Abstract
Background:
Previous studies described a parkinsonian personality characterized as rigid, introverted, and cautious; however, little is known about personality traits in
Objective:
To investigate personality in
Methods:
Using Cloninger’s biosocial model, we assessed personality in 193
Results:
PD patients showed low novelty seeking, high harm avoidance, and normal reward dependence and persistence scores. Harm avoidance was positively correlated with the severity of depression, anxiety, and apathy (rs = [0.435, 0.676],
Conclusion:
Personality in early PD patients allows us to recognize 3 patients’ phenotypes. Identification of such subgroups may help to better understand their natural history. Their longitudinal follow-up will allow confirming whether some personality features might influence disease evolution and treatment.
INTRODUCTION
Parkinson’s disease (PD) is a heterogeneous neurodegenerative disorder with a wider variability of motor and non-motor manifestations than previously thought [1–3]. Neuropsychiatric symptoms are common non-motor symptoms [4], including apathy, anxiety, and depression, and impulse control disorders as opposite ends of a hypo- and hyperdopaminergic behavioral spectrum [5]. Hypodopaminergic symptoms are related to the degeneration of ascending monoaminergic neurotransmitter systems, while the hyperdopaminergic symptoms are attributed to pulsatile dopaminergic stimulation and sensitization induced by antiparkinsonian drugs [5].
Since the beginning of the 20th century, several studies had described a parkinsonian personality, typically characterized as rigid, introverted, overcontrolled, cautious, conservative, punctual, serious, stoic, quiet, and industrious [6–12]. Most previous studies have assessed personality based on Cloninger’s biosocial model [6–11]. According to this model, personality is dependent on two main traits: temperaments and characters [13]. Temperaments (novelty seeking, harm avoidance, reward dependence, and persistence) are intended to be relatively stable personality dimensions related to genetics and neurotransmitters [13]. Individuals with high novelty seeking personality are often impulsive, extravagant, and irritable; those with high harm avoidance are pessimistic, fearful, shy, and fatigable; individuals with high reward dependence are sentimental, open, warm, and appreciative; and those with high persistence are often industrious, determined, ambitious, and perfectionistic [13]. Modulation of the four dimensions has been hypothesized to be related to dopamine, serotonin, noradrenaline, and glutamate, respectively [13].
PD patients were found to have less novelty seeking and more harm avoidance than control populations [6–11]. Applying another model of personality, the five-factor model, PD patients showed a higher level of neuroticism and lower levels of openness and extraversion [6, 12].
However, most studies have small sample sizes with heterogeneous disease duration and included mainly patients under dopaminergic medication [6–8, 10–12]. Thus, these studies do not reflect well the personality at the early stages of the disease. The possibility of studying patients with untreated PD allows us to rule out the potential confounding effect of dopaminergic treatment on neuropsychiatric symptoms. Likewise, little is known about the relationship between clinical and neuropsychiatric symptoms and personality traits in PD patients.
We intend to investigate personality in
METHODS
Participants
Patients were recruited from a multicenter, observational study on non-motor neuropsychiatric symptoms in
Assessment
Demographic data (age and sex), age at PD onset, disease duration, first motor sign (akinetic-rigid versus tremor), and side of symptoms onset (right versus left) were collected.
Personality traits were assessed using the Temperament and Character Inventory-Revised (TCI-R) questionnaire [14]. It has 240 questions rated from one (definitively false) to five (definitively true) and measures the four dimensions of temperament, based on Cloninger’s biosocial model. Each dimension is divided into 4 sub-dimensions. The final score in percentage ranges from zero to 100%. The higher is the score, the higher the patient presents the associated personality dimension, with 50% being the midpoint of the ranging spectrum. Personality is defined by the interaction of these four dimensions and combinations of extremes (high or low) dimensions correspond to personality disorders.
Patients were assessed with a detailed neurological and neuropsychological assessment including: the MDS-UPDRS [15] for motor and non-motor symptoms severity (MDS-UPDRS-I for Non-Motor Aspects of Experiences of Daily Living, range 0 to 52, MDS-UPDRS-IB for Part IB covering the non-behavioral non-motor symptoms such as sleep, pain, urinary and constipation problems, light-headedness and fatigue, range 0 to 28, MDS-UPDRS-II for Motor Aspects of Experiences of Daily Living, range 0 to 52, and MDS-UPDRS-III for motor examination, range 0 to 132), the Starkstein apathy scale [16] for apathy (range 0 to 42), the Beck depression inventory-2 (BDI-2) [17] for depression (range 0 to 63), the State-Trait Anxiety Inventory for anxiety trait (STAI-trait) and state (STAI-state) [18], each range 20 to 80, the Ardouin Scale of Behavior in Parkinson’s Disease (ASBPD) [19] for apathy, anxiety, depression (each item range from 0 to 4 : 4 = severe disorder; 3 = marked disorder; 2 = moderate disorder; 1 = mild disorder; 0 = absence of disorder) and hyperdopaminergic behaviors (calculated as the sum of single items (each range from 0 to 4) of hyperdopaminergic behaviors, e.g., hypomania, and impulse control disorders, range 0 to 52), the Impulsive Behavior Scale [20] (UPPS, range 0 to 180), the MATTIS Dementia rating scale [21] (range 0 to 144) and the Frontal Assessment Battery [22] (FAB, range 0 to 18) for cognition and the Neuropathic Pain Symptom Inventory [23] (NPSI, range 0 to 100) for pain assessment. Quality of life was evaluated by PDQ-39 [24] (the summary index (PDQ-39 SI) was calculated by the sum of dimension total scores divided by 8).
We defined the motor subtype by calculating a “tremor score” and a “non-tremor score” for each patient according to Lewis et al. [25]. The tremor score derived from the sum of MDS-UPDRS items 3.15 to 3.18 divided by 10. The non-tremor score derived from the sum of MDS-UPDRS items 3.1 to 3.3, 3.9, 3.10, and 3.12 to 3.14 divided by 12. The motor subtype was classified as tremor-dominant or akinetic-rigid type, when the tremor score was at least twice the non-tremor score or the inverse, respectively. The remaining patients were classified as mixed type.
Statistical analysis
All data analyses were performed using the JASP Statistics software. Descriptive data are expressed as mean (standard deviation) for continuous variables and as absolute numbers (percentage) for categorical variables. The normality of continuous variables was assessed with the Shapiro–Wilk test. Variables that failed the Shapiro-Wilk test were analyzed with nonparametric statistics. Correlational analyses between each personality dimension and scores on the motor and non-motor scales were carried out through a bivariate Pearson’s correlation analysis (r) if normality were verified; otherwise, a Spearman’s rank correlation analysis (rs). Secondly, to explore how different personality profiles and clinical variables clustered in early PD patients, we performed cluster analyses. A non-hierarchical (K-means) clustering analysis with z-score standardization was performed using as variables personality dimensions, motor- and non-motor data. MDS-UPDRS-IB, MDS-UPDRS-II, MDS-UPDRS-III, ASBPD score for depression, anxiety, and apathy, UPSS, and NPSI were introduced in the clustering analysis together with the four TCI-R dimensions. To avoid redundancies, we decided to exclude BDI-2, STAI, and Starkstein apathy scale given the high correlation coefficient between these scores and ASBPD depression, anxiety, and apathy scores (rs > 0.500). Since the small number of patients with an elevated hyperdopaminergic ASBPD score (9/193), this score was also not included in the clustering analysis, considering it would not contribute significantly to the differentiation of clusters. Determination of the optimal number of clusters was based on the Calinski-Harabasz pseudo-F value and Silhouette Score in successive steps of division in two, three, or four clusters. Then, for each variable included in cluster analysis as well as other clinical and demographic variables (age, sex, quality of life) we used parametric (one-way ANOVAs with Bonferroni
RESULTS
Of 198 de novo PD patients included in the study, full data were available for 193 patients for the analysis: 119 men (62%), mean age of 58.4 (8.8) years, mean age at PD onset 56.8 (9.1) years, 97 (50%) with an akinetic-rigid motor onset, mean OFF MDS-UPDRS-III 24.9 (9.7). None of the subjects had received levodopa or dopamine agonist medication but fifty (26%) were taking rasagiline. Table 1 shows the clinical characteristics of the studied group.
Demographic, motor, personality, and neuropsychological characteristics (
Values are mean (SD) or n.
Personality assessment (Table 1 and Supplementary Figure 1) revealed: a higher level of fear of uncertainty (62%), low mean novelty seeking (43%), and high mean reward dependence and persistence (59% and 59%, respectively). While only 31 patients had a novelty seeking score superior to 50%, 90, 161, and 147 patients had a harm avoidance, reward dependence, and persistence score superior to 50%, respectively. Figure 1 shows the descriptive data of personality assessment of our cohort and data from the French and Belgian normative population with 78 healthy French subjects (mean age (32.5±13.3)) [14] and 958 French-speaking participants of Belgium (546 women with a mean age of 33.6 (15.1) years) and 412 men with a mean age of 36.2 (14.9) years) [26]. Our PD patients showed low novelty seeking (42.6% vs. 48.4% and 46.6%), high harm avoidance (50.0% vs. 44.2% and 46.2%), and similar reward dependence (58.9% vs. 59.6% and 59.7%) and persistence (59.4% vs. 60.4% and 59.1%) than the French and Belgian normative population, respectively (no inferential statistics analysis was performed).
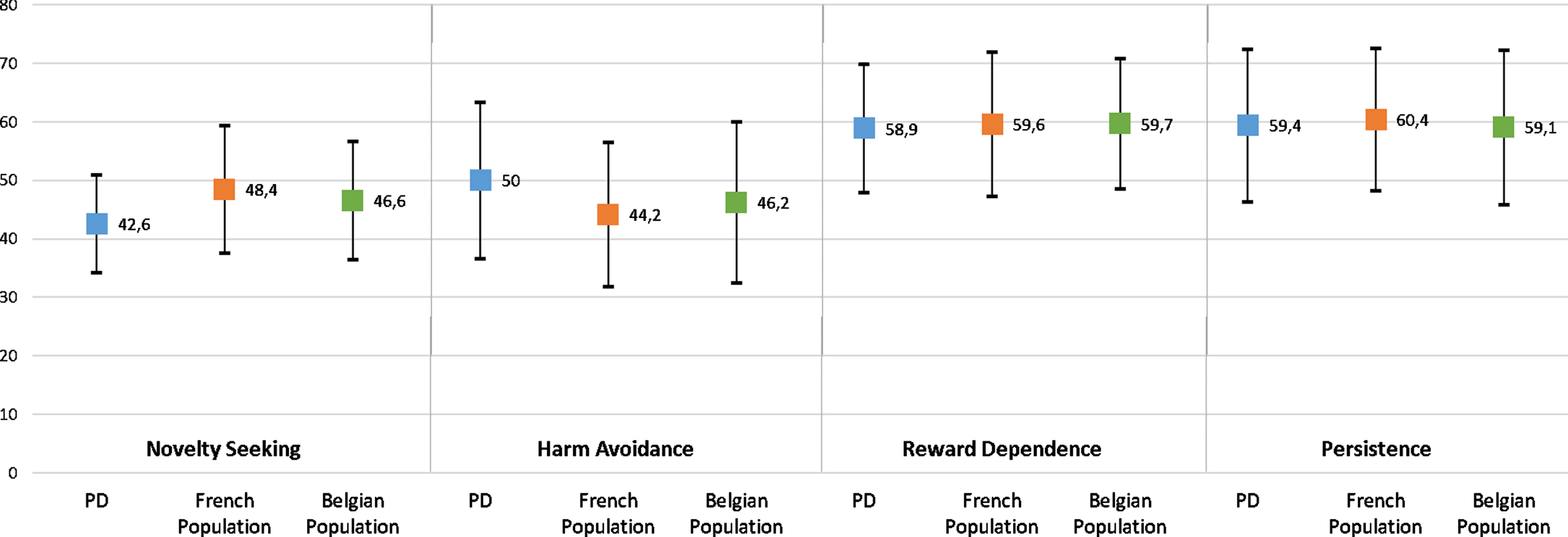
Personality traits of PD patients and healthy controls using TCI-R. Novelty seeking, harm avoidance, reward dependence, and persistence mean score (and SD) of de novo PD patients compared with results from validation studies of the TCI-R with healthy controls in French [14] and Belgian populations [26]. Our PD patients showed low novelty seeking (42.6% (8.4)) vs. 48.4% (10.9) and 46.6% (10.1)), high harm avoidance (50.0% (13.4)) vs. 44.2% (12.3) and 46.2% (13.8)), and similar reward dependence (58.9% (11.0)) vs. 59.6% (12.4) and 59.7% (11.2)) and persistence (59.4% (13.1)) vs. 60.4% (12.2) and 59.1% (13.2)) than the French and Belgian normative population, respectively (no inferential statistics analysis was performed).
At the time of evaluation forty-eight patients (25%) were significantly apathetic, 34 patients (18%) were significantly depressive and 50 patients (26%) were significantly anxious (ASBPD score≥2). Twenty patients had a past diagnosis of anxiety and fifty-four of depression (only 15 of them were still with depressive symptoms at the study evaluation). Only nine patients (4.7%) showed hyperdopaminergic behaviors (ASBPD score≥2 on one of the items of hyperdopaminergic behaviors). Of these nine patients, five had eating behavior, one creativity, one hobbyism, one risk-taking behavior, and one compulsive shopping.
Correlation analysis results are shown in Table 2. Harm avoidance was significantly positively correlated with the severity of depression, anxiety, and apathy (rs between 0.435 and 0.676) and with the MDS-UPDRS part I, IB, and II (rs between 0.349 and 0.508) and negatively correlated with quality of life (rs = –0.492). Additionally, a weak positive correlation was found between harm avoidance and women (rs = 0.257). Novelty seeking, reward dependence, and persistence were negatively correlated with apathy (rs between –0.274 and –0.375). A significant positive correlation was found between novelty seeking and UPSS (rs = 0.239).
Correlations between TCI-R dimensions and clinical features of PD patients
NS, novelty seeking; HA, harm avoidance; RD, reward dependence; P, persistence. Spearman’s rank correlation analysis (rs-values). Following Bonferroni correction for 80 multiple tests,
Clustering analysis divided our PD patients into three main clusters (Fig. 2 and Table 3). The clustering optimum was attained for the 3 clusters solution (Calinski-Harabasz pseudo-F = 37.165 and Silhouette score = 0.180). Cluster 1 included 51 patients with higher harm avoidance (63%) and lower novelty seeking (41%), cluster 2 included 34 patients with low novelty seeking (37%) but a median level of harm avoidance (53%), and cluster 3 included 108 patients with a lower harm avoidance (43%) and higher novelty seeking (45%), reward dependence (62%), and persistence (64%). Cluster 1 showed a higher severity of depression, anxiety, apathy, and impulsivity (ASBPD and UPSS) and an intermediate severity of motor symptoms (MDS-UPDRS-II and MDS-UPDRS-III). Cluster 2 had a higher severity in motor symptoms (MDS-UPDRS-II and MDS-UPDRS-III), with a higher severity in non-tremor items of MDS-UPDRS-III. Cluster 3 showed a lower severity of both motor and non-motor symptoms. Both clusters 1 and 2 had a higher MDS-UPDRS-IB score, covering other non-motor symptoms such as sleep, urinary, and constipation problems (
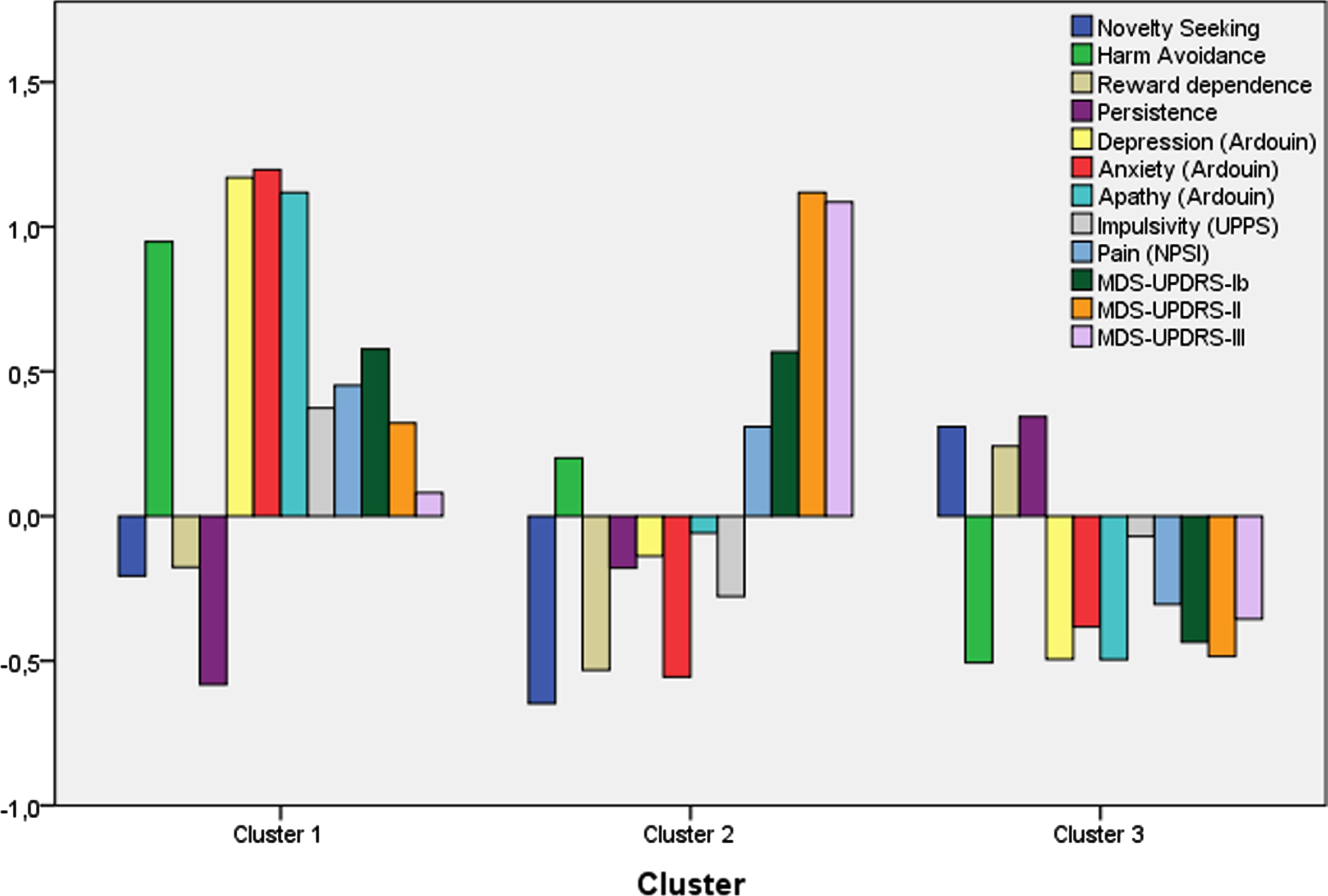
Clustering analysis of personality traits, motor- and non-motor symptoms. K-means clustering analysis included z-scores of 12 factors: personality dimensions (novelty seeking, harm avoidance, reward dependence and persistence), MDS-UPDRS part IB (non-behavioral non-motor symptoms), II and III, Ardouin score (ASBPD) for depression, anxiety and apathy, Impulsive Behavior Scale (UPPS) and Neuropathic Pain Symptom Inventory (NPSI). Since factors have different scales, cluster means for each factor are displayed as standardized scores relative to each overall mean. Three distinct clusters were found: cluster 1 (51 patients) with high harm avoidance, low novelty seeking, hypodopaminergic neuropsychiatric symptoms and an intermediate motor score severity; cluster 2 (34 patients) with low novelty seeking, and higher motor severity; and cluster 3 (108 patients) with low harm avoidance and high novelty seeking, reward dependence and persistence with lower symptoms severity.
Demographic and clinical features of early PD patients in the three clusters
MDS-UPDRS, MDS-sponsored Revision of the Unified Parkinson’s Disease Rating Scale; MDS-UPDRS-I, UPDRS part I, Non-Motor Aspects of Experiences of Daily Living; MDS-UPDRS-IB, UPDRS part I B, covering the non-behavioral non-motor symptoms such as sleep, pain, urinary and constipation problems, light-headedness and fatigue; UPDRS-II, UPDRS part II, Activities of Daily Living; UPDRS-III, UPDRS part III, Motor Examination; Ardouin Scale of Behavior in Parkinson’s Disease for depression, anxiety and apathy; UPPS, Urgency, Premeditation, Perseverance, Sensation Seeking, Impulsive Behavior Scale; NPSI, Neuropathic Pain Symptom Inventory. Tremor UPDRS III = sum of MDS-UPDRS items 3.15 to 3.18, non-tremor UPDRS III = sum of MDS-UPDRS items 3.1 to 3.3, 3.9, 3.10, and 3.12 to 3.14. Values are mean (SD) or n (%). aPearsons χ2 test; bFishers exact test; cKruskal-Wallis test with Dunn’s
TCI-R sub-dimensions were subsequently analyzed. Novelty seeking was significantly higher in cluster 3 compared with other clusters as did 2 of its 4 sub-dimensions: exploratory excitability (3 vs. 1, and 2,
DISCUSSION
This is the first study on personality in
Since previous studies of personality traits in PD patients are not entirely consistent, we speculated if, in fact, more than a particular parkinsonian personality, there are different personality traits that play a role in the presence of different clinical phenotypes.
Classification of patients according to personality traits, motor, and non-motor symptoms revealed the presence of 3 distinct subtypes of patients: i) group 1, with 26% of patients, had high harm avoidance and low novelty seeking traits and hypodopaminergic neuropsychiatric symptoms, impulsivity, and intermediate motor severity (= neuropsychiatric phenotype); ii) group 2, with 18% of patients, had a low novelty seeking trait and a higher motor severity, without neuropsychiatric symptoms but with similar severity of other non-motor symptoms as group 1 (= motor phenotype); iii) group 3, with 56% of patients, presented with lower harm avoidance, higher novelty seeking, reward dependence and persistence traits and lower motor severity and lower prevalence of non-motor symptoms (= benign phenotype). The 3 phenotypes did not differ in age, gender, or motor subtype. Although the motor subtype did not differ between the 3 phenotypes, the higher motor severity of the motor phenotype (group 2) was driven by a higher severity in non-tremor items of MDS-UPDRS-III, more than the tremor items score. While the neuropsychiatric phenotype (1) and motor phenotype (2) had a higher harm avoidance, characterized by excessive worrying, pessimism, shyness, doubtfulness, and easily fatigued, the benign phenotype (3) had a low harm avoidance, characterized by optimism, extrovert, confidence, and energy. Patients with the benign phenotype (high novelty seeking, reward dependence, and persistence) present with higher exploratory excitability and extravagance, openness to warm communication and attachment, and eagerness of effort and work-hardened.
Over the last decade, research has focused on non-motor symptoms and heterogeneity in PD [2–4, 30]. Disease manifestations and rates of progression vary greatly, and these differences are present since the disease onset [1–4, 30]. Increasing attempts are made to define different subtypes of PD patients, based on their baseline motor and non-motor characteristics, with different long-term prognoses [1–4, 29–32]. Non-motor symptoms have a key role in differentiating PD phenotypes: mood and apathy were associated with more severe motor symptoms [31] and RBD has been associated with poor prognosis [32]. More recently four different clusters of PD presentation were reported: 1) fast motor progression with symmetrical motor signs, poor olfaction, cognition, and postural hypotension; 2) mild motor and non-motor disease with intermediate progression rate; 3) severe motor disease, poor psychological well-being and poor sleep with intermediate motor progression; 4) slow motor progression, unilateral presentation, tremor-dominant [30]. However, none of these studies included personality in the classification of PD patients. Personality, as a constitutive characteristic of the subject, might help explain heterogeneity in PD. The follow-up of our cohort will help to understand whether our classification can help differentiate patient evolution and treatment response.
Cloninger’s defined novelty seeking as an individual’s tendency to action and exploratory behaviors, harm avoidance as a tendency to inhibit behaviors to avoid punishment, non-reward, and novelty, reward dependence as a tendency towards the maintenance of ongoing behaviors previously associated with reward and non-punishment and persistence as the ability to generate and maintain arousal and motivation internally, in the absence of immediate external reward [13]. Cloninger’s proposed a relationship between personality and monoaminergic neurotransmitter systems, linking the dopaminergic system to exploratory behaviors and response to novel stimuli (novelty seeking); serotoninergic system to inhibitory behaviors (harm avoidance); noradrenergic system to response to rewards and better maintenance (reward dependence) [13]. In PD patients, it has been claimed that the lower scores in novelty seeking are related to the dopaminergic deficit. The increased novelty seeking score after the introduction of dopamine agonist treatment shown in Bódi et al. [33] and higher novelty seeking scores in candidates for DBS treatment or advanced PD population [11] compared to controls and early untreated [9] PD patients reinforce this hypothesis. Later studies have shown that the association between different traits according to the Cloninger model and neurotransmitters is much less clear than originally stated [9, 34].
Cloninger’s model postulates harm avoidance and novelty seeking are also the most directly involved in susceptibility to mood disorders [26]. In our study, we found a prevalence of apathy, depression, and anxiety of 25%, 18%, and 26%, respectively, consistent with previous studies [4, 31]. These symptoms are commonly associated with each other. The dysfunction of the mesolimbic dopaminergic limbic circuit and mesolimbic serotonergic and noradrenergic circuits are implied in their pathophysiology [35]. In early-stage PD, serotonergic dysfunction has been shown to play a prominent role in the pathophysiology of apathy and mood symptoms [35]. Our results showed an association between personality and mood disorders. Harm avoidance was strongly positively correlated with the severity of depression, anxiety, and apathy in PD. Novelty seeking, reward dependence, and persistence were weakly negatively correlated with apathy, but not with depression or anxiety. Other smaller studies focusing on PD also revealed a strong association between high harm avoidance or increased neuroticism with depression and anxiety [36, 37]. Studies focusing on personality and apathy in PD are scarce [38]. Applying the TPQ, Pluck and Brown did not find significant differences in personality measures with high and low apathy scores [38]. The causal link between mood disorders and harm avoidance can be argued. Cloninger’s defended that personality dimensions are relatively stable through life and related to genetics and neurotransmitters. So, patients with higher harm avoidance can have a chronic maladaptive strategy that made them more susceptible to depression or anxiety. On the other hand, it is also possible that mood disorders lead to changes in personality and the co-occurrence of depression and anxiety and high harm avoidance scores may be a reflection of the intertwined nature of traits and states. Lhommée et al have previously demonstrated significant personality changes in PD patients after deep brain stimulation and a major reduction in antiparkinsonian treatment [10]. Harm avoidance increased, and extravagance (subdimension of novelty seeking) and persistence decreased after surgery, possibly related to the change from a preoperative hyperdopaminergic to a postoperative hypodopaminergic state [10].
Interestingly, the neuropsychiatric phenotype showed higher impulsivity scores, which is counterintuitive. The apathetic patient is not ready to make an effort, nor ready to wait for a reward and will need it right away, with a tendency to think and act rashly, which can be interpreted as “impulsive”. The distinct psychological process of dopamine-related motivation systems of ‘wanting’ a reward from the hedonically ‘liking’ the same reward could explain this paradox [39]. A disruption of goal-directed decision-making is proposed as another possible mechanism in a recent study showing that apathy and impulsivity correlated positively [40]. Hypodopaminergic behavioral symptoms and impulsivity might be predisposing factors for the development and severity of impulse control disorders, possibly induced by the dopaminergic sensitization in the mesolimbic circuit and probably related to a higher extent of the dopaminergic depletion in the ventral striatum [5]. The longitudinal follow-up of these patients will allow confirming whether some personality traits represent a risk factor to develop impulse control disorders.
Hyperdopaminergic ASBPD score was not included in the cluster analysis, which some could argue could be one important phenotype. The decision was taken, given the small number of patients with an elevated hyperdopaminergic score (9/193). In new set of cluster analysis with the hyperdopaminergic score, we found a lower statistical significance level of the different clusters (lower silhouette score and Calinski-Harabasz pseudo-F value and the hyperdopaminergic score was not significantly different between clusters). Impulse control disorders are a drug-induced complication and thus usually rare in untreated PD patients [5, 31], so it is not surprising that only 5% of our PD patients manifested moderate impulse control disorders. Additionally, compulsive eating was the most frequent symptom, which can also be related to depressed and anxious feelings (3 of the 5 patients were significantly depressive or anxious at the time of evaluation (ASBPD score≥2)).
Finally, harm avoidance was also negatively correlated with quality of life and neuropsychiatric phenotype showed the worse quality of life. Non-motor symptoms, in particular mood and apathy, represent a major determinant of quality of life in PD patients. Although personality traits were shown to contribute to many chronic diseases’ quality of life, fewer studies investigated it in PD [40]. High neuroticism was associated with worse quality of life in PD [41] but no studies applied Cloninger’s model.
Although it would be interesting to investigate the association between personality traits and cognition in PD patients, we did not include cognition in the model since one of the study inclusion criteria was a normal MATTIS Dementia rating scale score.
We acknowledge that our study has some limitations. First, our cohort is likely not representative of the whole PD population (relatively young, probably because of the recruitment in a tertiary care center). Secondly, the relatively small sample size and the lack of a control group may have limited our results and power of statistical analysis. Lastly, as in every study including early PD, we cannot exclude that a minority of patients may have atypical parkinsonism, mimicking PD.
In conclusion, our correlation and cluster analysis showed a strong relationship between personality, motor, and neuropsychiatric symptoms, allowing us to recognize 3 patients’ phenotypes (predominant neuropsychiatric, predominant motor, and benign). More than a unique parkinsonian personality, different traits might help explain heterogeneity in PD. Although, the association between different traits according to the Cloninger model and neurotransmitters is much less clear than originally stated, and the lack of strong biological data in support of this statement, we found that clinically personality, neurotransmitters imbalance, and neuropsychiatric symptoms seem to cluster in a way that may be of clinical value. Clinical recognition and characterization of these groups will allow a better understanding of the natural history of specific subtypes and better tailoring of treatment strategies. Their longitudinal follow-up will allow confirming whether some personality features might influence disease evolution and treatment. Combining clinical subtyping with imaging, and biological markers might help to optimize the definition of different subgroups with different evolution and response to treatment.
Footnotes
ACKNOWLEDGMENTS
We thank Chantal Delon-Martin (researcher from the Inserm, U1216, CHU Grenoble Alpes, Grenoble Institute Neurosciences) for the help in the statistical analysis.
The study was an Investigator-Initiated Study, where UCB provided financial support. The sponsor had no role in the study design, data analysis and interpretation, and in writing the manuscript.
CONFLICT OF INTEREST
B.M., E.L., A.B., H.K., E.S., P.P., V.F., S.M., F.D. declare that they have not received any sort of funding for the last 12 months. M.A. declares honoraria from Orkyn and Aguettant in the last 12 months. C.T. declares honoraria from Kyowa Kirin. JL.H. declares honoraria from advisory board de UCB, Abbvie, Medtronic. J.P.A. has served on advisory boards for Teva, UCB Pharma, AbbVie, and Zambon. He has received honoraria for presenting lectures from AbbVie, Medtronics, Novartis, Zambon, and UCB. E.M. has received honoraria from Abbott, Medtronic and Kyowa. S.T. declares honorarium from Orion, Boston. Grants from Neurodis, France Parkinson. P.K. declares grants from Swiss National Science Foundation, ROGER DE SPOELBERCH Foundation, Bertarelli Foundation, Annemarie Opprecht Foundation, Parkinson Schweiz, Michael J Fox Foundation, Aleva Neurotherapeutics, and Boston Scientific, personal fees (lecturing fees to employing institution/travel expenses to scientific meetings) from Boston Scientific, Bial, and Zambon outside the submitted work. A.C. declares research grants from France Parkinson Association and Medtronic, and lecturing fees from ABBVIE.
