Abstract
Background:
Verticality perception is frequently altered in Parkinson’s disease (PD) with Pisa syndrome (PS). Is it the cause or the consequence of the PS?
Objective:
We tested the hypothesis that both scenarios coexist.
Methods:
We performed a double-blind within-person randomized trial (NCT02704910) in 18 individuals (median age 63.5 years) with PD evolving for a median of 17.5 years and PS for 2.5 years and treated with bilateral stimulation of the subthalamus nuclei (STN-DBS) for 6.5 years. We analyzed whether head and trunk orientations were congruent with the visual (VV) and postural (PV) vertical, and whether switching on one or both sides of the STN-DBS could modulate trunk orientation via verticality representation.
Results:
The tilted verticality perception could explain the PS in 6/18 (33%) patients, overall in three right-handers (17%) who showed net and congruent leftward trunk and PV tilts. Two of the 18 (11%) had an outstanding clinical picture associating leftward: predominant parkinsonian symptoms, whole-body tilt (head –11°, trunk –8°) and transmodal tilt in verticality perception (PV –10°, VV –8.9°). Trunk orientation or VV were not modulated by STN-DBS, whereas PV tilts were attenuated by unilateral or bilateral stimulations if it was applied on the opposite STN.
Conclusion:
In most cases of PS, verticality perception is altered by the body deformity. In some cases, PS seems secondary to a biased internal model of verticality, and DBS on the side of the most denervated STN attenuated PV tilts with a quasi-immediate effect. This is an interesting track for further clinical studies.
Keywords
INTRODUCTION
Pisa syndrome (PS) defines a marked lateral trunk deviation, frequent in advanced Parkinson’s disease (PD) [1] with axial symptoms [2]. It represents a major source of disability [1, 3–5] and predicts early mortality [6].
Its management is often limited to rehabilitation [5, 7–11], which would benefit from tailored interventions based on the mechanisms underpinning PS. Recent reviews have synthesized hypothetical mechanisms [3, 12] involving basal ganglia (BG) and intervening in isolation or together: 1) asymmetry of outflow causing dystonia [13–15] or vestibulo-spinal asymmetry [16, 17]; 2) impaired sensory integration altering perceptions of head or trunk positions [17–21]; and 3) distorted sense of upright [12, 22–24]. These neural mechanisms might interact with a preexisting scoliosis [25].
The distorted sense of upright is an emerging interpretation [12, 26], supported by numerous studies that repeatedly found abnormal vertical estimates in PD [19, 26–33], and by analogy with other models of brain damage [34]. Individuals with PS would align their body to an internal vertical reference not matching the Earth vertical. Such determinism is possible, as the BG are involved in constructing the internal model of verticality via the thalamus, which is critically and broadly involved in graviception [35–38]. However, studies have failed to reach a definitive conclusion. The directional concordance between tilts in verticality estimates and trunk orientation remains unclear [22, 30]. Biases in vertical estimates are rarely systematically lateralized [23], contrary to what is expected for asymmetric brain lesions [34, 39]. In fact, most verticality abnormalities in PD are related to an uncertain perception of the vertical [19, 28–33].
Difficulties in understanding vertical perception in PD and PS could come from the coexistence of two different scenarios. Tilted vertical perception might cause PS in some patients and might be secondary to PS in others. Indeed in individuals with a deformed body geometry, vertical estimates may be aligned with the head or with the trunk according to the availability of intact vestibular or somaesthetic information [40–42] and even personal perceptive styles [43]. Our
Bilateral deep brain stimulation of subthalamic nuclei (STN-DBS) is an effective treatment for advanced PD and can be used for exploring BG functions. Assuming that some PD patients under chronic bilateral STN-DBS would align their trunk to a tilted internal model of verticality, inducing PS, the question was whether the PS could be experimentally attenuated by transiently eliciting a better verticality representation. The STN is indirectly connected to the thalamus via the globus pallidus [53, 54], so that asymmetrically modulating STN activity might affect the internal model of verticality [20]. Our
We recruited patients with PD and PS treated with bilateral STN-DBS. One part of the study was a cross-sectional analysis of the concordance between head, trunk, PV and VV orientations under chronic bilateral-on STN-DBS settings and their relationships with the main clinical features. Its reporting follows STROBE guidelines. The main part of the study consisted in an experimental intervention comparing trunk, PV and VV orientations under four conditions of STN-DBS. Its reporting follows CONSORT guidelines for within-person randomized non-pharmaceutical trials.
MATERIALS AND METHODS
Ethical and regulatory considerations
This study was conducted in accordance with the Declaration of Helsinki and was approved by an ethics committee (France Sud-Est-V, IDRCB:2011-A01533-38/1). All participants provided informed written consent. At the time we designed the study (2010), only multicentric randomized clinical trials were systematically registered at ClinicalTrials.gov, which explains the delay in declaring this pilot experimental study (NCT02704910). The recruitment of these infrequent particular patients (PS with bilateral STN-DBS) for a difficult experiment also took longer than expected. The trial was performed as planned in 2010 and accepted in 2011 by the ethics committee and was conducted, interpreted, and reported with integrity.
Participants
Inclusion criteria were idiopathic PD according to the “UK Parkinson’s disease Brain Bank criteria for idiopathic Parkinson’s disease” [57] with a diagnosis for more than 5 years; marked spontaneous lateral trunk tilt, found clinically obvious and consistent over time by neurologist expert in movement disorders (AC, VF, PK); treatment with bilateral STN-DBS; and able to tolerate complete transitory interruption of stimulation for several hours. Exclusion criteria were a history of vestibular or peripheral sensory deficits, dementia (score < 130/144 on the Mattis dementia rating scale) [58]; major depressive episode as defined by the Diagnostic and Statistical Manual of Mental Disorders 4-R criteria; and unstable medical problems. We excluded people with major unreducible postural deformities preventing installation of the apparatus used to measure verticality perception.
The sample size could not be calculated but was targeted at 20 PD participants, and 20 healthy individuals matched in age and sex served as controls (STN-DBS Bilateral-on). This was adapted to our monocentric recruitment (specific apparatus) and is usually sufficient for concluding on verticality perception in experimental studies [42, 60].
Clinical data with chronic Bilateral-on STN-DBS
The motor score (part III) of the Unified Parkinson’s Disease Rating Scale, UPDRS motor part [61] was assessed in PD participants off-medication. To determine whether motor symptoms were asymmetric, we calculated sub-scores for each hemibody (STN-DBS Bilateral-on) as the sum of the tremor (items 20-21), rigidity and bradykinesia (items 20–26) scores, with a difference≥2 defining an asymmetric PD [62].
Body orientation in the frontal plane, on- and off-medication
Trunk and head orientations in roll were first clinically measured in PD participants, on-medication and on-stimulation on day 0 by a trained practitioner (MJ) who was blinded to the study hypotheses. Participants were instructed to stand as upright as possible for several seconds while looking straight ahead at the landscape through a window. Trunk orientation was assessed by means of a plumb line, ruler, and rubber tape (Fig. 1A), allowing to calculate the tilt (in degrees) of the segment C7-S1 (Supplementary Methods). The trunk was considered upright within the ranges of normality defined by the instrumental measure (see below). Head orientation (Fig. 1B) was assessed in sitting position by measuring the tilt of the bi-pupillary line with respect to the horizontal plane [42] by using an inclinometer (Pro 360 Digital Protractor, resolution 0.1°). The head was considered upright for marginal tilts≤2° [42].
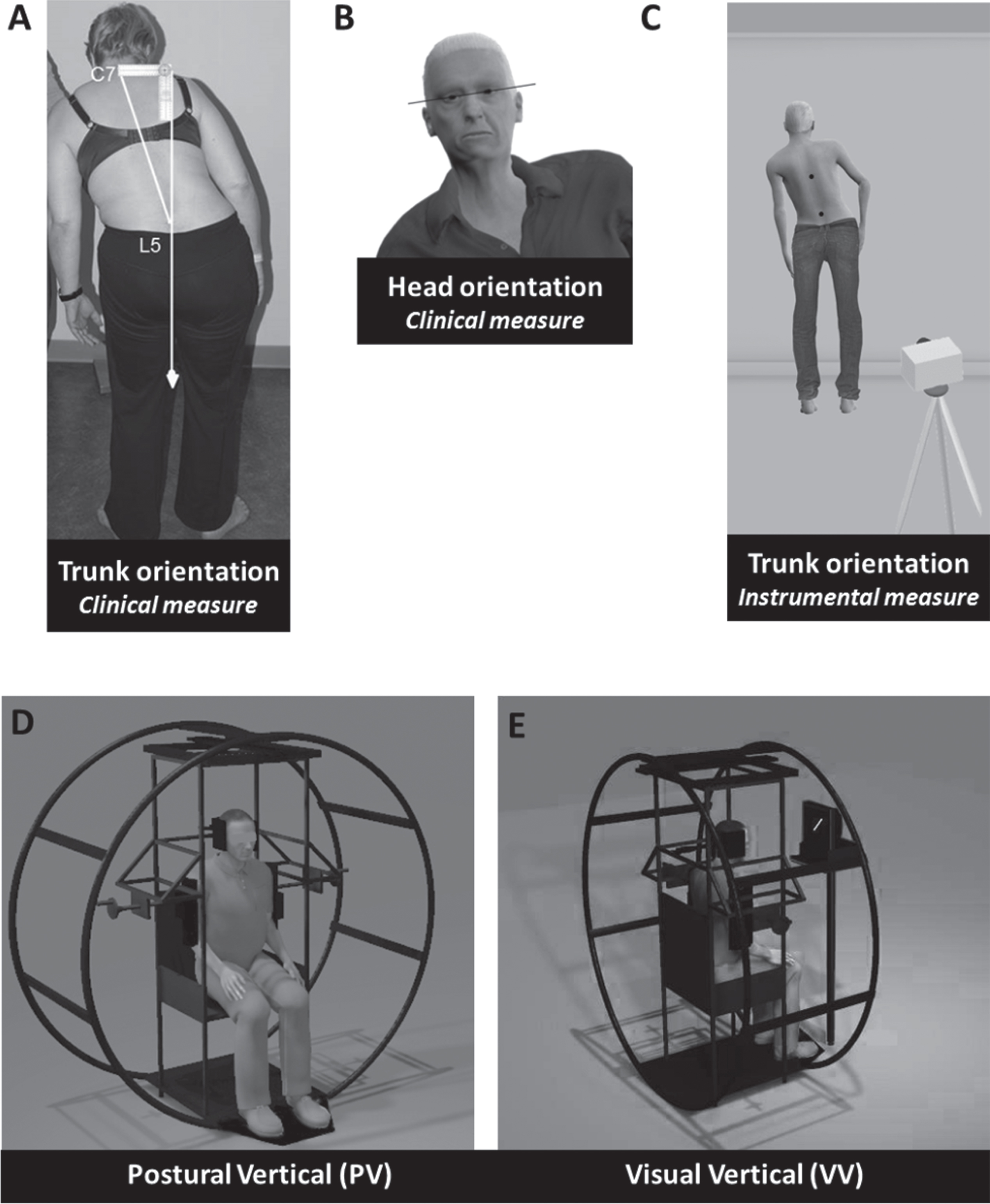
Schematic views of devices and tools used for assessing body orientation and verticality perception.
Trunk orientation was also quantified off-medication by a trained investigator (CP) who was blinded to the stimulation condition and clinical data of interest (Supplementary Results and Discussion). We used an instrumental measure of the segment T6-L3 (Fig. 1C), the spinal segment responsible for lateral trunk inclination [63]. PD Participants were instructed to stay as upright as possible for 30 s in two postural conditions (standing and sitting) and two visual conditions (with or without vision) (two trials each, for a total of eight trials) presented in a pseudo-random order (Supplementary Methods). Standing was considered the reference condition for the interventional trial, by analogy with the clinical assessment with the plumb line.
Verticality perception was explored in complete darkness by both PV and VV, in PD participants off-medication, by a trained investigator (CP) who was blinded to the stimulation condition and clinical data of interest (Fig. 1D, E; Supplementary Methods). For PV (Fig. 1D), participants seated in a wheel passively rolled in the frontal plane and indicated the body orientation they experienced as vertical [38]. For VV (Fig. 1E), participants seated upright indicated how to set a computerized luminous line to the earth vertical. The orientation of PV (PV-orient) and VV (VV-orient) was calculated as the algebraic mean of the six trials performed. PV and VV magnitudes were also expressed in absolute values.
Trunk orientation (instrumental measure) and verticality estimates were also assessed in healthy participants to determine range of normality for trunk orientation, PV and VV (Supplementary Results). For all orientations, a positive value indicated a tilt to the participants’ right.
The representation of the body schema was also assessed but not reported in this paper for clarity reasons (and length).
The presence of a scoliosis was analyzed on full radiography of the spine in standing (anterio-posterior and lateral) and supine (anteroposterior) positions. The detailed data for these measures were recently reported [25].
Trunk orientation in the sagittal plane was clinically determined by the C7 sagittal arrow provided by the horizontal distance between the C7 spinous process and the sacral fold with respect to the plumb line (cm), in a straightened-up position without help [64]. Although the criteria for defining camptocormia are debated, the involuntary sagittal inclination when standing was diagnosed as camptocormia with a C7 sagittal arrow > 8 cm, corresponding to the limit of normality in older people [64].
History of falls was merely assessed by asking patients if they had fallen in the past month [65].
STN-DBS modulation of trunk, PV and VV orientations in PS
Experimental design
We performed a double-blind within-person randomized trial investigating trunk orientation and verticality perception after a 12 h overnight withdrawal of dopaminergic treatment under four conditions of STN-DBS: Bilateral-on, Bilateral-off, Unilateral right (right-on/left-off) and Unilateral left (right-off/left-on). To facilitate data interpretation, unilateral stimulations were also expressed according to the side of the tilts observed in the Bilateral-off condition and named T runk, PV, or VV-Ipsi-off or -Ipsi-on. Stimulation parameters were those used for the chronic treatment: mean (SD) amplitude 2.7V (0.6), pulse width 62.5 (8.3) μs, frequency 135 (14.1) Hz. Stimulation was monopolar in 32 STN, bipolar in 2, and double monopolar in 2.
The experiment was organized over 1 week in the neurorehabilitation department. PD participants were tested on four consecutive mornings (days 1–4), one morning for each STN-DBS condition, according to a pseudo-randomly determined order (Latin square for the 16 first participants, then randomly for the 2 others). The STN-DBS parameters were adjusted or pretended to be adjusted (chronic Bilateral-on condition) at the beginning of each morning to maintain the similarity of interventions and ensure blinding for the patients and investigator in charge of assessments (CP). Each session lasted 3 h, with assessments performed in the same order. The UPDRS motor part was performed about 1 h after setting the stimulation condition. Trunk orientation was assessed 30 min later with conditions (standing/sitting, with/without vision) presented in a pseudo-random order. Measures of spatial estimates started about 2 h after setting the stimulation, with a pseudo-random order for PV, VV and body schema representation. The order was the same for the 4 days. Three investigators were involved: AC or BD to manage stimulation conditions and perform the UPDRS motor part, and CP who performed all measurements with blinding to the stimulation condition and clinical data of interest.
Stimulation parameters were restored or pretended to be restored (Bilateral-on condition) together with the medication as soon as all measures were collected. After a lunch break and a rest, participants had a light rehabilitation session in the afternoon that focused on gait, falls prevention, cognition, and adaption of technical aids if needed.
Outcome criteria and data analyses
The UPDRS motor part enabled assessment of the efficacy of the STN stimulation on PD cardinal signs under each stimulation condition. The main criteria for the trial was trunk orientation measured by the instrumental measure, chosen a priori for its accuracy and operator independence [66]. PV and VV were secondary criteria.
For trunk orientation, PV-orient, and VV-orient, the effects induced by switching on one or both sides of the STN-DBS were analyzed by reference to the tilt observed in the condition Bilateral-off STN-DBS. To analyze directional effects of the STN-DBS, we converted data signs so that greater positive values indicated a greater tilt remaining on the same side (worsening), whereas lower values indicated a lower tilt remaining to the same side (value remaining positive and meaning tilt improvement) or even a tilt to the opposite side (value becoming negative).
Statistical analyses
Statistical analysis involved using SPSS v23 (IBM Corp., Armonk, NY, USA). Data are expressed as mean (SD) or median [Q1;Q3] as appropriate (Gaussian distribution or not). Unless specified otherwise (Bonferroni correction indicated in the results), the significance level was fixed at p = 0.05, and effect sizes are given. Each part of the study had its own plan of statistical analyses.
Data for healthy participants were used to determine ranges of normality calculated according to data distribution (Supplementary Statistical Analyses). The effects of posture (standing/sitting) and vision (with/without) on trunk orientation were analyzed by two-way ANOVA in both healthy and PD participants. For PD participants, prevalence of abnormal trunk, PV and VV tilts was analyzed by chi-square test (Fisher’s exact test otherwise). Comparison of tilt magnitudes between controls and patients, or between postural and visual verticality estimates, involved the non-parametric T-test for independent series. Spearman correlation (rS) was used to test possible correlations between the magnitude of trunk and head tilts and PV and VV estimates.
STN-DBS effects were explored on the UPDRS motor scores to control that DBS was effective at the time of the experiment (Supplementary Results), on the trunk orientation (primary outcome), and on PV-orient and VV-orient (secondary outcomes) by using Friedman ANOVA and pairwise comparisons with a Bonferroni correction for multiple comparisons.
RESULTS
Figure 2 presents the flow diagram of the study. Between May 2012 and June 2015, we recruited 18 individuals with median age 63.5 [61;67] years, including 11 (61%) females, 16 right-handed (89%), with a PD evolving for a median of 17.5 [14;20] years, and asymmetry in 13/18 (72%). Patients were treated with bilateral STN-DBS for a median of 6.5 [4;10] years and implemented before the PS in 15/18 (84%). PS had been diagnosed for a median of 2.5 [1;4] years. Most patients (15/18) were able to walk without aid. There were 10/18 (56%) recurrent fallers, 6 reporting one or two falls per month and 4 several falls per week. Under bilateral stimulation, the median motor score of the UPDRS was 22.8 [17.1;31.1], much better than in the three other conditions of stimulation (Supplementary Materials). The median C7 sagittal arrow was 11.5 cm [8.0;15.0] without correlation with lateral trunk orientation (p = 0.28) or verticality perception (ps > 0.3). Eleven (61%) patients had camptocormia (C7 sagittal arrow > 8 cm under instructions to stand upright). Scoliosis was diagnosed in 11/18 (61%) patients, of moderate magnitude (median Cobb angle 17° [14;26]). All participants completed the whole experiment, and no data were missing. Controls were matched in terms of age (63 [58;65] years; p = 0.36) and sex (12 [60%] females, χ2 = 0.0, p = 0.94).
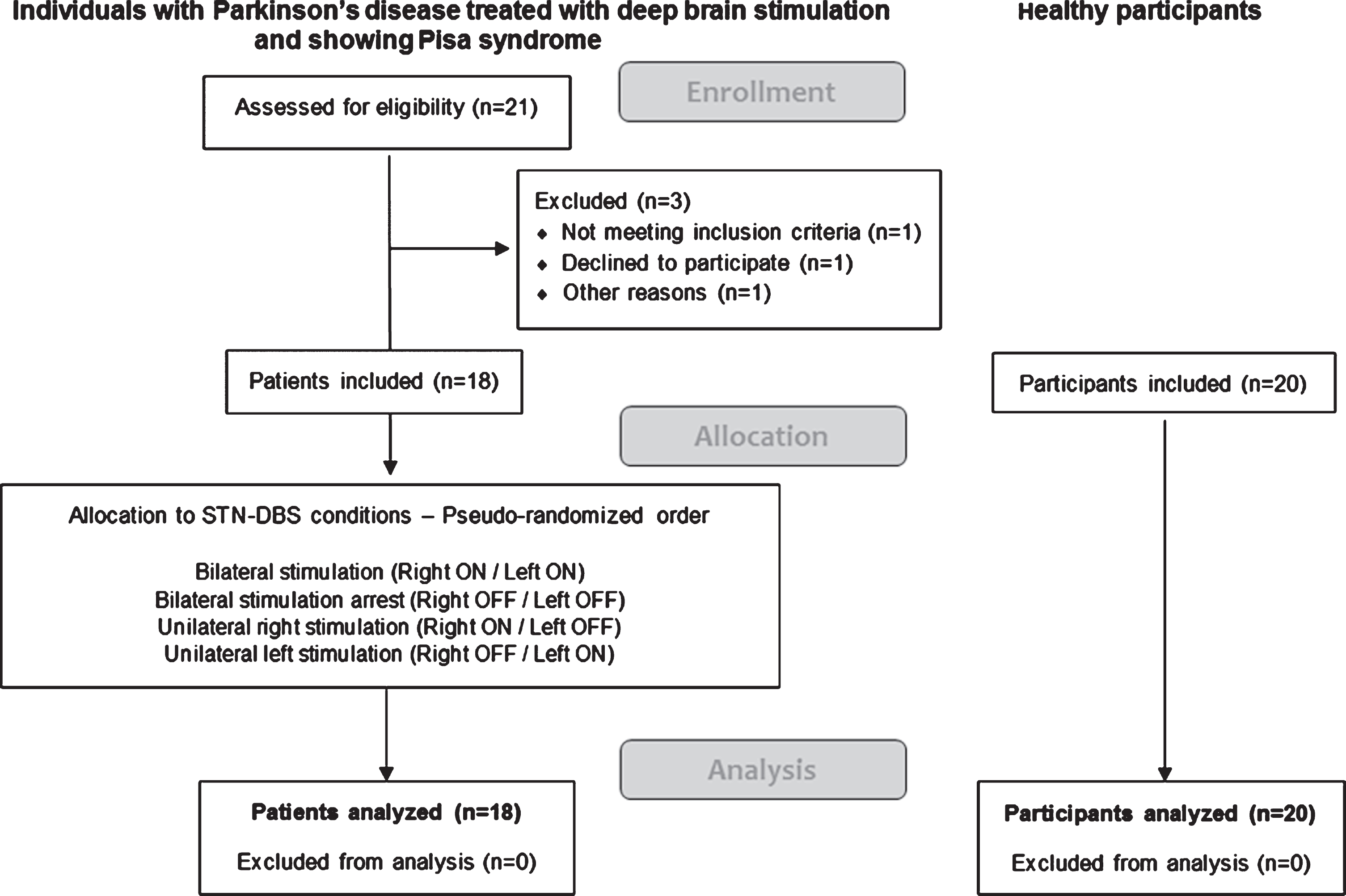
Flow chart of the study. STN-DBS: deep brain stimulation of subthalamic nuclei.
Trunk orientation
A two-way ANOVA on instrumental data (absolute values) showed that the magnitude of trunk tilts was greater standing than sitting (mean 6.9° [5.9] vs 3.3° [2.8]; F(1,68) = 10.92; p = 0.002, η2 = 0.138), without vision effect (F(1,68) = 0.246; p = 0.62) or interaction (F(1,68) = 0.009; p = 0.93). The lack of visual influence on trunk orientation allowed for analyzing DBS effects taking into account the four trials of trunk assessment, with and without vision. Only the standing posture was considered for further analyses. This position maximizes trunk tilt and corresponds to clinical assessment. Trunk tilts measured instrumentally in standing (instructions to be as upright as possible; with and without vision) were much greater in PD than healthy participants (4.7° [3.1;9.5] vs 0.9° [0.3;2.0]; Z = –4.678; p < 10-5).
According to the range of normality between –3° and +3° established in healthy participants (Supplementary Results), five participants were able to stand actively upright under instruction. The direction of the trunk tilts was unrelated to the side of the predominant PD symptoms as assessed at disease onset (10 individuals leaned toward the most affected body side and 8 toward the opposite side) or at the time of the experiment (among the 13 individuals with asymmetric symptoms, 5 leaned to the less-affected body side and 8 to the most-affected body side).
We found no systematic effect of STN-DBS on trunk orientation (χ2(3) = 1.267, p = 0.74). Individual and averaged data are presented in Fig. 3A and B.
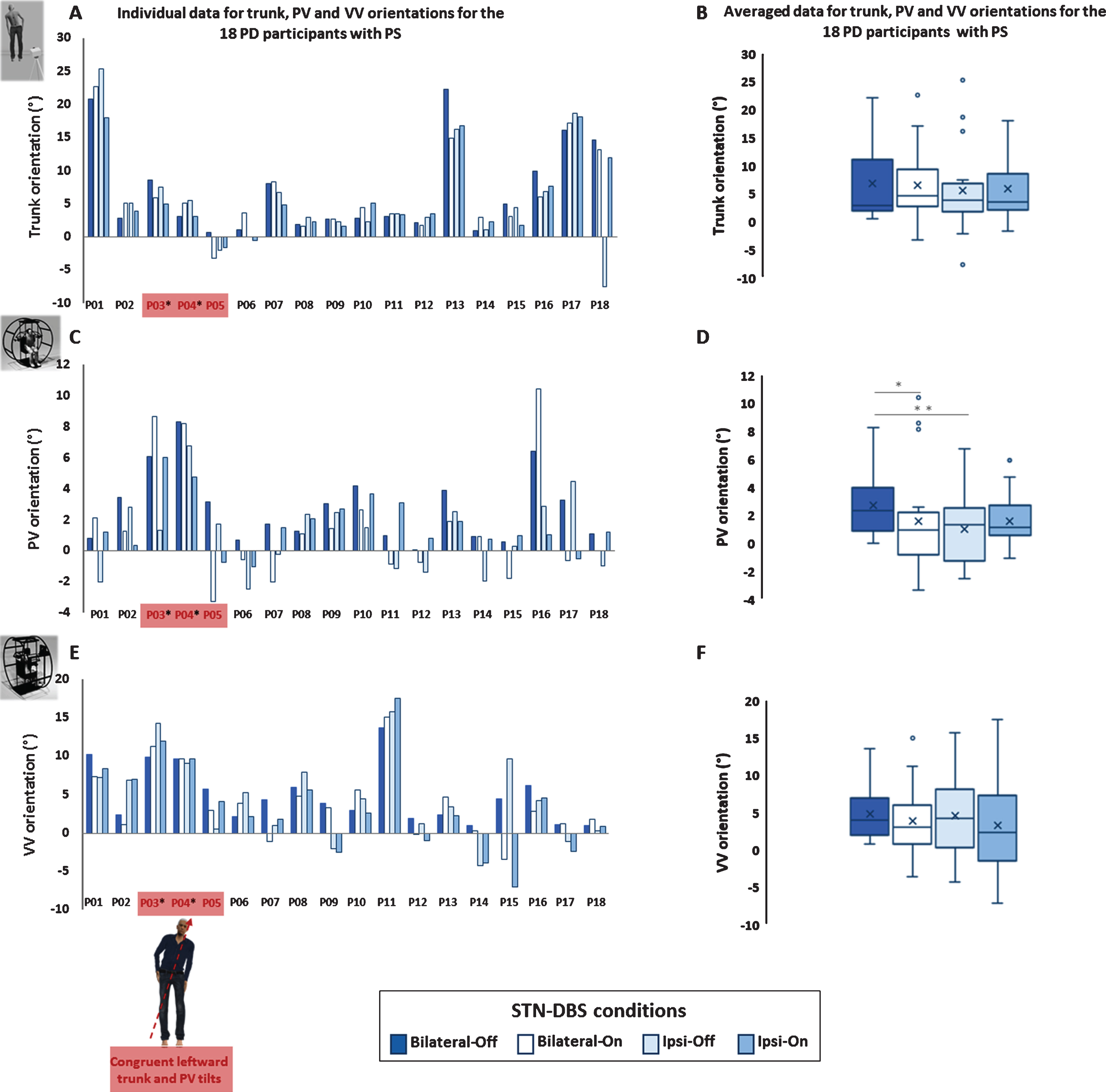
Effect of the deep brain stimulation of the subthalamic nuclei (STN-DBS), on trunk, postural vertical (PV) and visual vertical (VV) orientations under four DBS conditions (Bilateral-Off, Bilateral-On, Ipsi-Off and Ipsi-On) in the 18 Parkinson’s disease (PD) participants with Pisa syndrome (PS). Values in the three conditions of switching on one or both sides of the STN-DBS were converted over the reference condition: Bilateral-off STN-DBS, with signs converted so that greater positive values indicated a greater tilt remaining on the same side (worsening) and lower values indicated a lower tilt remaining on the same side (value remaining positive and meaning tilt improvement) or even a tilt to the opposite side (value becoming negative). A) Vertical bar graphs represent the individual data for mean trunk orientations in standing posture (with and without vision averaged data). B) Box-plots represent the effect of DBS conditions on the averaged trunk orientations. C) Vertical bar graphs represent the individual data for mean PV orientations. D) Box-plots represent the effect of DBS conditions on the averaged PV orientations. E) Vertical bar graphs represent the individual data for mean VV orientations. F) Box-plots represent the effect of DBS conditions on the averaged VV orientations. The three (17%) patients showing congruent orientations of trunk and PV tilts, as represented on the schematic representation, are indicated in red (P03, P04, P05). Among them, two patients, marked by an asterisk, presented a transmodal leftward PV-VV tilt congruent in magnitude and direction with the whole-body tilt. For box-plots, the X in the box represents the mean, the middle line the median, the ends of the box the lower quartile (Q1) and upper quartile (Q3) and whiskers the lower and upper adjacent values. Outlier data are shown as circles plotted beyond the whiskers. *p < 0.05; **p < 0.01.
Verticality perception
Normal verticality perception
Verticality perception (PV from –2° to +2° and VV from –4° to +4°, Supplementary Results) was normal in nine patients (50%). Thus, PS was not caused by a wrong verticality perception in half of patients of this series.
Biased verticality perception
Table 1 shows that in the whole series, the average magnitudes of PV and VV tilts were mild. Overall PV tilts were twice smaller than VV tilts (1.6° [0.9;2.5] vs 3.4° [1.4;5.5], p = 0.02), with no significant correlation between modalities (rs = 0.37, p = 0.13). Nine patients (50%) presented a biased verticality perception, with a prevalence of 6/18 (33%) for PV and 7/18 (39%) for VV. Among them, two showed a transmodal congruent verticality bias (PV and VV), and seven various patterns of isolated (n = 6) or incongruent (n = 1) verticality biases.
Postural and visual vertical data in healthy participants and Parkinson’s disease participants with Pisa syndrome
Data are n/N (%) or median [Q1; Q3]. PD, Parkinson’s disease; PS, Pisa syndrome. Normalized Uncertainty was calculated as the ratio of within-subject variability (SD) to orientation, expressing the robustness of the internal model of verticality regardless of any bias [36, 44].
Of note, PV tilts were only found in patients who leaned to the side of the initial PD symptoms: 6/10 vs 0/8 (p = 0.007). The four with PV tilted to the left had PV tilts of great magnitude (mean 7.6°), whereas the two with PV tilted to the right had marginal values of tilt (2.4°). Such a concordance was not found for VV tilts (5/10 vs 2/8, p = 0.2).
Modulating verticality perception by STN-DBS
As shown in Fig. 3C and D, PV orientation was modulated by some STN-DBS conditions in the 18 individuals with PD and PS (χ2(3) = 13.53, p = 0.003). Post-hoc comparisons showed that PV tilts were magnified under Bilateral-off (2.4° [0.9;4.0]) as compared with Bilateral-on (1.0° [–0.7;2.2]; p = 0.018) and PV-Ipsi-off (1.4° [–1.2;2.6]; p = 0.005).
In contrast, we found no systematic effect of the STN-DBS on VV orientation (χ2(3) = 2.732, p = 0.43). Individual and averaged data are in Fig. 3E and F.
Scenarios of body orientation as a function of verticality perception
These analyses were performed on clinical measurement of trunk orientation, which were strongly correlated with instrumental measurements (r = 0.88, p < 10-5), a result suggesting that both ways to assess trunk orientation in PD with PS were reliable.
Figure 4 presents individual data for head, trunk, PV, and VV orientations, showing different scenarios. Segmental body tilts were 6.5° [2.5; 9.8] for the head and 7.5° [4.1;9] for the trunk (Fig. 4A). Mismatches between head and trunk tilts were large (11.8° [6;18]), because of frequent opposite orientations, found in 10 patients (55%), in whom the magnitude of head and trunk tilts was inversely but not significantly correlated (rs = –0.602; p = 0.066).
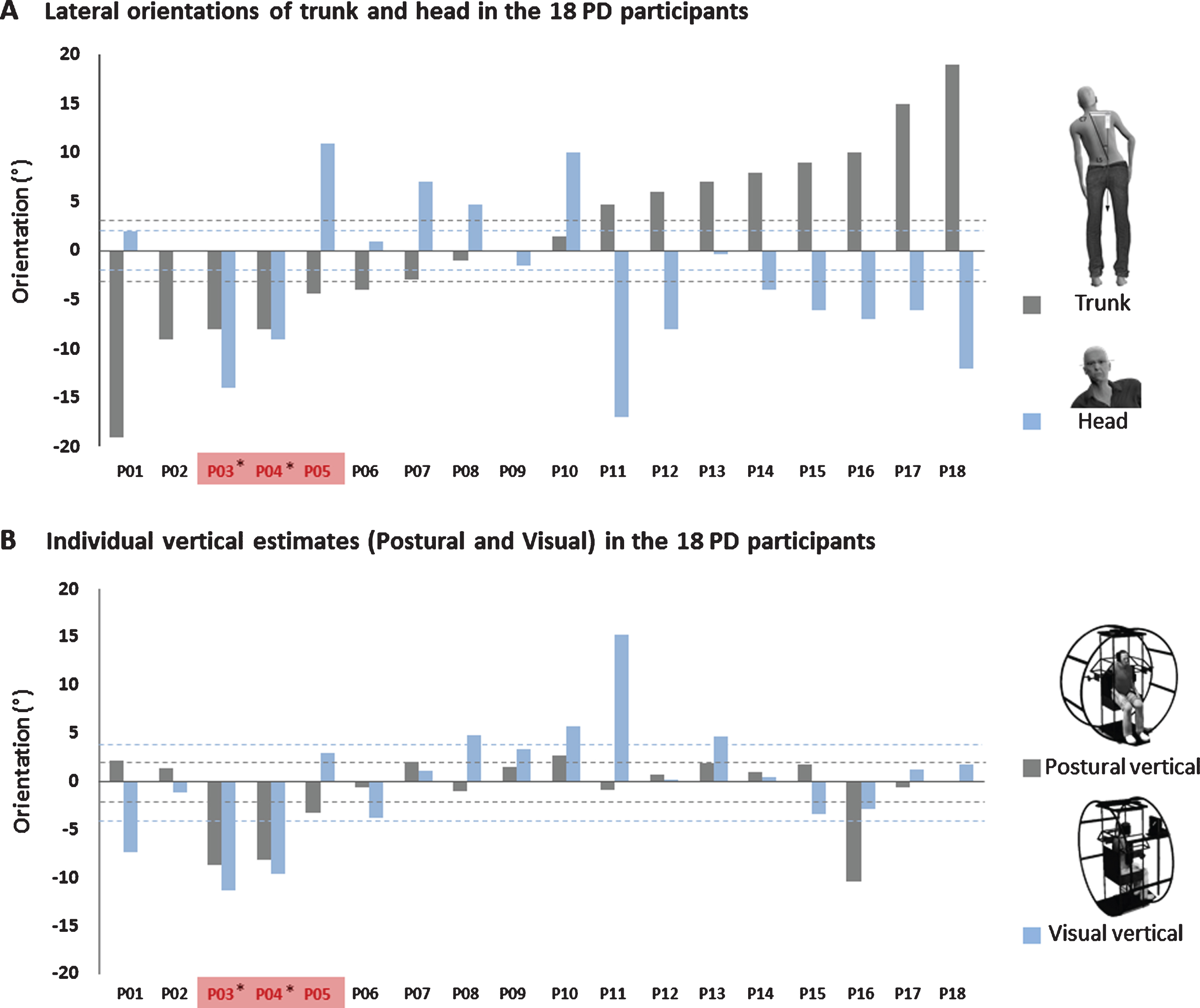
Individual data (each bar) for the 18 Parkinson’s disease (PD) participants with Pisa syndrome (PS) showing concordance in the roll plane between orientations of the trunk and head with those of verticality estimates (postural vertical [PV]), visual vertical [VV]). Data are expressed in raw signed values (negative for leftward tilt) and ranked according to increasing order of trunk tilt in degrees. Participants are named according to their order of appearance in the figures, from left (P01) to right (P018). A) Trunk tilt (dark grey bars) and head tilt (light blue bars) orientations. Only four individuals kept the head horizontal. The head was mostly tilted to the side opposite the trunk tilt, except in P03 and P04, with the head tilted to the same side. Dotted lines represent the ranges of normality for each body segments orientation (dark grey for trunk and light blue for head). B) PV (dark grey bars) and VV (light blue bars) estimates. Dotted lines represent the ranges of normality for each spatial estimate (dark grey for PV and light blue for VV). The three patients (17%) showing congruent orientations of trunk and PV tilts are indicated in red (P03, P04, P05). Among them, two patients, marked by an asterisk, presented a transmodal leftward PV-VV tilt congruent in magnitude and direction with the whole body tilt.
Ten participants showed a congruent orientation of the body (head, trunk or both) with verticality estimates (PV, VV, or both). Three presented a clear clinical picture. Among them, the only patient who stood perfectly upright (head and trunk) under instructions (P09), had normal verticality perception. Her verticality perception could not explain her spontaneous PS. The two other patients (11%), both right-handers, presented an outstanding clinical picture perfectly linking posture to verticality perception (P03 and P04). They showed a whole-body leftward tilt involving the head (P03 –14°, P04 –9°) and trunk (–8°, –8°), with congruent leftward tilts in PV (–8.7°, –8.2°) and VV (–11.3°, –9.6°). The left side was that of the predominant PD symptoms (i.e., opposite to the most denervated striatum). PS had developed 3 and 8 years, respectively, after STN-DBS, and at the time of the study, the patients showed a clear asymmetric PD. These findings strongly suggest the causal role of a biased internal model of verticality in their PS, involving both vestibular and somaesthetic graviception. Seven patients exhibited a less complete clinical picture, possibly linking PS to biased verticality perception. In four, all right handers, the trunk tilt was congruent with isolated PV tilt (P05) or isolated VV tilt (P01, P11, P13), with leftward trunk tilts in three. In three, the head tilt was congruent with isolated PV tilt (P16 who has a scoliosis) or isolated VV tilt (P08, P10).
Eight individuals presented incongruent orientations of the body and verticality estimates with a segmental body tilt despite a normal verticality perception. Their verticality perception could not explain the PS.
To sum up, the PS (trunk tilt) could be explained by a tilted verticality perception in 6/18 patients (33%), showing congruent trunk and PV and/or VV tilts (P01, P03, P04, P05, P11, P13). The clinical picture was complete in two of the six, who showed PV and VV tilts congruent in magnitude and direction with a whole body tilt (head and trunk). In the 12 other patients (67%), the PS could not be explained by verticality perception.
DISCUSSION
This double-blind within-person randomized trial is the first to investigate how each of the four conditions of a bilateral STN-DBS may modulate, in the very short term, the active body orientation with respect to gravity, in relation to verticality perception. This is also the first study to analyze the directional concordances between trunk, head, PV, and VV orientations in patients with PD and PS. This study brings convergent findings, confirming that BG are part of neural circuits involved in constructing the internal model of verticality, with a crucial role for the somesthetic graviception, positively modulated by the condition bilateral STN-DBS or unilateral on the side opposite to the PV biases.
The tilted verticality perception could explain the PS in 6/18 (33%) patients, with an outstanding clinical picture in 2/18 (11%), in whom the transmodal bias in verticality perception was likely the cause of the PS. This limited number of patients tempers the recent enthusiasm for the role played by verticality perception in inducing PS. Indeed, in our series, PS could not be explained by verticality perception in 67%of individuals, who had a normal verticality perception or presented associations between verticality estimates and head tilts only. Hence, other underlying mechanisms are at work in most PS cases, probably sensorimotor integration [20], dystonic [67] or orthopedic [25] isolated or combined factors [3, 12]. All these results taken together confirm our main hypothesis of the coexistence of two scenarios, accounting for the high frequency of PD patients with PS showing a wrong verticality perception. This situation calls for caution in interpreting the literature on verticality perception in PD. Of note, the head and trunk were mostly oriented in opposite directions, which does not fit with the picture of the Leaning Tower of Pisa (Supplementary Discussion).
PS may result from a biased internal model of verticality
We report a pronounced and indisputable bias in the internal model of verticality [38], very likely at the origin of the PS in two individuals (11%), who leaned leftward (head and trunk of similar magnitude) with a congruent (direction and magnitude) transmodal tilt of PV and VV. These cases suggest a dual alteration of vestibular and somaesthetic graviception, predominating in right BG. The most plausible interpretation is that these two patients unconsciously aligned their whole body on a wrong internal model of verticality, tilted leftward. Of note, this prevalence rate of about 10%is similar to that of pusher syndrome after hemisphere stroke [38, 68], the most striking human model with a wrong internal model of verticality [36, 38]. Besides sharing acronyms, pusher and Pisa syndromes likely share a dysfunction of the neural projections from the BG to the temporo-parietal junction as an underlying mechanism [38, 70]. In pushers, the internal model of verticality is almost exclusively biased after right hemisphere lesions [38], and both vertical estimates and trunk orientation are tilted to the side opposite (left hemiparesis) [38, 39]. In the current study, we found a similar lateralization, the two right-handed participants showing a pronounced bias in their internal model of verticality, leaning the whole body to the left, the body side of the predominant PD symptoms. Their predominant nigrostriatal denervation affected the right hemisphere, specialized in space processing. Post-stroke lateral inclination occurs suddenly and is overall obvious at the subacute stage, whereas in PD, the lateral inclination most often occurs insidiously and worsens with the disease course. This low progression of the disease with a less lateralized brain damage explains why no obvious pusher behavior is observed in PD.
Finally, our study extends findings known after stroke to PD. These two types of brain lesions with different location, asymmetry and courses, generalize the predominant contribution of the right hemisphere in constructing the internal model of verticality.
Somesthetic and vestibular graviception in PS
In most patients of our series, head and trunk on one hand and PV and VV on the other, were oriented in opposite directions or at least unrelated, which suggests that BG alterations of PD differentially affect somaesthetic and vestibular graviception. PV mainly tests somaesthetic graviception, which relies on tactile, muscular proprioceptive and interoceptive signals [36, 71], whereas VV mainly assesses vestibular graviception, which relies on otoliths [36, 72]. Our findings indicate that these two types of graviception follow different pathways and involve different structures within the BG, as is the case with cortical lesions [38]. The alteration of the specific structures and pathway of each modality might depend on how they are affected by the predominant PD side, the typology of the PD, and its disease duration and severity. Regardless of the PD, individual factors might also play a role. Further investigations with a large sample size are required.
In our study, modulating verticality perception by STN-DBS has been a way to better understand the role of BG in updating the internal model of verticality, by eliciting changes in somaesthetic graviception but not vestibular graviception. Indeed, PV tilts were attenuated when the STN-DBS was bilateral or switched off the side of PV tilt. This finding first indicates that the STN is more involved in the somaesthetic graviception than the vestibular graviception, for which it might act as a specific gateway. The STN are small nuclei that are indirectly connected via the globus pallidus to the ventroanterior thalamic nucleus and the ventrolateral thalamic nucleus [53, 54], the latter being structurally part of the posterolateral thalamus. Therefore, the STN are functionally connected to the posterolateral thalamus [73], a key structure for integrating somaesthetic and vestibular graviception [36–38, 74–76]. The STN are also an entry point for the BG [77] as it receives direct cortical inputs from motor and somatosensory cortical areas [78]. Furthermore, the anatomical proximity between the cortico-subthalamic projections of this direct pathway and the key regions of the neural circuits of the visual perception of verticality [79–81] support the involvement of STN in the neural circuits contributing to the internal model of verticality, mainly via somasthetic graviception. Such an interpretation agrees with the powerful modulation effect of STN-DBS on proprioception, in particular in dynamic tasks [20]. In addition, PV tilts better explained trunk tilts than did VV tilts, especially leftward trunk tilts. Several other mechanisms that may underpin PS include dystonia or scoliosis, as in P16, in whom the rightward trunk tilt was possibility due to scoliosis. No study has ever reported a modulation of the VV by adjusting STN-DBS in patients with PS. So our negative results for VV are the first to be reported. They do not contradict positive results reported in two case studies of peculiar individuals without PS [56, 82]. One study [56] tested the effect of a unilateral stimulator arrest on VV orientation in two PD patients with long-term bilateral STN-DBS complaining of a neck discomfort associated with an abnormal head tilt. Turning off the stimulator on the side opposite to the head tilt attenuated both the head and VV tilts. Of note, when the STN-DBS was switched on again, the voltage increase was immediately correlated with an increase in the VV tilt. The other study [82] tested the effect of the stimulator arrest in six individuals who received a chronic unilateral stimulation of the ventralis intermedius nucleus of the thalamus for tremor. VV was normal at baseline and altered at stimulator arrest. These two studies together with ours and some others reporting the existence of concordant VV biases and trunk tilts in PD with PS suggest that the vestibular graviception is also processed in the BG (outside the posterolateral thalamus) but to a lesser degree than somesthetic graviception.
Modulating DBS parameters to attenuate PS?
PS may occur before PD surgery [55], but in this series, developed long after for most patients. This situation could question a detrimental effect of long-term bilateral STN-DBS on the trunk orientation with respect to gravity. However, changing STN-DBS conditions did not modulate the trunk orientation, assessed in the very short term. Instead, we found that the PV tilts observed in the condition Bilateral-off were attenuated when the STN-DBS was on, either at both sides or at the side opposite to the PV biases. These latter conditions have in common stimulating the most denervated BG, possibly responsible for a contralesional PV tilt. This finding strongly suggests that the altered verticality perception observed, worse in the condition Bilateral-off, is rather a consequence of the disease course than an adverse effect of the STN-DBS.
The beneficial effect of the STN-DBS on PV tilts but not on trunk tilts is finally not so surprising. The limited number of patients in whom the PS was due to a wrong internal model of verticality explained that the STN-DBS effect obtained on PV had little chance to be transferred to the trunk in the whole series. However, Fig. 3A shows that, in some individuals (P03, P09, P13, P15, P16, P18), the trunk may have been better oriented in the Bilateral-on or unilateral-on STN-DBS than Bilateral-off condition. This was also reported in a recent single case experimental study [83]. Decreasing the voltage of STN-DBS on the side opposite to the trunk tilt rapidly attenuated the PS (from 12° to 4°), whereas the voltage increase rapidly worsened the PS. The lack of verticality perception measurements did not argue in favor or against an effect mediated by graviception modulation. In addition, PV tilts may be considered a neurobiological marker about how the brain constructs and updates the representation of the vertical. STN-DBS modulates this neurobiological marker, with a quasi-immediate effect, assessed in our study. One may assume that progressively adjusting the erect body posture onto a wrong internal model of verticality might take some time. This postural effect of a change in the somaesthetic graviception might be compensated by the capture of verticality cues from the surroundings and the awareness of body position in space [36, 44]. Thus, we cannot rule out that different temporal dynamics explain the effect on PV and not on the trunk. Finally, whether this trial provides false negative results is questionable. The short duration of the stimulation change (limited to few hours) might have not been sufficient to reveal an indirect postural effect secondary to a recalibration of the internal model of verticality. This idea agrees with the slow and often very late onset of postural disorders in PD. Perhaps it was too optimistic to expect an attenuation of the PS in the whole series in such a short time. Observations reporting changes in trunk orientation with DBS refer to the long term. Two studies have reported one patient each in whom PS was attenuated after chronic unilateral stimulation of the pedunculopontine nucleus, both after several months of stimulation [84, 85]. The stimulation was ipsilateral [84] or contralateral [85] to the PS, the cause of which was not detailed. Findings were interpreted as the result of stimulating suprasegmental parts of the BG via the bilateral and diffused projections of the pedunculopontine nucleus [84].
Future studies will have to bridge the gap between these short- and long-term effects, possibly explaining differences in the effects obtained by experimental and clinical settings.
Study limitations
This was an exploratory study with a limited sample size due to the selection of participants (PD with PS receiving long-term STN-DBS) via a monocentric recruitment imposed by a specific apparatus. The sample size was also adjusted because of the trial design, which was demanding in terms of availability for participants and examiners. It would have been interesting to specifically analyze the STN-DBS effect in a relevant subgroup of patients showing concordant trunk and PV tilts (i.e., in whom the PS was likely caused by an altered sense of upright). However, given the group’s heterogeneity, with a small subset of individuals in whom PS was secondary to a biased internal model of verticality, this was not possible without over-interpreting data. To simplify the procedure, we did not precisely quantify the spontaneous posture, in the roll plane or pitch plane. Instead, we favored a comprehensive assessment of the head and trunk lateral orientations, with and without vision, under the instruction to stand as upright as possible. It is unusual to instruct patients to stand as upright as possible when quantifying the trunk tilt of individuals with PS. However, this makes sense for examining patients in the posture they consider upright, if one wants to investigate a causal link with verticality representation and the PS. This instruction clearly reduced trunk tilts, the magnitude of which were smaller than those observed in the spontaneous posture of the patients in our study (not quantified), the usual condition in the literature [3, 12]. To limit the discomfort of participants and the duration of assessments, the number of vertical estimates were set at six per condition, the minimum ensuring sufficient reliability for verticality perception [86]. Besides a clear-cut scenario explaining PS, we found many marginal values that would have been better classified with 10 trials, given that ranges of normality slightly differed from our previous studies with 10 trials [36, 39]. These measurement biases, if any, might have underestimated the number of PS individuals with transmodal tilts of verticality representation but do not weaken our conclusion based on pronounced PV and VV tilts and clear whole-body tilts. One important limitation of the study is the lack of precise anatomical coordinates of the leads’ location, especially in the two individuals showing transmodal verticality tilts possibly at the origin of their PS. Indeed, most participants (15/18) had undergone surgery before 2010, when imaging reconstruction was not systematic. This limitation does not weaken the robustness of our conclusions; it only limits the possibility of better understanding the BG neural circuits devoted to graviception.
In conclusion, our study confirms the existence of two scenarios, one in which impaired verticality perception was secondary to the deformed body geometry due to the PS, and a second, less frequent scenario, found in only less than one third of individuals, in whom an altered graviception seemed to cause PS. Two individuals showed an outstanding clinical picture with a whole-body tilt (head and trunk) meeting the picture of the Leaning Tower of Pisa and a large transmodal verticality tilt (PV and VV) reflecting a dual alteration of vestibular and somaesthetic graviception. This clinical picture might help clinicians identify the cause of some PS and guide the treatment. STN-DBS elicited a quasi-immediate effect on PV orientation, attenuated when the most denervated BG were stimulated. This might lead to promising perspectives to recalibrate in the long term a biased internal model of verticality underpinning PS.
Footnotes
ACKNOWLEDGMENTS
We thank Pierre Pollak for the fruitful discussion that led to the working hypotheses and the study conception. The authors are grateful to all Parkinson’s disease and control participants for their willingness and their time and the associations France Parkinson and Handinnov for their support.
This work was supported by the Association France Parkinson (IPOLAP) for the neurology team, and PHRC VERTICAM for the rehabilitation team.
CONFLICT OF INTEREST
Pr. Krack reports grants from Swiss National Science Foundation, ROGER DE SPOELBERCH Foundation, Catalyst/Bertarelli Foundation, Michael J Fox Foundation, Annemarie Opprecht Foundation, Parkinson Schweiz, research grants from Boston Scientific, and Aleva, lecturing fees paid to employing institution from Boston Scientific, as well as reimbursement of travelling expenses to scientific meeting by Zambon, all support outside the submitted work.
Pr. Pérennou is Chief Editor of the Annals of Physical and Rehabilitation Medicine, consultant for IPSEN, MERZ, and Allergan in the last 5 years, without any link with the current paper.
