Abstract
Background:
Motor phenotype (MP) can be associated with a different prognosis in Parkinson’s disease (PD), but it is not fixed and can change over time.
Objective:
Our aim was to analyze how the MP changed over time and to identify factors associated with the changes in PD patients from a multicenter Spanish PD cohort.
Methods:
PD patients who were recruited from January-2016 to November-2017 (baseline visit; V0) and evaluated again at a 2-year±30 days follow-up (V2) from 35 centers of Spain from the COPPADIS cohort, were included in this study.MP was calculated at both visits based on Jankovic classification in TD (tremor dominant), IND (indeterminate), or PIGD (postural instability and gait difficulty). Sociodemographic and clinical data were collected, including serum biomarkers.
Results:
Five hundred eleven patients (62.57±8.59 years old; 59.2%males) were included in the study. At V0, MP was: 47.4%(242/511) TD; 36.6%(187/511) PIGD; 16%(82/511) IND. Up to 38%(194/511) of the patients changed their phenotype from V0 to V2, being the most frequent from TD to IND (8.4%) and from TD to PIGD (6.7%). A worse cognitive status (OR = 0.966) and less autonomy for activities of daily living (OR = 0.937) at V0 and a greater increase in the globalNMS burden (OR = 1.011) from V0 to V2 were associated with changing from TD to another phenotype after 2-year follow-up.
Conclusion:
The MP in PD can change over time. With disease progression, the percentage of cases with non-tremoric MP increases. PD patients who changed from TD to postural instability and gait difficulty increased NMS burden significantly.
INTRODUCTION
Parkinson’s disease (PD) is a complex disorder with a wide variety of symptoms that have a negative impact on a patient’s quality of life (QoL) and independence for activities of daily living (ADL). A very important aspect is that PD is a clinically heterogeneous disease, as symptoms and prognosis can be very different among patients [1]. It has even been suggested that PD is thus not just a dopaminergic motor disease, but a dysfunctional multi-neurotransmitter pathway driven central and peripheral nervous system disorder that ought to be considered a syndrome and not a disease [2]. In this context, it is of great value to identify different phenotypes with prognostic value. Three motor phenotypes have been described in PD: tremor-dominant (TD), postural instability and gait difficulty (PIGD), and indeterminate (IND) subtype [3]. These phenotypes have been associated with different cognitive trajectories, motor outcomes, and biomarkers profiles [4, 5]. Previous studies have shown that the TD variant has a slower rate of progression with less deterioration regarding QoL [6, 7], and some serum markers have been identified as linked to TD [8]. However, data from some prospective follow-up cohorts indicate that the motor phenotype is not fixed and can change over time, with patients, initially identified as TD, changing to another phenotype after only a 1-year follow-up [8, 9]. Simuni et al. observed in 323 de novo PD patients from the Parkinson’s Progression Markers Initiative (PPMI) cohort that 39%PIGD and 18%TD shifted subtypes after 12 months independently of dopaminergic treatment [9]. More recently, Luo et al. found using data from the PPMI (N = 115) and BioFIND (Fox Investigation for New Discovery of Biomarkers in PD) (N = 162) studies that motor subtypes are not fixed and change with progression of the disease and with treatment [10]. In studies with long-term follow up, more than 50%of TD patients can shift to another motor phenotype [11]. Although some studies did not really find significant differences of symptoms and dopaminergic innervation between TD and PIGD patients after a certain period of follow up, importantly, patients who switched from the TD subtype to the PIGD subtype during disease progression experienced a worse clinical prognosis [12].
In this framework, we suggest the hypothesis that TD patients have a better prognosis and that those TD patients who change to a non-TD phenotype overtime would present a worse outcome. Moreover, factors with prognostic value associated with a change from a TD to a non-TD phenotype and on the contrary, with a stable-TD motor phenotype, may be identified. The aims of the present study was: 1) to identify the motor phenotype in PD patients from a multicenter Spanish PD cohort and factors associated with it; 2) to analyze the change in the motor phenotype after 2-year follow-up; 3) to compare the progression of PD patients with different phenotypes including those subjects who changed from TD phenotype to non-TD phenotype after 2-year follow-up vs. those ones remaining as TD patients (stable TD patients); 4) to identify independent factors associated with having a stable TD motor phenotype and with a change from TD to another phenotype after 2-year follow-up; 5) to analyze the value of serum biomarkers (SB) regarding the motor phenotype. Importantly and compared to previous papers, we provide here a very complete analysis conducted in a multicenter cohort with robust data in which many variables, motor and non-motor in addition to serum biomarkers, were included, filling these data a gap in the literature.
METHODS
PD patients who were recruited from January-2016 to November-2017 (baseline visit; V0) and evaluated again at a 2-year±30 days follow-up (V2) from 35 centers of Spain from the COPPADIS cohort [13], were included in this study. Methodology about COPPADIS-2015 study can be consulted in https://bmcneurol.biomedcentral.com/articles/10.1186/s12883-016-0548-9 [14]. This is a longitudinal-prospective, 5-year follow-up study designed to analyze natural progression of PD in which patients diagnosed with PD according to UK PD Brain Bank criteria without dementia were included [13].
Information on sociodemographic aspects, factors related to PD, comorbidity, and treatment was collected. Motor status, non-motor symptoms (NMS), QoL and disability was assessed at V0 and at V2 using different validated scales: Hoenh & Yahr (H&Y) [15]; UPDRS-III and UPDRS-IV [16]; Freezing of Gait Questionnaire [FOGQ]) [17]; Parkinson’s Disease Cognitive Rating Scale (PD-CRS) [18]; Non-Motor Symptoms Scale (NMSS) [19]; Beck Depression Inventory-II (BDI-II) [20]; Parkinson’s Disease Sleep Scale (PDSS) [21]; Neuropsychiatric Inventory (NPI) [22]; Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease-Rating Scale (QUIP-RS) [23]; Visual Analog Scale-Pain (VAS-Pain) [24]; Visual Analog Fatigue Scale (VAFS]) [25]; the 39-item Parkinson’s disease Questionnaire (PDQ-39) [26]; PQ-10 [27]; the EUROHIS-QOL 8-item index (EUROHIS-QOL8) [28]; ADLS (Schwab & England Activities of Daily Living Scale) [29]. In patients with motor fluctuations, the motor assessment was made during the OFF state (without medication in the last 12 hours) and during the ON state [14]. The assessment was only performed without medication in patients without motor fluctuations.
Classification of motor PD phenotype
Motor phenotype was calculated based on a previously published formula using 4 items from the Unified Parkinson's Disease Rating Scale (UPDRS) part II (UPDRS-II) and 4 items from the part III (UPDRS-III) [3]. From UPDRS-II, items were: 2.13 (falling unrelated to freezing); 2.14 (freezing when walking); 2.15 (walking); and 2.16 (tremor). From UPDRS-III, items were: 3.20 (tremor at rest); 3.21 (action or postural tremor of hands); 3.29 (gait); 3.30 (postural stability). Tremor score was calculated as the mean of the summation of items 2.16, 3.20, and 3.21 whereas PIGD score as the mean of the summation of items 2.13, 2.14, 2.15, 3.29 and 3.30. The motor phenotype was defined according to the result of the next quotient: TD score / PIGD score: 1)≥1.5, tremor dominant (TD); ≤1, postural instability and gait difficulty (PIGD); > 1 but < 1.5, indeterminate (IND). All patients were assessed during the OFF state (without medication in the last 12 hours) when the motor phenotype was defined. The motor phenotype was defined at both visits, V0 and V2. A stable motor phenotype was defined as the same phenotype at both V0 and V2 (TD-TD; IND-IND; PIGD-PIGD). A non-stable motor phenotype was defined as a motor phenotype changing from V0 to V2 (TD-IND, TD-PIGD, IND-TD, IND-PIGD, PIGD-TD, PIGD-IND). The definition of a stable phenotype throughout the 2-year follow-up was carried out, on the one hand, with the intention of answering clearly the question of how many patients changed their phenotype and, secondly, to define the stable-TD phenotype based on the suggested hypothesis.
Serum biomarkers determination
Blood sample collection for the determination of different SB included S-100b protein, tumor necrosis factor (TNF)-a, interleukin (IL)-1, IL-2, IL-6, vitamin B12, methylmalonic acid, homocysteine, uric acid, ultrasensitive CRP (US-CRP), ferritin, and iron. SB levels were determined from frozen blood samples obtained from subjects participating in the COPPADIS-2015 study from 9 centers of Spain. The extraction of the sample was carried out no longer than 3 months after the first clinical assessment (V0) in the absence of infections and/or fever. The analysis was conducted at a common laboratory: REFERENCE LABORATORY (http://www.reference-laboratory.es). Different methods were used: visible spectrophotometry (iron); immunoluminescence (S-100b protein, ferritin, vitamin B12, and homocysteine); enzimoimmunoassay (IL-1, IL-2, and TNF-a); immunoassay (US-CRP); mass spectrometry (methylmalonic acid); enzymatic technique (uric acid). Outliers were excluded from the analysis.
Data analysis
Data were processed using SPSS 20.0 for Windows. Data were expressed as a percentage or mean±standard deviation. For comparisons between groups, the Student’s t-test, Mann-Whitney-Wilcoxon test, Chi-square test, or Fisher test were used as appropriate (distribution for variables was verified by one-sample Kolmogorov-Smirnov test). Marginal homogeneity test was used for determining if the frequency of different motor phenotypes changed significantly or not from V0 to V2. General linear model (GLM) repeated measures were used to test whether the mean differences of scores in different scales applied between the two visits (V0 and V2) were significant with regard to the phenotype. Age, gender, disease duration, H&Y stage, and levodopa equivalent daily dose (LEDD) at V0 [30] were included as covariates. Cohen's d formula was applied for measuring the effect size. It was considered: small effect = 0.2; medium effect = 0.5; large effect = 0.8. Firstly, we analyzed differences between groups at baseline: TD vs. IND vs. PIGD; TD vs. non-TD. Secondly, differences between those patients with a stable-TD motor phenotype (TD-TD) and without stable-TD motor phenotype (TD-IND; TD-PIGD; IND-IND; IND-TD; IND-PIGD; PIGD-PIGD; PIGD-IND; PIGD-TD) were analyzed. Regarding the hypothesis of a better outcome in stable-TD PD patients, binary regression models were used for determining independent factors associated with stable-TD motor phenotype (being TD at V0 and staying as TD at V2), and with the change from TD phenotype at V0 to non-TD phenotype at V2. Any variables with univariate associations with p-values < 0.20 were included in a multivariable model, and a backwards selection process was used to remove variables individually until all remaining variables were significant at the 0.10 level except age at onset, gender, disease duration, H&Y stage, and LEED, which were included as covariates given the heterogeneity of the sample. A p-value < 0.05 was considered significant.
Standard protocol approvals, registrations, and patient consents
Approval from the Comité de Ética de la Investigación Clínica de Galicia from Spain (2014/534; 02/DEC/2014) was obtained. A written informed consent from all participants was signed. COPPADIS-2015 was classified by the AEMPS (Agencia Española del Medicamento y Productos Sanitarios) as a Post-authorization Prospective Follow-up study with the code COH-PAK-2014-01.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request. No computer coding was used in the completion of the current manuscript.
RESULTS
Five hundred eleven patients (62.57±8.59 years old; 59.2%males) from the COPPADIS cohort with a mean disease duration of 5.52±4.27 years were included in the study. At V0, motor phenotypes were: 47.4%(242/511) TD; 36.6%(187/511) PIGD; 16%(82/511) IND (Fig. 1). Compared to baseline, a significant change (p = 0.009) was observed in the motor phenotype at V2:39.5%(202/511) TD; 39.2%(200/511) PIGD; 21.3%(109/511) IND. Up to 38%(194/511) of the patients changed their phenotype from V0 to V2:8.4%(43/511) from TD to IND; 7.6%(39/511) from TD to PIGD; 6.7%(34/511) from PIGD to IND; 6.5%(33/511) from PIGD to TD; 4.9%(25/511) from IND to PIGD; 3.9%(20/511) from IND to TD. PIGD motor phenotype was the most stable (67.9%with the same phenotype at V2) and IND the least (42.7%with the same phenotype at V2) (Fig. 1).
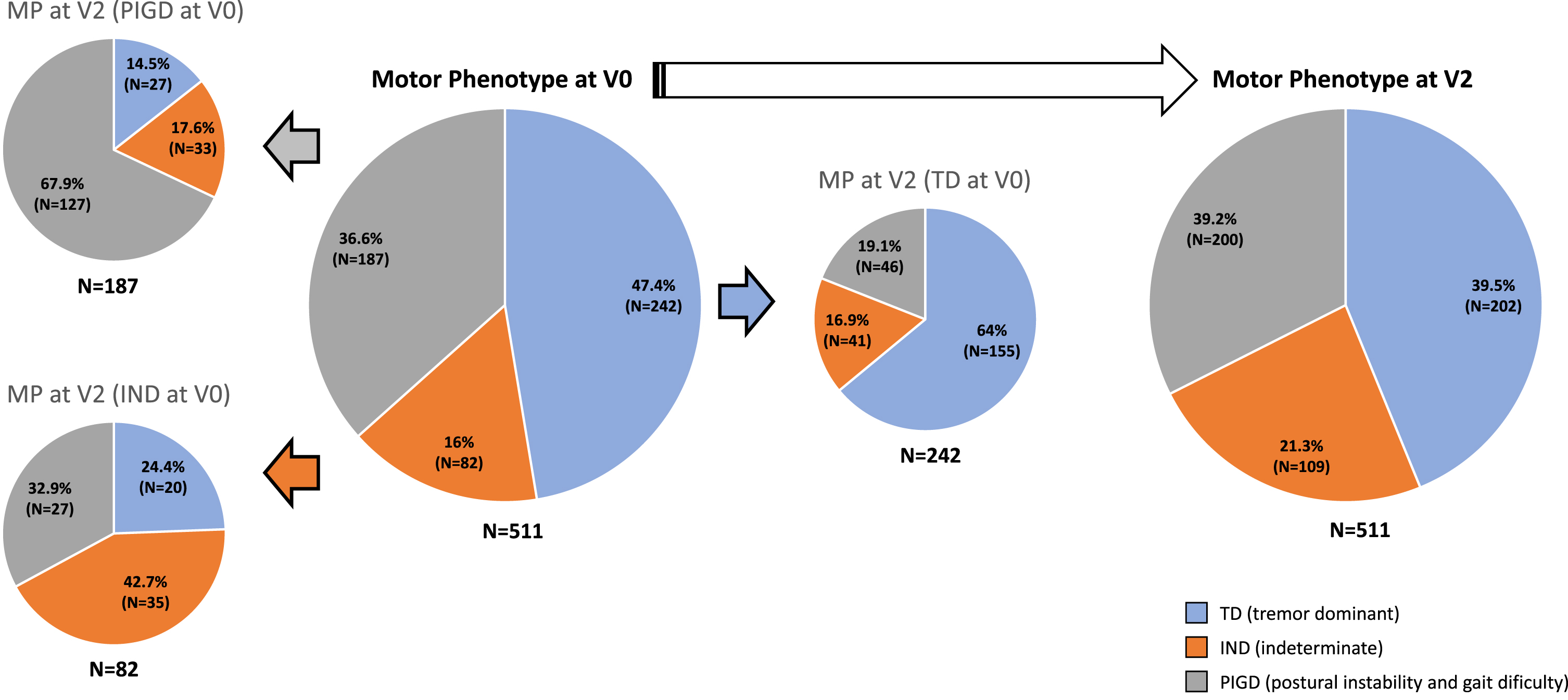
Percentage of patients with different motor phenotype (TD vs. PIGS vs. IND) at V0 and at V2 (difference between both visits, p = 0.009). For patients with different phenotype at V0, motor phenotype at V2 is shown. IND, indeterminate; PIGD, postural instability gait difficulty; TD, tremor dominant.
Compared to other motor phenotypes at baseline, TD patients were receiving lower LEDD and daily dose of levodopa, had lower disease duration, were in an earlier H&Y stage, and in general had a better status in terms of motor symptoms / complications (UPDRS-III; UPDRS-IV; FOGQ), NMS (NMSS; BDI-II; VAS-PAIN; VASF-Physical; VASF-Mental), independence for ADL (ADLS), and QoL (PDQ-39SI; PQ-10; EUROHIS-QoL) (Table 1). Regarding SB, lower serum levels of S-100b protein, and methylmalonic acid were detected in TD compared to non-TD patients at V0:0.06±0.04 vs. 0.17±0.78 (p = 0.023) and 0.14±0.09 vs. 0.21±0.18 (p = 0.011), respectively. When the change from V0 to V2 in motor symptoms, NMS, QoL and autonomy for ADL was compared between TD patients at V0 and those patients without TD motor phenotype at V0, only the increase was significantly greater in TD patients on VAS-PAIN (Cohen's d = 0.31 vs. 0; p = 0.005) and VASF-Physical (Cohen’s d = 0.29 vs. 0.02; p = 0.039) (Table 1).
PD-related variables in patients regarding the motor phenotype
Results are expressed ad mean±SD or %. ANOVA, Mann-Whitney-Wilcoxon and Chi-square tests were applied for assessing the relation with variables at V0. General linear model (GLM) repeated measure were applied for analyzing the relation with the change of variables from V0 to V2 (age, gender, disease duration, H&Y-OFF at V0, and LEDD at V0 were included as covariates except for analyzing the change from V0 to V2 in LEDD –only the other 4 variables); pa, TD vs. IND vs. PIGD; pb, TD vs. non-TD. ADLS, Schwab & England Activities of Daily Living Scale; BDI, Beck Depression Inventory-II; EUROHIS-QOL8, European Health Interview Survey-Quality of Life 8 Item-Index; FOGQ, Freezing Of Gait Questionnaire; H&Y, Hoenh & Yahr; LEED, levodopa equivalent daily dose; NMS, non-motor symptoms; NMSB, non-motor symptoms burden; NMSS, Non-Motor Symptoms Scale; NPI, Neuropsychiatric Inventory; PD, Parkinson's disease; PD-CRS, Parkinson’s Disease Cognitive Rating Scale; PDQ-39SI, 39-item Parkinson’s Disease Quality of Life Questionnaire Summary Index; PDSS, Parkinson’s Disease Sleep Scale; QoL, Quality of life; QUIP-RS, Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease-Rating Scale; UPDRS, Unified Parkinson’s Disease Rating Scale; VAFS, Visual Analog Fatigue Scale; VAS-Pain, Visual Analog Scale-Pain.
When PD patients with stable-TD motor phenotype were compared to patients with another phenotype (non stable-TD), to have a stable-TD motor phenotype was associated with lower doses of medication (LEED and daily dose of levodopa), a shorted disease duration, an earlier H&Y stage, and a better status in terms of motor symptoms/complications (UPDRS-III; UPDRS-IV; FOGQ), cognition (PD-CRS), NMS (NMSS; BDI-II; PDSS; VAS-PAIN; VASF-Physical; VASF-Mental), independence for ADL (ADLS), and QoL (PDQ-39SI; PQ-10; EUROHIS-QoL) (Table 2). Specifically, global NMS burden (NMSS total score) at V0 was lower in stable-TD patients compared to non stable-TD patients (35.7±34.84 vs. 48.66±37.89; p < 0.0001), but the increase in the NMSS total score from V0 to V2 was significantly higher (p = 0.010) in non stable-TD patients (Coehn’s effect = 0.41) than in stable-TD patients (Coehn's effect = 0.1) as well (Table 2 and Fig. 2A). By domains, the score of all domains increased from V0 to V2 significantly in non stable-TD patients except urinary symptoms but only cardiovascular symptoms in those patients with stable-TD phenotype (Supplementary Table 1 and Fig. 2B). Something similar was observed with the UPDRS-III-OFF, presenting a lower UPDRS-III-OFF score at baseline in stable-TD patients compared to non stable-TD patients (20.09±10.39 vs. 22.99±10.59; p = 0.002) and a lesser increase in the score from V0 to V2 (Coehn’s effect = 0.51 vs. 0.29; p = 0.013) as well (Table 2). With regard to SB, in stable-TD patients compared to non stable-TD patients, lower serum levels of S-100b protein (0.06±0.03 vs. 0.14±0.66; p = 0.006) and methylmalonic acid (0.12±0.41 vs. 0.19±0.17; p = 0.006) were detected.
PD-related variables in patients with TD stable motor phenotype (TD at V0 and at V2) compared to the rest of patients (IND stable motor phenotype, PIGD stable motor phenotype or non-stable phenotype)
Results are expressed ad mean±SD or %. Mann-Whitney-Wilcoxon and Chi-square tests were applied for assessing the relation with variables at V0. General linear model (GLM) repeated measure were applied for analyzing the relation with the change of variables from V0 to V2 (age, gender, disease duration, H&Y-OFF at V0, and LEDD at V0 were included as covariates except for analyzing the change from V0 to V2 in LEDD –only the other 4 variables). ADLS, Schwab & England Activities of Daily Living Scale; BDI, Beck Depression Inventory-II; EUROHIS-QOL8, European Health Interview Survey-Quality of Life 8 Item-Index; FOGQ, Freezing Of Gait Questionnaire; H&Y, Hoenh&Yahr; LEED, levodopa equivalent daily dose; NMS, non-motor symptoms; NMSB, non-motor symptoms burden; NMSS, Non-Motor Symptoms Scale; NPI, Neuropsychiatric Inventory; PD, Parkinson's disease; PD-CRS, Parkinson’s Disease Cognitive Rating Scale; PDQ-39SI, 39-item Parkinson’s Disease Quality of Life Questionnaire Summary Index; PDSS, Parkinson’s Disease Sleep Scale; QoL, Quality of life; QUIP-RS, Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease-Rating Scale; UPDRS, Unified Parkinson’s Disease Rating Scale; VAFS, Visual Analog Fatigue Scale; VAS-Pain, Visual Analog Scale-Pain.
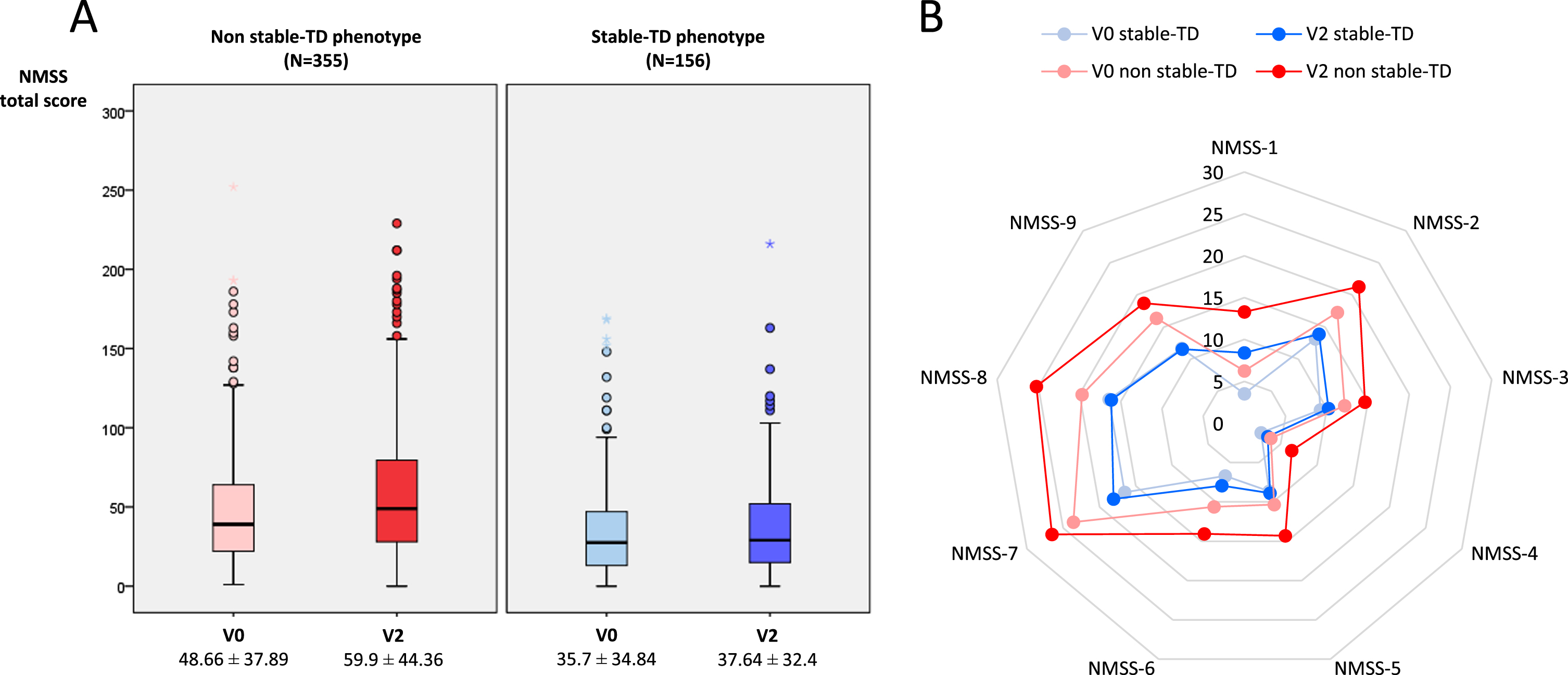
A) Change in NMSS total score from V0 (baseline) to V2 (2-year follow-up±1 month) in PD patients without stable-TD phenotype (N = 335) and with stable-TD phenotype (N = 156). V2 vs. V0 in patients without stable-TD phenotype, p < 0.0001; V2 vs. V0 in patients with stable-TD phenotype, p = 0.439. B) Change from V0 to V2 in the score of different domains of the NMSS in patients without stable-TD phenotype (p < 0.05 for all domains except urinary symptoms) and with stable-TD phenotype (p < 0.05 only for cardiovascular symptoms). Data and p values are shown in Supplementary Table 1. NMSS, Non-Motor Symptoms Scale; TD, tremor dominant.
Factors independently associated with having a stable TD motor phenotype (TD-TD) were to be older at symptoms onset (OR = 1.075; p = 0.013), to have a higher score at V0 on UPDRS-III-OFF (OR = 1.098; p = 0.005), PD-CRS (OR = 1.050; p = 0.004), and ADLS (OR = 1.100; p = 0.005), to have a lower FOGQ score (OR = 0.754; p = 0.014), and to have lower serum levels of methylmalonic acid (OR = 0.001; p = 0.038) at V0 (R2 = 0.52; Hosmer and Lemeshow test, p = 0.351; Omnibus test, p < 0.0001) (Table 3; N = 511). When each continuous variable was considered as dichotomous variable in the same model, the next results were observed: age at onset > 60 years, OR = 3.99 (95%CI, 1.417–11.239; p = 0.009; R2 = 0.53); UPDRS-III score > 30 at V0, OR = 7.051 (95%CI, 1.646–30.205; p = 0.009; R2 = 0.51); FOGQ total score < 5 at V0, OR = 8.762 (95%CI, 1.650–46.533; p = 0.011; R2 = 0.52); PD-CRS total score > 90 at V0, OR = 2.656 (95%CI, 1.036–6.81; p = 0.042; R2 = 0.49); ADLS score > 90%at V0, OR = 3.095 (95%CI, 1.190–8.051; p = 0.021; R2 = 0.5); serum levels of methylmalonic acid < 0.20 (mcmol/L) at V0, OR = 4.611 (95%CI, 1.058–20.001; p = 0.042; R2 = 0.51) (Fig. 3). Independent factors associated with the change from TD motor phenotype to another phenotype at V2 were to be receiving a higher LEDD at V0 (OR = 1.001; p = 0.009), to have a lower score at V0 on PD-CRS (OR = 0.966; p = 0.008) and ADLS (OR = 0.937; p = 0.011), and to suffer a greater increase in the NMSS total score from V0 to V2 (OR = 1.007; p = 0.007) (R2 = 0.28; Hosmer and Lemeshow test, p = 0.299; Omnibus test, p < 0.0001) (Table 4; N = 242). Specifically, an increase greater than 20 points from V0 to V2 on the NMSS total score tripled the probability of changing from TD to another phenotype (OR = 3.224; 95%CI, 1.309–7.943; p = 0.011; R2 = 0.25).
Binary regression model about factors associated with stable TD motor phenotype (i.e., TD phenotype at V0 and at V2)
Dependent variable: stable TD motor phenotype (i.e., TD phenotype at V0 and at V2). OR and 95%IC are shown. aunivariate analysis; bmultivariate analysis (R2 = 0.52; Hosmer and Lemeshow test, p = 0.351; Omnibus test, p < 0.0001). ADLS, Schwab & England Activities of Daily Living Scale; FOGQ, Freezing Of Gait Questionnaire; H&Y, Hoehn & Yahr; LEDD, levodopa equivalent daily dose; PD-CRS, Parkinson’s Disease Cognitive Rating Scale; UPDRS, Unified Parkinson’s Disease Rating Scale.
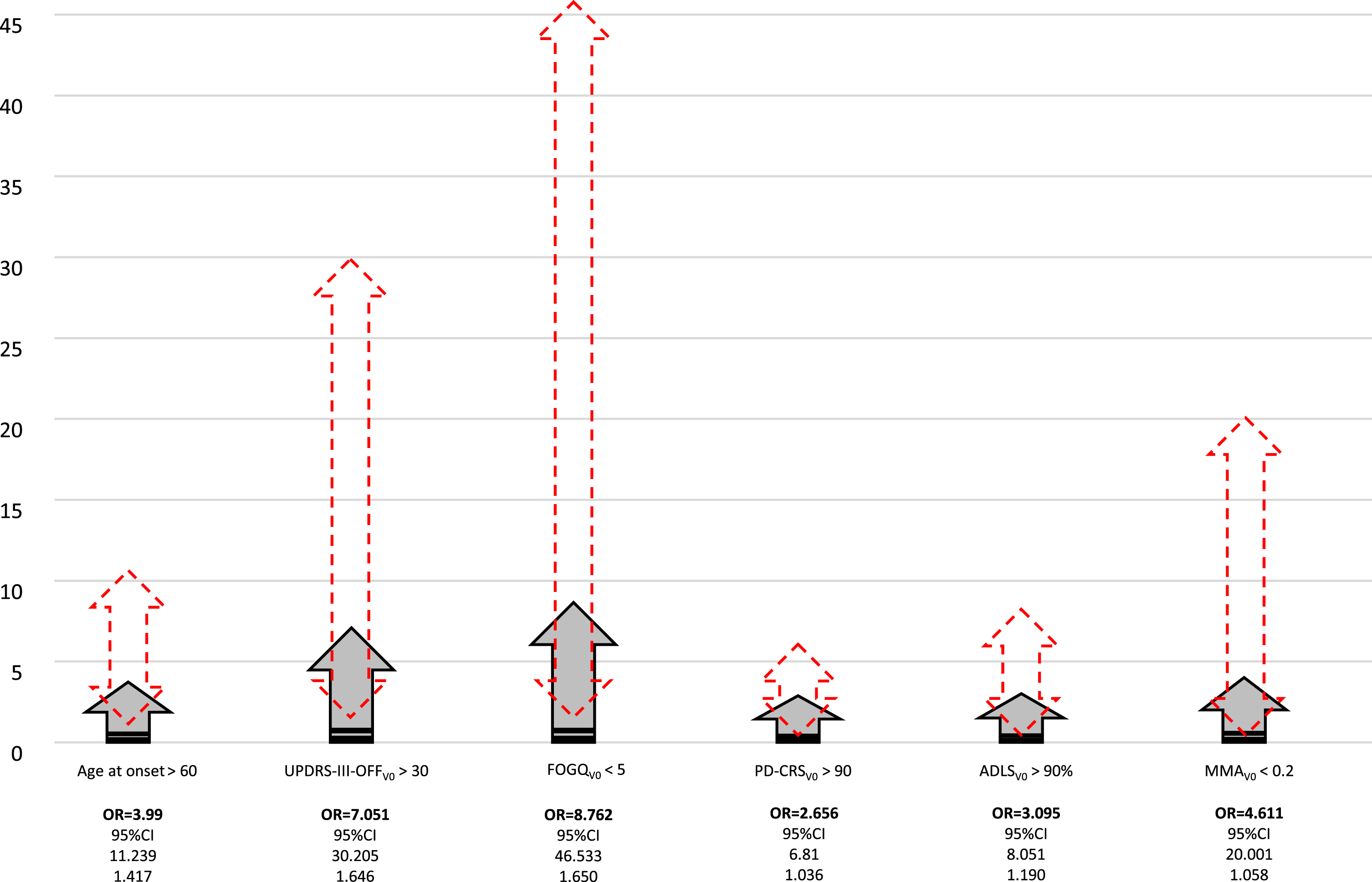
Factors independently associated with having a stable TD motor phenotype (i.e., TD at V0 and again TD at V2). Grey arrows show the OR. Red arrows with discontinue line represent the 95%CI. ADLS, Schwab & England Activities of Daily Living Scale; FOGQ, Freezing Of Gait Questionnaire; MMA, methylmalonic acid; OR, odds ratio; PD-CRS, Parkinson’s Disease Cognitive Rating Scale; UPDRS, Unified Parkinson’s Disease Rating Scale.
Binary regression model about predictors of the change from TD motor phenotype at V0 to non-TD motor phenotype at V2
Dependent variable: change in motor phenotype from stable TD at V0 to another phenotype at V2 (i.e., TD-IND, TD-PIGD). OR and 95%IC are shown. aunivariate analysis; bmultivariate analysis (R2 = 0.28; Hosmer and Lemeshow test, p = 0.299; Omnibus test, p < 0.0001). ADLS, Schwab & England Activities of Daily Living Scale; FOGQ, Freezing Of Gait Questionnaire; H&Y, Hoehn&Yahr; LEDD, levodopa equivalent daily dose; PD-CRS, Parkinson’s Disease Cognitive Rating Scale; UPDRS, Unified Parkinson’s Disease Rating Scale.
DISCUSSION
The present study shows that motor phenotype in PD patients is unstable and, in up to 40%of the patients, it changed after 2-year follow-up. We identified a better cognitive status, more independence for ADL, less gait problems, a greater score on the UPDRS-III, and lower serum levels of methylmalonic acid as independent factors associated with stable-TD motor phenotype. Moreover, a greater impairment from baseline to 2-year follow-up in the global NMS burden was associated with the change from TD to non-TD motor phenotype.
The frequency of different motor phenotypes in previous studies varies depending on the classification used and, since PIGD phenotype is related to disease duration [16], on the time from symptoms onset [8–11, 32]. For example, frequency of PIGD phenotype was 17.5%in a study from the PPMI cohort (N = 320) with an average disease duration starting at 6.7 months after diagnosis [9] but up to 52.7%in 226 PD patients with a median disease duration of 2 years [33]. Jankovic et al. reported 29.1%of PIGD cases in 118 patients from the Deprenyl and Tocopherol Antioxidative Therapy of Parkinsonism (DATATOP) cohort with an average disease duration of 1.25 years, which was the basis of the TD/PIGD classification [3]. However, Ren et al. recently found up to 56.2%of PIGD in 283 newly diagnosed, untreated PD patients using the Jankovic classification [34]. Another important factor that can cause variability is the motor state in which the patient is evaluated, since the phenotype can change if the patient is evaluated during the ON or during the OFF state [10]. In practice, the patient can sometimes be in the semi-ON or semi-OFF state. Importantly, in our study all the patients were classified as TD, IND or PIGD while they were in the OFF state, by protocol. Algorithms developed to distinguish TD and PIGD phenotypes use motor exam (Part II and III) scores from UPDRS or the revised Movement Disorder Society- UPDRS (MDS-UPDRS) [3, 36]. Differentiation between TD and PIGD parkinsonism allows clinicians to tailor interventional strategies that accommodate the person’s unique symptomatology and the proper changing needs. However, motor phenotype is unstable and changes throughout the progression of the disease, as it is more probable to have a PIGD phenotype in the long-term. Of 423 patients from the PPMI, the percentage of PIGD was lower than 20%at baseline, 23.4%after 1 year, and 30.5%after 5 years, and the contrary for TD, higher than 70%, 65.7%and 60.3%, respectively [10]. Motor phenotype can vary even after only 1-month follow-up. Using the Jankovic classification, Ren et al. [34] recently observed that up to 45.4%and 27.5%of TD and PIGD patients, respectively, had an unstable motor phenotype, changing to another phenotype only 1 month after the first evaluation. In fact, fair consistency was observed between the Jankovic, Schiess, and Kang classification systems in this study, as the Schiess classification was the most stable and the Jankovic classification was the most unstable [34]. We observed 47.4%TD and 36.6%PIGD cases at baseline with a mean time from symptoms onset of 5.5 years, being unstable 36%and 32.1%of TD and PIGD patients, respectively, after 2 years. Our findings seem to align with previous reports and suggest that a significant portion of patients switches from the TD subtype to the PIGD subtype during disease progression [9, 34].
On the other hand, recent clinical and neurobiological research suggests the existence of non-motor subtypes in PD: Park Cognitive; Park Apathy; Park Depression/Anxiety; Park Sleep; Park Pain; Park Fatigue; Park Autonomic [35]. Brendel et al. [36] identified three unique subtypes using data from the BioFIND cohort with clustering analysis: subtype I, characterized by mild symptoms, both motor and non-motor; subtype II, characterized by an intermediate severity, with a high tremor score and mild non-motor symptoms. Subtype III, with more severe motor and non-motor symptoms. Although different biological markers may be useful in the future when classifying PD for its management (i.e., genetic, neuroimaging, biochemical, etc.), in Brendel study there was no clear difference in demographics, biomarker levels, and genetic risk scores. Unfortunately, non-motor phenotypes were not analyzed in our cohort.
The presence of non-stable-TD phenotype was associated in our cohort with more severe disease including motor and non-motor affectation, greater dependence for ADL, and a worse QoL. Wu et al. reported that PIGD phenotype had a higher prevalence of NMS and worse QOL than TD phenotype in a cross-sectional study conducted in 301 patients with early PD [39]. Specifically, Huang et al. observed that patients with the PIGD subtype had more severe sleep impairment, fatigue, and urinary disturbance compared with patients with the TD subtype [40]. Ren et al. observed in 119 de novo PD patients with the PIGD phenotype more severe cardiovascular symptoms, sleep impairments, mood disturbances, and pain after comparing with 73 TD and 34 IND patients [33]. In line with previous evidence [33, 39–41], a greater global NMS burden (NMSS total score) and pain (VAS), fatigue (VAS), and cognitive (PD-CRS), mood (BDI-II) and sleep (PDSS) problems at baseline were linked to non-TD phenotype in our cohort. However, a key aspect is that PIGD phenotype is related to longer disease duration and many confounded factors exist because motor and NMS burden progress over time. Proof of this is that a lower score on the UPDRS-III scale was associated with the TD phenotype, but after adjusting to other covariates including axial symptoms and time from symptoms onset, the relationship inverted significantly. Presenting tremor can lead to an increase in the scale score compared to not presenting it, which would range from 0 (without tremor) to a maximum of 20 points.
Interestingly, a better cognitive status and lower serum levels of methylmalonic acid were identified as independent factors associated with stable TD phenotype. Although cognitive impairment was not associated with PIGD subtype in 422 patients from the PPMI cohort, Urso et al. found postural instability as an independent predictor of cognitive impairment [42]. One previous study suggested that PIGD seems to correlate with cognitive impairment, in which underlying mechanism might be involved in Aβ1–42 aggregation in the brain and relevant neurochemical disturbance featured by the depletion of HVA in CSF [43]. Levodopa exposure is associated with methylmalonic acid elevation, and lower serum levels could be associated with less severe disease requiring less medication, as in TD patients [44]. Although Lian et al. observed increased ferritin levels in the serum compared with the PD-PIGD and control groups [8] suggesting that iron metabolism and relevant inflammation might modulate clinical phenotypes of PD, we did not find differences in ferritin and iron serum levels or any other SB between groups. By the contrary, an inverse relationship between cognitive status and the risk of changing from TD to non-TD phenotype was found in our cohort. Moreover, patients switching from the TD subtype to the PIGD subtype during disease progression had not only a worse clinical prognosis but also a greater impairment in the global NMS burden. Biomarker studies point toward non-TD PD as representing more advanced and diffuse neurodegeneration than TD PD, encompassing dopaminergic and non-dopaminergic as well as synuclein and non-synuclein (Abeta) pathologies [45]. Finally, another factor related to phenotype was age at onset, tripling the probability of stable-TD phenotype when it was higher than 60. Previous studies reported that tremor at presentation is twice as common in those with onset over 64 years as compared to those with onset under 45 (early onset PD) and becomes more common with increasing age at onset [46].
The present studies have some important limitations. It was a secondary data analysis of the COPPADIS cohort and analyses were limited in the number of data available. The information about motor phenotype at baseline and after 2-year follow-up was recorded in 511 out of 689 PD patients (74.2%). However, this is a limitation observed in other prospective studies analyzing the change in phenotype, with maintenance rates of 75%at 1-year [32]. Moreover, the follow-up time is not long and the period in which the motor phenotype was defined was based on the COPPADIS study methodology, with all the information collected (motor symptoms, NMS, QoL and disability) with different scales 2 years after the baseline visit. Based on the COPPADIS project [14], we will plan to carry out an analysis with a 5-year follow-up. It would be of great interest to define what could be considered a stable phenotype in future publications. As it has been previously commented, we used the Jankovic classification, which is more unstable than others, and non-motor phenotypes were not included. Our sample was not fully representative of the PD population due to inclusion and exclusion criteria at baseline (i.e., age limit, no dementia, no severe comorbidities, no second line therapies, etc.) [14]. Instead of a specific tool for assessing comorbidity, like Charlson Index or others, the total number of non-antiparkinsonian medications was used as a surrogated marker of comorbidity [47]. By the contrary, the strengths of our study include the large sample size and the extensive clinical and demographic information recorded with many justified variables included in the models in a multicenter prospective longitudinal cohort. Despite this, the regression models used to identify independent factors associated with stable-TD and unstable-TD motor phenotype explains 52%and 28%of the of the variance, respectively, but it was either low as well or not provided or analyzed in other studies [9–12, 34]. Maybe, other biomarkers of PD per se (genetic, biochemical, neuroimaging, etc.) not included could explain part of the variance. Furthermore, a lower sample size in the model for determining what factors were independently associated with the change from TD phenotype at V0 to another phenotype at V2 could explain the lower value of R2.
In conclusion, this study shows in a large cohort of patients that the motor phenotype in PD is unstable, and that the change from a tremoric to non-tremoric phenotype is associated with a significant increase in the global burden of non-motor symptoms. Cognition, gait and serum levels of methylmalonic acid may be biomarkers related to a specific motor phenotype.
Footnotes
ACKNOWLEDGMENTS
We would like to thank all patients and their caregivers who collaborated in this study. Many thanks also to Fundación Española de Ayuda a la Investigación en Parkinson y otras Enfermedades Neurodegenerativas (Curemos el Parkinson; http://www.curemoselparkinson.org), Alpha Bioresearch (![]() ), and other institutions helping us.
), and other institutions helping us.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
FINANCIAL DISCLOSURES
Santos García D. has received honoraria for educational presentations and advice service by AbbVie, UCB Pharma, Lundbeck, KRKA, Zambon, Bial, Italfarmaco, and Teva.
Cores Bartolomé C. has received honoraria for educational presentations and advice service by Lundbeck and UCB Pharma.
Jesús S. has received honoraria from AbbVie, Bial, Merz, UCB, and Zambon and holds the competitive contract “Juan Rodés” supported by the Instituto de Salud Carlos III. She has received grants from the Spanish Ministry of Economy and Competitiveness (PI18/01898) and the Consejería de Salud de la Junta de Andalucía (PI-0459-2018).
Aguilar M: UCB and Schwabe with assistance to a Congress; Nutricia with assistance to a congress and payment of lecture.
García Caldentey J. has received honoraria for educational presentations and advice service by Qualigen, Nutricia, AbbVie, Italfarmaco, UCB Pharma, Lundbeck, Zambon, Bial, and Teva.
Caballol N. has received honoraria from Bial, Italfármaco, Qualigen, Zambon, UCB, Teva and KRKA and sponsorship from Zambon, TEVA and AbbVie for attending medical conferences.
Legarda I. has received honoraria for educational presentations and advice service by AbbVie, UCB Pharma, Zambon, Bial, and Teva.
Hernández Vara J. has received travel bursaries and educational grants from AbbVie and has received honoraria for educational presentations from AbbVie, Teva, Bial, Zambon, Italfarmaco, and Sanofi-Genzyme.
Cabo I. has received honoraria for educational presentations and advice service by AbbVie, Zambon, and Bial.
López Manzanares L: Compensated advisory services, consulting, research grant support, or speaker honoraria: AbbVie, Acorda, Bial, Intec Pharma, Italfarmaco, Pfizer, Roche, Teva, UCB, and Zambon.
Ávila Rivera MA. has received honoraria from Zambon, UCB Pharma, Qualigen, Bial, and Teva, and sponsorship from Zambon and Teva for attending conferences.
Puente V. has served as consultant for AbbVie and Zambon; has received grant/research from AbbVie.
Dotor García-Soto J: Compensated advisory services, consulting, research grant support, or speaker honoraria: Merck, Sanofi-Genzyme, Allergan, Biogen, Roche, UCB and Novartis.
Solano Vila B. has received honoraria for educational presentations and advice service by UCB, Zambon, Teva, AbbVie, Bial.
Álvarez Sauco M. has received honoraria for educational presentations and advice service by AbbVie, UCB Pharma, Zambon, Bial, and Teva.
Vela L. has received honoraria for educational presentations and advice service by AbbVie, UCB Pharma, Lundbeck, KRKA, Zambon, Bial, and Teva.
Escalante S. has received honoraria for educational presentations and advice service by AbbVie, Zambon, and Bial.
Cubo E: Travel grants: AbbVie, Allergan, Boston; Lecturing honoraria: AbbVie, International Parkinson’s disease Movement Disorder Society.
Carrillo Padilla F. has received honoraria from Zambon (SEN Congress assistance).
Martínez Castrillo JC. has received research support from Lundbeck, Italfarmaco, Allergan, Zambon, Merz, and AbbVie. He has received speaking honoraria from AbbVie, Bial, Italfarmaco, Lundbeck, Krka, TEVA, UCB, Zambon, Allergan, Ipsen, and Merz.
Sánchez Alonso P. has received honoraria for educational presentations and advice service by AbbVie, UCB Pharma, Lundbeck, KRKA, Zambon, Bial, and Teva.
Alonso Losada MG. has received honoraria for educational presentations and advice service by Zambon and Bial.
López Ariztegui N. has received honoraria for educational presentations and advice service by AbbVie, Italfarmaco, Zambon, and Bial.
Gastón I. has received research support from AbbVie and Zambon and has served as a consultant for AbbVie, Exelts, and Zambon.
Kulisevsky J: (1) Consulting fees: Roche, Zambon; (2) Stock / allotment: No; (3) Patent royalties / licensing fees: No; (4) Honoraria (e.g., lecture fees): Zambon, Teva, Bial, UCB; (5) Fees for promotional materials: No; (6) Research funding: Roche, Zambon, Ciberned; Instituto de SaludCarlos III; FundacióLa Maratóde TV3; (7) Scholarship from corporation: No; (8) Corporate laboratory funding: No; (9) Others (e.g. trips, travel, or gifts): No.
Blázquez Estrada M. has received honoraria for educational presentations and advice service by AbbVie, Abbott, UCB Pharma, Allergan, Zambon, Bial, and Qualigen.
Seijo M. has received honoraria for educational services from KRKA, UCB, Zambon, Bial; travel grants from Daiichi and Roche.
Ruiz Martínez J. has received honoraria for educational presentations, attending medical conferences, and advice service by AbbVie, UCB Pharma, Zambon, Italfarmaco, Bial, and Teva.
Valero C. has received honoraria for educational services from Zambon, AbbVie and UCB.
Kurtis M. has received honoraria from Bial, the Spanish Neurology Society, and the International and Movement Disorders Society.
de Fábregues O. has received honoraria for educational presentations and advice service by Bial, Zambon, AbbVie, KRKA, and Teva.
González Ardura J. has recieved honoraria for speking from italofarma, Krka, Genzyme, UCB, Esteve, Psyma iberica marketing research SL and Ferrer, course grant from Teva and travel grant from Merck.
López Díaz L. has received honoraria from UCB, Lundbeck, and KRKA.
Martínez-Martin P. has received honoraria from National School of Public Health (ISCIII), Editori-al Viguera and Takeda Pharmaceuticals for lecturing in courses, and from the International Parkinson and Movement Disorder Society (MDS) for management of the Program on Rating Scales.
Mir P. has received honoraria from AbbVie, Abbott, Allergan, Bial, Merz, UCB, and Zambon and have received grants from the Spanish Ministry of Economy and Competitiveness [PI16/01575] co-founded by ISCIII (Subdirección General de Evaluación y Fomento de la Investigación) and by Fondo Europeo de Desarrollo Regional (FEDER), the Consejería de Economía, Innovación, Ciencia y Empleo de la Junta de Andalucía [CVI-02526, CTS-7685], the Consejería de Salud y Bienestar Social de la Junta de Andalucía [PI-0437-2012, PI-0471-2013], the Sociedad Andaluza de Neurología, the Jacques and Gloria Gossweiler Foundation, the Fundación Alicia Koplowitz, the Fundación Mutua Madrileña.
No other authors have anything to disclose.
COPPADIS STUDY GROUP
Adarmes AD, Almeria M, Alonso Losada MG, Alonso Cánovas A, Alonso Frech F, Alonso Redondo R, Álvarez I, Álvarez Sauco M, Aneiros Díaz A, Arnáiz S, Arribas S, Ascunce Vidondo A, Aguilar M, Ávila MA, Bernardo Lambrich N, Bejr-Kasem H, Blázquez Estrada M, Botí M, Borrue C, Buongiorno MT, Cabello González C, Cabo López I, Caballol N, Cámara Lorenzo A, Canfield Medina H, Carrillo F, Carrillo Padilla FJ, Casas E, Catalán MJ, Clavero P, Cortina Fernández A, Cosgaya M, Cots Foraster A, Crespo Cuevas A, Cubo E, de Deus Fonticoba T, de Fábregues-Boixar O, Díez-Fairen M, Dotor García-Soto J, Erro E, Escalante S, Estelrich Peyret E, Fernández Guillán N, Gámez P, Gallego M, García Caldentey J, García Campos C, García Moreno JM, Gastón I, Gómez Garre MP, Gómez Mayordomo V, González Aloy J, González-Aramburu I, González Ardura J, González García B, González Palmás MJ, González Toledo GR, Golpe Díaz A, Grau Solá M, Guardia G, Hernández Vara J, Horta-Barba A, Idoate Calderón D, Infante J, Jesús S, Kulisevsky J, Kurtis M, Labandeira C, Labrador MA, Lacruz F, Lage Castro M, Lastres Gómez S, Legarda I, López Ariztegui N, López Díaz LM, López Manzanares L, López Seoane B, Lucas del Pozo S, Macías Y, Madrigal Lkhou E, Mata M, Martí Andres G, Martí MJ, Martínez Castrillo JC, Martinez-Martin P, McAfee D, Meitín MT, Menéndez González M, Méndez del Barrio C, Mir P, Miranda Santiago J, Morales Casado MI, Moreno Diéguez A, Nogueira V, Novo Amado A, Novo Ponte S, Ordás C, Pagonabarraga J, Pareés I, Pascual-Sedano B, Pastor P, Pérez Fuertes A, Pérez Noguera R, Planas-Ballvé A, Planellas L, Prats MA, Prieto Jurczynska C, Puente V, Pueyo Morlans M, Puig Daví A, Redondo Rafales N, Rodríguez Méndez L, Rodríguez Pérez AB, Roldán F, Ruíz De Arcos M, Ruíz Martínez J, Sánchez Alonso P, Sánchez-Carpintero M, Sánchez Díez G, Sánchez Rodríguez A, Santacruz P, Santos García D, Segundo Rodríguez JC, Seijo M, Sierra Peña M, Solano Vila B, Suárez Castro E, Tartari JP, Valero C, Vargas L, Vela L, Villanueva C, Vives B, Villar MD.
