Abstract
Background:
Neuropsychiatric symptoms (NPS) are the most common non-motor symptom in Parkinson’s disease (PD).
Objective:
To investigate the association between the burden of NPS and motor prognosis in patients with PD.
Methods:
We enrolled 329 drug-naïve patients with PD, who was non-demented and followed-up≥2 years after their first visit to the clinic with baseline dopamine transporter (DAT) imaging and neuropsychiatric inventory (NPI) scores. We performed a survival analysis and a linear mixed model analysis to assess longitudinal motor outcomes according to the NPI total score.
Results:
The Kaplan-Meier analysis showed no difference in the development of levodopa-induced dyskinesia and wearing-off according to the NPI total score. However, higher burden of NPI total score was associated with earlier freezing of gait (FOG) development in the time-dependent Cox regression models after adjusting for age at symptom onset, sex, disease duration, Unified PD Rating Scale motor score, baseline Mini-Mental State Examination score, DAT activity in the posterior putamen and levodopa-equivalent daily dose (LEDD) (Hazard ratio 1.047, p = 0.002). A linear mixed model analysis revealed that patients with a higher NPI total score had a more rapid LEDD increment (NPI×time, p = 0.003). Among 52 patients with PD who eventually developed FOG during the follow-up period, there was a significant correlation between the NPI total score and time with FOG development (γ= –0.472; p = 0.001) after adjusting for confounding factors.
Conclusion:
The present study demonstrated that the severity of NPS is a predictor of early freezing and motor progression in patients with PD.
INTRODUCTION
Although motor symptoms in Parkinson’s disease (PD) often receive the most attention in terms of treatment, non-motor symptoms are frequent and have a major impact on the quality of life of patients with PD [1]. Among the non-motor symptoms, neuropsychiatric symptoms (NPS) such as depression, anxiety, apathy, and psychosis are the most common, and they have a detrimental effect on the burden placed on caregivers as well as the risk of hospitalization [2, 3]. The prevalence of NPS depends on the patients’ cognitive status, and it is approximately exhibiting 56%in early untreated patients with PD and 89%in PD dementia [2, 4].
The relationship between NPS and cognition is well established in patients with PD [5, 6]. Several studies have demonstrated that depression, anxiety, and apathy are closely associated with future cognitive decline in patients with PD [7–9]. A recent study revealed that mild behavioral impairment (MBI), which has been proposed as sustained and impactful NPS acquired later in life [10], is an early and important marker for cognitive impairment and brain atrophy in patients with PD [11].
In terms of the relationship between NPS and motor outcomes, we previously reported that the presence of depression would reflect poor motor compensation in de novo patients with PD [12]. Although one recent study reported that higher motor severity at baseline was associated with worsening NPS over time [13], no studies have comprehensively focused on the effects of NPS on longitudinal changes in parkinsonian motor symptoms in PD. We hypothesized that NPS negatively influences long-term motor outcomes in drug-naïve patients with PD. To test this, we explored the clinical parameters for disease progression with regards to the motor aspects of PD [14], including levodopa-induced dyskinesia (LID), wearing-off, freezing of gait (FOG), and longitudinal increases in doses of dopaminergic medications according to the severity of NPS.
MATERIALS AND METHODS
Participants
We recruited 329 patients with drug-naïve PD who visited the movement disorders outpatient clinic at Yonsei University Severance Hospital between April 2009 and April 2018. PD was diagnosed according to the clinical diagnostic criteria of the UK PD Society Brain Bank [15]. All patients underwent dopamine transporter (DAT) imaging using [18F] N-(3-fluoropropyl)-2β-carbonethoxy-3β-(4-iodophenyl) nortropane positron emission tomography (18F-FP-CIT PET) scans and had decreased DAT availability in the posterior putamen. Parkinsonian motor symptoms were evaluated using the Unified Parkinson’s Disease Rating Scale Part III (UPDRS-III) at the first visit [16]. The Korean version of the Mini-Mental State Examination (MMSE) was used to assess general cognition [17]. The exclusion criteria were as follows: unavailable or remote (>6 months from diagnosis) neuropsychiatric evaluation, long symptom duration (>3 years); MMSE score < 24 or PD-dementia on the basis of detailed neuropsychological tests; major psychiatric disorders; follow-up < 2 years; FOG at the initial visit. This study was approved by the Yonsei University Severance Hospital institutional review board, and the need for informed consent was waived due to the retrospective nature of the study.
Quantitative analyses of 18F-FP-CIT PET
Image acquisition and quantitative analyses of 18F-FP-CIT PET data were performed according to the methods reported in our previous study [18]. DAT activity in the posterior putamen (DAT-PP) was defined as (mean standardized uptake value of the posterior putamen volume of interests [VOI] –mean standardized uptake value of the occipital VOI)/(mean standardized uptake value of the occipital VOI). Because DAT-PP is known to be closely associated with the severity of parkinsonian motor symptoms, DAT activity in this region was included in the data analysis.
Assessment of NPS
We assessed the NPS of patients by interviewing their caregivers using the Korean version of the neuropsychiatric inventory (NPI) [19]. The NPI evaluated 12 behavioral and psychological symptoms on the basis of observations within the past month: delusions, hallucinations, agitation/aggression, depression/dysphoria, anxiety, elation/euphoria, apathy/indifference, disinhibition, irritability/lability, aberrant motor behavior, sleep and nighttime behavior disturbances, and appetite and eating changes. Each symptom was rated retrospectively based on the patients’ condition. The severity of each manifestation was classified into grades (ranging from 0, absent, to 4, frequent). The NPI score of individual NPS was calculated by multiplying the severity by the frequency on a scale ranging from 0 to 12. Higher scores indicate greater psychopathology. The NPI total score was calculated by the sum of the individual NPI score. To perform sensitivity analysis, we dichotomized patients with PD using cut-off score of 4: NPI (+) group (NPI total score≥4; n = 98) and NPI (–) group (NPI total score < 4; n = 231) [20, 21]. We also grouped neuropsychiatric symptoms into three factors: Psychosis (delusions and hallucinations), Hyperactivity (agitation/aggression, elation/euphoria, disinhibition, irritability/lability, and aberrant motor disturbance), and Mood (depression/dysphoria, apathy, anxiety, sleep and night-time behavior change, appetite and eating change) [22].
Assessment of the development of LID, wearing-off and FOG
The participants visited our outpatient clinic every 3–6 months, and two movement disorder experts (Y.H.S. and P.H.L.) carefully assessed the presence of LID, wearing-off and FOG through a history obtained from patients and caregivers or by direct neurological examination at every visit [14]. The date on which the patients with PD or their caregivers reported that LID occurred or the date on which LID was first seen in the clinic was regarded as the date of occurrence of LID. The onset of wearing-off was defined as the time point when patients first complained of end-of-dose deterioration or when we first decided to increase the dosing schedule of levodopa from 3 times to 4 times a day. Nocturnal or early morning off symptoms were not regarded as a proof of wearing-off. FOG was defined as an unintentional and temporary phenomenon where the feet fail to progress forward, despite the intention to walk, and various subtypes were identified: hesitation at the initiation of walking, freezing on turning, freezing in restricted areas, destination freezing, and open-space hesitation in the absence of stimuli likely to result in FOG [23]. Two movement disorder specialists inspected the patients’ gait at the outpatient clinic and specifically asked the patients about the characteristic sensation of the feet becoming “glued to the floor” at every visit.
Longitudinal changes in the doses of dopaminergic medication
All participants visited the outpatient clinic for at least 2 years after their first visit, and the doses of their medications were adjusted to control the motor symptoms effectively by P.H.L. and Y.H.S., according to the patients’ responses. The patients visited the outpatient clinic with 3 to 6-month intervals between visits. We calculated the levodopa-equivalent daily dose (LEDD) of PD medication based on a previously reported method [24].
Statistical analysis
To compare the baseline demographic characteristics between the groups, Mann Whitney U tests were used for continuous variables, and Pearson’s χ2 tests were used for categorical variables. Cox regression models were used to estimate the hazard ratios (HRs) and 95%confidence intervals (CIs) for the development of LID, wearing-off, or FOG according to the NPI total score, while adjusting for age at symptom onset, sex, disease duration, UPDRS-III score, baseline MMSE score, DAT-PP and LEDD (i.e., LEDD at the time of LID, wearing-off, FOG onset, or at the last visit to the outpatient clinic in patients without LID, wearing-off, or FOG). Regarding FOG, the assumption of proportionality was rejected because the time-dependent covariate analysis results were statistically significant (p = 0.008). Thus, we used a time-dependent Cox regression model to assess the effects of NPS on the development of FOG based on a 5-year time point [23] (i.e., follow-up period within 5 years vs. after 5 years) after adjusting for age at symptom onset, sex, disease duration, UPDRS-III score, baseline MMSE score, DAT-PP and LEDD at FOG onset in patients with FOG or at the last visit to the outpatient clinic in those without FOG. A sensitivity analysis was performed using groupwise comparison. The time from the diagnosis of PD to the onset of LID, wearing-off, or FOG was assessed with Kaplan-Meier estimates. Log-rank and Breslow tests were used to compare the Kaplan-Meier plots between the groups. In terms of FOG, we further assessed whether the PD groups according to the presence of NPS affect the development of FOG using Cox regression and time-dependent Cox regression models. In addition, the relationships between the NPI total score and time to the development of FOG were assessed using Pearson correlation and partial correlation analyses for the patients with PD who eventually developed FOG during the follow-up period after adjusting for age at symptom onset, sex, disease duration, UPDRS-III score, baseline MMSE score, and DAT-PP. A linear mixed model was used to compare the rates of the longitudinal LEDD changes according to the NPI total score. A linear mixed model was used to compare the longitudinal changes in LEDD. We regarded time as a continuous variable, up to 60 months when > 40%of the patients (n = 158) were followed up. Participants were added as random effects and age at symptom onset, sex, disease duration, UPDRS-III score, baseline MMSE score, and DAT-PP as fixed effect terms. The effect of the NPI total score on longitudinal changes in LEDD was tested using an NPI×time interaction term. For the sensitivity analysis, we further assessed a group [NPI (+) vs. NPI (–)]×time interaction term while treating time as a categorical variable with a 6-month interval.
Statistical analyses were performed using SPSS (version 25.0; IBM Corporation, Armonk, NY, USA) and R (v4.0, http://www.r-project.org). Results with a two-tailed p value < 0.05 were considered statistically significant.
Data availability
The de-identified data that support the findings of this study are available from the authors upon reasonable request.
RESULTS
Demographic and clinical characteristics
The demographic and clinical characteristics of the enrolled patients are shown in Table 1. The median age at symptom onset was 67 (interquartile range [IQR], 61–73) years and 51.7%of the patients were females. The median disease duration was 12 (IQR, 5–19) months, UPDRS-III score at the time of PD diagnosis was 21 (IQR, 15–17), and the median years of education was 9 (IQR, 6–12) years. The median MMSE score at baseline was 27 (IQR, 26–29) and the median NPI total score was 1 (IQR, 0–5). Groupwise comparison of the demographic and clinical characteristics are shown in Supplementary Table 1. The NPI (+) group was significantly older than the NPI (–) group. As expected, the baseline MMSE was significantly lower in the NPI (+) group than in the NPI (–) groups. The frequency of individual NPS is shown in Supplementary Figure 1A. Among 195 PD patients with one or more individual NPS, 104 (53.3%) patients had two or more NPS simultaneously. Additionally, when illustrating a Venn diagram of 195 patients with one or more individual NPS according to three factors, 45 (23.1%) patients had two or more factors of NPS (Supplementary Figure 1B).
Demographic characteristics
Values are expressed as median (interquartile range) or number (percentage). DAT, dopamine transporter MMSE, Mini-Mental Status Examination; NPI, neuropsychiatric inventory; PD, Parkinson’s disease; UPDRS-III, Unified Parkinson’s Disease Rating Scale Part III.
The effect of NPS on the development of LID and wearing-off
During the follow-up period (4.84±1.77 years), LID and wearing-off developed in 55 (16.7%) and 62 (18.8%) patients with PD, respectively. The Cox regression model showed that the NPI total score did not significantly affect the development of LID (HR, 0.961; 95%CI, 0.915–1.008; p = 0.102) and wearing-off (HR, 0.990; 95%CI, 0.956–1.026; p = 0.582; Table 2) after adjusting for the effects of potential confounders, including age at symptom onset, sex, disease duration, UPDRS-III score, baseline MMSE score, DAT-PP, and LEDD. Sensitivity analysis showed that LID and wearing off-free survival (Ploghboxrank = 0.261 and 0.430) did not differ between the NPI (+) and NPI (–) groups (Fig. 1).
Multivariate time-dependent Cox Hazard models for prediction of the development of LID, wearing-off, and FOG
Results of Cox regression and time-dependent Cox regression analyses for the development of LID, wearing-off, and FOG after controlling for age at symptom onset, sex, UPDRS-III, baseline MMSE score, DAT activity in the posterior putamen, and LEDD. DAT, dopamine transporter; FOG, freezing of gait; LEDD, levodopa-equivalent daily dose; LID, levodopa induced dyskinesia; MMSE, Mini-Mental Status Examination; NPI, neuropsychiatric inventory; UPDRS-III, Unified Parkinson’s Disease Rating Scale Part III.
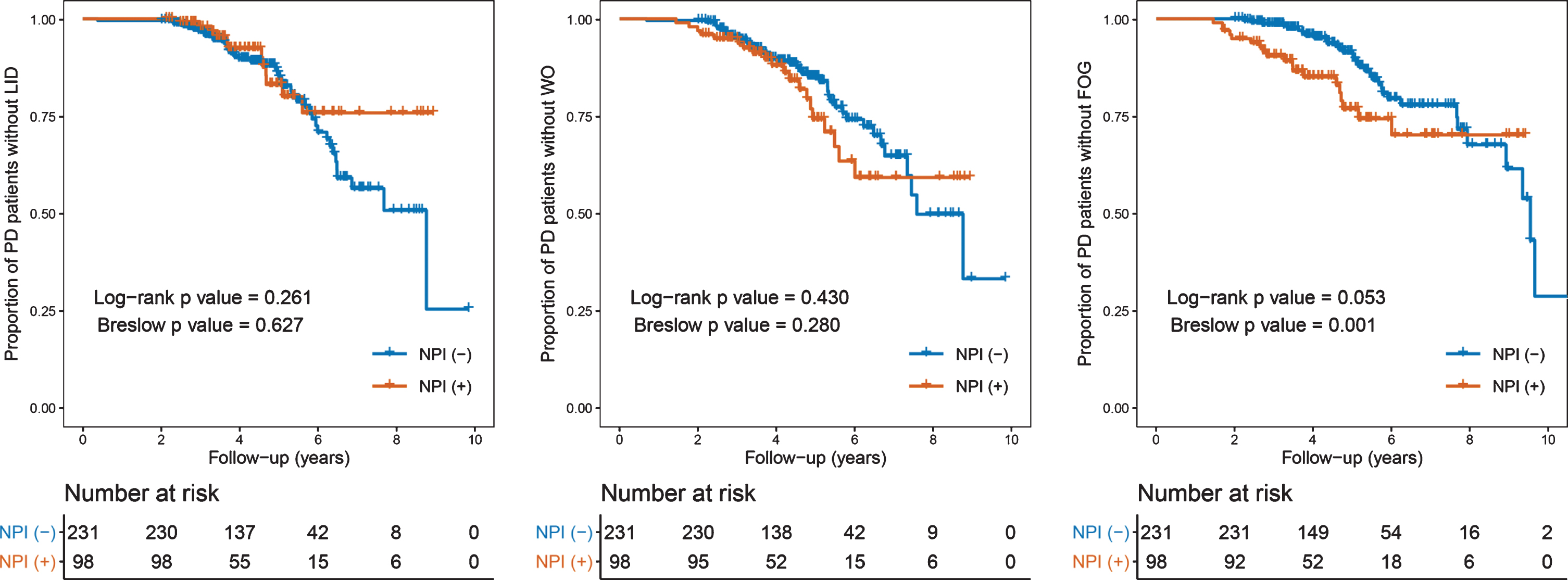
Curves of Kaplan-Meier estimates of the onset of LID, wearing-off, and FOG after the diagnosis of PD according to the PD groups. The crosses in the graphs indicate censored data. FOG, freezing of gait; LID, levodopa-induced dyskinesia; NPI, Neuropsychiatric Inventory; PD, Parkinson’s disease; WO, wearing-off.
The effect of NPS on FOG development
During the follow-up period, 52 patients (15.8%) developed FOG among all the patients with PD. The Cox regression model showed that the NPI total score did not significantly affect the development of FOG (p = 0.100) after adjusting for the effects of potential confounders. The time-dependent Cox regression model based on the 5-year time point revealed that the NPI total score was a significant predictor of the development of FOG within 5 years (HR, 1.047; 95%CI, 1.018–1.078; p = 0.002; Table 2) after adjusting for confounding factors. In sensitivity analysis, the Kaplan-Meier curves showed that the difference in FOG-free survival between the NPI (+) and NPI (–) groups appeared prominent during earlier years (PBreslow = 0.001) but waned over time (Ploghboxrank = 0.053, Fig. 1). Additionally, the Cox regression model showed that the risk of FOG development during the follow-up period was significantly higher in the NPI (+) group than in the NPI (–) group (HR, 2.222; 95%CI, 1.201–4.113; p = 0.011; Supplementary Table 2). The risk of FOG development within 5 years was more prominent in the NPI (+) group than in the NPI (–) group (HR, 3.885; 95%CI, 1.856–8.130; p < 0.001; Supplementary Table 2). Further evaluation after grouping NPI into three factors showed that both hyperactivity and mood/apathy were associated with the early development of FOG; however, the models using total NPI score as an independent variable had higher C-index and lower AIC values than those using hyperactivity or mood/apathy, indicating that total NPI score was more explanatory for the early development of FOG than an individual cluster of NPS (Supplementary Table 3). Psychosis was not into analysis due to its low prevalence.
Relationship between the NPI total score and time with the development of LID, wearing-off and FOG
Among 55 patients with PD who eventually developed LID and 62 patients with PD who developed wearing-off during the follow-up period, a correlation analysis revealed that the NPI total score was not significantly associated with the time taken for the development of LID (γ= –0.148, p = 0.281) and wearing-off (γ= –0.228, p = 0.075). Among 52 patients with PD who eventually developed FOG during the follow-up period, correlation analysis revealed that there was a significant association between the NPI total score and time taken for the development of FOG (γ= –0.455; p = 0.001), and the relationship remained relevant even after adjusting for confounding factors (γ= –0.472; p = 0.001; Table 3 and Fig. 2).
Correlation analysis between the NPI total score and time to the development of motor complication
Values are expressed as the Pearson correlation coefficient. Partial correlation analysis was performed after adjusting for age of symptom onset, sex, disease duration, UPDRS-III score, baseline MMSE score, and DAT activity in the posterior putamen. DAT, dopamine transporter; FOG, freezing of gait; LID, levodopa induced dyskinesia; MMSE, Mini-Mental Status Examination; NPI, neuropsychiatric inventory; UPDRS-III, Unified Parkinson’s Disease Rating Scale Part III.
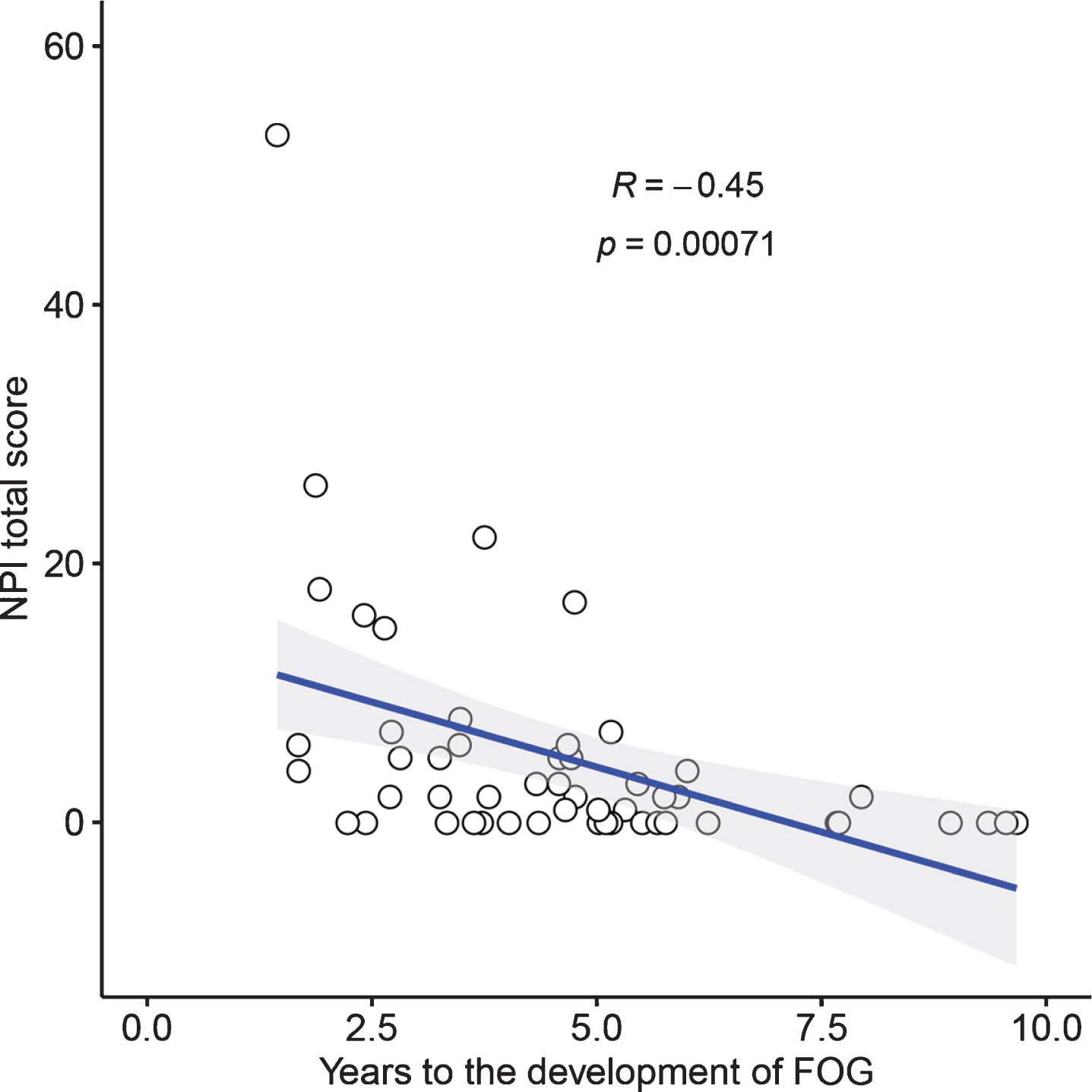
Scatter plot of NPI total score and time taken for FOG development. FOG, freezing of gait; NPI, Neuropsychiatric Inventory.
Longitudinal assessment of the changes in LEDDs
All enrolled patients with PD were treated with dopaminergic medications for at least 2 years. The NPI×time interaction term in the linear mixed model was statistically significant (p = 0.003) after adjusting for age at symptom onset, sex, disease duration, UPDRS-III score, baseline MMSE score, DAT-PP, NPI total score, time, and NPI×time, indicating that patients with high NPI total score had a rapidly increased LEDD (Table 4). Both hyperactivity and mood/apathy were associated with longitudinal LED increase, however the models using total NPI score as an independent variable had lower AIC values than those using hyperactivity or mood/apathy, indicating that total NPI score was more explanatory for the longitudinal LED increase than an individual cluster of NPS (Supplementary Table 3). Sensitivity analysis showed that the NPI (+) group required higher doses of dopaminergic medications compared to the NPI (–) groups after adjusting for age at symptom onset, sex, disease duration, UPDRS III score, baseline MMSE, DAT-PP, group, time, and group×time (Fig. 3 and Supplementary Table 4).
Longitudinal changes of levodopa-equivalent daily doses in patients with Parkinson’s disease according to neuropsychiatric burden
Results of linear mixed models for LED after controlling for age of symptom onset, sex, UPDRS-III, baseline MMSE score, DAT activity in the posterior putamen, NPI, time, and NPI×time. DAT, dopamine transporter; LEDD, levodopa-equivalent daily dose; MMSE, Mini-Mental State Examination; NPI, neuropsychiatric inventory; PD, Parkinson’s disease; UPDRS, UPDRS-III, Unified Parkinson’s Disease Rating Scale Part III.
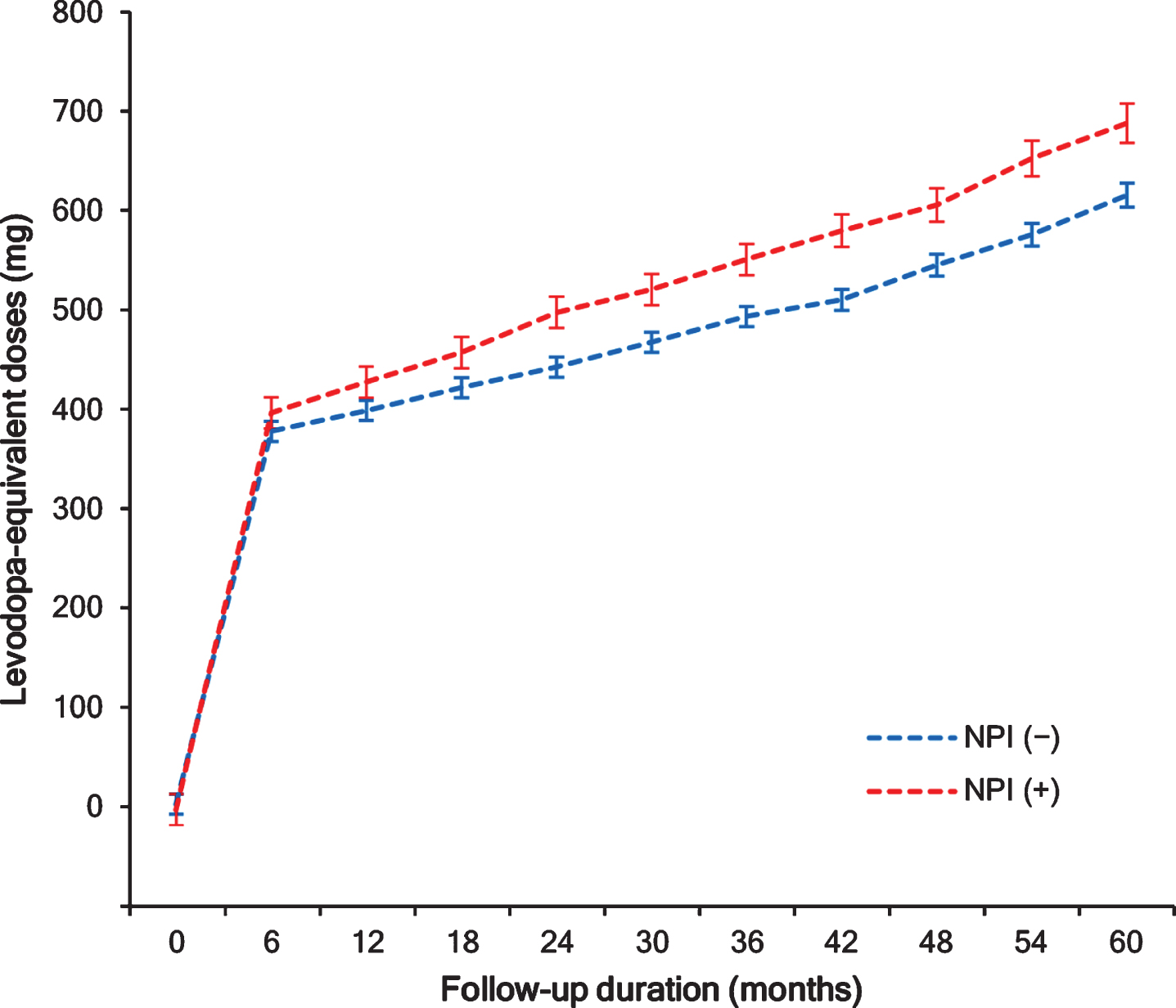
Longitudinal increases in levodopa-equivalent daily doses. A linear mixed model analysis showed that the NPI (+) group required higher doses of dopaminergic medications compared to the NPI (–) groups. NPI, Neuropsychiatric Inventory.
DISCUSSION
In this study, we investigated the effects of NPSs on the longitudinal motor prognosis in patients with PD. The major findings were as follows. First, a higher burden of NPS was independently associated with the risk and timing of the development of FOG. Second, a higher burden of NPS required a rapid longitudinal increase in LEDD. Third, the NPS burden was not associated with the development of wearing-off and LID. Our results suggest that the burden of NPS at baseline would have a significant impact on the longitudinal motor prognosis in patients with PD.
The present study demonstrated that the NPI total score at the time of PD diagnosis was an independent predictor of FOG development in the future. Sensitivity analysis using a cut-off score 4 of the NPI total score also showed that the PD group with a higher burden of NPS had an increased risk of FOG development. Moreover, we confirmed this relationship using correlation analysis among patients with PD who eventually developed FOG, which showed significant correlation between the NPI total score and time taken for FOG development. Previously, several studies have reported that individual NPS such as anxiety or depression is an important factor for the development of FOG in patients with PD [25, 26], even though there are some studies with contrasting results [27, 28]. In this study, considering that individual patients with PD would simultaneously experience various NPS such as apathy, abnormal perception, anxiety, and depression [29], we used the NPI sum, where a higher score indicates greater psychopathology, to assess individual NPS comprehensively. Indeed, among PD patients with one or more individual NPS, more than half of the patients simultaneously had two or more individual NPS in this cohort, which suggests that individual NPS overlaps with each other. In terms of the pathogenesis of FOG, ample evidence suggests that FOG is a complex network disorder comprised of basal ganglia dysfunction derived from the loss of nigrostriatal dopaminergic neurons and dysfunction in basal ganglia-cortical pathways encompassing motor, cognitive, and limbic functions [30, 31]. Of those, several studies have demonstrated that structural and functional changes in the limbic system are closely involved in the development of FOG. Gilat et al. showed that patients with PD who experienced FOG had altered functional connectivity between the amygdala, putamen, and frontoparietal network, and the connectivity networks were significantly associated with FOG severity and a subjective fear of falling [32]. Additionally, some studies revealed that panic attacks and physiologic increases in the heart rate were closely linked to the onset of FOG episodes, which implies that neurobehavioral systems, such as the limbic system may play an important role in the development of FOG [33, 34]. Thus, considering that microstructural alterations in the limbic network already begin in apathic and depressive patients with de novo PD [35], it is plausible that the baseline burden of NPS may act as an independent predictor for the risk and timing of FOG development through the mediation of limbic circuits.
In contrast to FOG, there was no difference in the development of LID and wearing-off according to the NPI total score. The underlying mechanism leading to FOG, LID, or wearing-off is complex and are likely distinct from each other. Several lines of evidence have suggested that neurodegeneration across the locomotor system, including the cortico-cortical, motor-related cortico-fugal, and cortico-striatal tracts as well as the cerebellar and pedunculopontine nuclei, is related to FOG [36], whereas the role of presynaptic nigrostriatal dopamine deficits in the development of FOG remains uncertain [14]. Nigrostriatal dopaminergic degeneration is the most important predisposing factor in LID and wearing-off [14], and striato-cortical sensorimotor pathways appear to be more closely linked to the development of these motor complications rather than the limbic corticostriatal pathway [37, 38]. In a similar manner, LID and wearing-off are generally dopamine-related or -responsive symptoms, whereas the dopamine responsiveness of FOG and NPS is somewhat heterogeneous [39]. In this regard, distinct possible mechanisms responsible for LID, wearing-off, and FOG may lead to discordant associations between the burden of NPS and individual motor complications.
Interestingly, we found that a higher NPI total score or the NPI (+) group was associated with a more rapid increase in longitudinal LEDD, even after adjusting for disease duration, UPDRS-III score, and DAT-PP. These results suggest that motor progression assessed by the requirement of dopaminergic medications would be affected by the burden of NPS at baseline in patients with PD. So far, only a few studies have reported the association between individual NPS and parkinsonian motor deficits. Ravina et al. demonstrated that patients with PD who had comorbid depression required increased doses of symptomatic therapy. We also reported that depression was associated with a rapid increase in LEDD in patients with de novo PD. Additionally, the presence of apathy is significantly associated with more severe motor symptoms [40, 41]. In further analyses using each cluster of NPS factors, models using the NPI total score was superior to models using each cluster of NPS factors in terms of predicting the longitudinal LEDD increase as well as the early development of FOG. Taken together, our data provide evidence that the baseline burden of total NPS, encompassing individual NPS comprehensively, would be a predictor of motor prognosis in PD. Although the pathomechanism explaining the relationship between NPS burden and motor progression is uncertain, a neural reserve operating in patients with PD may mediate this association. We previously reported that patients with PD who had MBI were associated with significantly higher UPDRS-III scores, even at similar DAT-PP than those without MBI, which suggests that the presence of MBI may indicate a small neural reserve in PD [42]. Moreover, we identified the motor reserve network composed of the basal ganglia, inferior frontal cortex, insula, and cerebellar vermis in patients with early-stage PD, and we demonstrated that the motor reserve network strength are associated with the individual’s capacity to cope with PD-related pathologies [43]. Some studies have revealed that there are structural or functional alterations in the inferior frontal and insular cortices in patients with PD who exhibited apathy and depressive symptoms [44, 45], which implies that neural networks of individual NPS may overlap with those of the motor reserve. In addition, psychosocial factors such as psychological stress [46] and social isolation [47] may also affect motor prognosis in patients with PD. In the future, clinical and imaging studies using sophisticated statistical approaches are needed to unravel the pathomechanisms underlying the relationship between the burden of NPS and motor progression.
Our study has some limitations. First, we used the NPI total score as an indicator of the burden of NPS. The NPI only assesses individual NPS over the past month, and a recently developed and validated instrument to assess neuropsychiatric burden, the MBI Checklist [10], may be more accurate than NPI, as the MBI Checklist requires a history of 6 months of new-onset NPS. Additionally, due to the retrospective nature of this study, information on longitudinal changes in NPS was lacking. Association between changes in NPS over time and motor outcomes in patients with PD should be assessed in future studies. Second, we defined LID, wearing-off, or FOG when clinicians detected signs or symptoms of these, which may limit the generalization of the results in terms of quantitative and objective methodology. Finally, even though a consensus for clinically meaningful endpoints has not been established, LEDD and its longitudinal changes to control parkinsonian motor symptoms may indirectly reflect the severity of PD and disease progression [48]. However, since global disability in patients with PD is complexly influenced by motor and non-motor symptoms [49], an increase in LEDD may not accurately reflect the progression of PD.
In conclusion, the baseline burden of NPS in drug-naïve patients with PD is closely related to the early development of FOG and rapid motor deterioration. These findings suggest that assessing the total burden of NPS is crucial for predicting motor outcomes in patients with PD. Future studies are needed to clarify whether the early intervention of NPS improves motor outcome in patients with PD.
Footnotes
ACKNOWLEDGMENTS
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (NRF-2019R1A2C2085462)
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
