Abstract
Neuropsychiatric symptoms in Parkinson’s disease (PD) are highly prevalent and profoundly disabling, often emerging even before the onset of motor symptoms. As the disease progresses, these symptoms usually become increasingly impairing and are now recognized as having the greatest impact on quality of life not only for patients but also for caregivers. In recent years, there have been significant advances in the diagnosis and management of neuropsychiatric symptoms. However, there are still substantial gaps in therapeutic approaches and algorithms, with limited pharmacological and nonpharmacological treatment options currently available. One of the main reasons for this is the complex molecular and neural bases of these symptoms, which involve both dopaminergic and nondopaminergic neurotransmission systems and extend far beyond the nigrostriatal pathway. As a result, the drugs currently recommended for treating neuropsychiatric symptoms in PD are few and supported by limited evidence. In this context, the experience of the treating neurologist remains critical in selecting the most appropriate individualized therapy. The aim of this paper is to review the available therapeutic options and provide an overview of current research efforts, particularly those focusing on pharmacological treatments.
Plain language summary
Neuropsychiatric symptoms in Parkinson’s disease (PD) are very common and significantly impact daily life. These symptoms often appear even before motor problems start and tend to get worse as the disease progresses. They are now considered to have the biggest effect on quality of life, not just for patients but also for their caregivers. In recent years, there have been important advances in diagnosing and managing these symptoms. However, treatment options (both medications and non-drug approaches) are still quite limited. This is partly because these symptoms are caused by complex changes in the brain that go beyond the usual dopamine system involved in PD. As a result, there are very few drugs available for treating these symptoms, and the evidence supporting their use is limited. In this challenging context, the neurologist’s expertise is essential for tailoring treatments to each patient. This paper aims to summarize the treatments currently available and highlight ongoing research, especially in pharmacological therapies.
Introduction
Parkinson’s disease (PD) has evolved from being considered a purely motor disorder, characterized by the degeneration of dopaminergic nigrostriatal neurons, to a condition where non-motor symptoms (NMS) play a more prominent role, and where disease pathology extends far beyond dopaminergic circuits and the nigrostriatal pathway. 1 It is well established that the basal ganglia, which remain central to the disease, are involved not only in motor circuits but also in circuits related to cognition, behavior, and emotions. 2 However, α-synuclein pathology and neurodegeneration are often found in other cortico-subcortical areas of the central nervous system and also in the peripheral nervous system, even in the early and premotor stages of the disease.3,4 This widespread involvement contributes to the diverse array of NMS observed in PD.
NMS were initially regarded as late complications of the disease, but many of these symptoms can precede the onset of motor symptoms, in what is now recognized as prodromal PD. 5 Developing at least one or typically a combination of NMS is almost inevitable throughout the disease course, and their absence after 5 years of disease progression is a red flag according to the latest Movement Disorders Society (MDS) PD diagnostic criteria. 6 In the advanced stages of PD, they become increasingly impactful, emerging as the major contributors to reduced quality of life and caregiver burden. 7
Neuropsychiatric symptoms such as cognitive impairment, psychosis, depression, anxiety, apathy, and impulse control disorders (ICD) are, as a group, the most common NMS in PD. 8 While any of these symptoms can occur at any moment of the disease, depression and anxiety are typically reported in the earlier stages, whereas cognitive impairment and psychosis, particularly in the form of dementia and complex hallucinations or delirium, are more characteristic of advanced stages (Figure 1). 1 These neuropsychiatric symptoms can significantly disrupt social and family dynamics, leading to a negative impact on both quality of life and caregiver burden.8 –11 In many cases, they may also become the primary reason for requiring nursing home care. 12 Additionally, some of these symptoms, such as cognitive impairment, hallucinations or apathy, are also predictors of more aggressive disease phenotypes.13,14

Neuropsychiatric symptoms in PD. They are represented with bars indicating the earliest stage of the disease at which they may manifest.
Early detection and assessment of neuropsychiatric symptoms are essential for the design of therapeutic approaches, for improving patients’ quality of life, and for reducing caregiver burden. Furthermore, they enable a more accurate prognosis of disease progression, helping patients and their families anticipate and prepare for future needs. The objective of this review is to provide an overview of published evidence to help clinicians in managing patients with neuropsychiatric symptoms in PD.
Search strategy
For this narrative review, we conducted a nonsystematic literature search of peer-reviewed articles published in English up to November 2024. The search was performed using the PubMed database. The terms used for this search included: (“Parkinson*” AND (“treatment” OR “therap*” OR “management” OR “pharmacological” OR “non-pharmacological”)), combined with (“neuropsychiatric” OR “psychiatric”), “depression,” “anxiety,” “cogniti*,” (“psychosis” OR “hallucinations” OR “delusion”), “apathy,” and (“impulse control OR impuls*”). Additional studies were identified through references in the included manuscripts and those deemed relevant were selected.
Cognitive impairment
Cognitive impairment can manifest at any stage of the disease and is categorized into three phases based on its impact on functional disability: PD-subjective cognitive decline (PD-SCD), PD-mild cognitive impairment (PD-MCI), and PD-dementia (PDD).15 –17 One-third of patients will exhibit PD-MCI at the time of the diagnosis, and up to 80% may develop PDD over the course of the disease.18 –20 Fronto-striatal functions, including attention, working memory, planning and set-shifting, are commonly affected earlier in the disease, whereas posterior cortical functions, such as memory, language, and visuospatial abilities, are more frequently impaired during the transition to or in already established PDD.21,22 While PD-MCI criteria were recently established, there is a need to actualize those for PDD.17,23,24
Multiple mechanisms have been implicated in the development of PD-associated cognitive impairment, including protein aggregation (mainly Lewy pathology but also Alzheimer’s Disease and other pathology), genetic predisposition (SNCA, GBA1, MAPT H1 haplotype, or apoE4), and various neurotransmitter dysfunction (dopamine, acetylcholine, noradrenaline, or serotonin).25 –31 Progression patterns of cognitive impairment are highly heterogeneous, but certain mechanisms such as cholinergic dysfunction, cortical spread of aggregated proteins, or genetic factors have been associated with posterior cortical phenotypes, which are linked to more aggressive cognitive decline and to an increased risk of developing PDD.32,33
Pharmacological interventions
Cognitive impairment in PD can significantly affect the quality of life of both patients and caregivers not only in later stages of dementia but also in the early stages, when patients have preserved insight to recognize and feel frustrated by even mild impairments.34,35 Unfortunately, pharmacological treatments to improve cognition are very limited with only a few symptomatic options and no therapies to modify its progression. 36
Currently, rivastigmine is the only symptomatic treatment that has demonstrated benefits in PDD (Table 1). 37 Other acetylcholinesterase inhibitors, such as donepezil or galantamine, are also considered potentially useful for improving cognition in PDD.36,38 In contrast, despite the fact that multiple clinical trials have been conducted with rivastigmine, rasagiline, atomoxetine, or donepezil, none of these drugs has demonstrated clear symptomatic benefits in PD-MCI.36,39 –44
Treatment recommendations for neuropsychiatric symptoms in PD: insights from published literature, expert consensus, and clinical experience.
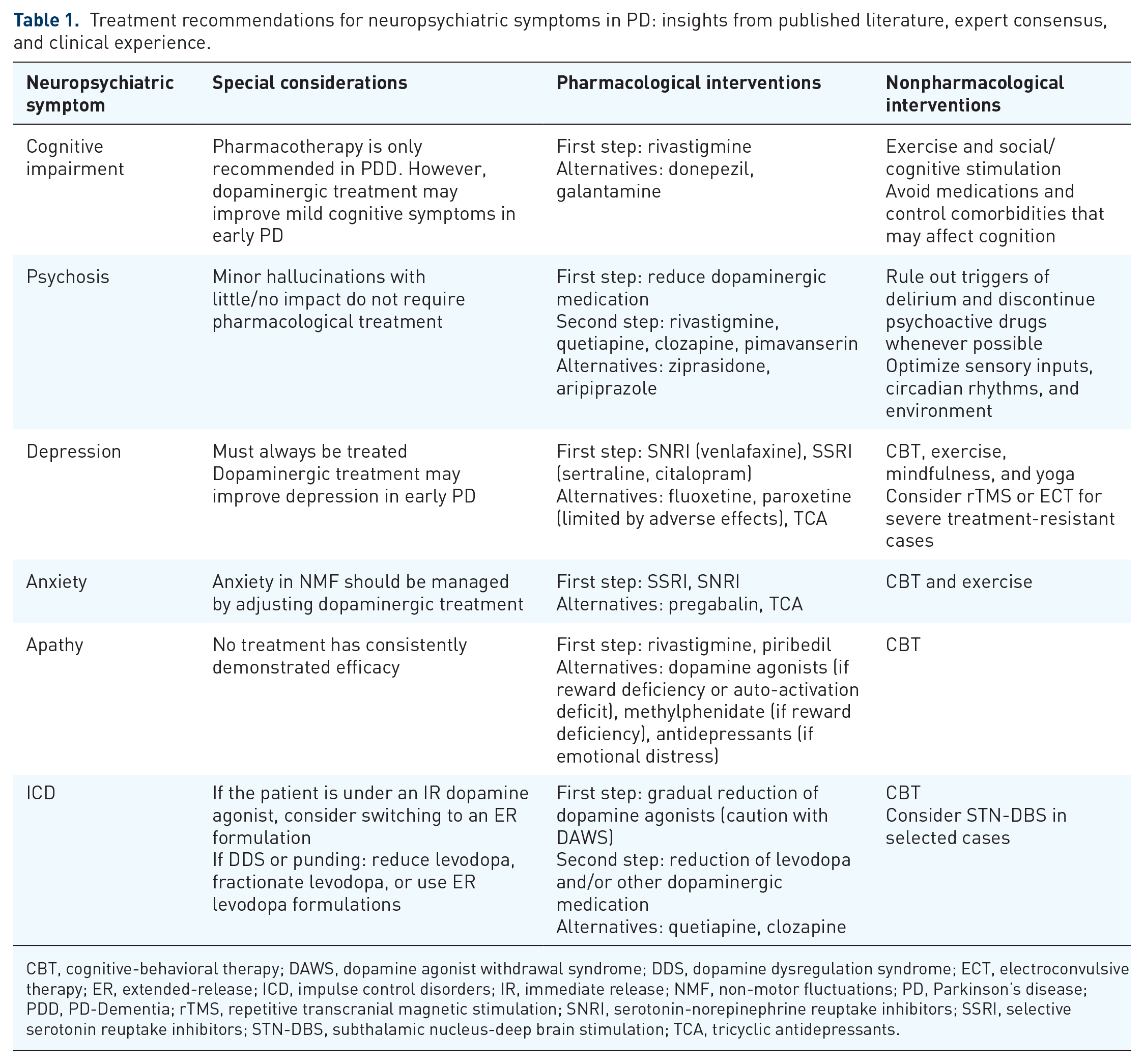
CBT, cognitive-behavioral therapy; DAWS, dopamine agonist withdrawal syndrome; DDS, dopamine dysregulation syndrome; ECT, electroconvulsive therapy; ER, extended-release; ICD, impulse control disorders; IR, immediate release; NMF, non-motor fluctuations; PD, Parkinson’s disease; PDD, PD-Dementia; rTMS, repetitive transcranial magnetic stimulation; SNRI, serotonin-norepinephrine reuptake inhibitors; SSRI, selective serotonin reuptake inhibitors; STN-DBS, subthalamic nucleus-deep brain stimulation; TCA, tricyclic antidepressants.
Both symptomatic and disease-modifying therapies for cognitive impairment in PD remain unmet needs, and ongoing or recently completed trials are investigating novel therapeutic targets beyond dopamine and cholinergic systems (Table 2). These include ANAVEX2-73 (blarcamesine), a Sigma-1 receptor agonist (NCT03774459, NCT04575259); DAAOI-P, a D-amino acid oxidase inhibitor and NMDA receptor subunit 1 facilitator (NCT04470037); ATH-1017 (fosgonimeton), a positive modulator of the hepatocyte growth factor neurotrophic system (NCT04831281); or SAGE-718, a positive allosteric modulator of the NMDA receptor (NCT04476017), among others. Additionally, there are trials evaluating the efficacy of classic drugs, such as donepezil, in PD-MCI using large multicenter cohorts (NCT05709301). In the future, clinical trials targeting the earlier phases of PD-associated cognitive impairment, such as PD-SCD or PD-MCI, will provide a more optimal window for evaluating the effectiveness of drugs aimed at delaying disease progression.45,46
Active clinical trials of drugs targeting neuropsychiatric symptoms in PD as a primary outcome (from ClinicalTrials.gov).

The following search terms were introduced in ClinicalTrials.gov as of December 14, 2024: Parkinson’s Disease AND Cognitive impairment, Parkinson’s Disease AND (Psychosis OR Hallucinations OR Delusions), Parkinson’s Disease AND Depression, Parkinson’s Disease AND Anxiety, Parkinson’s Disease AND Apathy, and Parkinson’s Disease AND Impulse control disorders.
ICD, impulse control disorders; PD, Parkinson’s disease; RCT, randomized clinical trial.
Non-pharmacological interventions
In view of the limited pharmacological options, it remains crucial to avoid the use of medications that may exacerbate cognitive impairment, such as anticholinergics or sedating drugs, as well as controlling comorbid conditions that may contribute to cognitive impairment, such as orthostatic hypotension, cerebral small vessel disease, or sleep disorders.47 –49
Additional nonpharmacological strategies, including cognitive and social stimulation, physical exercise, and noninvasive brain stimulation should also be considered in these patients. Although cognitive training has shown potential benefits in some randomized clinical trials (RCT), a recent meta-analysis found no compelling evidence of meaningful cognitive improvements in PD-MCI or PDD.36,50,51 Physical exercise has shown benefits in improving cognition in PD and given its broad positive effects in the disease, should always be a recommendation.52,53 While noninvasive brain stimulation techniques, such as repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation cannot yet be recommended for clinical use, they remain promising tools for the future. 54 Emerging technologies are also exploring the possibility of replacing basal forebrain cholinergic neurons with stem cell-derived cells as a potential treatment for PDD. 55 Overall, these nonpharmacological approaches show potential but require further investigation to fully establish their effectiveness.
Psychosis
PD psychosis (PDP) encompasses a spectrum of illusions, hallucinations, and delusions in patients with PD. 56 It usually presents as minor hallucinations (typically visual), which occur in over 40% of patients, even in the prodromal phases.56 –59 As PD progresses, hallucinations tend to become more complex, with a progressive loss of insight, particularly in patients with PDD.60,61 Delusions are a common PD symptom in advanced stages, but may also occur isolated in younger and cognitively unimpaired patients. 62
PDP is thought to result from disrupted intrinsic connectivity between attentional networks and the default mode network (DMN), which affects bottom-up processing systems. Dysfunction in the ventral attentional network may compromise visual processing, leading to an increased dependence on the posterior DMN, which is involved in introspection, mental imagery, and episodic memory retrieval.56,63 This disruption leads to impairments across several processes, including visual perception and recognition, multisensory integration, semantic categorization and meaning attribution of stimuli, reality monitoring, and attention. 56
Mesocorticolimbic dopaminergic degeneration was considered a key factor in the development of PDP. 64 Nevertheless, other neurotransmitters such as the cholinergic degeneration within the nucleus basalis of Meynert and the pedunculopontine nucleus, both essential components of the visuoperceptive system, as well as the upregulation of 5-HT2A and 5-HT3 receptors in the striatum and cortex, are also important contributors to PDP and have therapeutic implications.65 –67
Pharmacological interventions
PDP may not require treatment if symptoms have little or no impact on psychosocial function. 68 However, as the disease progresses, symptoms may become disruptive and require specific management (Table 1).
After ruling out potential infections or other triggers of delirium, the first step is to reduce dopaminergic stimulation and increase cholinergic innervation. 56 Although the evidence is limited, a recommended sequence of medication reduction is anticholinergics, followed by selegiline, dopamine agonists, amantadine, catechol-O-methyltransferase (COMT) inhibitors, monoamine oxidase B (MAO-B) inhibitors, and finally levodopa.69,70 Benzodiazepines, opioids, and other psychoactive drugs should also be discontinued whenever possible. 56
If these strategies do not adequately control PDP, the addition of specific medications should be considered. The biological mechanisms underlying PDP have led to the use of drugs that block dopaminergic or serotoninergic receptors, or those that enhance cholinergic neurotransmission. 56 Atypical antipsychotics are the most commonly used medications due to their low D2 receptor activity and their high 5-HT2A receptor activity. 56 Although clozapine is the only antipsychotic classified as clinically useful by the MDS Evidence-Based Medicine (MDS-EBM) Committee, quetiapine, which is considered possibly useful, is the most frequently prescribed drug.36,71 Other atypical antipsychotics, such as ziprasidone and aripiprazole, may be used as an alternative in selected patients.72,73 Haloperidol, risperidone, and olanzapine should be avoided due to their higher risk of motor worsening.36,74,75
Beyond antipsychotic agents, other drugs with different mechanisms of action allow to avoid the risks of worsening Parkinsonian symptoms. Enhancing cholinergic neurotransmission with rivastigmine is a useful and safe approach, particularly in patients with PDD. 76 Pimavanserin, a selective 5-HT2A receptor inverse agonist and antagonist with no dopaminergic, adrenergic or muscarinic affinity, has shown significant improvement in PDP without negative motor effects.77,78 Similar to clozapine, pimavanserin is classified as clinically useful by the MDS-EBM, but for the moment it is only available in a few countries like the United States or India. 36 Studies conducted decades ago demonstrated the utility of 5-HT3 antagonists such as ondansetron in PDP, and a current RCT is evaluating its efficacy (NCT04167813) (Table 2). 79 Finally, and also based on the serotoninergic hypothesis, a recent study suggested the potential efficacy of ulotaront, a trace amine-associated receptor 1 agonist with 5-HT1A receptor agonist activity. 80
Nonpharmacological interventions
General measures including restoring sensory inputs (adequate lighting, and vision or hearing aids), maintaining circadian rhythms, or keeping an adequate familial environment, should always complement pharmacological treatment in PDP. 81
Depression
Depression is one of the most common NMS in PD with a prevalence of 35%–50%.82,83 Between 5%–20% of patients will develop a major depressive disorder and an additional 10%–30% will develop minor/subsyndromal forms of depression, frequently preceding the onset of motor symptoms.82,83 In contrast with other NMS, such as apathy or cognitive impairment which generally worsen during the course of the disease, depression in PD remains stable. 84 Risk factors include non PD-specific (age, female gender, personal history of anxiety or depression, family history of depression, worse functioning on activities of daily living, and cognitive impairment) and PD-specific characteristics (higher levodopa doses, severe motor fluctuations, autonomic dysfunction, and sleep disturbances).85 –87
A unified pathophysiological model for depression in PD (DPD) is lacking, but it is believed to result from a combination of psychosocial, psychological, and neurobiological factors. 84 There is compelling evidence indicating that DPD arises from dysfunction in both dopaminergic and nondopaminergic neurotransmitter systems. 84 Multiple studies have demonstrated a link between serotonin and noradrenaline with DPD, as well as early degeneration of these neurons, which explains the early onset of DPD in many patients.3,84 Furthermore, DPD is associated with motor symptoms and may also manifest as nonmotor fluctuations, highlighting the involvement of the dopaminergic system. 84 Anatomically, DPD results from disruptions in mesolimbic circuits, primarily involving the anterior cingulate cortex, orbitofrontal cortex, amygdala, thalamus, and ventral striatum, which are key regions for emotional regulation and reward processing.84,88
Due to the significant impact on quality of life, caregiver burden, and overall disability, DPD should be actively treated regardless of clinical severity. 89
Pharmacological interventions
The efficacy and tolerability of selective serotonin reuptake inhibitors (SSRI), serotonin-norepinephrine reuptake inhibitors (SNRI), and tricyclic antidepressants (TCA) have been extensively investigated and demonstrated in RCTs, and several options are recommended for managing DPD (Table 1). 90 However, in the absence of large comparative trials specific to DPD, the choice of antidepressant remains pragmatic and is based on clinical judgment. 91
Citalopram, sertraline, paroxetine, and fluoxetine are SSRIs currently categorized as possibly useful by the MDS-EBM, despite their potential to exacerbate tremor or parkinsonism (<5%) and the caution required when combined with MAO-B inhibitors due to the risk of serotonin syndrome.36,48 Notably, paroxetine has the strongest anticholinergic effect, making sertraline (which may also improve motor symptoms), citalopram, and fluoxetine the preferred options.92 –94 TCAs are the antidepressant group with the second-highest number of RCTs after SSRIs, and three of them (nortriptyline, desipramine, and amitriptyline) have been categorized as possibly useful by the MDS-EBM.36,93,95,96 However, their frequent anticholinergic and cardiac side effects, especially in older adults and patients with orthostatic hypotension or cardiovascular disease, limit their clinical use. 91 Although SNRIs have been less studied, venlafaxine is the only antidepressant classified as clinically useful by the MDS-EBM, and some authors suggest superiority of dual or predominantly noradrenergic reuptake inhibitors over SSRIs.36,84,94 In clinical practice, other antidepressants such as duloxetine, trazodone, mirtazapine, or bupropion are frequently used despite limited evidence in PD. 36
Dopaminergic therapy, especially in the early stages of the disease, can also alleviate depressive symptoms.91,97 Pramipexole has shown significant antidepressant effects in a RCT and is classified as clinically useful by the MDS-EBM.36,98 However, the inconsistent results observed with rotigotine and ropirinole, combined with the demonstrated increased risk of ICD in patients with DPD treated with pramipexole, highlight the need for caution when prescribing dopamine agonists in this population.99 –102 MAO-B inhibitors have limited evidence supporting their use for DPD.36,103,104
Other pharmacological agents, including 5-hydroxytryptophan, atomoxetine (selective norepinephrine reuptake inhibitor), or agomelatine (melatonin 1 and 2 receptor agonist and neutral 5-HT2C antagonist), have been assessed in a limited number of studies and require further evidence to establish their utility.93,105,106
Nonpharmacological interventions
Cognitive-behavioral therapy (CBT), including telemedicine delivery, has demonstrated efficacy for DPD, classifying as possibly useful by the MDS-EBM, and is the most widely used form of psychotherapy.36,107,108 A course of psychotherapy is a recommended for the initial treatment of clinically significant depression, either alone or in combination with antidepressants. 48
Interest in the use of brain stimulation therapies for mood disorders in PD is growing. High-frequency rTMS targeting the left dorsolateral prefrontal cortex has shown overall short-term benefits for depression in a recent meta-analysis, and has been categorized as possibly useful by the MDS-EBM.36,109 Studies on the effects of deep brain stimulation (DBS) on DPD have yielded mixed results.110,111 While, subthalamic nucleus-DBS (STN-DBS) has generally been associated with favorable outcomes for depression when targeting more anterior, medial and ventral areas, some studies have suggested greater improvement with globus pallidus interna-DBS (GPi-DBS).111 –114 Electroconvulsive therapy has demonstrated effectiveness for treatment-resistant DPD, but is reserved for selected cases due to its potential side effects, including cognitive impairment, delirium, and exacerbation of autonomic symptoms. 115
Other nonpharmacological interventions such as physical activity, mindfulness or yoga have also shown benefits for depressive symptoms and should also be considered as part of a comprehensive treatment plan.116,117
Anxiety
Anxiety affects over one-third of patients with PD and is defined as a generalized response to uncertain or nonimmediate threats, often characterized by excessive worry, hypervigilance, and physiological arousal.118,119 Among the various anxiety disorders, generalized anxiety is the most prevalent in PD. 118 It frequently precedes the onset of motor symptoms, worsening in parallel with disease progression, and often manifesting or exacerbating as part of nonmotor fluctuations.120 –122 These symptoms not only impair the quality of life of patients but also increase significantly caregiver burden, making anxiety a high research priority in PD management, after balance and falls.34,123,124
A broad neural network is implicated in the pathophysiology of anxiety in PD, encompassing structures such as the amygdala, hippocampus, and nucleus accumbens, along with connections to cortical regions, including the prefrontal and insular cortices, among others. 125 While serotonin seems to be the primary neurotransmitter deficiency in anxiety, its association with nonmotor fluctuations also suggests a strong involvement of the dopaminergic system.125,126
Pharmacological interventions
First-line medications used for treating anxiety in PD include those that have proven effective in the general population, such as SSRIs or SNRIs, while other drugs such as pregabalin, TCAs, or buspirone, may serve as alternatives (Table 1).127 –129 Medications known to be highly effective in managing anxiety, such as benzodiazepines or quetiapine, should be used with caution due to their risk of dependence, their lower tolerability, and their higher risk of falls.128,130 Given the potential role of dopamine deficits in PD-related anxiety, pramipexole may also be considered as a treatment option. 131 If anxiety is identified as a nonmotor fluctuation, adjusting dopaminergic medications is the most efficacious therapy. 123
Despite the abovementioned recommendations, most drugs (including paroxetine, venlafaxine, desipramine, citalopram, or atomoxetine, among others) have not been tested in RCTs or have not shown efficacy in RCTs where anxiety was a secondary outcome.93,94,123,132 The only pharmacological RCT specifically designed for anxiety in PD investigated the effects of buspirone, which, while effective in a small group of patients, was poorly tolerated due to adverse effects. 129 Currently, only one phase II RCT with mirtazapine (NCT06530290) is investigating the effects of medications for anxiety in PD, while many commonly used antidepressants have yet to be evaluated (Table 2).
Nonpharmacological interventions
The scarcity of scientific evidence and the potential adverse reactions to medications make nonpharmacological therapies an option to consider for all patients with anxiety. CBT is the most widely used form of psychotherapy in general population and has demonstrated efficacy with observable functional brain changes in PD.133 –135 Considering the significant impact of anxiety on caregiver burden, involving caregivers in CBT interventions has been suggested as a potentially beneficial approach. 136 A recent meta-analysis has also highlighted the significant impact of exercise on PD-related anxiety. 137 As already mentioned, exercise should always be a recommendation in PD given its broad favorable effects. 138 Despite heterogeneous results, there is growing evidence that STN-DBS may also improve anxiety in both the short and long term, especially when stimulation is directed closer to the ventral STN.137,139 –142 Additional nonpharmacological interventions, such as mindfulness and noninvasive brain stimulation have also shown promise, though further research is required to establish their efficacy.143,144
Apathy
Apathy is defined as a state of decreased motivation resulting in reduced goal-directed activities, not explained by cognitive impairment, emotional distress, or reduced consciousness, although it often overlaps with both depression and cognitive impairment. 145 Its prevalence in PD ranges from 35%–70%, sometimes preceding the diagnosis, and is typically associated with reduced quality of life, increased dependence, and heightened caregiver stress. 146
Apathy encompasses behavioral, affective, and cognitive features and is conceptualized as the inability of the basal ganglia to validate relevant signals transmitted from the prefrontal cortex. 147 It has been associated with cerebral regions involved in reward and executive processing, such as the left nucleus accumbens, lateral prefrontal cortex, orbitofrontal cortex, and parietal cortex. 148 Apathy has also been linked to deficits in multiple neurotransmitter systems, especially dopaminergic loss in mesolimbic and mesocortical circuits, as well as in serotoninergic and cholinergic pathways. 149
Pharmacological interventions
Apathy is a challenging symptom, with limited evidence supporting specific treatments. Piribedil and rivastigmine are the only drugs categorized as possibly useful by the MDS-EBM (Table 1). 36 Piribedil is a selective D2/D3 dopamine agonist that demonstrated benefits in patients who became apathetic after STN-DBS. 150 Trials with other dopamine agonists, such as pramipexole and ropinirole, although methodologically poor and not specifically aimed at clinical apathy, have shown positive effects on apathy, while rotigotine has yielded inconsistent results.151 –154 A RCT with rivastigmine demonstrated benefits for apathy in patients without dementia or depression. 155
Small studies with apathy as a secondary outcome have shown that methylphenidate, which enhances mesocorticolimbic stimulation by selective inhibition of dopamine reuptake, or candesartan, an angiotensin receptor blocker, might be potentially useful in improving apathy in PD.156,157 A RCT with safinamide did not found significant differences between treated and untreated groups, but post-hoc analyses revealed a positive trend, warranting further investigation in larger studies. 158 Other studies with SNRIs (duloxetine) and SSRIs (paroxetine and escitalopram) have not demonstrated significant effects on apathy, despite the known emotional component present in some patients. 159
Non-pharmacological interventions
Non-pharmacological therapies for apathy in PD offer promising but very limited evidence. Key elements of CBT, such as exercise and behavioral activation, have shown potential in reducing apathy, although it remains unclear whether these are direct effects or secondary to improvements in other neuropsychiatric symptoms. 160
Current evidence suggests that STN-DBS may exacerbate apathy in PD. This could be due to postsurgical medication adjustments or the direct effects of neuromodulation.161,162 However, the low methodological quality and heterogeneity of existing studies highlight the need for well-designed studies to better understand these associations.
Impulse control disorders
ICD are primarily considered an iatrogenic complication of dopaminergic medications, particularly dopamine agonists that preferentially bind to D3 receptors. 163 These disorders are characterized by the inability to resist the temptation to perform certain behaviors, which are executed repetitively with a lack of self-control, often resulting in negative consequences for the patient or their environment. 164 The most common ICD in PD include pathological gambling, compulsive shopping, hypersexuality, binge eating, or hobbyism. Their severity may range from mild to significant disruptions in social, occupational, and financial functioning.165,166 Dopamine dysregulation syndrome (DDS) and punding require special therapeutic considerations, as they are most commonly observed with high doses of levodopa, which results in pulsatile stimulation of D1 and D2 receptors. 167
ICD affect up to 20%–45% of PD patients, can arise years after the initiation of any dopaminergic therapy, and often become chronic, showing poor response to dose reduction or discontinuation of the triggering medication.163 –165 Risk factors for ICD include male gender, younger age at disease onset, longer disease duration, apathy, depression, tobacco, and personal or family history of gambling or alcoholism.99,164 –166,168,169 Studies using 18F-FDG-PET imaging and feedback-related negativity have also been able to address metabolic and neurophysiological markers of ICD that are present even before the onset of symptoms.170,171
The pathophysiology of ICD is primarily linked to dysfunctions within the dopaminergic system. Sensitization of D2/D3 receptors in the midbrain and anterior cingulate cortex, along with reduced dopamine transporter availability in the striatum, disrupt reward and impulse regulation pathways.172,173 These alterations compromise the ability of the brain to control goal-directed behavior, contributing to the development of compulsive actions.
Pharmacological interventions
Given the lack of therapeutic evidence for ICD, management strategies are mostly based on clinical experience, making it difficult to reach a consensus on treatment algorithms (Table 1). 164
The first-line strategy is to gradually reduce dopamine agonists, adjusting the speed of tapering to the severity of ICD. 164 Immediate stop is generally not recommended and clinical monitoring is essential due to the risk of dopamine agonist withdrawal syndrome (DAWS), a condition characterized by a significant worsening of NMS (such as anxiety, depression, drug craving, or insomnia) which can occur even with slow taperings. 164 For persistent ICD symptoms, reducing levodopa or other dopaminergic medications, such as MAO-B or COMT inhibitors, may be necessary. In cases of DDS and punding, initial strategies include lowering the total levodopa dose, increasing dose fractionation, or transitioning to extended-release formulations. 167
If dopaminergic reduction is insufficient, adjunctive second-line strategies must be considered. RCTs have only been conducted for two drugs. Amantadine showed potential efficacy for pathological gambling, but was found to exacerbate ICD in other studies.174,175 Similarly, despite improvements on a PD-specific ICD scale, naltrexone did not show significant differences between treatment and placebo groups and was associated with adverse events. 176 Therefore, these drugs are not currently recommended for treating ICD.36,91,164 Currently, pimavanserin is under investigation for ICD in an active RCT (NCT03947216) (Table 2).
Although clozapine and quetiapine have only shown positive results in case reports, they are frequently prescribed by experts, and are recommended as adjunctive second-line drugs in selected cases.164,177 –179 The role of SSRIs remains controversial, as observational studies link them to increased ICD in PD, while evidence from non-PD population suggests potential benefits for compulsive behaviors like pathological gambling and trichotillomania.180,181
Additional strategies include switching from immediate-release to extended-release dopamine agonists to achieve more continuous dopaminergic stimulation, although this approach requires further validation.163,182,183 Two prospective studies demonstrated a reduction in ICD following the initiation of levodopa–carbidopa intestinal gel, independent of dopamine agonist dose reduction, supporting that a switch from pulsatile to a more continuous dopaminergic stimulation may result in ICD improvement.184,185
Nonpharmacological interventions
The involvement of committed caregivers is essential and has been identified as the most significant positive predictor for remission. 164 Additionally, addressing legal or financial difficulties through social work support, and assessing psychiatric comorbidities, insight, and legal capacity, are strongly recommended. 164
Currently CBT is the only nonpharmacological intervention classified as possibly useful by the MDS-EBM, based on a single study that demonstrated a significant reduction in ICD severity.36,186
Most observational studies, including large prospective cohorts, suggest that ICD tend to decrease after bilateral STN-DBS, while studies on unilateral STN-DBS or GPi-DBS report mixed results.187 –190 The efficacy of bilateral STN-DBS is believed to be a consequence of the reduction in dopaminergic medication, rather than a direct effect of the stimulation. 164
Status quo, challenges, and future directions
Currently, the available tools to manage neuropsychiatric symptoms in PD are very limited. Moreover, their effectiveness remains modest, and the impact of these symptoms both in patients and caregivers continues to be profound, particularly in the late stages of the disease.
Cognitive impairment is one of the most devastating neuropsychiatric symptoms and is often accompanied by complex hallucinations and delusions. In PDP, significant progress has been made with the commercialization of pimavanserin, a promising drug that offers the advantage of avoiding the motor side effects of neuroleptics. 78 However, in the realm of cognitive impairment, the situation has remained largely unchanged for over 20 years, with only one drug available that has modest effects and does not halt the progression of symptoms. 37 These symptoms progressively compromise brain functions, leading ultimately to complete functional dependency. The main challenge in developing molecules for symptomatic treatment, and especially in developing molecules aimed at modifying the course of cognitive decline, lies in the fact that its pathophysiology, like that of PD itself, remains largely elusive. Advances in biomarker research, along with ongoing studies on novel molecules, are expected to synergistically enhance our understanding of the molecular basis of cognitive impairment in PD, paving the way for the development of new therapeutic targets.
Although depression is one of the neuropsychiatric symptoms with the most RCT conducted, there are no large comparative studies, and the choice of antidepressant remains largely dependent on the neurologist. 91 In the case of anxiety, the challenge is compounded by the lack of RCTs in patients with PD, and treatment selection is based on results from studies in individuals without PD. The underlying mechanisms of anxiety in PD, which likely differ from those in the non-PD population, along with the higher risk of intolerance due to falls, exacerbation of motor symptoms, cognitive decline, or dysautonomia, suggest that findings from RCTs conducted in general population may not be directly applicable to PD.36,123 Therefore, there is a need for RCTs that establish a priority order for antidepressants in DPD, as well as RCTs specifically targeting anxiety as a primary outcome.
Apathy is a similarly debilitating but more complex and poorly understood symptom from a neuropsychological perspective. To date, there are only two recommended treatments with very modest efficacy, and the reality is that they offer no guarantee of symptomatic improvement. Apathy has been previously conceptualized as an entity with four distinct subdomains, each linked to unique neural substrates and contributing differently to apathy in each patient: decrease in emotional resonance (reward deficiency syndrome), depression (emotional distress), decrease in cognitive interests (executive dysfunction), and absence of spontaneous activation of mental processes (auto-activation deficit). 147 A treatment approach that discriminates between these four proposed subdomains could provide a promising foundation for enrichment strategies in future clinical trials. These strategies may help guide an individualized approach to the management of apathy.
Finally, ICD represent one of the greatest treatment challenges in PD, with patients caught between a rock (ICD) and a hard place (DAWS and/or worsening of motor symptoms). Achieving the right balance can be complicated and may require the use of adjunctive medications, often neuroleptics, that are not officially approved for this indication. Combining pharmacological treatment with strong caregiving support and psychiatric support may be essential.
This review is not without limitations. We acknowledge that sleep and appetite disorders, as well as aberrant motor behavior, are sometimes classified as neuropsychiatric disorders in PD. However, due to space limitations and to maintain clarity for the reader, we decided not to include these symptoms in the present review. More importantly, the primary limitations of this narrative review are the lack of a systematic approach to study selection, which may introduce selection bias, as well as the absence of a formal quality assessment of the included articles. However, the limited number of studies on treatment of neuropsychiatric symptoms in PD makes management highly dependent on the clinician experience. In this context, a narrative review can still be very valuable, as it provides a broad overview of current knowledge and highlights areas where standardized guidelines are lacking.
Conclusion
Neuropsychiatric symptoms in PD are more prevalent and have a much greater functional impact than previously thought, which has driven the development of numerous diagnostic and assessment tools aimed at improving clinical management and research. Despite these advancements, there are still many gaps in their treatment, with limited medications available for many of these symptoms, or with many medications with low percentage of therapeutic success. In this context, nonpharmacological therapies are highly valuable. The lack of understanding of the molecular basis of these symptoms in particular, and of PD in general, as well as the complexity of their neural substrates, significantly hinders the development of new molecules and the design of high quality RCTs, which remain scarce. Advancing knowledge of specific biological mechanisms will be crucial for identifying new therapeutic targets for neuropsychiatric symptoms in PD.
