Abstract
Background:
Parkinson’s disease (PD) is a heterogeneous movement disorder with patients manifesting with either tremor-dominant (TD) or postural instability and gait disturbance (PIGD) motor subtypes. Small nerve fiber damage occurs in patients with PD and may predict motor progression, but it is not known whether it differs between patients with different motor subtypes.
Objective:
The aim of this study was to explore whether there was an association between the extent of corneal nerve loss and different motor subtypes.
Methods:
Patients with PD classified as TD, PIGD, or mixed subtype underwent detailed clinical and neurological evaluation and corneal confocal microscopy (CCM). Corneal nerve fiber density (CNFD), corneal nerve branch density (CNBD), and corneal nerve fiber length (CNFL) were compared between groups, and the association between corneal nerve fiber loss and motor subtypes was investigated.
Results:
Of the 73 patients studied, 29 (40%) had TD, 34 (46%) had PIGD, and 10 (14%) had a mixed subtype. CNFD (no./mm2, 24.09 ± 4.58
Conclusion:
Greater corneal nerve loss occurs in patients with PIGD compared with TD, and patients with a higher CNFD or CNFL were more likely to have the TD subtype. CCM may have clinical utility in differentiating different motor subtypes in PD.
Keywords
Introduction
Parkinson’s disease (PD) is a clinically heterogeneous disease with patients manifesting a diverse spectrum of motor and non-motor symptoms and signs. 1 Motor symptoms such as resting tremor, bradykinesia, and postural instability are characteristic features of PD. 2 Patients can be divided into different clinical motor subtypes with features that are tremor-dominant (TD) or they may predominantly have postural instability and gait disturbance (PIGD). 3 Patients with the TD subtype respond relatively satisfactorily to levodopa administration, are less likely to develop dyskinesia, 4 and have a relatively better prognosis than those with PIGD.5,6 While patients with the PIGD subtype are less responsive to levodopa therapy, progress more rapidly,5,6 and have greater cognitive dysfunction 7 with a lower quality of life.
PD is a complex multi-system neurodegenerative disease, with not only impairment of the central dopaminergic nigrostriatal system but also peripheral nerve degeneration.8,9 Both large-medium (Aα/β) fiber and small (Aδ and C) fiber damage have been demonstrated in PD.10,11 Large fiber dysfunction has been attributed to levodopa therapy, while small fiber neuropathy is thought to be intrinsic to the neurodegenerative process in PD.11–13
Corneal confocal microscopy (CCM) is an
In this study, CCM was performed in PD patients to explore whether there was an association between the extent of corneal nerve loss and different motor subtypes. We speculated that the PIGD subtype might show greater corneal nerve fiber loss compared with the TD subtype.
Materials and methods
Participants
Patients with PD were recruited from the Department of Neurology, Henan Provincial People’s Hospital between March 2017 and December 2020. Age, sex, and education level were evaluated in all subjects.
PD patients were diagnosed clinically according to the 2015 Movement Disorder Society clinical diagnostic criteria for Parkinson’s disease. 20 Clinically diagnosed PD and clinically probable PD were recruited. Parkinson-plus syndromes, such as multiple system atrophy, progressive supranuclear palsy, cortical-basal ganglia degeneration, dementia with Lewy bodies, and secondary parkinsonism (vascular, drug-induced, inflammatory, immune-mediated, normal pressure hydrocephalus, infectious, traumatic, etc.), were excluded. Any participants with a suspected family history of a movement disorder were excluded. Healthy controls were recruited from volunteers or spouse of PD patients who had no history of neurodegenerative disease. Subjects younger than 40 or older than 85 years of age were excluded from the study. Subjects with a history of corneal disease, eye surgery, glaucoma, eye inflammation, or thyroid eye disease were excluded. Other causes of peripheral neuropathy were excluded based on a history of excess alcohol use (>150 ml/d) and an assessment of vitamin B12 and folate, serum electrophoresis to exclude multiple myeloma, and cryoglobulinemia and macroglobulinemia and an oral glucose tolerance test to exclude impaired glucose tolerance and diabetes. The clinical features of each participant were carefully evaluated by an experienced neurologist (H-Q.Y.) who was specialized in movement disorders. All subjects agreed to participate in the study and written informed consent was obtained. The study was approved by the Ethics Committee of Henan Provincial People’s Hospital (No. 202148).
Clinical evaluation
The assessment of motor and non-motor symptoms was performed in the ‘ON’ state. PD symptoms were assessed with part I, II, III and IV sub-scales of the Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS), and Hoehn and Yahr (H-Y) staging was undertaken for all PD patients. 21 Age at onset was defined as the time when the first cardinal motor sign(s) were noted by either the patient or family members. Disease duration was defined as the time interval between presentation with first motor symptoms and enrollment into the present study. The Scale for Outcomes in Parkinson’s Disease for Autonomic Symptoms (SCOPA-AUT), a reliable and validated questionnaire, was undertaken in patients with PD to detect autonomic dysfunction. Montreal cognitive assessment (MoCA, Beijing Version) was used to assess the cognitive status. Quality of life was evaluated with the 39-item Parkinson’s disease questionnaire (PDQ-39) with a higher score indicating a poorer quality of life. Levodopa equivalent daily dose (LEDD) was assessed according to the levodopa conversion formula. 22 Briefly, 100 mg levodopa = 133 mg entacapone = 1 mg pramipexole = 5 mg ropinirole = 10 mg selegiline = 1 mg rasagiline = 100 mg amantadine.
Motor subtypes
Motor subtype classification in PD patients was performed according to Stebbins
CCM
A Heidelberg Retina Tomograph III with a Rostock Cornea Module (HRT III RCM; Heidelberg Engineering GmbH, Heidelberg, Germany) was used to acquire images of the central corneal sub-basal nerve plexus. To minimize the impact of motor symptoms especially tremor on the results, CCM was completed in PD patients when they were in the ‘ON’ state. Topical lidocaine was used to anesthetize the eye of each subject and they were seated comfortably and instructed to fixate on an outer fixation light. The TomoCap was correctly positioned on the cornea by visualizing with the CCD camera. An experienced examiner took images at the level of the sub-basal nerve plexus in the central cornea using the ‘section’ mode according to an established protocol. 24 Four to six optimal quality CCM images from the central cornea of each eye were selected and analyzed using a validated, manual purpose-written software (CCMetrics, Imaging Science and Biomedical Engineering, Manchester, UK). 25 Three parameters were analyzed: (a) corneal nerve fiber density (CNFD) – the number of main nerve fibers per square millimeter; (b) corneal nerve branch density (CNBD) – the number of primary branches originating from the main nerve; and (c) corneal nerve fiber length (CNFL) – the sum of length of all nerve fibers and branches per square millimeter. 26
Statistical analysis
The Shapiro–Wilk test was used to assess the normality of data. For normal variables, numbers are expressed as mean ± standard deviation (SD). Analysis of variance with Bonferroni’s
Logistic regression was used to examine the relationship between CCM parameters and motor subtypes, adjusting for H-Y stage, SCOPA-AUT, and UPDRS-II. Variables for inclusion were carefully chosen, given the number of events available and univariate relationship. The receiver operating characteristic (ROC) curve was used to analyze the capability of corneal nerve parameters for distinguishing the PIGD subtype from the TD subtype. All analyses were carried out using SPSS version 22.0 (IBM Corporation, Armonk, NY, USA). Scatter plots and ROC curve were generated using GraphPad Prism version 8.0 (GraphPad Software, Inc, San Diego, CA, USA). The value of
Results
Clinical and demographic profiles
In this study, 87 PD patients were enrolled and 73 were finally included. Eight patients with impaired glucose tolerance, four with corneal disease, and two with multiple system atrophy were excluded. The age of the PD group (52.05% male) was 62.67 ± 7.67 years with age at onset of 59.15 ± 8.02 years and disease duration of 3 (2–4) years. The average H-Y stage of the PD group was 2 (1–3). In the control group (
Of the 73 patients with PD, 29 (40%) were classified with TD, 34 (46%) with PIGD, and 10 (14%) had a mixed subtype. There were no significant differences in age, sex, age at onset, disease duration, LEDD, UPDRS-I, UPDRS-III, and MoCA scores between the different motor subtypes. As shown in Table 1, H-Y stage [3 (2–4)
Demographic characteristics of participants.

CNBD, corneal nerve branch density; CNFD, corneal nerve fiber density; CNFL, corneal nerve fiber length; LEDD, levodopa equivalent daily dose; MoCA, Montreal cognitive assessment; NA, not available; PDQ-39, the 39-item Parkinson’s disease questionnaire; PIGD, postural instability and gait disturbance; SCOPA-AUT, the scale for outcomes in PD for autonomic symptoms; TD, tremor-dominant; UPDRS, Unified Parkinson’s Disease Rating Scale.
Data are expressed as mean ± SD or median (interquartile range). For normally distributed variables, analysis of variance (ANOVA) with Bonferroni’s
Differences between TD and PIGD.
Differences between TD and mixed.
CCM parameters in different motor subtypes with PD
CNFD [no./mm2, 24.09 ± 4.58
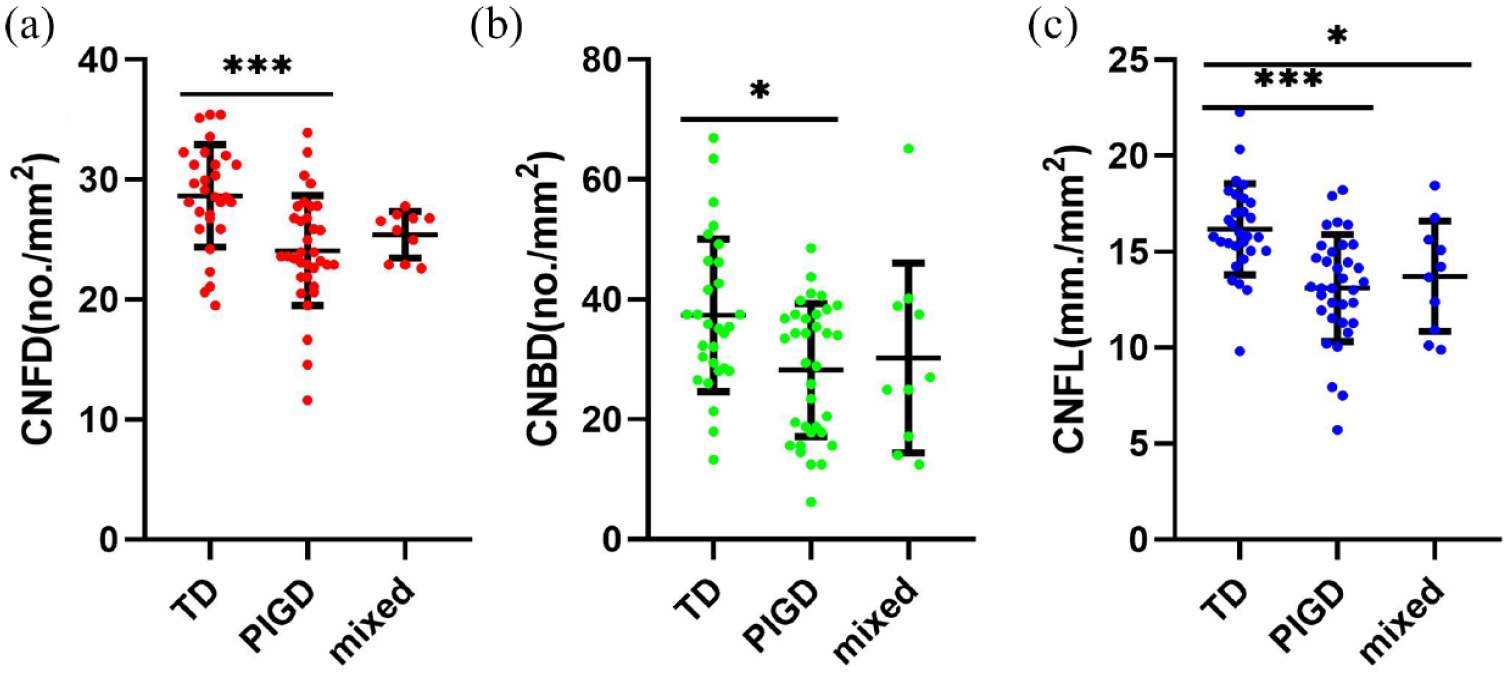
CCM measurements in PD patients with different motor subtypes. Scatter plots of (a) CNFD, (b) CNBD, and (c) CNFL in TD, PIGD, and mixed groups.
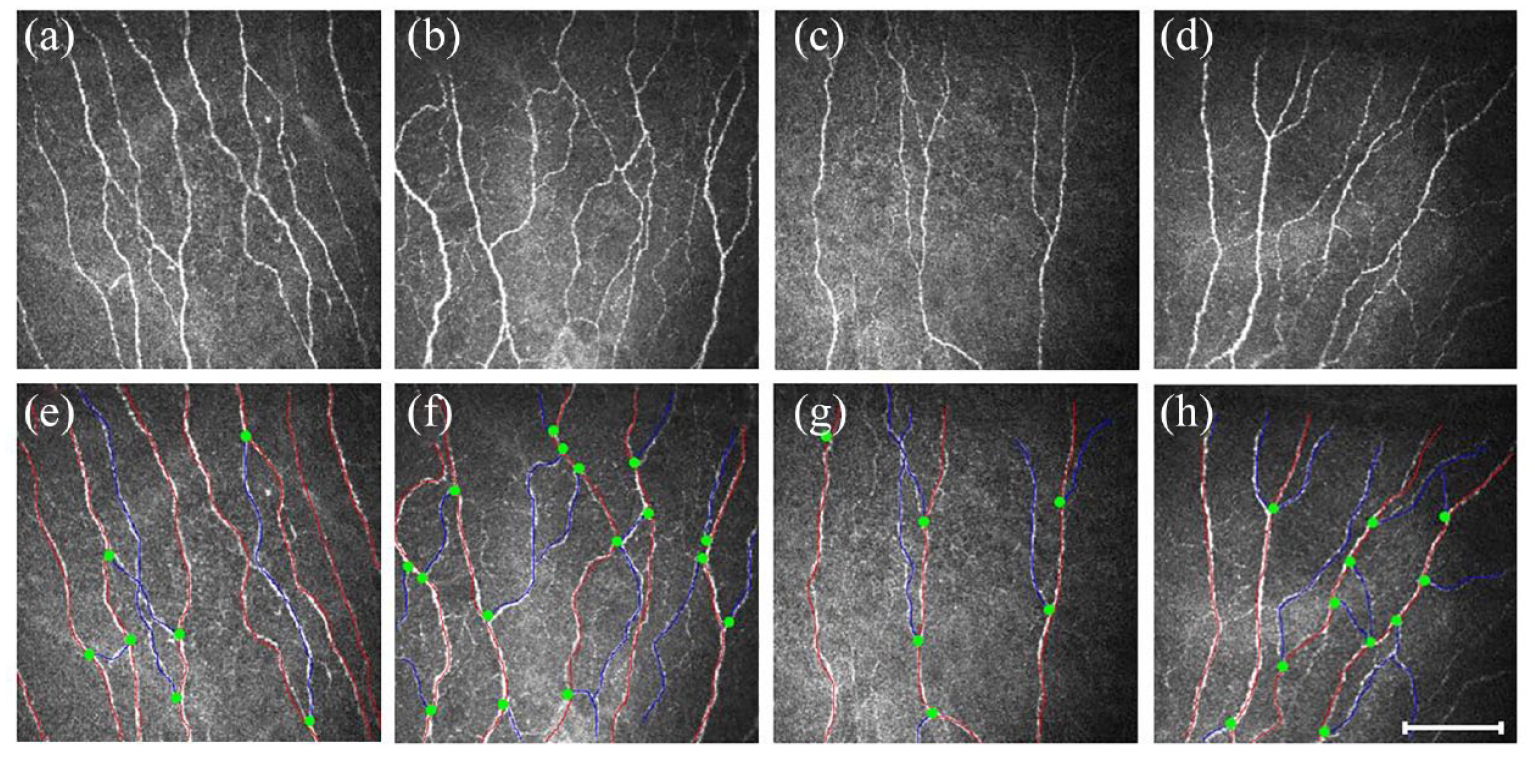
Representative CCM images in (a, e) a healthy control and patient with (b, f) TD, (c, g) PIGD, and (d, h) mixed disease. (a–d) Corneal nerve fibers are linear homogeneous and highly reflective. (e–h) Nerve fiber trunks are highlighted in red, green dots indicate the origin of the branches, and blue and red lines combined indicate the CNFL. Scale bar = 100 µm.
Logistic regression analysis
The logistic regression analyses between corneal nerve parameters and motor subtypes are summarized in Table 2. Univariate logistic regression analysis with motor subtypes as the dependent variable showed a significant association with CNFD [odds ratio (OR) = 1.278,
Multivariate multinomial logistic regression analysis for the independent association between CNFD and CNFL and motor subtypes with PIGD subtype as the reference.

CI, confidence interval; CNFD, corneal nerve fiber density; CNFL, corneal nerve fiber length; OR, odds ratio; PIGD, postural instability and gait disturbance; SCOPA-AUT, the scale for outcomes in PD for autonomic symptoms; TD, tremor-dominant; UPDRS, Unified Parkinson’s Disease Rating Scale.
Results from univariable logistic regression.
Results from multivariable logistic regression adjusted for H-Y stage, SCOPA-AUT, and UPDRS-II.
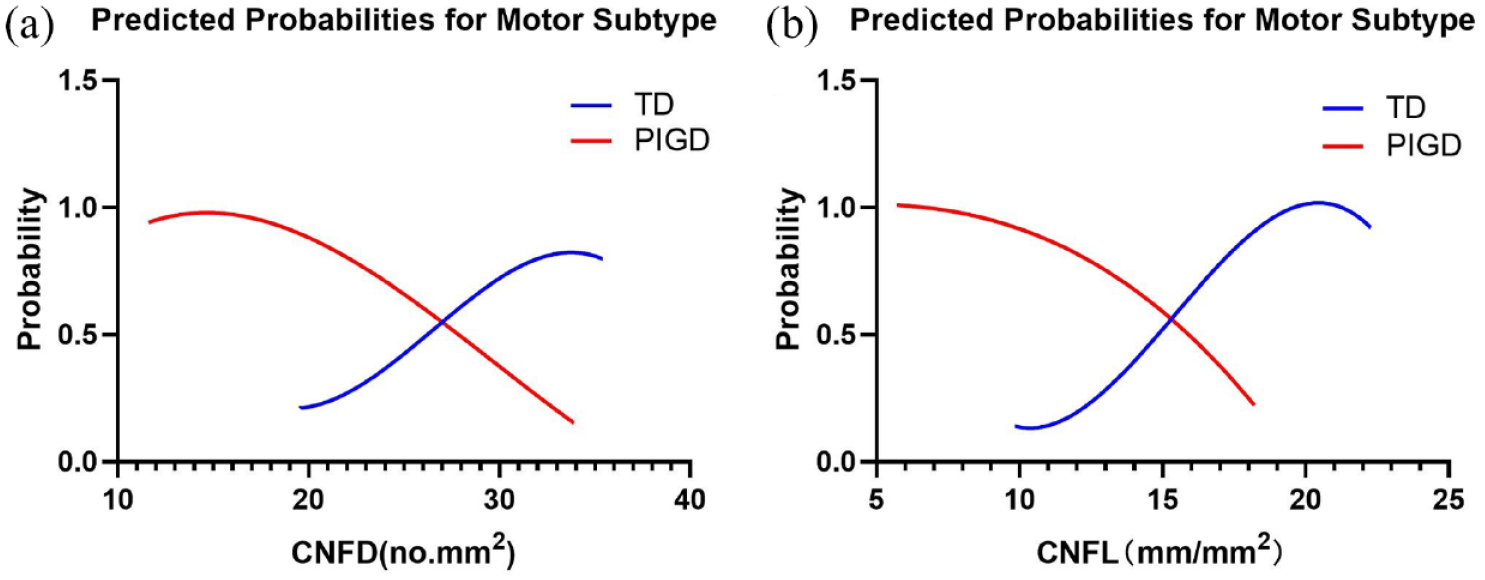
Relationship between (a) CNFD and (b) CNFL and the probability of motor subtypes after correction for confounders.
Diagnostic utility of CCM
ROC analysis showed that CNFD, CNBD, and CNFL could distinguish between TD and PIGD subtypes with an area under the curve (AUC) of 77.69% (95% CI, 65.65–89.73%), 67.70% (95% CI, 54.47–80.93%), and 81.74% (95% CI, 71.08–92.41%), respectively. Using a CNFD cutoff of <27.98 no./mm2, the sensitivity and specificity for TD was 85.29% and 65.52%, respectively. Using a CNBD cutoff of <25.96 no./mm2, the sensitivity and specificity for TD were 44.12% and 89.66%, respectively. Using a CNFL cutoff of <15.02 mm/mm2, the sensitivity and specificity for TD were 76.47% and 79.31%, respectively. A combination of all three corneal nerve parameters increased the AUC to 83.16% (95% CI, 72.60–93.73%), with a sensitivity and specificity of 85.29% and 79.31%, respectively (Figure 4).
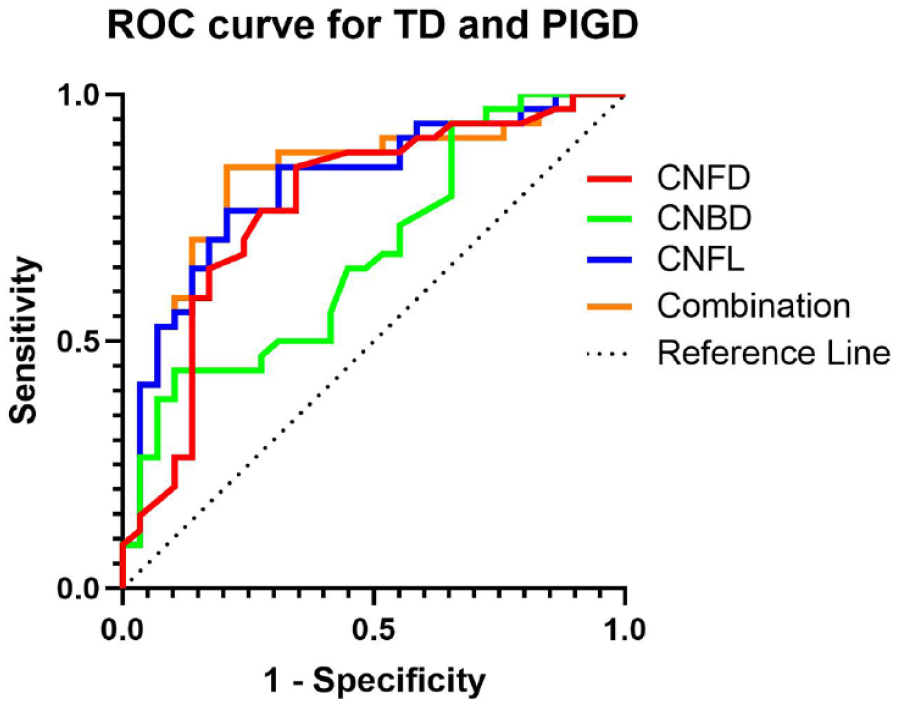
The ROC for CCM distinguishing TD from PIGD.
Discussion
PD is a clinically heterogeneous disease, and the motor phenotype may be a good predictor of disease progression. Not only do the PIGD subtype have an increased risk of falls and gait freezing27,28 but they also have a poorer prognosis with more rapid disease progression, compared with the TD subtype. 5 At present, subtype diagnosis is based primarily on clinical evaluation and reliable biomarkers to differentiate subtypes are lacking.
In the early,
The presence of peripheral neuropathy is associated with a tripling in the frequency of falls in PD patients. 33 Peripheral neuropathy has also been shown to be an independent marker of a severe PD phenotype, with worse cognitive function, and motor and autonomic symptoms. 34 We previously reported a lower CNFD and higher CNBD indicative of concomitant small fiber degeneration and regeneration in patients with mild-moderate PD.17,35 Furthermore, the severity of corneal nerve loss correlated with the severity of cognitive, autonomic, and motor dysfunction.17,18 In this study, we show that patients with the PIGD subtype have greater corneal nerve loss evidenced by a lower CNFD, CNBD, and CNFL compared with the TD motor subtype. Furthermore, patients with the PIGD subtype had more severe disease severity (higher H-Y stage), autonomic dysfunction (higher SCOPA-AUT score), impairment in activity of daily living (increased UPDRS-II score), and a decreased quality of life (increased PDQ-39 score) compared with the TD subtype, consistent with other studies.5,27,36 We have also shown that combined corneal nerve parameters have an excellent diagnostic utility for differentiating the TD from the PIGD subtypes, with an AUC of 0.832, far superior to plasma neurofilament light chain, with an AUC of only 0.656. 31 Furthermore, logistic regression analyses showed that CNFD and CNFL were associated with the TD and PIGD motor subtypes after adjusting for confounders. We speculate that small fiber neuropathy combined with progressive dopaminergic dysfunction may contribute to faster progression and a poorer prognosis in the PIGD subtype of PD.
The present findings add to the growing body of data showing corneal nerve fiber loss in a range of neurodegenerative diseases, including dementia,15,37 amyotrophic lateral sclerosis, 38 Friedreich’s ataxia, 39 multiple sclerosis,40,41 and PD.16–19 Indeed, progressive corneal nerve fiber loss is evident in subjects with mild cognitive impairment (MCI) and dementia, 15 and is associated with cognitive dysfunction and functional independence. 37 Corneal nerve fiber loss has also been reported in patients with amyotrophic lateral sclerosis and related to disease severity. 38 In patients with Friedreich’s ataxia, corneal nerve fiber loss correlates with increasing clinical severity scores. 39 Corneal nerve loss also occurs in patients with multiple sclerosis, 40 and progressive loss is associated with progression of neurological disability. 41
We acknowledge limitations in our study including a single-center and cross-sectional design and the lack of inclusion of an akinetic rigid (AR) subgroup of patients with PD. We also acknowledge that patients may shift between motor subtypes with disease progression,42,43 which requires further analysis in a longitudinal study. The sample size of this study was relatively small, and future larger studies should include more patients with the mixed subtype and with the AR subtype. The PD patients were all clinically diagnosed, without a pathological diagnosis, and therefore we cannot exclude the possibility that some patients may eventually develop atypical parkinsonism. There was overlap of CCM parameters between groups, and therefore, a single CCM examination is not sufficient for clinical differentiation.
In conclusion, PD patients with the PIGD subtype have evidence of greater corneal nerve fiber loss compared to the TD subtype. CCM had excellent diagnostic utility with a high sensitivity and specificity to distinguish the PIGD from the TD subtype. CCM may represent a rapid convenient ophthalmic imaging technique to identify motor subtypes in PD. Our findings warrant larger, longitudinal studies to establish the utility of CCM in the monitoring and prognostication of PD subtypes.
Supplemental Material
sj-docx-1-tan-10.1177_17562864231165561 – Supplemental material for The severity of corneal nerve loss differentiates motor subtypes in patients with Parkinson’s disease
Supplemental material, sj-docx-1-tan-10.1177_17562864231165561 for The severity of corneal nerve loss differentiates motor subtypes in patients with Parkinson’s disease by Ning-Ning Che, Qiu-Huan Jiang, Shuai Chen, Si-Yuan Chen, Zhen-Xiang Zhao, Xue Li, Jian-Jun Ma, Jie-Wen Zhang, Rayaz A. Malik and Hong-Qi Yang in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors thank the patients and healthy individuals for their participation in this study. They also thank the Manchester Biomedical Research Centre for access to the CCMetrics and ACCMetrics software.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
