Abstract
Background:
Cognitive impairment (CI) is common in Parkinson’s disease (PD) and an important cause of disability. Screening facilitates early detection of CI and has implications for management. Preclinical disability is when patients have functional limitations but maintain independence through compensatory measures.
Objective:
The objective of this study was to investigate the relationship between scores on the Mini-Mental State Examination (MMSE) and Montreal Cognitive Assessment (MoCA) with levels of PD severity and disability.
Methods:
PD patients (n = 2,234) in a large observational study were stratified by disease severity, based on Total Unified Parkinson’s Disease Rating Scale (Total UPDRS) and Hoehn and Yahr (HY) stage. Using MMSE (n = 1,184) or MoCA (n = 1,050) and basic (ADL) and instrumental activities of daily living (IADL) scales for disability, linear regression analysis examined associations between cognitive status and disability.
Results:
Cognition and disability were highly correlated, with the strongest correlation between IADL and MoCA. Only 16.0% of mean MMSE scores were below threshold for CI (28) and only in advanced PD (Total UPDRS 60+, HY≥3). MoCA scores fell below CI threshold (26) in 66.2% of the sample and earlier in disease (Total UPDRS 30+, HY≥2), corresponding with impairments in ADLs.
Conclusion:
In a large clinical dataset, a small fraction of MMSE scores fell below cutoff for CI, reinforcing that MMSE is an insensitive screening tool in PD. MoCA scores indicated CI earlier in disease and coincided with disability. This study shows that MoCA, but not MMSE is sensitive to the emergence of early cognitive impairment in PD and correlates with the concomitant onset of disability.
INTRODUCTION
More than 40% of individuals with Parkinson’s disease (PD) have evidence of mild cognitive impairment (MCI) at onset [1] and the cumulative prevalence of PD dementia (PDD) reaches more than 80% after 20 years of disease [2]. While neuropsychological testing is the gold standard for diagnosing CI in PD, cognitive screening instruments are routinely used in clinical settings to detect cognitive impairment and to measure global cognitive function [3]. The PD quality measures from the American Academy of Neurology recommend screening for cognitive impairment in PD at least annually [4]. Identifying CI informs management, impacts prognosis [5] and is a marker for disability in PD [6, 7]. The presence of CI also has implications for quality of life for both the patient and caregiver and is associated with increased caregiver burden [8–12].
Several tools are available to screen for CI including the Mini-Mental State Examination (MMSE) [13, 14] and the Montreal Cognitive Assessment (MoCA) [15–17]. The MMSE is limited by insensitivity to executive dysfunction [18] and ceiling effect in PD [19, 20]. More than half of persons with PD (PwP) who have normal MMSE scores are found to be impaired by other cognitive assessments [21, 22]. Despite these limitations, the MMSE continues to be used widely, both in clinical practice and as inclusion/exclusion criteria for clinical trials [23, 24]. The primary objective of this study was to investigate the relationship between scores on common cognitive screening tools (MMSE and MoCA) with levels of PD progression, quantified by Total Unified Parkinson Disease Rating Scale (Total UPDRS) and Hoehn and Yahr (HY) stage. The secondary objective was to investigate the correlations between cognitive scores and disability, including basic (ADLs) and instrumental (IADLs) activities of daily living. The study aims to show the inter-relationships between cognitive function, disease severity, and disability in PD.
METHODS
Study population
The University of Maryland PD and Movement Disorders Center has conducted the HOME (Health Outcomes Measurement) Study since 2002, a prospective longitudinal database of consenting PwP, with clinician-reported assessments, patient-reported outcome measures, and annual cognitive assessments collected at routine clinic visits. The University of Maryland, Baltimore Institutional Review Board provided approval for the HOME Study.
Over 80% of PwP visiting the center agreed to participate. Participants were clinically diagnosed with idiopathic PD by a movement disorders specialist using the UK Brain Bank diagnostic criteria [25] and the diagnosis was verified at every office visit. Individual participants were included in this study if they had at least one visit with MMSE or MoCA, Total UPDRS, HY and disability assessments. Every visit for which complete assessments were available were included in the data analysis, thus individual participants may appear more than once.
Measures
Demographic information including age, gender, race, education, and PD duration (time since PD diagnosis) was collected at the time of enrollment. Trained research assistants, medical assistants, or neurologists performed cognitive assessments with the MMSE from 2002–2009, and the MoCA since 2009. Only one cognitive screening test was administered at each visit. Examiners added a point to the total MoCA score for people with less than or equal to 12 years of education [15]. PD progression was assessed with the Total UPDRS and HY staging by the treating neurologist at each visit. Research assistants supervised patient-reported completion of the Older Americans Resource and Services Disability Subscale (OARS) [26], which includes seven items for ADLs and seven items for IADLs. All patients reported their best and worst function on each item.
We based the cognitive screening score cutoffs on the existing literature in PD with the objective of selecting clinically meaningful values for cognitive screening. We selected a cut-off of below 26 for the MoCA [6, 27–29]. This is based on studies showing good discrimination between normal cognition and cognitive impairment at an optimal screening score of < 26, with 90% sensitivity and 75% specificity [28]. While some studies suggest this cutoff may be too sensitive [30, 31], these studies were performed in community-based samples that spanned typical aging and neurodegenerative diseases thus had more varied educational backgrounds and cognitive impairment profiles compared to that of this PD cohort. The selection of the optimal cut-off for MMSE was more challenging, as this is heavily debated in the literature. Some studies suggest that any value below 30 is abnormal [28], however this would be too sensitive and not specific in many populations—in fact the mean score for every category in our cohort would be abnormal by this criteria. Based on this uncertainty, we used two MMSE cut-offs: a high cutoff below 28 and a low cut-off below 26. The higher cut-off (below 28) is the most accurate score identified in PD-specific populations to discriminate between cognitively normal and cognitively impaired PwP [28] and is the suggested conversion for a MoCA score of 26 [32]. The lower cut-off (MMSE below 26) was selected because it is the value most often used in recent and on-going clinical trials in PD [23, 24].
Statistical analysis
Each occasion with a cognitive assessment, disease severity, and disability ratings from a single visit was considered one entry. Using cross-sectional data, PwP were stratified by disease severity based on Total UPDRS scores (< 20, 20–29, 30–39, ... , ≥80) and HY stage. Mean MoCA and MMSE scores were calculated for each Total UPDRS subgroup and each of the HY stages. Post-hoc tests examined differences between Total UPDRS subgroups and HY stages. Analyses of variance (ANOVA) models tested for differences between MMSE or MOCA scores across all Total UPDRS subgroups and HY stages. Linear regression analyses tested the correlation between Total OARS, OARS ADL and IADL subscores, and the measures of cognition.
RESULTS
Demographic and clinical characteristics of participants
The sample comprised 1,184 patients whose first cognitive assessment was the MMSE and 1,050 patients whose first assessment was the MoCA, resulting in a total of 2,686 MMSE records and 2,062 MoCA records, based on more than one record for a subgroup of patients. A total of 361 individuals entered the study with an MMSE as the first cognitive assessment and were included again when MoCA was performed at subsequent visit(s). The majority in both the MMSE and MoCA groups were male, white, and college-educated (Table 1). The mean Total UPDRS was higher in the MMSE group compared to the MoCA group (Table 1). In both groups, the mean time since PD diagnosis (disease duration) was 7 years. Overall, 35.7% of patients reported motor fluctuations, the majority of whom (66.3%) were tested during the “on” state. There were small, but statistically significant baseline differences between the MoCA and MMSE groups for age, education, Total UPDRS, and HY stage.
Patient demographics, disease features, and baseline assessments
∧Demographic data determined at the time of enrollment. *p =0.001 and **p < 0.0001.
Correlation between cognition and disability
Measures of cognition showed a high correlation with disability (Table 2); all values were statistically significant (p < 0.0001). The correlation between cognition and disability was higher for IADLs than ADLs and strongest (r = 0.60) for MoCA and IADLs.
Correlation Between Function (OARS) and Cognition (MMSE and MoCA)
Cognitive screening assessments across the disease spectrum
Mean MMSE scores ranged from 24 to 29 across subgroups on the Total UPDRS (Table 3). None of the disease severity subgroups had a mean MMSE below the cutoff score of 26 for CI, except for the most severe UPDRS 80 + group. Similarly, using the high MMSE cut-off of below 28, mean MMSE scores were impaired only in the advanced UPDRS 60–69, 70–79, and 80 + subgroups, underscoring the ceiling effect of this test (Fig. 1). Mean MoCA scores ranged from 19 to 27 across the Total UPDRS subgroups (Table 3) and mean MoCA scores fell below the cutoff of 26 for CI in all Total UPDRS subgroups of 30–39 and higher (Fig. 1).
Mean MMSE and MoCA Associated with Incremental Total UPDRS Subgroups
Mean cognitive test scores for each Total UPDRS subgroup. Cognitive scores associated with impairment highlighted in light gray. High cutoff MMSE scores highlighted in dark gray. Darkened lines indicate statistically significant between-group differences.
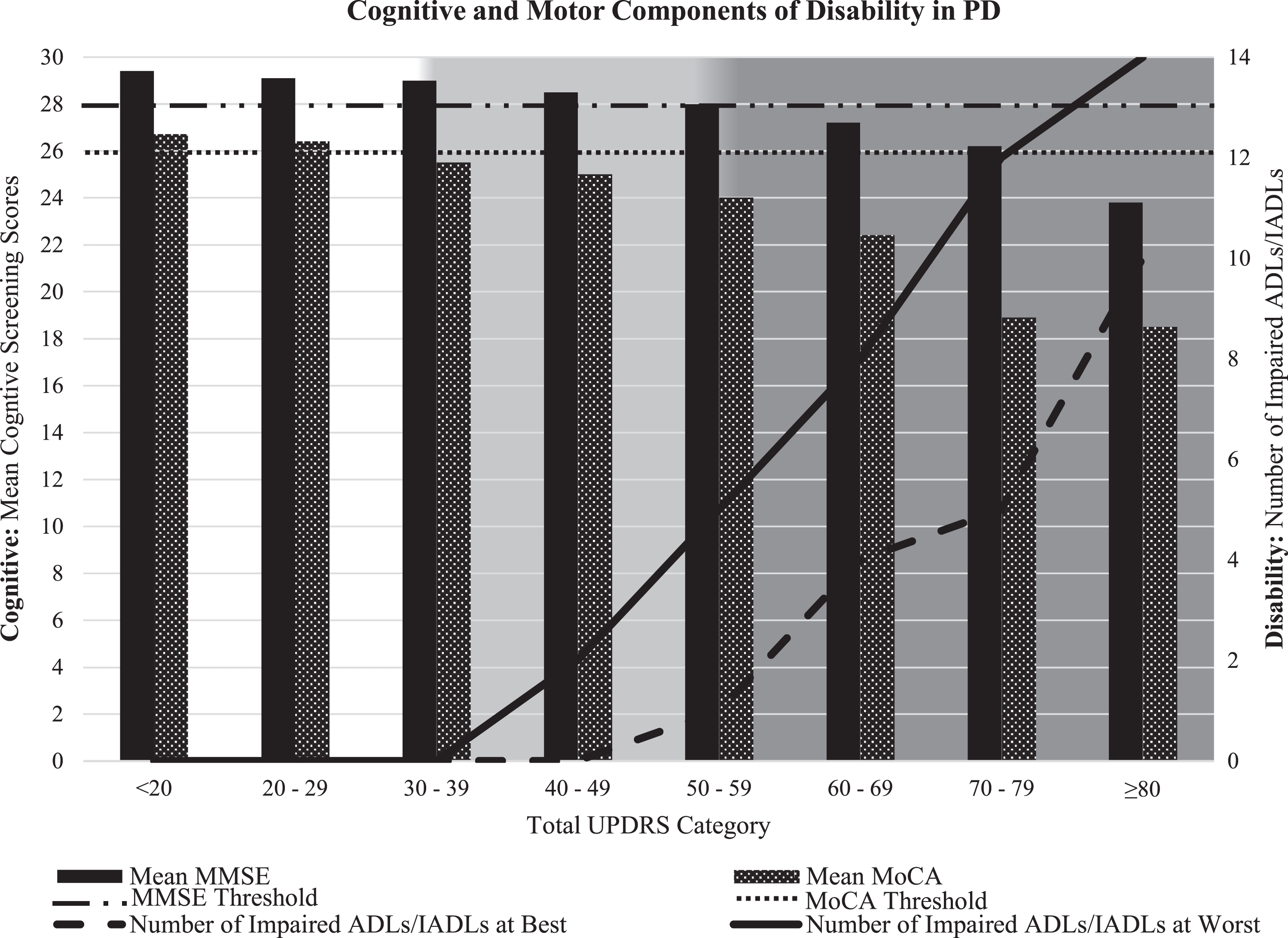
Cognitive Function (mean MMSE and MoCA scores) and Disability (number of impaired ADLs/IADLs) by incremental Total UPDRS Subgroups illustrate the combined effects of cognitive and physical impairments on disability in PD. Disability is measured by the OARS (Older Americans Resources and Services Disability Subscale) comprised of 14 items, including 7 ADLs and 7 IADLs [31]. All patients reported their best and worst function on each item. Preclinical disability (light gray shading) is defined by less than two impaired ADLs and Disability (dark gray shading) is defined as two or more impaired ADLs. MMSE cognitive impairment cut-off designated at 28 and MoCA at 26.
Post-hoc t-tests showed that mean MMSE scores differed significantly (p < 0.01) between subgroups with a Total UPDRS score above 30 even though mean MMSE scores were not below the high cutoff for cognitive impairment (< 28) until subgroups above 60 or≥80 for the low MMSE cutoff (< 26) (Table 3). Differences in mean MoCA scores were significant between UPDRS subgroups 20–29 and 30–39 and between all subgroups from 40–49 to 70–79 but did not differ significantly between the lowest UPDRS subgroups < 20, between 30–39 and 40–49, or above 70–79 (Table 3).
A closer examination of cognitive scores across Total UPDRS categories, using an MMSE cutoff below 26 and a MoCA cutoff below 26, revealed that the mean MMSE score was below the threshold for CI in only 94 of 2,440 records (3.9%), compared to 810 of 1,223 (66.7%) for those who were assessed with the MoCA. Using a higher MMSE cutoff of 28, still only 391 of 2440 records (16.0%) were below the threshold for cognitive impairment.
Mean MMSE scores ranged from 26 to 30 across HY stages (Table 4). Mean MoCA scores ranged from 21 to 27 (Table 4). Mean MMSE scores differed significantly (p < .05) between each HY stage. Mean MoCA scores differed significantly between stages 2.5, 3, and 4–5. Mean MMSE scores remain above the high cutoff for CI through advancing disease stages until HY 3, whereas MoCA scores nearly reach the CI threshold of 26 even in the earliest disease stages.
Mean MMSE and MoCA Scores at Different Stages of PD Motor Severity (HY Stages)
Mean levels of cognitive function (MoCA and MMSE) across worsening PD motor severity (HY Stages). Cognitive scores associated with impairment highlighted in light gray. High MMSE cutoff highlighted in dark gray.
Notably, the mean MoCA score dropped below the CI threshold at Total UPDRS 30–39 and between HY stage 2 and 2.5; the transition from HY 2 to 3 has been previously identified as the critical period for emergence of disability [33].
For both the MMSE and MoCA, the standard deviations increased with greater disease progression as measured by Total UPDRS and HY, indicating a broader range of cognitive function with disease progression, though sample sizes are smaller.
DISCUSSION
This study used the largest sample in the literature to date to investigate the relationship between common cognitive screening assessments (MMSE and MoCA) and PD progression. We found that 2/3 of MoCA scores fell below the cutoff for CI compared to 4–16% for the MMSE. Mean MMSE scores varied little across the range of disease progression and did not fall below the threshold for cognitive impairment (MMSE < 26) until the most advanced stages on the Total UPDRS and HY—an observation that held even when using a higher threshold of MMSE < 28. In contrast, MoCA scores were highly sensitive to cognitive change in PD, where the mean MoCA score was close to the CI threshold at the lowest ranges of the Total UPDRS and HY. This study describes the inter-relationships between cognitive function, PD symptom severity, and functional status to show the contribution of cognitive impairment to disease progression and disability.
The wider score range and markedly reduced ceiling effect demonstrate the greater sensitivity of the MoCA compared to the MMSE for cognitive screening in PD. These factors have been previously reported [18–22]; however, the magnitude of the comparative sensitivity to detect CI in PD demonstrated in this study is striking. The MMSE has been reported to be a better predictor of dementia and to track better with PD progression [20, 34]. Our results call into question these reported advantages of the MMSE and add to the evidence that it is inadequate for CI screening in PD. The Movement Disorders Society Rating Scales Review Committee includes the MoCA as one of three global cognitive screening tools recommended for use in PD clinical practice, along with the Mattis Dementia Rating Scale 2nd Ed and the Parkinson’s Disease Cognitive Rating Scale, whereas the MMSE only has a “suggested” designation [3]. Despite this, the MMSE continues to be used as eligibility criteria in many recently published clinical trials and numerous active studies on clinicaltrials.gov [23, 24]. While many studies emphasize the superiority of the MoCA over the MMSE as a screening tool, the optimal tool for tracking cognitive change over time is less established. It has been suggested that MMSE is superior to MoCA in tracking change in PD [35], but other studies show no difference between the MMSE and MoCA in assessing change [20, 37]. Our cross-sectional study showing worsening of MoCA scores with increasing PD progression and strong correlations between MoCA scores and disability suggest that MoCA may be useful longitudinally and warrants further investigation.
Cognitive function showed moderate to high correlations with disability. Notably, MoCA assessments that reached the threshold for CI corresponded to Total UPDRS 30–39 and HY stage 2–3, the same period that we previously identified as a critical stage where disability emerges [33]. There are both motor and cognitive components to disability in PD, however functionally relevant, practical cognitive assessments to detect this are limited [9, 38]. Cognitive impairment is associated with more rapid progression of motor disability [39, 40] and advanced PD is associated with postural instability and falls [41], increased disability, and reduced quality of life [7–10]. Before this transition, there is a critical time for potential intervention—a period of “preclinical disability”—when patients have functional limitations but can compensate successfully and maintain functional independence in daily activities [33]. We previously reported that loss of independence in ADLs (emerging between Total UPDRS 30 to 40 and HY 2 to 3), corresponds to the onset of gait and balance impairment [33]. This study shows that this inflection point from independent to dependent function not only corresponds to increasing motor dysfunction but also to the emergence of cognitive impairment, as measured by the MoCA (Fig. 1). The figure shows that UPDRS 30–39 corresponds with a drop of the mean MoCA score below the threshold of cognitive impairment, in contrast to the mean MMSE score that fails to drop below its corresponding threshold until UPDRS≥60. Patient-reported disability, based on the mean number of impaired ADLs and IADLs, shows the convergence of cognitive impairment with the onset of disability at best function. Increasing disability based on the progression of cognitive and motor dysfunction is observed at each successive increment of cognitive and motor impairment. Therefore, the transition from Total UPDRS 30 to 40 and HY 2 to 3 is an important marker of both motor and nonmotor (cognitive) progression. Clinicians can use this insight to provide important anticipatory guidance and proactively employ interventions during this period to delay disability, including increasing dopaminergic therapy, minimizing medications with deleterious cognitive effects, and recommending rehabilitation and exercise.
The strengths of this study include the use of naturalistic data from clinical practice and a very large sample size. We acknowledge that the MMSE and MoCA are both screening tools and abnormal scores on these instruments are not specific for PD-related cognitive impairment. However, we aimed to use easily obtainable information from standard clinical tools to identify relevant transition points in PD progression. The advantages of the MoCA over the MMSE are driven in part by the increased weight of executive function tasks [20]. However, executive dysfunction is not specific to PD and can be caused by a myriad of other factors such as sleep disturbance, medications, vascular disease, etc. [42]. There was greater variation in MoCA scores overall and with disease progression, which is likely to be based on the intrinsic test properties of the MoCA, the underlying disease process, and differences in sample sizes. Limitations include recruitment from an academic movement disorder center whose patients are disproportionately white and highly educated, thus limiting generalizability. Specific data regarding duration of motor symptoms was not available for a significant portion of the sample and thus could not be included in analysis. There were small statistical differences between the MMSE and MoCA cohorts including age, education, and disease stage. Also, MMSE and MoCA assessments were done in sequential periods (2002–2009 and 2009-present) rather than concurrently, which may introduce bias into the sample. The data were cross-sectional, so results reflect observations across a population, rather than changes in patients over time.
This study presents the results of cognitive screening assessments spanning all stages of disease in the largest PD cohort studied to date. In summary, our results highlight several key points. First, this study adds to existing evidence that the MoCA is superior to the MMSE as a screening tool in PD. Cognitive change was identified in two-thirds of PD patients with the MoCA as compared to less than a fifth of patients identified with the MMSE. Second, the period of emergence of cognitive impairment as measured by the MoCA corresponds to the period in PD progression when disability emerges. This confluence illustrates the combined effects of motor and cognitive function in managing daily activities. Therefore, the emergence of CI portends the gradual transition from independent to dependent function, marking a critical target for intervention. These results highlight the importance of routine cognitive assessment with sensitive tools like the MoCA. Future directions include longitudinal validation of cognitive assessments and development of new measures that capture the combined effects of motor and cognitive impairments on PD-related disability.
Footnotes
ACKNOWLEDGMENTS
This research was conducted at the University of Maryland Parkinson Disease and Movement Disorders Center as part of the HOME (Health Outcomes Measurement) Study and is supported by The Rosalyn Newman Foundation.
CONFLICT OF INTEREST
AS: The author has no conflict of interest to report.
AGB: The author has no conflict of interest to report. Dr. Gruber-Baldini receives grant funding from the National Institutes of Health (R01AG059651; R01 AG059651, P30 AG028747-13S3, T32 AG000262, R21) and a grant from Gilead Science, Inc.
FRVC: The author has no conflict of interest to report.
JMS: The author has no conflict of interest to report.
SGR: The author receives grant support from the NINDS; book royalties from Springer, Oxford, Informa; chair of the Data Safety Monitoring Board for Enterin; reviewer for UpToDate; Consultant for Best Doctors
MJA: The author receives research support from the NIA (R01AG068128, P30AG047266), the Florida Department of Health (grant 20A08), and as the local PI of a Lewy Body Dementia Association Research Center of Excellence. She receives royalties from the publication of the book Parkinson’s Disease: Improving Patient Care and she has received honoraria for presenting at AAN and International Parkinson and Movement Disorder Society annual meetings.
LMS: The author receives research funding from the National Institutes of Health (1R01AG059651-01; 3P30AG028747-13S3), the Maryland Innovation Initiative, the Rosalyn Newman Foundation and the Eugenia and Michael Brin Family, and receives publishing royalties from Oxford University Press and Johns Hopkins University Press.
