction [24]. Mice were held by the mid section of the tail and suspended for 6–8 s as described previously [20]. The degree of clasping of the hindlimbs was recorded by video and analyzed by a blinded observer using the following criteria: 0 (no clasping) indicates flexible and freely moving hindlimbs; 1 indicates inward clasping of one hindlimb or partial inward clasping of both hindlimbs; 2 indicates inward clasping of both hindlimbs but with some flexibility; 3 indicates no flexibility and complete and immediate inward clasping of the hindlimbs. A visual guide used for scoring can be found here [25].
Inverted grid
The inverted grid test was used to measure grip strength as described previously [26]. The apparatus used consisted of a 1 cm2 wire grid stretched over a wooden frame (37 cm×29 cm) with sides long enough to prevent mice from climbing to the other side. Mice were placed in the center of the grid which was immediately turned upside down (head over tail) and suspended ∼40 cm over a box containing soft nesting material. The time elapsed before mice let go of the grid and fell (to a maximum of 5 min) was recorded. The grid was cleaned with alcohol between mice.
GI tests
Transit time test
The carmine red test was used to measure whole-gut transit time as described previously [27]. Mice received an oral gavage of 100
Fecal water content
Fecal water content was investigated as a measure of gastrointestinal function. Fecal pellets were collected from mice individually and stored in pre-weighed 1.5 mL microcentrifuge tubes at –20°C until further use. Tubes containing fecal pellets were weighed, lyophilized at < –20°C for 16 h, and weighed again. The percent H2O content was calculated by subtracting the final fecal weight from the initial fecal weight and dividing by the initial weight.
Behavioral tests
Open field test (OFT)
The OFT was used to measure motor function and anxiety-like behavior [28]. Mice were placed in the center of a clear box (49 Liter Tote, Home Depot: 40 cm×30 cm×56 cm) which had a base divided in a grid with a designated open field of 22 cm by 11 cm. Individual mice were allowed to explore freely for 5 min while their movement was recorded by video. The box was cleaned with alcohol between mice. Tracking and scoring for the open field test was performed using ANY-maze software.
Tail suspension test
The tail suspension test was used to assess depression-like behavior as described previously [29, 30]. Briefly, mice were suspended by the tail using adhesive lab tape and video recorded for 6 min. The time spent immobile was recorded by a blinded observer.
Cylinder test
The cylinder test was used to measure spontaneous activity behavior [31]. Mice were placed in a novel environment (1 L transparent cylinder) and video recorded for 5 min. The number of rearing events (defined as the clear placement of both forepaws on the side of the cylinder) was then recorded.
16S sequencing
Fecal pellets were collected from mice and stored at –80°C until further use. DNA was extracted using the QIAamp PowerFecal DNA kit (QIAGEN 12830) according to the manufacturer’s instructions. A bead beater (FastPrep-24; MP Biomedicals) was used to homogenize fecal samples. DNA was stored at –20°C until further use. Indexed, barcoded primers (515F: GTGCCAGCMGCCGCGGTAA; 806R: GGACTACHVHHHTWTCTAAT) were used to amplify the bacterial 16S rDNA V4 region. The library was pooled and sequenced using paired-end 300-cycle reads with a v3 Reagent Kit on an Illumina MiSeq platform. Raw sequences were trimmed to 250 bp and processed using
Serotonin quantification
Serotonin levels were determined in colonic tissue using the Serotonin ELISA Assay kit (Eagle Biosciences SER39-K01). Fecal contents were manually removed during tissue collection. Tissue was collected in 1X PBS containing 0.1% ascorbic acid (Sigma) and protease inhibitors (complete™, EDTA-free Protease Inhibitor Cocktail; Sigma) and stored at –80°C until further use. Tissue was thawed and homogenized using tungsten beads and a Mixer Mill MM400 (Retsch; 25 Hz for 2 min). Samples were then spun on a microcentrifuge (16,000×
Statistical analysis
In all figures, data are presented as the mean±SD. Statistical analyses and visualizations were performed in GraphPad Prism 8 unless otherwise indicated. Multiple groups were compared by the Kruskal-Wallis test. The Mann-Whitney U test was used when comparing two groups and the Wilcoxon test was used for paired analyses. A two-sided
RESULTS
Transgenic mice display motor deficits as early as 6 weeks of age
We compared the performance of TG mice with sex- and age-matched WT mice on a variety of motor tests at 6, 14, and 22 weeks of age. As previously shown [22], TG mice showed a significantly reduced latency to fall off the accelerating rotarod at 6 weeks (
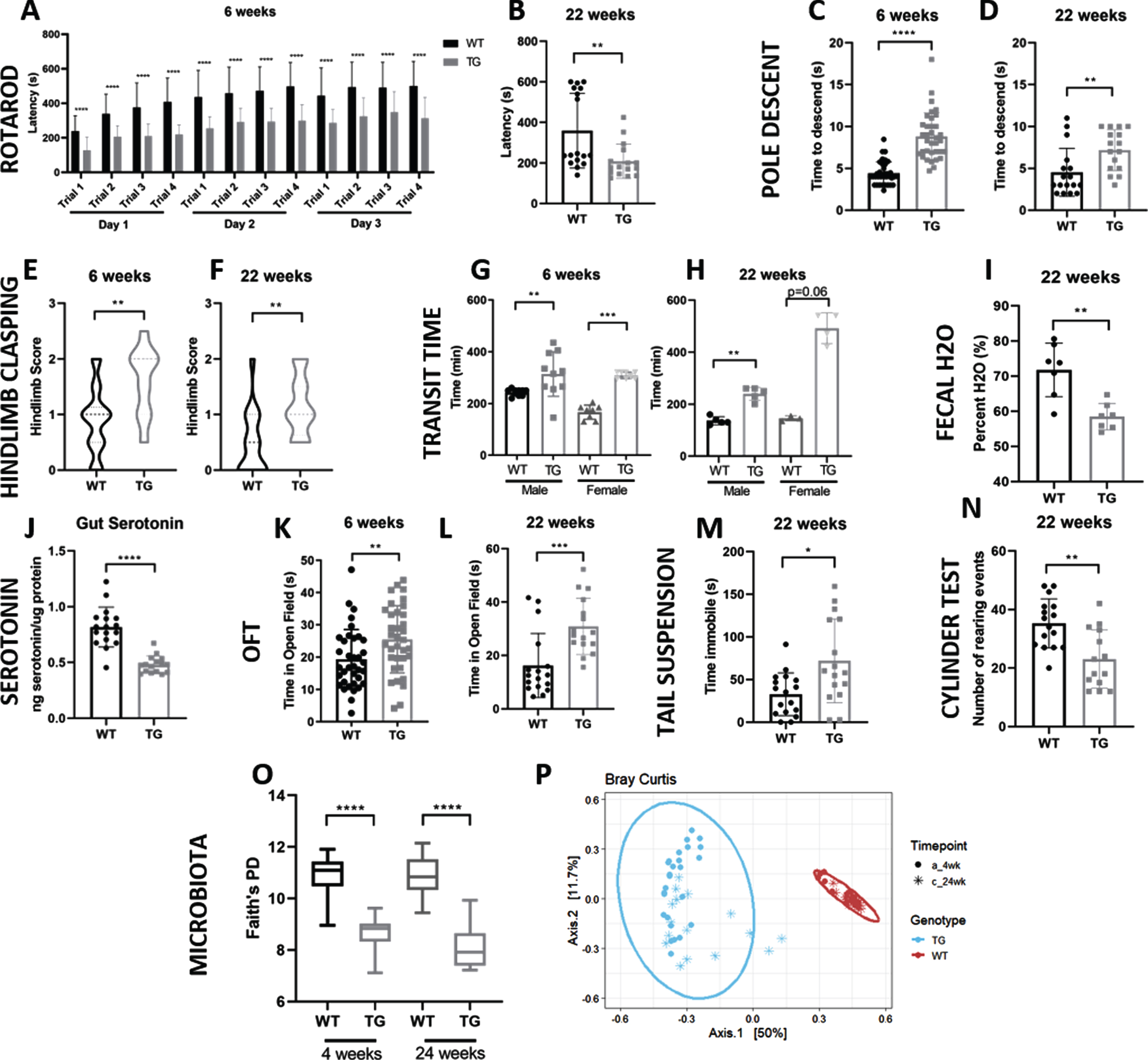
TG mice display multiple PD-like phenotypes when compared to WT mice. A,B) Latency to fall in the Rotarod test at 6 and 22 weeks of age. Data at 22 weeks represents the average of 12 trials over 3 days for each mouse. C, D) Time to descend pole at 6 and 22 weeks. E, F) Hindlimb clasping reflex score at 6 and 22 weeks. G, H) Whole-gut transit time at 6 and 22 weeks as indicated by carmine red test. I) Fecal water content at 22 weeks. J) Serotonin levels in colonic tissue normalized to total protein content. K, L) Time spent within the open field zone at 6 and 22 weeks. M) Time spent immobile during the tail suspension test. (N) Number of rearing events during rearing test. O) Gut microbiota alpha diversity as shown by Faith’s phylogenetic diversity. P) Gut microbiota beta diversity as shown by Bray Curtis dissimilarity. Data points indicate individual mice; error bars indicate standard deviation of the mean. *
To further examine signs of gross motor function, we performed a pole descent test. Descent time provided a proxy of motor ability with longer descent time indicative of increased motor impairment. TG mice required a significantly increased time to descend the pole (
Collectively, the rotarod, pole descent, and hindlimb clasping tests revealed gross motor deficits in TG mice, with these Parkinsonian-like motor symptoms already apparent by 6 weeks of age. TG mice showed no difference in time to fall from an inverted grid (Supplementary Figure 1A) indicating that motor deficits were likely not a result of differences in grip strength. Similarly, TG and WT mice of each sex were similar in weight over the experimental time frame (although female mice diverged in weight at 18 weeks of age; Supplementary Figure 1I, J) implying that body size differences were also not likely responsible for motor differences.
We next investigated whether motor symptoms progressed from an early age (6 weeks) to late adulthood (22 weeks) by performing paired analyses for individual mice. While the latency to fall off a rotarod decreased significantly over time for both TG and WT mice (
Transgenic mice display gastrointestinal dysfunction as early as 6 weeks of age
Gastrointestinal dysfunction has been linked to Parkinson’s disease in human patients and has been found to occur early in disease progression [7]. To investigate gastrointestinal function, transit time and fecal water content were compared between age-matched TG and WT mice at multiple timepoints. We first investigated the whole-gut transit time by the carmine red test. TG mice displayed a significantly increased transit time, indicative of increased constipation, as early as 6 weeks of age (males:
Transgenic mice display altered behavior starting as early as 6 weeks of age
To investigate whether TG mice displayed other non-motor PD like symptoms, namely behavioral alterations, we compared the behavior of age-matched TG and WT mice through a variety of tests. We performed the OFT to measure anxiety-like behavior (more time within the open field zone (OFZ) indicates less anxiety [40]). TG mice spent a significantly greater time within the OFZ (
Transgenic mice differ in gut microbiota composition from wild-type mice
We next investigated whether the motor, GI, and behavioral alterations observed in TG mice were associated with compositional differences in gut bacteria, and if bacterial composition changed over time. Consequently, we performed 16S sequencing on fecal pellets collected from WT and TG mice at 4 weeks of age (early time point) and 24 weeks of age (late time point). Alpha diversity is a measure of compositional heterogeneity within a sample; at both time points, TG mice showed a significantly lower alpha diversity (Faith’s PD
Antibiotic treatment has minimal effect on transgenic mouse PD-like phenotype
In light of the early-onset altered gut microbial community that TG mice exhibit, we then aimed to test whether modulating the gut microbiota would affect the PD-like symptoms. To this end, a subset of TG and WT mice were treated with a cocktail of broad-spectrum antibiotics and profiled for motor, gastrointestinal, and behavioral phenotype. Antibiotic efficacy was assessed by 16S rDNA sequencing of representative fecal samples collected during treatment (data not shown).
Interestingly, TG mice treated with antibiotics displayed no changes in motor phenotype as assessed by rotarod, pole descent test, or hindlimb clasping score (Fig. 2A-C). We next turned to the GI tract to investigate the effect of oral antibiotic treatment on the non-motor symptoms. Since antibiotic treatment can induce diarrhea [41], we measured the transit time two weeks after treatment to avoid immediate effects of antibiotics. While antibiotic treatment significantly increased transit time in WT mice (
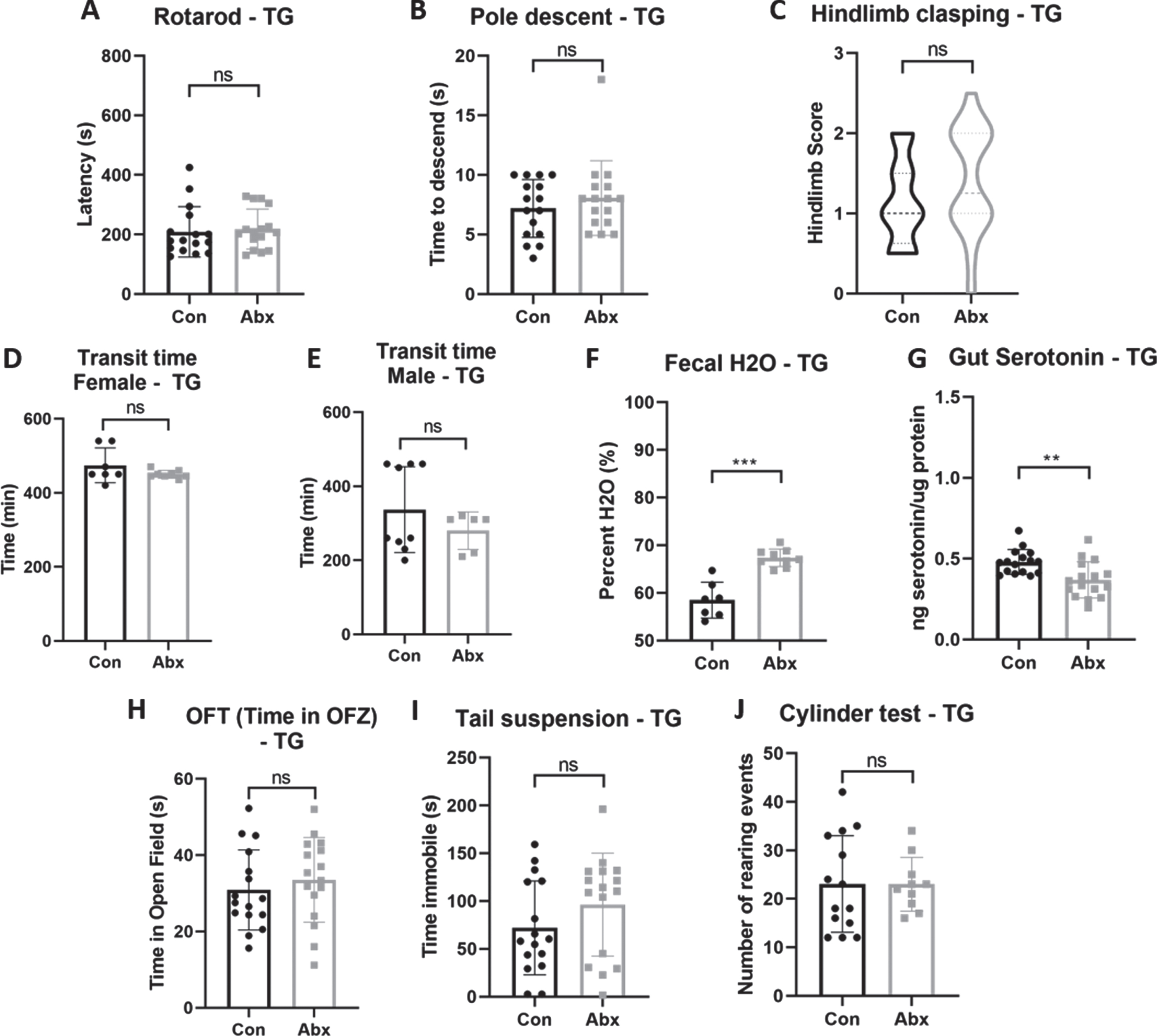
Effect of antibiotic treatment on motor performance, GI function, and behavior in TG mice. A) Latency to fall off of rotarod, (B) time to descend pole, (C) hindlimb clasping reflex score, (D) whole-gut transit time test in females and (E) males, (F) fecal water content, (G) serotonin levels in colonic tissue normalized to total protein content, (H) time spent within open field zone in OFT, (I) time spent immobile during the tail suspension test, (J) number of rearing events during rearing test. All data presented here are from 22 weeks of age. Data points indicate individual mice; error bars indicate standard deviation of the mean. *
Transfer of commensal species by cohousing has minimal effect on transgenic mouse PD-like phenotype
Since depletion of the gut microbiota through antibiotics resulted in a minimal effect on disease phenotype, we next investigated whether the addition of bacterial species or broad shifts in community composition might have an effect on PD symptoms. Three-week-old female TG and WT mice were cohoused (2:2) for a period of 4 weeks to facilitate the transfer of commensal gut bacterial species. A subset of cohoused mice was then split (to investigate the stability of the transferred microbiota), while the remaining mice were cohoused for another 4 weeks (see Fig. 3A for the experimental setup). Prior to cohousing, TG and WT mice exhibited distinct microbiota communities as assessed by Bray Curtis beta diversity (Fig. 3B). After 4 weeks of cohousing, the microbiota of cohoused (but not control) TG mice shifted to closely resemble that of WT mice (Fig. 3C). TG mice that remained cohoused, as well as those that were split, remained clustered with WT mice at the 8-week time point (Fig. 3D). This demonstrates that the TG microbiota shifted towards the WT composition as a result of cohousing and that the transferred bacterial community remained relatively stable up to 4 weeks after cohousing. Interestingly, there appeared to be no shift in the WT microbiota towards the TG community in WT cohoused mice. Specific changes in bacterial abundance at the family level among the TG experimental groups are given in Supplementary Table 1.; no significant differences between the cohoused and split groups were identified.
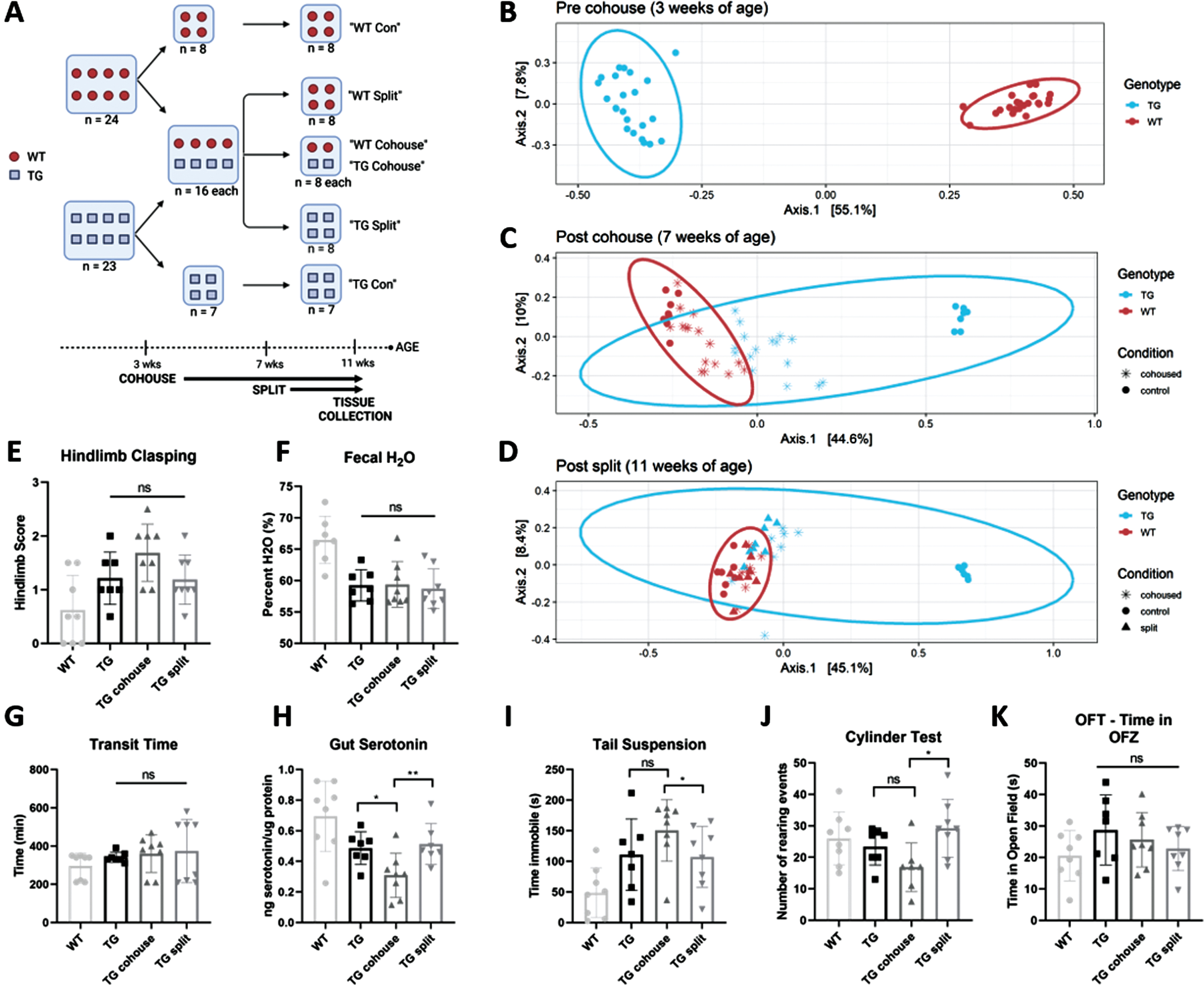
Effect of cohousing on motor performance, GI function, and behavior in TG mice. A) Experimental groups and timeline. B) PCoA plot of Bray Curtis beta diversity at baseline (prior to cohousing), (C) after 4 weeks of cohousing, and (D) 4 weeks after splitting a subgroup from cohousing. E) Hindlimb clasping reflex score, (F) fecal water content, (G) whole-gut transit time, (H) serotonin levels in colonic tissue normalized to total protein content, (I) time immobile in tail suspension test, (J) number of rearing events in cylinder test, (K) time spent within the open field zone of the OFT. Motor, GI, and behavioral tests performed at 11 weeks of age. Data points indicate individual mice and represent data from two experimental cohorts; error bars indicate standard deviation of the mean. *
TG cohoused mice showed no difference in hindlimb clasping score compared to control non-cohoused mice (Fig. 3E); rotarod and pole descent performance also remained unchanged (data not shown) suggesting that overall, motor symptoms were not affected by cohousing-induced microbiota alterations. We next investigated GI function; fecal water content and GI transit time were also not significantly different in cohoused TG mice (Fig. 3F, G). However, cohoused TG mice showed significantly lower gut serotonin levels than both control and split TG mice (
DISCUSSION
In this work, we have used the previously described TG
Model characterization (motor, GI, and behavioral phenotype)
We have observed a similar reduced latency to fall in the rotarod test as Kuo et al. (2010) [22]; however, unlike this previous report, no change in total distance travelled during the OFT was observed. One reason for this unexpected difference in motor performance in the OFT could be the use of a 5-min testing period in our study compared to the 10-min testing period used previously [22]. We have expanded the characterization of motor deficits by multiple testing methods to solidify the motor impairment. These motor deficits were likely not a result of inherent differences in grip strength or body size between the mouse strains. While female TG mice were significantly greater in weight than their sex-matched WT controls starting at 18 weeks of age, this divergence in weight appeared after motor differences were observed; therefore, this difference in weight is presumably not a factor in the interpretation of the motor deficits. Notably, we observed the appearance of motor symptoms as early as 6 weeks of age, with no transgene-specific progressive decline in motor function up to 22 weeks of age. Kuo et al. (2010) [22] noted that motor impairments became most striking in extremely aged mice (18 months of age) which was outside the scope of this study; however, it might be expected that a similar decline would be observed if the mice used in this study were aged to this same degree. To our knowledge, Kuo and colleagues did not perform pairwise statistical comparison of motor performance for their control mouse strains over time so it is unclear to what degree this progression of motor impairments could be attributed to general aging. The discovery that this mouse model displays motor deficits as early as 6 weeks of age provides rationale for future studies using this model to be shorter in time frame.
The gastrointestinal dysfunction at 22 weeks of age previously reported for this model [22] was replicated in this study; however, we observed slowed transit time as early as 6 weeks of age. Although the delayed transit time in TG mice did not appear progressive until 22 weeks of age, TG mice displayed decreased fecal water content starting at 14 weeks of age indicating partial progression of GI symptoms. The early and persistent GI dysfunction co-occurred with age-related progressive motor symptoms indicating that this model effectively represents early PD with GI involvement.
The behavioral alterations described in this study represent novel findings for this model and indicate its potential usefulness in studying PD-related mood disorders. Kuo et al. (2010) [22] found no differences in exploratory behavior in the OFT; however, we observed that TG mice spent more time within the OFZ indicating that TG mice display a more exploratory and active phenotype. A decrease in time spent within the open zone has been used to suggest greater anxiety-like behavior [28, 40]. However, in this study the control (WT) mice spent less time within the open zone than TG mice; if the WT mouse behavior is assumed to be a “healthy” baseline then the TG mouse performance in this test may be considered as a deviation from normal mouse behavior. Notably, the altered behavior displayed by TG mice in the OFT was exacerbated by age in a PD-dependent manner, indicating that the behavioral phenotype in these mice is progressive. The increased time spent immobile in the tail suspension test indicates a more depression-like [30] phenotype for TG mice while the decreased number of rearing events during the cylinder test indicate lower levels of spontaneous activity [31] and may signify less interest in the surrounding environment, or apathy-like behavior. These subsequent behavioral tests were only performed at the endpoint (22 weeks of age) to minimize stress to the mice; however, future experiments could provide valuable insight into the progression of the depression-like and apathy-like phenotype in this model. Overall, the results of the behavioral tests indicate that behavioral alterations are present in this model, some of which may model the mood disorders observed as comorbidities in human PD.
The behavioral alterations observed here are consistent with the decreased anxiety-like behavior displayed by other alpha-synuclein transgenic mouse models [42, 43]. This points towards a potential link between alpha-synuclein and behavioral alterations in PD. Notably, our work does not elucidate whether behavioral alterations are driven by general overexpression of alpha-synuclein or by the A53T mutant form in particular; future studies using different control mouse strains (ex. PAC-Tg(
Some caution should be taken in the interpretation of the behavioral alterations observed in this model as only a few behavioral tests were performed. A more robust conclusion could be made using additional tests (light-dark box test, elevated plus maze, forced swim test) although the results presented here afford rationale for continued behavioral investigation in this model. Extrapolations towards human mood disorders from mouse behavioral data should always be tempered, although in this case the wealth of data on mood alterations in human PD [44–48] provides added confidence in the interpretation of results.
The TG mice described here display several aspects of PD-like disease including GI dysfunction and behavioral alterations indicating their potential value as a model of PD with multiple system involvement.
Model characterization (gut microbiota)
TG mice displayed gut microbiota compositions with lower alpha diversity than WT mice at both 4 weeks and 24 weeks of age. A lower alpha diversity has been used as an indicator of a more “dysbiotic” gut community [49]. TG samples also clustered separately from WT samples in a PCoA plot of Bray Curtis beta diversity, indicating that these two mouse strains exhibit distinct microbial communities. The bacterial family
The analysis of changes to the microbiota between 4 and 24 weeks of age revealed PD-dependent age-related microbial shifts.
Antibiotics
To explore whether broad changes to the gut microbiota of TG mice would affect the disease phenotype, mice were treated with a cocktail of antibiotics via their drinking water for a total of 8 weeks throughout adulthood. Although there were slight trends towards worsening motor and behavioral symptoms, overall, antibiotics did not significantly affect the disease phenotype of TG mice. Fecal water content was increased and transit time showed a trend towards decrease in antibiotic-treated TG mice. Although the transit time differences were not significant, potentially due to the small sample size for this test, it is notable that in WT mice antibiotic treatment had an opposite and significant effect. Furthermore, antibiotic-treated WT mice showed no difference in fecal water content. This indicates that TG and WT mice respond differently to oral antibiotics in terms of GI function, implying that their distinct microbiotas may play different roles in modulating GI function. The gut microbiota has been shown to increase colonic serotonin levels [54, 55]; in support of this, colonic serotonin was significantly decreased in both TG and WT mice treated with antibiotics. However, the decrease in gut serotonin did not match the contrasting changes in transit time due to antibiotic treatment observed between genotypes, indicating a disconnect between transit time and gut serotonin in these mice.
A previous study using this same cocktail of antibiotics in a different transgenic mouse model of PD (administered from ∼5–12 weeks of age) found that treatment attenuated PD pathophysiology [20]. While Sampson and colleagues [20] found that antibiotic treatment improved PD-like symptoms in their mouse model, in this study we have shown trends towards a worsening PD-like phenotype which indicates that there are perhaps baseline differences in the role the microbiota plays in these two genetic mouse models. As well, these two PD mouse models are likely host to distinct baseline gut microbial communities that may respond differently to the same cocktail of antibiotics. Both the TG mice in this study and the mice used by Sampson et al. (2016) [22] are alpha-synuclein transgenic models, however the TG mice used in this study maintain the natural human
Cohousing
Since microbial depletion did not appear to have a drastic effect on the PD phenotype, we were interested to see whether microbial transfer could rescue the PD phenotype. To do so, we cohoused WT mice with TG mice as described above. Interestingly, the microbial community shifts were toward the WT composition, and were persistent (as evident by the group split after 4 weeks of cohousing). This implies that TG mice have a lower colonization resistance, which likely linked to their lower alpha diversity [61]. Even though a successful transfer was documented, it did not affect the PD phenotype, further supporting the notion that the gut microbiota does not play a significant role in this PD model. While mice that were cohoused for a full 8 weeks showed differences in behavior and gut serotonin levels when compared to mice that were split after only 4 weeks of cohousing, both of these groups of mice had very similar microbiota composition. Therefore, these changes are likely not a result of microbiota alterations and could be due to acute stress in the mice that were still being housed with WT mice during testing. Although newly-weaned female mice were chosen for cohousing studies to minimize social stress, studies have shown that exposure of mice to social stress can affect behavior [62]. Studies have also indicated that external gut microbes colonize differently depending on the sex of the recipient mouse [63], therefore, the restriction of this cohousing work to female mice only also presents a limitation in the generalizability of the conclusions. In this study we showed that substantial changes to the gut microbiota, via two discrete methods, had a minor effect on the PD-like phenotype of TG mice implying that the gut microbiota does not play a significant causal role in this model.
The gut microbiota and PD
Our findings that the gut microbiota is minimally involved in a mouse model of PD contrasts somewhat with published literature. Multiple animal studies have indicated a role for the microbiota in PD [20, 64–67]. Upon deeper inspection, many of these studies demonstrated a microbial intervention (probiotic or fecal microbial transfer (FMT)) that improved PD pathophysiology in a toxin-induced PD mouse model. In fact, two of these studies showed that while the beneficial effect of the intervention was dependent on the gut microbiota, antibiotic treatment alone had a minimal effect on PD pathophysiology [21, 68]. This provides some consistency with the absence of an effect due to antibiotics observed in our study, although our antibiotic regimen was longer. Three of these studies appear to show a causal role for the PD microbiota in promoting disease phenotype through the administration of FMT from PD mice (or human PD patients) into healthy or PD mice, and showing that PD pathophysiology was replicated or exacerbated [20, 67]. Importantly, this inferred causality from human-to-mouse FMT studies has recently been called into question [70]. The use of cohousing to transfer commensal fecal species can be thought of as a milder and less invasive form of FMT, although our study differs from the reported literature in that a “healthy” microbiota was not associated with a reduced PD-like phenotype. Recent discussions in the field have called for a more rigorous approach to determining causality for the microbiota in human disease states [71, 72]. An important consideration in determining causality is the stability of the causal relationship under different background conditions [73]; our study provides an important critical perspective to the field. It has been shown that specific gut bacterial species can induce alpha-synuclein aggregation in animal models [74]; therefore, a more targeted approach to modulating the microbiota than used here may better elucidate gut-brain interactions in PD.
While gut inflammation has been found to promote pathophysiology in a Thy-1-promoter-driven alpha-synuclein (A53T) mouse model [75], one recent study found that DSS treatment of an LPS plus paraquat mouse model of PD did not affect neurodegeneration despite inducing an increase in inflammation and microglial activation [76], both mechanistic pathways implicated in other PD studies. The study by Dwyer et al. (2021) [69] and the work presented here demonstrate the heterogeneity of PD, and highlight a need for more rigorous investigations into the importance of the gut microbiota and gut-to-brain communication pathways in PD.
Study limitations
Given that a major finding of this study was that modulations to the gut microbiota had a minimal effect on the PD phenotype in this transgenic mouse model, we were unable to identify potential mechanisms behind the role of the gut microbiota in PD. We initially investigated gut serotonin levels as a potential mechanism behind differences in transit time since serotonin is thought to be involved in GI motility, secretion, and vasodilation [38]. While TG mice displayed an increased transit time and decreased gut serotonin levels, the differences in gut serotonin levels in TG mice treated with antibiotics or cohoused with WT mice were not accompanied with the expected differences in transit time. Therefore, we chose not to pursue serotonin signaling as a potential mechanism in PD-related GI dysfunction further. Indeed, the role of serotonin in peristalsis has come into question recently [77], and other GI neurotransmitters and signaling molecules are likely more involved in modulating transit time in transgenic mice. The gut microbiota has been suggested to impact host serotonin levels [78], and the results of our antibiotic treatment experiment support the importance of the microbiota in serotonin signaling.
A potential weakness of this study is the control mouse strain chosen. We chose to attempt to replicate the Snca+/+ control used by Kuo et al. (2010) [22] as this strain best represented normal healthy mice while maintaining a similar mixed background to the TG mice. Indeed, Snca+/+ mice appeared to display the greatest differences in PD phenotype when compared to the A53T TG strain [22]. However, comparison to the
The TG mice used in this study have been previously shown to not exhibit significant loss of dopaminergic neurons [22]. Given that in human PD patients an estimated 30–50% of dopaminergic neurons are lost prior to the onset of motor symptoms [79], this may appear inconsistent. However, the finding of motor deficits in the absence of neurodegeneration is actually common in genetic models of PD—particularly in alpha-synuclein-overexpressing mouse models [31, 81]—indicating that perhaps in a murine context, alpha-synuclein can disrupt dopaminergic cell function in the absence of cell death. Furthermore, the TG mice in this study appear to have normal motor ability when observed by eye and motor deficits only become apparent under testing. Therefore, it may be possible that these mice represent an early stage of PD where symptoms are only detectable under sensitive testing (to which prodromal PD patients would not be subject).
We provided a more in-depth characterization of the
Footnotes
ACKNOWLEDGMENTS
We would like to thank the staff at the UBC Animal Care facility for their expertise and assistance with animal work. We would also like to thank Dr. Cheryl Wellington for the generous sharing of mouse behavioral equipment and analysis tools. Thank you to members of the Finlay lab for thought-provoking discussions and technical assistance. And, we would like to acknowledge the use of BioRender.com to create images used in this manuscript. This work was supported by operating grants from the Canadian Institute for Health Research (CIHR) (FDN-159935).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
