Abstract
Background
Accessible screening tools are crucial for the early detection of cognitive impairment in Parkinson's disease (PD), especially in contexts where in-person assessments are not feasible.
Objective
To assess the clinical usability of telephone-based cognitive screening (TBCS) tools in PD patients.
Methods
Forty-two non-demented PD individuals and N = 103 healthy controls (HCs) underwent a TBCS battery including the Telephone Interview for Cognitive Status (TICS), the Telephone-based Frontal Assessment Battery (t-FAB) and sub-tests from the Telephone-based Verbal Fluency Battery (t-VFB) and Telephone Language Screener (TLS). PD individuals were also administered the Montreal Cognitive Assessment (MoCA) in person and assessed for behavior and cognitive-driven functional independence via caregiver-report questionnaires administered over the telephone (Neuropsychiatric Inventory, NPI and Amsterdam IADL Questionnaire – 30-item version, A-IADL-Q-30, respectively). Correlations were run to test the construct and ecological validity of TBCS tests in PD individuals; ROC analyses were run to test the capability of TBCS tests to discriminate PD individuals from HCs and PD individuals with a defective MoCA from those performing normally (PD-MCI vs. PD-CN).
Results
The vast majority of TBCS measures were significantly associated with the MoCA and A-IADL-Q-30 scores, supporting convergent and ecological validity, respectively; TBCS measures also diverged from the NPI. TBCS measures of global cognition and executive-attentive measures discriminated PD individuals from HCs with acceptable accuracy (AUC = 0.71–0.75), showing, by contrast, a better performance in differentiating PD-MCI from PD-CN groups (AUC = 0.74–0.89).
Conclusion
TBCS tests are clinimetrically sound tools for identifying cognitive deficits in PD.
Plain language summary title
Can telephone tests help identify cognitive problems in people with Parkinson's disease?
Plain language summary
People with Parkinson's disease often face challenges with memory and attention, which can affect their daily lives. Finding these problems early is important, but traditional in-person tests can be hard to arrange, especially for people who live far away or have difficulty moving. This study looked at whether simple tests done over the telephone could help identify cognitive problems in people with Parkinson's disease. We tested 42 people with Parkinson's and 103 healthy individuals using phone-based tools that check memory, attention, language, and planning. We also compared the phone test results with in-person tests and questionnaires filled out by caregivers about how well patients manage daily activities. The results showed that phone tests gave information similar to in-person tests and caregiver reports. They were able to tell apart people with Parkinson's from healthy individuals, and especially to identify those with more severe cognitive difficulties. These findings suggest that telephone tests are a useful and reliable way to check cognitive problems in people with Parkinson's disease. This could make it easier for specialists to detect and monitor these problems, especially when in-person visits are not possible.
Keywords
Introduction
Parkinson's disease (PD) is a progressive neurodegenerative condition characterized by both motor and non-motor symptoms, including sleep disorders, autonomic dysfunction, as well as cognitive impairment and behavioral disorders. 1 Cognitive impairment is a non-motor disturbance common even at an early stage in PD. 2 It ranges from mild cognitive impairment (PD-MCI) to dementia (PD-D), affecting approximately 40% of patients, particularly as the disease progresses. 3 The most severely impaired cognitive functions include executive functions, attention, memory, visuo-spatial abilities and language, detrimentally affecting patients’ functional independence.4,5
To improve the diagnosis and management of cognitive impairment in PD, specific guidelines for PD-MCI 6 and PD-D 7 have been developed, aimed at standardizing diagnostic criteria, facilitating early identification, and optimizing patient care. The Montreal Cognitive Assessment (MoCA) is a rapid cognitive screening tool that evaluates multiple domains and contributes to early detection and monitoring of cognitive deficits.8,9 A study conducted in New Zealand demonstrated its ability to detect dementia and MCI in this population. 10 Its brevity and ease of administration make it suitable for both clinical and research settings where time and resources may be limited.11,12 This screener, even if administered within a relatively short time interval, yields a score that is not biased by possible learning effects. 13
Although an early and accurate diagnosis of cognitive impairment in PD is crucial, several factors - including limited specialist availability, financial costs, geographic constraints, and difficulties in patient mobility - can restrict patients’ access to a comprehensive neuropsychological assessment.
Moreover, many individuals with PD do not undergo systematic cognitive assessment until symptoms have become significantly debilitating, delaying interventions that could enhance quality of life and potentially slow functional decline. Therefore, it is essential to develop more accessible and easily administered screening tools that enable earlier identification of at-risk individuals and facilitate timely referral for comprehensive diagnostic assessment.
Due to the barriers that some patients face in accessing in-person neuropsychological services, remote assessment tools have gained importance. The COVID-19 pandemic significantly accelerated the adoption of telemedicine, including in neuropsychology, as it became a necessary response to the limitations imposed by social distancing and the need for remote healthcare.14,15
Tele-neuropsychology has emerged as a promising solution, utilizing technologies such as videoconferencing, web-based applications, and telephone-based evaluations to provide high-quality diagnostic work-ups remotely. 16 Tele-neuropsychology includes telephone-based cognitive screening (TBCS) as a rapid and practical method for identifying populations at risk of cognitive disorders or dementia. 17 However, research on TBCS tests in people with PD remain limited.18,19 In addition to improving accessibility, such tools may help characterize cognitive functioning in a broader range of patients, considering individual variability in clinical expression.
The review by Binoy, Lithwick Algon 20 highlights that remote cognitive testing is an effective method for cognitive assessment in PD. Key advantages include improved accessibility to a broader range of patients, reduced costs, and the ability to tailor sessions to individual needs. Importantly, remote assessments facilitate follow-up, which is crucial given the progressive nature of PD and the need for continuous monitoring. 20 However, several challenges remain, including concerns about data security, the potential exclusion of more impaired individuals, technical issues, interference from caregivers, and the lack of normative data. Moreover, it is still uncertain whether remote tests measure the same cognitive constructs as in-person assessments. 20
A preliminary validation of a telephone version of the Montreal Cognitive Assessment (T-MoCA) was conducted to assess its ability to identify mild cognitive impairment in people with PD. 19 While the T-MoCA showed potential as a screening tool when in-person assessment is not feasible, it demonstrated weak correlations with standard cognitive measures, leading the authors to recommend cautious interpretation and use in clinical practice. 19
Given the above premises, this study aims to evaluate the clinical usability accuracy of a selected set of standardized TBCS tests in people with PD, in the view of determining whether these tools can serve as a valid alternative to traditional in-person cognitive screening procedures. To this end, the study focuses on testing the convergent, divergent, and ecological validity of a set of TBCS tests, as well as on their ability to discriminate people with PD from healthy controls and cognitively normal PD individuals (PD-CN) from PD-MCI ones.
Methods
Participants
A total sample of 145 participants was recruited between November 2023 and November 2024, all native Italian speakers or individuals with an excellent command of the Italian language, aged between 38 and 91 years. The sample was divided into two groups: 42 people with PD and 103 healthy controls (HCs).
All participants were recruited through non-probabilistic sampling. Exclusion criteria for the HC were: 1) a negative history for neurological or psychiatric disorders; 2) current use of psychotropic medications; 3) severe and/or uncompensated general-medical conditions; 4) uncorrected hearing impairments. For the PD group, the main inclusion criterion was a confirmed diagnosis of PD according to the Movement Disorder Society–Sponsored Revision of the Unified Parkinson's Disease Rating Scale (MDS-UPDRS) criteria. 21 Individuals with uncorrected hearing deficits, organ or systemic failure, hypophonia, severe behavioral disorders or a diagnosis of dementia were excluded.
Participants provided informed consent and data were treated according to current regulations. The study protocol received approval from the Ethics Committee of the IRCCS Istituto Auxologico Italiano (ID: 23C308) and by the local Institutional Review Board of Milan A1 (Approval No. 57/2025).
Materials
Upon recruitment, people with PD underwent an in-person cognitive assessment with the MoCA. The telephone-based assessment was then conducted at least seven days, but no more than six months, after the MoCA being administered. For the telephone-based assessment, a test battery was administered consisting of the following instruments:
Telephone Interview for Cognitive Status (TICS).22,23 The test consists of 11 items assessing Orientation (at personal, spatial, and temporal levels; range = 0–12), Attention and Executive Functions (backward counting, “serial 7s” and abstraction; range = 0–9), Memory (immediate recall of a 10-word list and semantic memory; range = 0–12), and Language (naming to description of nouns, repetition of sentences and comprehension of a simple command; range = 1–8). An additional Delayed Recall task of the 10-word list was also administered at the end of the TICS (range = 0–10). The total score ranges from 1 to 41, and from 1 to 51 with the addition of the Delayed Recall task. Telephone-based Frontal Assessment Battery (t-FAB).
24
The t-FAB assesses executive functions such as cognitive flexibility and inhibition. The total test score ranges from 0 to 12 and comprises all of the tasks enclosed within the in-person version of the FAB
25
except for Programming and Environmental autonomy ones. It thus includes the following items: Conceptualization, Mental flexibility, Sensitivity to interference and Inhibitory control, with the last two being adapted to be administered over the telephone. Besides the total score, two subscores are calculated: the t-FAB-1, including Conceptualization and Mental flexibility tasks, and the t-FAB-2, including Sensitivity to interference and Inhibitory control ones. Telephone-based Verbal Fluency Battery (t-VFB).
26
The t-VFB measures the ability to generate words under specific conditions. The test, which is a telephone-based adaptation of Costa, Bagoj
27
VFB, consists of 3 different verbal fluency subtests, each consisting of 3 trials: phonemic, semantic and alternate verbal fluency (t-PVF/-SVF/-AVF). The tests consist of generating the highest number of words according to the different criteria in 60 s. Productions of proper names, place names, numbers or inflected words with the same suffix are not considered valid. In the t-PVF tests, subjects are asked to generate words starting with letters “F”, “A” and “S”. In the t-SVF, the three criteria are “Colors”, “Animals” and “Fruits”. Finally, in the t-AVF, subjects are asked to continuously alternate letter-cued words with category-cued words as follows: “A/Color”, “F/Animal”, “S/Fruit”. For the purpose of this study, the following trials of the t-VFB were addressed: “F” and “S” items from the t-PVF, “Animals” and “Fruits” from the t-SVF, and “F/Animals” and “S/Fruits” for the t-AVF. Telephone Language Screener (TLS).
28
The test comprises different tasks assessing phonological, lexical-semantic, and morpho-syntactic components, as well as an extra Backward Digit Span task. For the purpose of this study, three subtests were addressed: Backward Digit Span (BDS), Naming to Description of Nouns (NtD-N) and Naming to Description of Verbs (NtD-V). For the Backward Digit Span, in accordance with Pasotti, De Luca
29
two scores are calculated, namely the total number of correctly reported sequences (range = 0–8), this being a measure of sustained attention during task execution, and the longest remembered sequence (range = 0–6), this being a proper measure of phonological working memory. Naming to description tests require subjects to name an object or an action from a given verbal description; through these tests, the lexical-semantic component is targeted.
In the group of people with PD, two semi-structured interviews were also proposed to caregivers over the telephone for the assessment of behavioral disorders and functional independence:
Neuropsychiatric Inventory (NPI).
30
This proxy-report, semi-structured interview allows for the assessment of behavioral disorders that typically occur in neurodegenerative conditions. It consists of 12 sections inquiring about delusions, hallucinations, agitation, depression, anxiety, apathy, irritability, euphoria, disinhibition, aberrant motor behavior, sleep changes and eating disorders. For each positive cluster, proxies are asked to rate the frequency (range = 0–4) and severity (range = 0–3) of symptoms; the total NPI score (range = 0–144) is computed by summing the Frequency*Severity measure of each positive symptom cluster. Amsterdam IADL Questionnaire – 30-item version (A-IADL-Q-30).
31
The questionnaire consists of 30 items that assess the degree of independence in performing complex instrumental tasks, as well as the impact of cognitive and motor difficulties on daily living.
32
For the purpose of this study, a Classical Test Theory-based scoring system was embraced, with each item being assigned a score from 0 to 4, where 4 indicating higher levels of autonomy and 0 that subject is not able anymore to complete that task autonomously. In order to account for non-applicable items (e.g., tasks that were never carried out by the patient), the total A-IADL-Q-30 score was computed by averaging all applicable items (range = 0–4).
HCs were solely administered TBCS tests, whilst proxy-report tools were additionally administered to patients’ carers.
Procedure
Prior to TBCS test administration, a standardized protocol – which has been previously described in detail22,26,28 – was implemented in order to 1) take appropriate measures for controlling the testing setting and 2) ensure a good quality of the telephone call. Setting control measures included the involvement of a caregiver that could ensure that no source of distractions (e.g., TV on, loud noises, etc.) or suggestions/facilitations (e.g., calendars, paper-and-pencil to write down stimuli for memory tasks, etc.) were present within the room where the examinee was located. This proxy was also required in order to confirm address information provided by the examinee – which was the outcome of spatial orientation questions of the TICS-Orientation –, as well as to provide technical support whenever necessary. As to the sound-check procedure, a set of standard tasks were administered in order to verify that no echoes, delays or other audio distortions were present from both the examinee's and the examiner's standpoints. Examinees were instructed not seek or receive assistance from the proxy.
The battery of tests was administered in the same order to all participants: TICS, t-FAB, t-VFB, TLS.
The administration of TBCS tests lasted approximately 30 min. After TBCS tests being administered, the people with PD were asked to actively involve the caregiver in the completion of two questionnaires: NPI and A-IADL-Q-30. This additional phase lasted approximately 30 min.
Statistical analysis
Data distribution for each variable of interest was assessed via skewness and kurtosis indices, with values exceeding |1| for skewness and |3| for kurtosis being considered suggestive of non-Normality. 33 Given the predominance of non-Normally distributed variables, non-parametric methods were applied for running construct and ecological validity analyses.
Specifically, construct validity of TBCS measures was explored via Spearman's coefficients against the MoCA and the NPI for convergent and divergent validity testing, respectively. Similarly, Spearman's correlations were run against the A-IADL-Q-30 in order to investigate the ecological validity of TBCS tests. Bonferroni's correction was applied within the abovementioned correlational sets by addressing k = 4 multiple tests – with 4 being the number of tests (i.e., TICS, t-FAB, t-VFB and TLS) –, thus resulting in an αadjusted = 0.013.
Moreover, receiver-operating characteristic (ROC) analyses were run to test the classificatory accuracy of each TBCS measure in discriminating both the whole PD cohort from HCs and PD-CN from PD-MCI individuals. TBCS scores adjusted for relevant demographic confounders according to current Italian norms22,23,26,28 were entered into the abovementioned ROC analyses in order to level out inter-individual variability in background features. For the purpose of the second set of ROC analyses (i.e., PD-CN vs. PD-MCI), MCI was defined, in alignment with Level-I MD criteria 6 as a defective/borderline demographically-adjusted MoCA score according to current Italian norms. 34 Specifically, participants were classified as PD-MCI if they presented with an Equivalent Score 0 (defective) or 1 (borderline) on the MoCA, following the criteria established by Capitani and Laiacona 35 and the corresponding normative framework. 36 This procedure should not be interpreted as equivalent to a Level II diagnosis, which requires a comprehensive neuropsychological assessment. 6 AUC values >0.7 were deemed as indexing an acceptable level of discriminatory accuracy. 37 No multiplicity correction was applied to the both of the above-mentioned sets of ROC analyses.
Analyses were run via SPSS 28 (IBM Corp., 2024) and jamovi 2.6 (the jamovi project, 2024). Missing values were excluded pairwise.
Results
Demographic and background clinical characteristics of people with PD and HCs are reported in Table 1. The two groups were matched for education, although people with PD proved to be significantly older than HCs. Moreover, the two groups were unbalanced for sex – with a higher proportion of males in the PD cohort when compared to HCs and the HC group including a higher number of females when compared to PD patients. Within the PD group, disease duration was highly variable, ranging from 1 to 23 years since onset. Additionally, 26.2% of people with PD had undergone deep brain stimulation (DBS) surgery. The time elapsed between surgery and the TBCS evaluation ranged from 5 to 26 months, with a mean of ∼16 months. Finally, the mean interval between the in-person and TBCS assessments was of ∼64 days, corresponding to approximately two months.
Background characteristics of PD and HC groups.
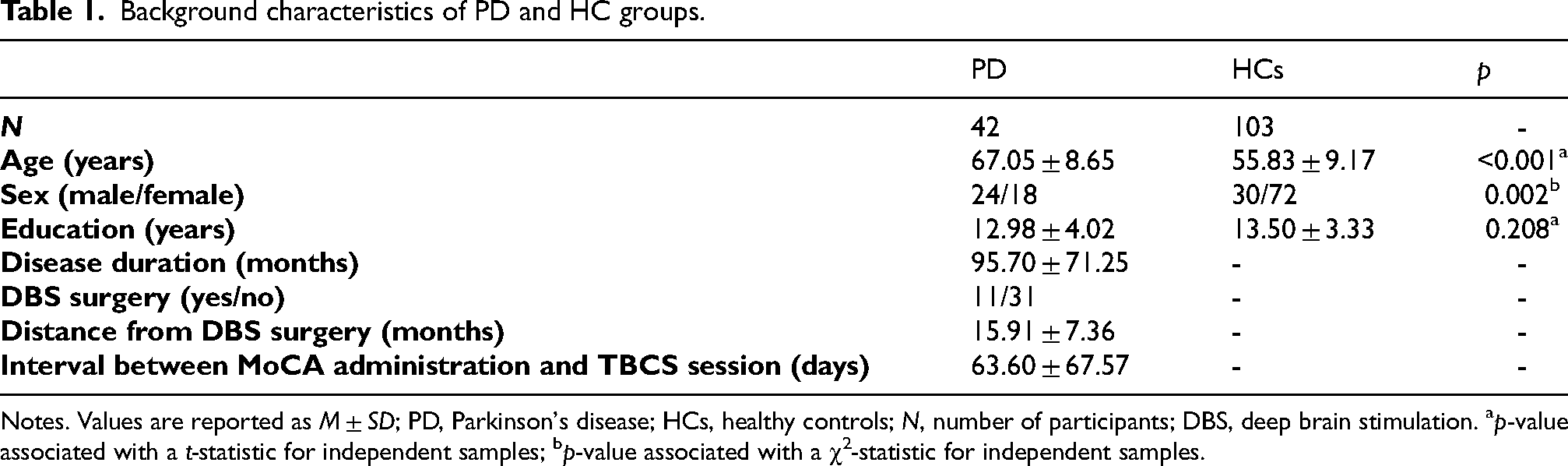
Notes. Values are reported as M ± SD; PD, Parkinson's disease; HCs, healthy controls; N, number of participants; DBS, deep brain stimulation. ap-value associated with a t-statistic for independent samples; bp-value associated with a χ2-statistic for independent samples.
Patients’ and HCs’ neuropsychological data are summarized in Table 2. Overall, PD patients scored lower than HCs on all TBCS measures. Among people with PD, the highest percentages of impaired performances were observed as to the Memory (11.9%) and Delayed Recall (16.7%) subtests of the TICS, the Conceptualization + Mental flexibility subtest of the t-FAB (14.3%), and the alternate fluency tasks of the t-VFB (28.6%). By contrast, the percentage of HCs performing defectively on TBCS tests was overall negligible (i.e., < 2%), except for semantic fluency (4.9%) and alternate fluency (3.9%) tasks of the t-VFB.
Neuropsychological measures of PD and HC groups.

Notes. MoCA, Montreal Cognitive Assessment; TICS, Telephone Interview for Cognitive Status; t-FAB, telephone-based Frontal Assessment Battery; t-FAB-1, Conceptualization + Mental flexibility tasks; t-FAB-2-M, Sensitivity to interference + Inhibitory control tasks; t-VFB, Telephone-based Verbal Fluency Battery; t-PVF, Telephone-based Phonemic Verbal Fluency; t-SVF, Telephone-based Semantic Verbal Fluency; t-AVF, Telephone-based Alternate Verbal Fluency; TLS, Telephone Language Screener; BDS-T, Backward Digit Span-Total; BDS-WM, Backward Digit Span-Working Memory; NtD-N, Naming to Description of Nouns; NtD-V, Naming to Description of Verbs; NPI, Neuropsychiatric Inventory; A-IADL-Q-30, Amsterdam IADL Questionnaire −30-item version. ap-values associated with Mann-Whitney's U-statistic run on continuous test scores; bdata available for N = 38 patients; cdata available for N = 40 patients.
At αadjusted = 0.013, no associations were detected in patients between TBCS measures and disease duration (rs ≤ 0.34, p ≥ .030).
Table 3 shows Spearman's correlation coefficients for convergent, divergent and ecological validity testing in the PD cohort. At αadjusted = 0.013, all TBCS measures proved to be positively associated with the MoCA, except for the t-SVF-Animals, both TLS-BDS measures and the TLS-NtD-N. Moreover, all TBCS measures diverged from the NPI. As to ecological validity analyses, significant, positive correlations were detected between A-IADL-Q-30 scores and the TICS and the TICS with Delayed Recall, both t-SVF and t-AVF scores, as well as the TLS-NtD-V.
Spearman's coefficients between TBCS measures and convergent (MoCA), divergent (NPI) and ecological validity (A-IADL-Q-30) measures.
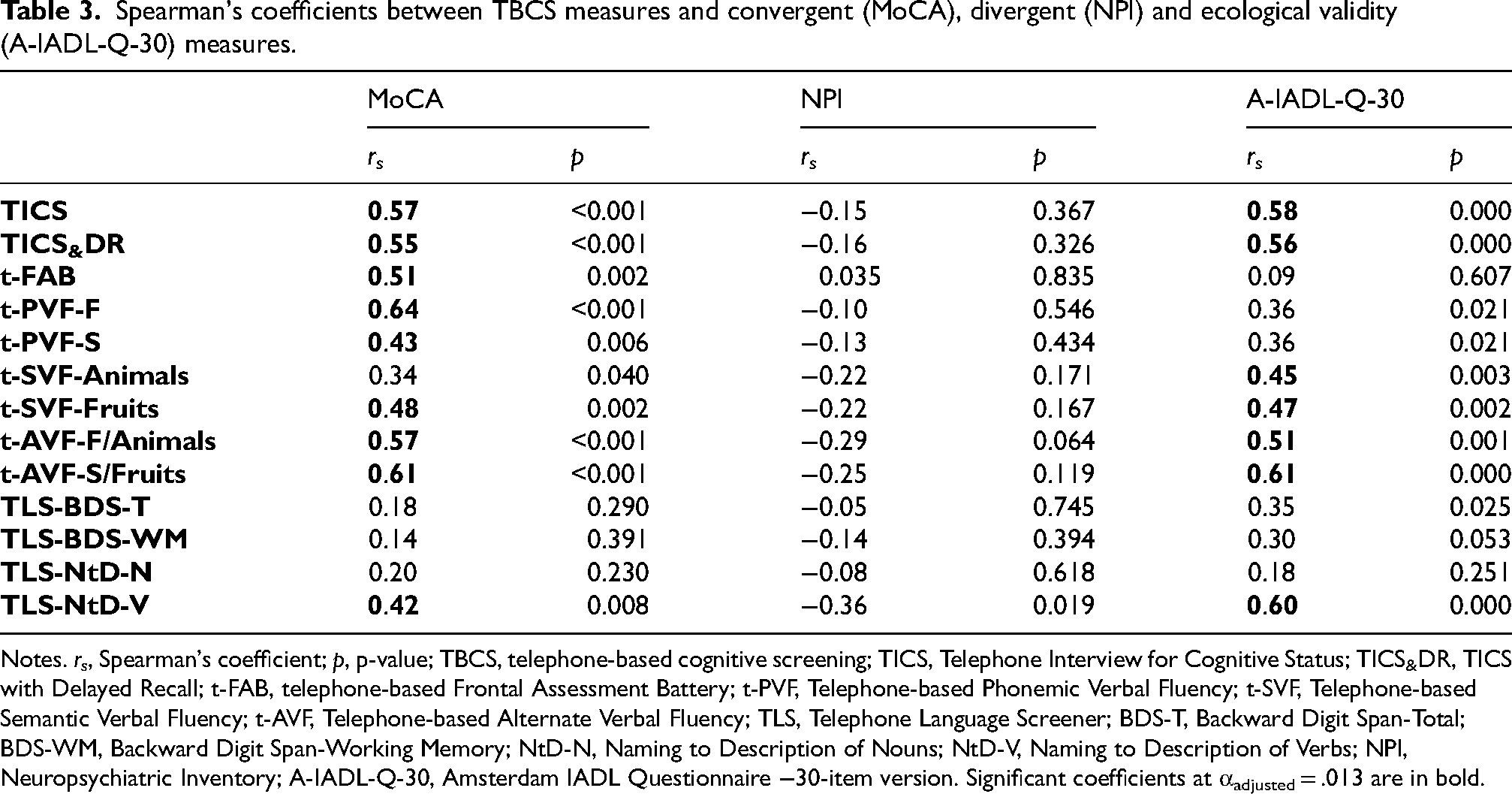
Notes. rs, Spearman's coefficient; p, p-value; TBCS, telephone-based cognitive screening; TICS, Telephone Interview for Cognitive Status; TICS&DR, TICS with Delayed Recall; t-FAB, telephone-based Frontal Assessment Battery; t-PVF, Telephone-based Phonemic Verbal Fluency; t-SVF, Telephone-based Semantic Verbal Fluency; t-AVF, Telephone-based Alternate Verbal Fluency; TLS, Telephone Language Screener; BDS-T, Backward Digit Span-Total; BDS-WM, Backward Digit Span-Working Memory; NtD-N, Naming to Description of Nouns; NtD-V, Naming to Description of Verbs; NPI, Neuropsychiatric Inventory; A-IADL-Q-30, Amsterdam IADL Questionnaire −30-item version. Significant coefficients at αadjusted = .013 are in bold.
The results of ROC analyses for the discrimination of the whole PD cohort from HCs are presented in Table 4. Notably, only a few tests achieved an acceptable level of accuracy in discriminating patients from HCs, namely, the TICS (AUC = 0.73) and the TICS with Delayed Recall (AUC = 0.721), the t-PVF-F (AUC = 0.75), both t-AVF tests (AUC = 0.70–0.73) and the TLS-NtD-N (AUC = 0.716).
ROC analyses for the comparison between people with PD and HCs.
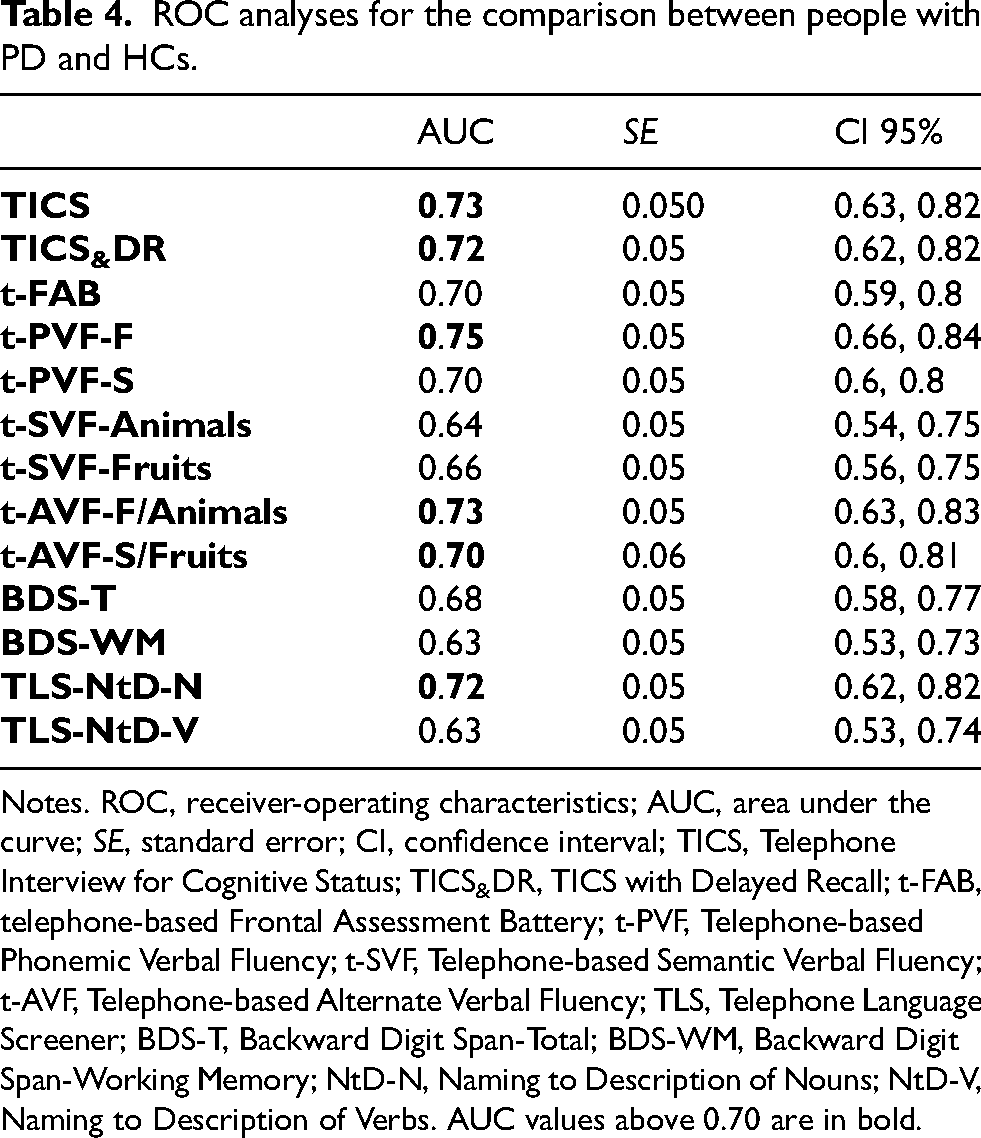
Notes. ROC, receiver-operating characteristics; AUC, area under the curve; SE, standard error; CI, confidence interval; TICS, Telephone Interview for Cognitive Status; TICS&DR, TICS with Delayed Recall; t-FAB, telephone-based Frontal Assessment Battery; t-PVF, Telephone-based Phonemic Verbal Fluency; t-SVF, Telephone-based Semantic Verbal Fluency; t-AVF, Telephone-based Alternate Verbal Fluency; TLS, Telephone Language Screener; BDS-T, Backward Digit Span-Total; BDS-WM, Backward Digit Span-Working Memory; NtD-N, Naming to Description of Nouns; NtD-V, Naming to Description of Verbs. AUC values above 0.70 are in bold.
In Table 5, the results of the ROC analyses concerning the discrimination between PD-CN and PD-MCI patients (defined as those with a borderline/defective MoCA scores) are shown. The TBCS tests that exhibited a moderate level of accuracy in distinguishing between these groups were the TICS (AUC = 0.89) and TICS with delayed recall (AUC = 0.85), the t-PVF-F (AUC = 0.74), the t-SVF-Fruits (AUC = 0.783), both t-AVF tests AUC = 0.77–0.84) and “S/Fruits” (AUC = 0.84), and eventually the name to description of verbs task of the TLS (AUC = 0.83).
ROC analyses for the comparison between PD-MCI and PD-CN individuals.
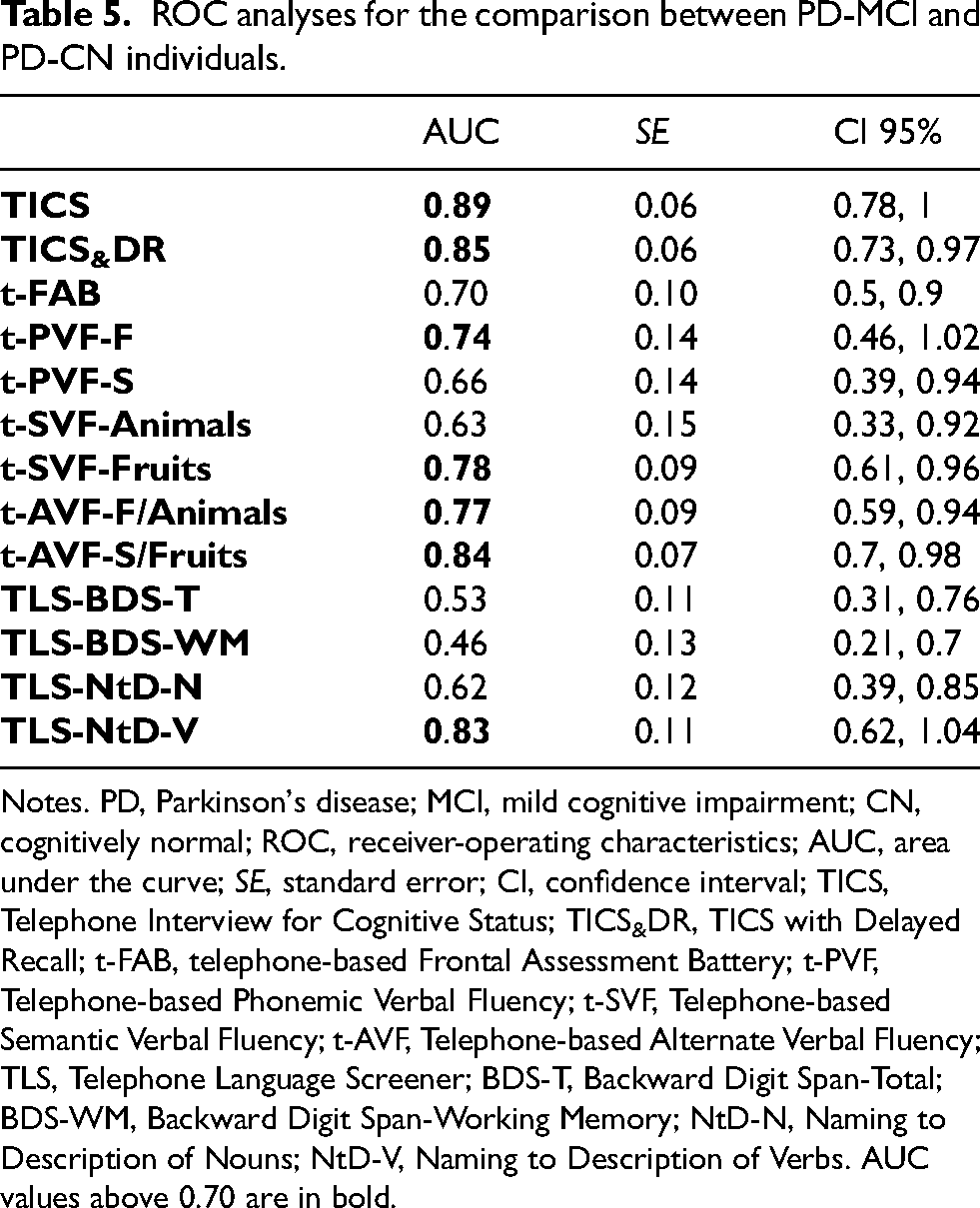
Notes. PD, Parkinson's disease; MCI, mild cognitive impairment; CN, cognitively normal; ROC, receiver-operating characteristics; AUC, area under the curve; SE, standard error; CI, confidence interval; TICS, Telephone Interview for Cognitive Status; TICS&DR, TICS with Delayed Recall; t-FAB, telephone-based Frontal Assessment Battery; t-PVF, Telephone-based Phonemic Verbal Fluency; t-SVF, Telephone-based Semantic Verbal Fluency; t-AVF, Telephone-based Alternate Verbal Fluency; TLS, Telephone Language Screener; BDS-T, Backward Digit Span-Total; BDS-WM, Backward Digit Span-Working Memory; NtD-N, Naming to Description of Nouns; NtD-V, Naming to Description of Verbs. AUC values above 0.70 are in bold.
Discussion
This study investigated the clinical usability of TBCS tests in people with PD, with the aim of evaluating their construct and ecological validity, as well as their classificatory accuracy with regard to both the discrimination of PD individuals from HCs and the detection of PD-MCI. Given the high prevalence of cognitive impairment in PD - affecting over 40% of patients 4 -, the early identification and monitoring of cognitive impairment in this population remain essential components of effective clinical management. Cognitive decline in PD follows a heterogeneous and progressive course, often beginning with subtle impairments in attention, executive functions, and visuospatial abilities, and evolving into more global deficits over time.4,38 The identification of such non-motor symptoms, especially in the early stages, might be challenging, and thus requires tools that are both sensitive and practical for real-world application.
In this context, the TBCS tools examined in the present study offer a promising alternative to traditional in-person cognitive screening procedures, with the potential to improve access to first-level cognitive evaluations and properly guide potential diagnostic work-ups. In fact, several TBCS tests herewith addressed proved to 1) converge with a gold-standard, in-person screener – i.e., the MoCA –, 2) diverge from a measure of behavioral changes (i.e., the NPI), 3) be associated with patients’ cognitive-driven independence in everyday-life scenarios – as assessed by the A-IADL-Q-30 –, and thus to be ecologically valid, 4) be able to discriminate PD individuals from HCs and 5) effectively detect, among PD individuals, those with Level I-classified MCI. The above was particularly true for two tests, namely the TICS – assessing global cognitive efficiency – and the t-AVF – assessing set-shifting abilities.
Upon in-presence screening assessment through the MoCA, the percentage of subjects who performed below the established cut-off 36 corresponded to ∼16%. Similar percentages emerged by applying normative thresholds to TBCS tests assessing global cognition (TICS), memory (TICS-Memory; TICS-Delayed Recall) and attentive-executive functions (t-FAB-2; t-AVF) – this suggesting that TBCS tests yield a proportion of cognitive impairment that is consistent with that yielded by traditional, in-person screeners.
The findings are consistent with the literature, which identifies executive dysfunction and memory impairment as core features of cognitive decline in PD.39,40 These cognitive functions may be compromised even at the early stages of PD, as shown by a study in which a cohort of newly diagnosed PD individuals who had not yet started treatment, reported more pronounced impairments in verbal memory and processing speed/attention. 39
The convergent validity of the telephone-based tools was supported by significant correlations with MoCA scores. Specifically, the TICS (with and without Delayed Recall), the t-FAB, the t-VFB, and the verb-naming subtest of the TLS demonstrated moderate to strong associations with the MoCA, indicating that they effectively capture similar cognitive constructs.
As for the TICS, the correlation with the MoCA is explained by the presence in both tests of tasks related to verbal memory, attention and language. 41 The t-FAB and t-VFB, on the other hand, address executive functions and verbal fluency tasks,24,26 domains that are also represented within the MoCA. Finally, the TLS naming task assesses lexical retrieval for verbs and shows a correlation with the MoCA, which although broader in object naming, shares the assessment of lexical access as a fundamental linguistic function. 28 The MoCA is widely recognized as one of the recommended tests for screening of cognitive impairment in PD, due to its ability to comprehensively assess several crucial cognitive functions. 12 The significant correlations observed in this study suggest that TBCS tests may serve not only as complementary tools but also as valid first-level screening options, particularly in contexts where in-person MoCA administration is not feasible.
It should be noted that the interval between the in-person and telephone MoCA assessments, which showed variability among participants (M = 63.6 days, SD = 67.6), ranging from a few days to six months. This variability was mainly due to organizational reasons and participant availability. Cognitive decline in Parkinson's generally progresses slowly, with measurable changes typically occurring on an annual rather than a monthly timescale.40,42 Moreover, the follow-up study by Goldman et al. (2018) 43 demonstrated that, over a 6–12 month period, MoCA scores tend to remain relatively stable, with only modest changes that are often not clinically significant. Nonetheless, some studies highlight substantial clinical variability: while most PD individuals follow a slow trajectory, a subset may experience a more accelerated course, often associated with factors such as older age, greater motor severity, or vascular comorbidities.42,44 Longer intervals could have introduced cognitive changes and thereby attenuated correlations, leading to an underestimation of convergent validity. The fact that, despite this, most TBCS measures still showed moderate-to-strong associations with the MoCA provides further evidence of the robustness of our findings.
After confirming convergent validity, no correlations emerged between TBCS tests and behavioral changes as measured by the NPI. This supports the divergent validity of the TBCS tests, suggesting they are not confounded by behavioral symptoms. Moreover, although behavioral symptoms are common in PD, particularly in early stages they may not substantially impact cognitive test performance. 39 Furthermore, the absence of strong associations was consistent with expectations, also considering that patients with clinically significant neuropsychiatric symptoms were excluded from the sample.
Importantly, ecological validity was demonstrated through significant positive correlations between performance on TBCS measures and the A-IADL-Q-30, a validated proxy-based questionnaire assessing the degree of independence in performing complex instrumental tasks. 31 As an informant-based tool, the A-IADL-Q-30 may be influenced by caregiver-related factors such as burden or depressive symptoms, 45 although previous studies suggest that these potential confounding effects are limited, 46 thus supporting the robustness of the instrument. The significant correlations support the notion that the current TBCS tests represent a valid estimate of PD individuals independence in everyday-life scenarios. In fact, domains such as attention, working memory, and executive functioning – commonly impaired in PD – are known to predict reduced functional independence,47,48 and the current findings are consistent with this body of evidence.
A relevant set of findings from the present report are those yielded from ROC analyses, which appeared to support the capability of TBCS test to discriminate people with PD from HCs. Several test and subtests - particularly the TICS, alternate fluency tasks of the t-VFB, and the TLS noun-naming task - achieved AUC values above 0.7, indicating moderate accuracy in distinguishing people with PD from HCs.
These results are aligned with the literature, according to which people with PD show poorer performances when compared to HCs on cognitive tests that cover the vast majority of the areas covered by the PD-MCI criteria – attention and working memory, executive functions, memory and language. 49
The final aim of this report was to assess the capability of TBCS tests to discriminate PD-MCI from PD-CN, with these groups being defined based on their demographically adjusted scores on the MoCA. 34 Results showed that the TICS, t-VFB, and the verb-naming task of the TLS demonstrated acceptable-to-moderate levels of accuracy in differentiating between the two groups, in accordance with Swets’ (1988) guidelines. 37 In particular the TICS, TICS&DR, t-AVF-S/Fruits, TLS-NtD-V emerged as the most clinically promising measures (all AUC > 0.80).
Regarding the TICS, its optimal level of discriminatory accuracy is in line with the literature showing that comprehensive cognitive screening batteries, as long as they adequately capture PD-specific cognitive impairment profiles – e.g., executive, attentive and memory deficits – are able to identify PD-MCI. 12 Moreover, adding the Delayed Recall task did not improve its diagnostic accuracy, in line with previous findings from the general population. 22 The t-VFB also showed high accuracy in distinguishing PD-MCI from PD-CN, consistent with a recent report on the diagnostic properties of the in-person version of the VFB in this population. 50 Its strong correlation with both linguistic and executive functions further supports its validity. 26 Similarly, the TLS verb-naming task showed good discriminative accuracy, in alignment with the notion of in-person verb-naming tests being sensitive to cognitive decline in people with PD. 51
In contrast, the t-FAB showed a low level of accuracy, which diverges from recent findings on the in-person version of the FAB in people with PD. 52 This discrepancy may be due to the reduced number of items in the telephone version compared to the in-person version. In addition, modality-specific limitations - such as the absence of visual cues, reliance on auditory-only presentation, and reduced interaction - may have further constrained the ability of the t-FAB to fully capture executive functioning.
Some methodological limitations must be acknowledged. The control and patient groups were not matched for age and sex, with PD individuals being older and more frequently male than controls. While all test scores were adjusted for demographic variables according to Italian normative data, and this likely mitigated the impact of group differences, future studies would benefit from recruiting an HC group that is accurately matched for sex and age to that of PD individuals. At the same time, it has to be acknowledged that our findings of HCs being less frequently males and younger when compared to the PD cohort somehow reflect the epidemiology of PD itself – which is an age-related neurodegenerative disorder with a male predominance. 53 Hence, whilst problematic at a methodological level for the specific aims of this study, the above-mentioned, between-group demographic imbalance is an expected occurrence that mirrors real-life scenarios. Additionally, the sample size, while adequate for the scopes of the present study, should be expanded to increase the level of generalizability of these findings. Such a consideration mostly applies to potential subgroup analyses, such as that addressing people with PD who did or did not underwent DBS surgery. In fact, in this cohort, the small number of patients undergone this procedure (11 out of 43) prevented us from performing statistically sound and practically meaningful subgroup analyses. Given that DBS might affect patients’ cognitive status 54 it is relevant for future investigations on the topic to take into account such a confounding factor in replicating our analyses.
A final point of interest concerns the practical advantages of telephone-based cognitive screening. This modality allows for flexible, remote administration, which is particularly beneficial for people with PD who may face mobility limitations or reside in areas with limited access to specialized care. It also reduces the logistical and economic burden of in-person visits, making it an appealing option for routine monitoring or triage in both clinical and research contexts. Given the growing emphasis on telemedicine, especially following the COVID-19 pandemic, the availability of validated remote cognitive screening tools becomes increasingly important. In this broader landscape, recent work has focused on the development of multidimensional screening instruments,55,56 as well as video-based MoCA adaptations and tablet-based cognitive platforms.20,57 While these approaches offer promising avenues for more comprehensive and interactive assessments, they require technological resources and digital literacy that may not be available to all patients. By contrast, telephone-based screening remains a low-barrier and valid option, particularly when in-person or technology-intensive methods are not feasible, thus positioning TBCS as a complementary tool within the evolving field of remote cognitive assessment in PD. However, TBCS also present inherent limitations compared to video- or tablet-based approaches. By relying exclusively on auditory presentation, telephone-based tasks cannot capture visuospatial or graphomotor abilities, which are often relevant in PD. Moreover, the lack of visual support and reduced interaction may constrain the assessment of higher-level executive functions. These limitations suggest that TBCS should not be regarded as a replacement for comprehensive or technology-based assessments, but rather as a complementary and pragmatic option in settings where such alternatives are not feasible.
Conclusion
In conclusion, the findings of this study support the clinical utility of TBCS in people with PD. These tools demonstrate adequate validity, ecological relevance, and classificatory accuracy, and represent a viable alternative to in-person screening procedures for identifying and monitoring cognitive impairment in PD. Their integration into clinical practice could enhance the early detection of cognitive impairment in this population, thus supporting timely intervention, and ultimately improving the quality of care and life for individuals living with PD.
Footnotes
Acknowledgements
The authors would like to thank Dr Beatrice Curti, Dr Giulia De Luca, and Dr Arianna Moreschi for their support in data collection and statistical analyses. We thank Giunti Psychometric for allowing us to employ the Telephone Interview for Cognitive Status for experimental purposes. We thank the Amsterdam University Medical Center (UMC) for providing us with and allowing us to employ the Italian translation of the Amsterdam IADL Questionnaire – 30-item version (A-IADL-Q-30); the A-IADL-Q-30 is freely available for use by academic users and not-for-profit organizations, and requests can be forwarded via the following link: ![]() .
.
Ethical Considerations
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of IRCCS Istituto Auxologico Italiano (ID: 23C308) and by the local Institutional Review Board of Milan A1 (Approval No. 57/2025). All subjects provided informed consent.
Author contributions
Conceptualization: R.F.; Methodology: R.F., E.N.A.; Formal analysis: E.N.A.; Investigation: AD, F.V.; Data curation: AD, F.V., E.N.A.; Writing - original draft preparation: AD; Writing - review and editing, AD, E.N.A., D.M., A.M., C.M., S.Z., F.P., G.S., F.C., E.M., E.Z., F.M., B.P., and R.F.; Supervision: R.F. All authors have read and agreed to the published version of the manuscript.
Funding
This research was partially funded by the Italian Ministry of Health to the Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico (Ricerca Corrente 2025; Grant Number: 01) and by the PRIN Grant (Code: 20224CACME).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.N.A. has edited the official manual of the Italian version of the Telephone Interview for Cognitive Status (TICS) on behalf of Giunti Psychometrics and perceive royalties from Giunti Psychometrics when such a manual is purchased. Giunti Psychometrics holds the copyright of the Italian TICS. E.N. Aiello serves as an Editorial Board member for BMC Neurology. B. Poletti received fees for consulting services and/or speaking activities from Liquidweb S.r.l., and is Associate Editor for Frontier in Neuroscience. The other authors have declared no competing interests.
Data availability statement
Datasets related to the present study cannot be made publicly available as including sensitive information but can be made available upon reasonable request from interested researcher to the Corresponding Author.
