Abstract
Background:
Ankylosing spondylitis (AS) is an immune-mediated, chronic inflammatory rheumatic disorder. The etiology of Parkinson’s disease (PD) is multifactorial; however, inflammation is receiving an increasing amount of attention as an underlying cause of the neurodegenerative process of PD.
Objective:
We performed a nationwide longitudinal, population-based matched cohort study to assess the association with the later development of parkinsonism in Korea.
Methods:
This study was conducted using records from the Health Insurance Review and Assessment Service database. The cumulative incidence rate of PD was estimated. Fine–Gray subdistribution hazard models were used to identify hazards associated with PD development based on the presence of AS. Exposure to anti-inflammatory drugs was measured and analyzed to determine the protective effect of these medications. Additionally, the hazard ratio (HR) for atypical parkinsonism was estimated.
Results:
The results of the Fine–Gray subdistribution hazard model revealed that the HR for PD development in the AS group was 1.82 (95%confidence interval [CI], 1.38–2.39, p < 0.001). A significant decrease in PD development was observed in patients with AS taking non-steroidal anti-inflammatory drugs (NSAIDs). The HR for atypical parkinsonism in the AS group was 3.86 (95%CI, 1.08–13.78, p < 0.05).
Conclusion:
We found that AS was associated with an increased risk of PD and atypical parkinsonism. NSAIDs used for AS control have some protective effects against PD. Further studies assessing whether biological treatment mitigates PD risk in patients with high activity are warranted.
INTRODUCTION
Ankylosing spondylitis (AS) is an immune-medi-ated, chronic inflammatory rheumatic disorder cau-sed by enthesopathy and arthritis of the axial skeleton that causes severe chronic pain [1]. No cure is available for AS, and typically, the onset occurs early in socially active young adults; thus, patients with AS experience decreased quality of life with long-lasting emotional and functional impairments [2, 3]. The development of AS is mediated by multiple cell types and pathogenic pathways. It is strongly associated with increased production of tumor necrosis factor-α (TNF-α) and interleukins [4, 5]. Recent research has highlighted the importance of the gut–immune axis, the local immunometabolic environment, in the disease pathogenesis of AS [6]. A recent study reported an increased risk of ischemic heart disease, depression, dementia, and stroke in patients with AS [7–9].
AS is characterized by chronic pro-inflammatory immune activity, a trait that is now suggested to be the fundamental element of neurodegenerative disorders. The aforementioned gut–immune axis is a recently focused concept of pathogenesis of AS, and intestinal inflammation may be of particular relevance in the pathogenesis of parkinsonism, including Parkinson’s disease (PD) and multiple system atrophy (MSA) [10]. Although the etiology of parkinsonism is multifactorial, inflammation is increasingly receiving attention as the underlying cause of the neurodegenerative process of this condition. Dysfunction of the immune system and immune-metabolic environment may be involved in the pathogenesis of parkinsonism [11]. The relationship between PD and autoimmune diseases that chronically expose individuals to high concentrations of inflammatory cytokines has been studied [12, 13]. In this study, we hypothesized that the patients with AS are at a high risk of developing PD. We investigated the risk of PD in patients with AS and the possible protective effect of medications used for AS. Recently, Yeh et al. reported that patients with AS had an increased risk of developing PD in Taiwan [14]. However, this study did not consider the association with atypical parkinsonism, such as MSA, progressive supranuclear palsy (PSP), and corticobasal degeneration (CBD). Therefore, we performed a nationwide, longitudinal population-based matched cohort study to assess the association with later development of parkinsonism, including PD, MSA, PSP, and CBD in patients with AS in Korea.
METHODS
Data source
We analyzed a nationwide database, the Health Insurance Review and Assessment Service (HIRA) database, from 2009 to 2019. The Korean National Health Insurance (NHI) system is a nationwide insurance system operated by the Korean government and covers up to 100%of the population, including 97%of health insurance and 3%of medical aid. All healthcare providers submit claims data, including diagnostic codes (the International Classification of Disease, Tenth Revision codes [ICD-10 codes]), procedure codes, and demographic information, to the HIRA to request reimbursement for medical costs from the NHI service. Thus, the medical records of almost all patients at healthcare institutions are prospectively recorded in the HIRA database.
Study population
Since 2004, the Korean government has operated a registration program for patients with rare intractable diseases such as AS. We identified all patients diagnosed with AS between January 2009 and December 2014 from the HIRA database. Patients aged ≥20 years with AS were defined as patients with primary or secondary diagnosis of AS (ICD-10 code: M45) and registration code for AS (V140) in the program for rare intractable diseases. We excluded patients who were diagnosed with AS in 2009 to ensure that the AS group included only patients with new episodes. A matched cohort of patients without AS was randomly selected by matching patients by sex, age, year of diagnosis, and insurance type to the AS group at a ratio of 1 : 4. The primary outcome of this study was a new diagnosis of PD; thus, patients with PD diagnosis at baseline were excluded from both the groups.
Other variables
The primary outcome variable was PD development. We selected patients with a primary or secondary diagnosis of PD (ICD-10 code: G20) for more than two times until December 31, 2019. In addition, demographic characteristics such as age and sex were analyzed. Patients were categorized based on age at AS diagnosis: 20–34 years, 35–49 years, 50–64 years, and ≥65 years. Comorbidity was defined based on the Charlson comorbidity index (CCI), which has been used to represent various comorbidities in previous epidemiological studies. Diseases included in the CCI are congestive heart failure, myocardial infarction, cerebrovascular disease, peripheral vascular disease, connective tissue disease, chronic lung disease, ulcer, chronic liver disease, severe liver disease, dementia, diabetes, hemiplegia, moderate or severe kidney disease, tumor, leukemia, lymphoma, moderate or metastatic solid tumor, and acquired immunodeficiency syndrome. We categorized CCI scores into the following four ranges: 0, 1–2, 3–4, and ≥5. In addition to the CCI score, data on hypertension, defined by ICD-10 codes, were extracted. The main pharmaceutical substance code was used to determine the drug use for AS. The main substances for each drug were as follows: celecoxib, meloxicam, aceclofenac, naproxen, ibuprofen, dexibuprofen, ketoprofen, diclofenac, etodolac, and piroxicam were used as non-steroidal anti-inflammatory drugs (NSAIDs); sulfasalazine and methotrexate were used as immunosuppressant therapy; etanercept, infliximab, adalimumab, golimumab, and secukinumab were used as TNF-α inhibitors. Health care use was defined as the total number of inpatient or outpatient visits within 1 year before AS diagnosis or inclusion date.
Statistical analyses
The baseline characteristics of both the groups are presented as numbers with percentages for categorical variables and means with standard deviations for continuous variables. A generalized estimating equation was used to compare the distributions of baseline demographic characteristics, selected comorbidities, and health care visits between the two groups. Given the observational nature of the study, exact matching was used to adjust for the potentially confounding effects of imbalances in baseline prognostic factors. Variables considered for the exact matching were age, sex, insurance type, and year of enrollment. The exact matching was performed using the authors’ own R code for each year from 2010 to 2014. The incidence of PD was calculated as the number of PD patients identified during follow-up divided by the total person-years of follow-up for each group. Death was a competing event for PD. Hence, we performed a competing risk analysis. The cumulative incidence rate of PD was estimated, and a Gray’s test was used to determine the differences in PD incidence. To identify hazards associated with PD development based on the presence of AS during the 10-year follow-up period (January 1, 2010 to December 31, 2019), the Fine–Gray subdistribution hazard model was used and the hazard ratios (HRs) and 95%confidence intervals (CIs) were calculated. This model estimates subdistribution hazard based on cumulative incidence functions. Thus, the Fine–Gray subdistribution hazard model can be applied to evaluate a direct relationship between covariates and the cumulative incidence. [15] A two-sided p-value of < 0.05 was considered statistically significant. Statistical analyses were performed using the statistical software SAS System for Windows version 9.4 (SAS Institute Inc, Cary, NC) and R version 3.4.4 (R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
Characteristics of patients
This study enrolled 91,050 patients (AS group: 18,210 patients and comparison group: 72,840). The sociodemographic and medical characteristics of both the groups after exact matching are presented in Supplementary Table 1. No significant differences were noted between the two groups for age, sex, insurance type, and year of enrollment. Patients with AS had more comorbidities than did those without AS. As for medications, the most frequently prescribed medication in the AS group was NSAIDs (88.4%), followed by immunosuppressants (64.6%) and TNF-α inhibitors (25.7%). The mean number of health care visits in the AS group was 23.1±25.0 and that in the comparison group was 16.0±23.0 (p < 0.0001).
Risk of PD development in patients with ankylosing spondylitis
Table 1 displays the incidence rates and HRs for PD development. During the 10-year follow-up period, the incidence rates of PD in the AS and comparison groups were 0.67 and 0.43 per 1000 person-years, respectively. The results of the Fine–Gray subdistribution hazard model revealed that the HR for PD development in the AS group was 1.82 (95%CI, 1.38–2.39, p < 0.001) The relationship between AS and PD development was assessed according to sex and found to be significant in both sexes. The results of the Fine–Gray subdistribution hazard model revealed that the HRs for PD development in men and women were 1.86 (95%CI, 1.34–2.59, p < 0.001) and 1.70 (95%CI, 1.03–2.80, p < 0.05), respectively. The HRs for PD development according to age group are presented in Supplementary Table 2. In the AS group, the rate of PD development was high in patients aged ≥50 years (50–64 years, HR = 2.35, 95%CI, 1.41–3.91, p < 0.05; ≥65 years, HR = 1.50, 95%CI 1.03–2.16, p < 0.05).
Calculated hazard ratios of PD in the AS and control groups
AS, Ankylosing spondylitis; PD, Parkinson’s disease. Incidence rate is the incidence of PD per 1000 person-year. Adjusted for comorbidities, number of hospital visit, and medications. *p < 0.05, **p < 0.001.
The cumulative incidence curves and the Gray’s test for PD development stratified by sex and age showed that the rate of PD development was higher in the AS group than in the control group (Supplementary Figures 1 and 2).
Supplementary Table 3 presents the HR for PD development according to follow-up duration after AS diagnosis. During the 10-year follow-up period, the risk of PD development was consistently higher in the AS group compared to the comparison group. The HRs for the development of PD in the AS group at 1 and 10 years of follow-up were 3.51 (95%CI, 1.83–6.76, p < 0.001) and 1.82 (95%CI, 1.38–2.39, p < 0.001), respectively.
Risk of PD development after AS diagnosis according to medication
Figure 1 presents the HR for PD development according to medication use in patients with AS. No significant relationship was found between PD development and use of medications, including immunosuppressants and TNF-α inhibitors, in patients with AS (p > 0.05). On the contrary, a significant decrease in PD development was observed in patients with AS receiving NSAIDs compared with those who did not take NSAIDs (HR = 0.51, 95%CI, 0.39–0.67, p < 0.001). An inverse dose-response relationship was found between the duration of NSAID use and PD development (Fig. 2, Supplementary Table 4).
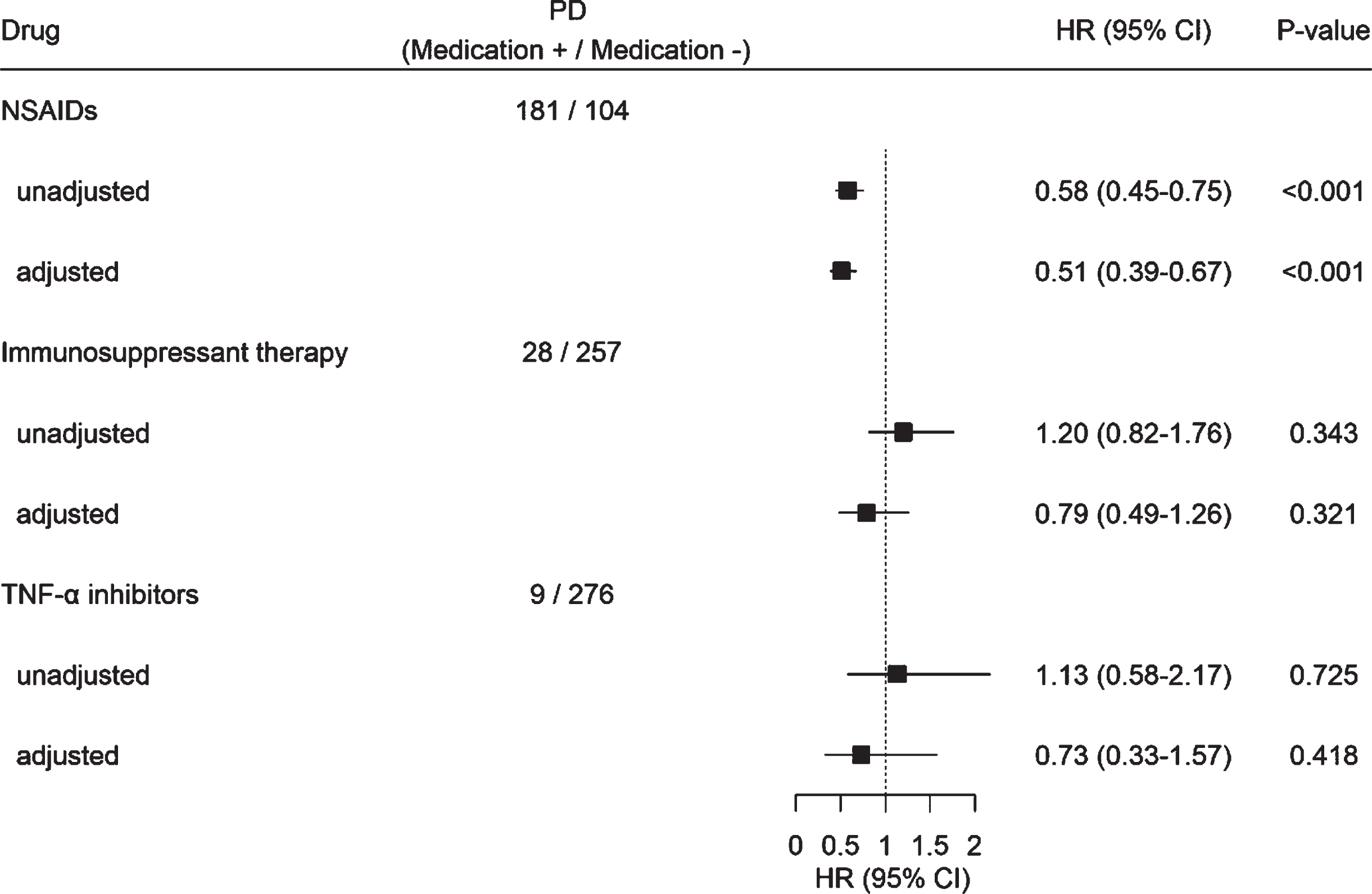
Hazard ratio for PD development according to medication use in patients with ankylosing spondylitis further adjusting for comorbidities and the number of hospital visit.
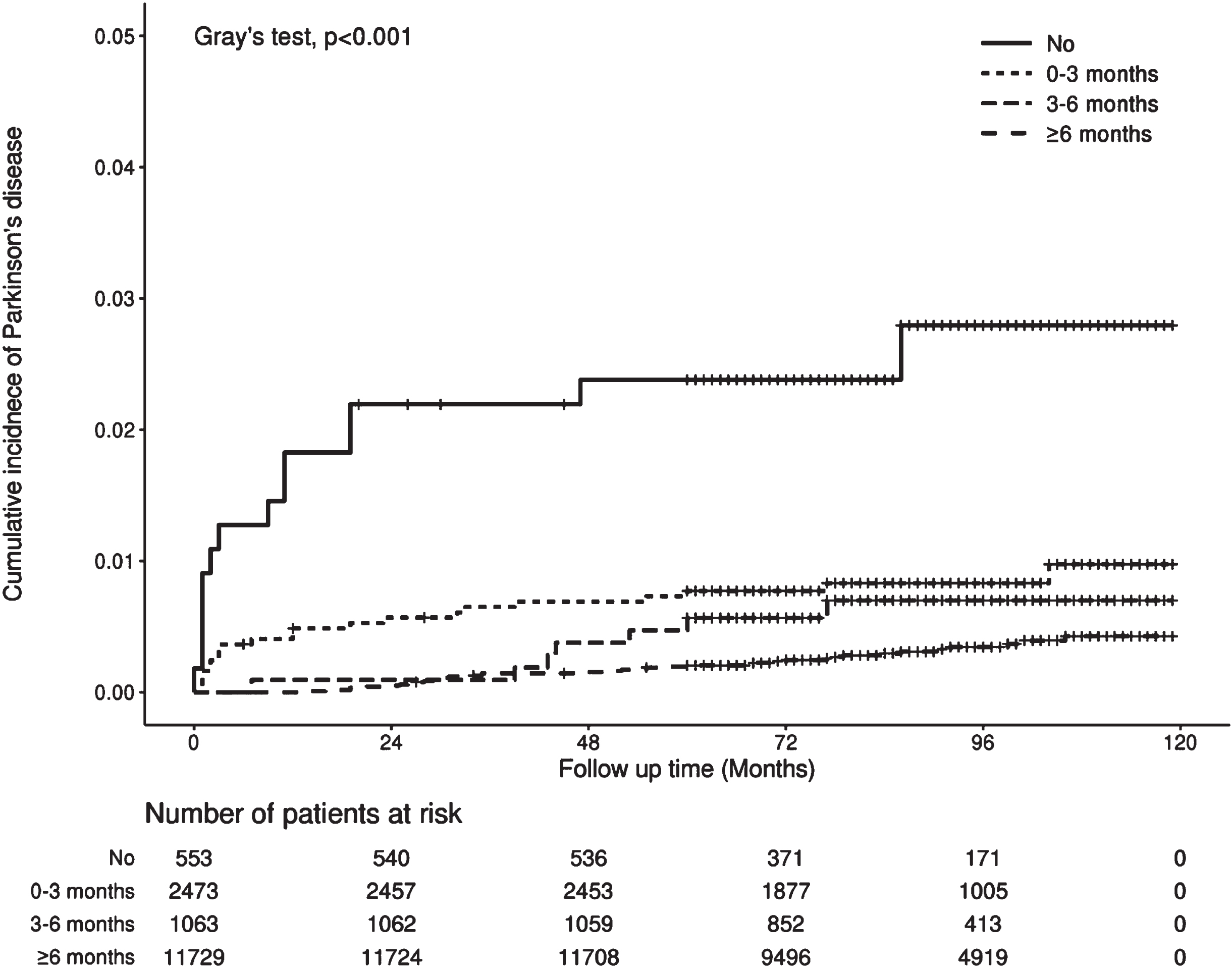
Comparison of cumulative incidence of Parkinson disease in patients with ankylosing spondylitis according to the cumulative duration of NSAIDs use (Gray’s test, p < 0.001). The cross symbol in the graph indicates censored data, which means event (death) other than PD occurred at that time.
Risk of atypical parkinsonism development after ankylosing spondylitis diagnosis
The results of the Fine–Gray subdistribution hazard model showed an HR of 3.86 (95%CI, 1.08–13.78, p < 0.05) for the risk of atypical parkinsonism in the AS group. Subgroup analyses by specific types of atypical parkinsonism revealed no statistically significant results (Table 2).
Calculated hazard ratios of atypical parkinsonism in the AS and control groups
AS, Ankylosing spondylitis; PD, Parkinson’s disease; MSA, Multiple system atrophy; PSP, Progressive supranuclear palsy; CBD, Corticobasal degeneration. Incidence rate is the incidence of PD per 1000 person-year. Adjusted for comorbidities, number of hospital visit, and medications. *p < 0.05, **p < 0.001.
DISCUSSION
In this large, population-based cohort study, we investigated the risk of PD and atypical parkinsonism in patients with AS using data from the Korean HIRA database. This study revealed that the HR for PD in the AS group was higher than that in the control group. Therefore, we suggest that AS is a risk factor for the development of PD. Similarly, Yeh et al. reported that patients with AS had an increased risk of PD in Taiwan [14]. However, our study sample was larger than those of previous studies. Moreover, we analyzed the association with atypical parkinsonism. Compared with previous studies, our study adopted more sophisticated adjustment methods such as Fine–Gray subdistribution hazard models and adjustment for the number of hospital visits. Fine–Gray subdistribution hazard models are increasingly being used for the analysis of time-to-event outcomes in the presence of competing events [16]. After exact matching, patients with AS had significantly more comorbidities than did the matched control patients based on the CCI. Healthcare visits were more frequent in patients with AS than in matched control patients. We adjusted the number of healthcare visits to ameliorate surveillance bias. Naturally, patients diagnosed with AS had more hospital visits than did those without AS diagnosis; therefore, other diseases may be more likely to occur in patients with AS than in patients without AS. After adjusting for comorbidities, number of hospital visits, and medications, AS was associated with an increased risk of PD development. Currently, neuroinflammation is being considered in the debate on the physiopathogenesis of PD [17]. Systemic inflammation, a major component of AS, is thought to contribute to neuronal inflammation and dopaminergic neuron loss in PD through infiltration and accumulation of immune cells from the periphery [18]. In addition, postmortem analyses of human samples and experimental animal studies have indicated that the activation of glial cells increases the levels of proinflammatory factors such as TNF, interleukin (IL)-1β, IL-2, IL-4, and IL-6 (regional inflammation), a common feature of the PD brain. Proinflammatory cytokine profiles in the cerebrospinal fluid and peripheral blood, as well as in the striatum and substantia nigra pars compacta, of patients with PD and animal models of PD have been studied [19]. Similar findings were reported in humans. TNF-α, a proinflammatory cytokine involved in AS, is present in high titers in the blood, cerebrospinal fluid, and striatum of patients with PD [20]. Evidence suggests autoimmune and genetic im-mune dysfunction involvement in PD [21]. A nationwide population-based study showed an increased risk of PD among patients with autoimmune diseases [12]. Recently, genome-wide conjunctional analysis identified 17 novel loci that overlap between PD and autoimmune diseases, suggesting that these PD-associated loci contribute to PD through immune defects and that immune dysfunction is not simply the end product of this neurodegeneration process. These findings strongly support the presence of an interaction between the immune system and neurodegeneration in PD [22].
In fact, PD and atypical parkinsonism are often difficult to distinguish clinically and share a similar spectrum of symptoms. Although their pathophysiology is unclear, it has been suggested that there are both similarities and differences. We assessed the association with later development of atypical parkinsonism in addition to PD. Unfortunately, the number of atypical parkinsonism cases was extremely small to accurately analyze the HR. However, patients with AS had a tendency of high HR for developing MSA. Atypical parkinsonian syndromes are synucleinopathies and tauopathies, that is, disorders characterized by abnormal deposition of α-synuclein and tau proteins, respectively. The site of deposition is correlated with clinical features [23, 24]. A specific pattern of synuclein aggregates can be identified in the enteric nervous system in patients with PD and is most likely associated with inflammatory processes and intestinal barrier dysfunction [25] which might be associated with the gut–immune axis that was recently implicated in the pathogenesis of AS.
Next, we hypothesized that the anti-inflammatory effect of the drug used for AS could have a protective effect against the development of PD. To address the effect of medication on the risk of PD, the HR of PD according to medication in patients with AS was analyzed. Medications used for AS are usually NSAIDs, immunosuppressants, and biological agents such as TNF-α inhibitors. The results showed that NSAIDs reduced the risk of PD, while the other category of medications did not show a significant protective effect against the development of PD. These data might be explained by the protective effect of long-term NSAID treatment in patients with rheumatoid arthritis [26]. Similarly, a previous meta-analysis of NSAID use and risk of PD suggested that the use of NSAIDs was significantly associated with lowered PD risk [27]. Immunosuppressants and TNF-α inhibitors did not exhibit a significant protective effect against PD development in this study. On the contrary, a previous study reported early exposure to anti-inflammatory and anti-TNF-α therapy was associated with reduced PD incidence in patients with inflammatory bowel disease and suggested that targeting peripheral TNF-α is sufficient to reduce neuroinflammation [28]. This difference in findings is presumed to be the result of the difference in the severity of AS between the cohorts of the studies. Generally, NSAIDs are recommended for the treatment of active AS in adults; however, in addition to NSAIDs, biological agent is used for the treatment of active AS [29]. Therefore, patients using biological agents probably had highly active AS and were likely to experience limited protective effect of the medication. Another suggestion is that the relatively small number of events in the anti-TNF-α therapy exposed cohort compared with that in the cohort using NSAIDs contributed to the wide confidence intervals.
This study had several limitations that should be acknowledged. AS and other comorbidities were defined by diagnostic codes in the claims data from the NHI system without clinical or laboratory data; hence, we cannot exclude misclassification. In addition, the severity of AS measured by clinical symptom scores could not be analyzed because of the innate limitation of the claims data. We could not obtain information on family history of PD and risk factors for PD, such as smoking habits, alcohol consumption, occupation, and lifestyle [30, 31].
And the number of patients taking immunosup-pressant therapy and TNF-α inhibitors are significantly smaller. Therefore, the study of the effect of these drugs could be underpowered. Last, the number of patients with atypical parkinsonism was too small to accurately analyze HR, and dementia with Lewy body was not included in the analysis. Future studies with larger sample sizes with dementia with Lewy body inclusion are needed to elucidate the relationship between AS and atypical parkinsonism.
CONCLUSION
We found that AS was associated with an increased risk of PD and atypical parkinsonism. NSAIDs used for AS control have some protective effects against PD development. However, a prospective study assessing whether different immune modulators are beneficial in reducing PD risk in patients with AS is warranted. Further studies with a larger sample of patients with atypical parkinsonism (such as MSA, PSP, and CBD) should be performed.
Footnotes
ACKNOWLEDGMENTS
This study was supported by the National Re-search Foundation of Korea (NRF) grant funded by the Korean government (MSIT) [grant number 2020R1C1C1006867).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
