Abstract
INTRODUCTION
Constipation and other non-motor symptoms (NMS) are often present at the prodromal stage of Parkinson’s disease (PD), which has led to an increased interest in gastrointestinal (GI) function in this disorder [1, 2]. It has been hypothesized that PD may initiate in gastrointestinal autonomic nerve endings [3], and pathological alpha-synuclein inclusions are detectable in the GI tract up to 20 years before PD diagnosis [4]. Nevertheless, our current knowledge of the underlying causes of constipation is limited.
Constipation is most often diagnosed using questionnaires and based on the presence of specific subjectively perceived symptoms. As recently reviewed, the prevalence of subjective constipation in PD varies from 8% to 70%, probably in part caused by differing definitions of constipation across questionnaires [5, 6]. In a clinical setting, subjective constipation is important in the light of symptom treatment and relief. Yet, objective colonic dysfunction may be much more frequent than subjective constipation, and preliminary evidence suggest that subjective constipation correlates poorly with objective dysfunction [6].
The aim of the present study was to investigate colonic transit time (CTT) and volume as objective markers of colonic function in PD and to correlate these measures with subjectively perceived questionnaire-based GI symptoms and gastric emptying time (GET) measured by scintigraphy.
MATERIALS AND METHODS
Ethics statement
The study was approved by the Central Denmark Region Committee on Health Research Ethics (No. 1-10-72-255-14). All participants provided written informed consent.
Subjects
A total of 32 PD patients and 26 healthy control subjects without neurological disorders were included. For all subjects data were collected on colonic volume, olfaction, and gastrointestinal NMS according to ROME III criteria and the Cleveland constipation scoring system (CSS). In a subset of subjects (24 PD, 17 controls) we also assessed CTT, GET, and the NMS Questionnaire (NMSQuest) was administered. We had a few missing data points in the study (see supplementary Table 1). Neither PD patients nor control subjects were selected based on the presence or degree of constipation symptoms. Exclusion criteria were: systemic and gastrointestinal disorders (except constipation), prior gastrointestinal surgery, psychiatric disease, significant medical conditions, including renal, heart, or liver failure, substance abuse, allergies towards specific active ingredients, and pregnancy.
PD patients were diagnosed by movement disorder specialists according to UK Brain Bank criteria [7]. Dopamine transporter SPECT was previously performed in 17 PD patients, all were pathological. The patients received the following anti-parkinsonian medication: none (n = 1), levodopa (n = 4), dopamine agonist (n = 4), levodopa + dopamine agonist (n = 9), levodopa + MAOB-inhibitor (n = 3), levodopa + COMT-inhibitor (n = 2), levodopa + MAOB-inhibitor + dopamine agonist (n = 2), levodopa + MAOB-inhibitor + COMT-inhibitor (n = 1), levodopa + dopamine agonist + COMT-inhibitor (n = 1), MAOB-inhibitor + dopamine agonist (n = 5). The median levodopa equivalent daily dose (LEDD) in the PD group was 487.5 mg (range 0–1750 mg). Five PD patients and 1 control subject received laxative treatment, and continued this throughout the study (Psyllium Husk, Movicol, Magnesia).
Proton pump inhibitors were withdrawn >3 days prior to examinations. All examinations and questionnaires were performed in the “on” state, except the Unified Parkinson’s Disease Rating Scale part III (UPDRS-III), which was performed after >12 hours of medication abstinence.
Colonic transit time
Colonic transit time (CTT) was evaluated using the radio opaque marker (ROM) technique [8]. One capsule containing 10 ROM was ingested each morning (8 a.m.) for six consecutive days. Twenty-four hours after ingestion of the last capsule, a computed tomography (CT) was performed on a Siemens Symbia T16 SPECT/CT system (Siemens AG, Erlangen, Germany) including intravenous contrast medium (Visipaque 270 mg/mL; 2 mL/kg). All participants fasted for solid food and liquids for a minimum of 8 hours prior to the CT. Half a glass of water was allowed in the morning for ingestion of anti-parkinsonian medication.
The number of ROM in the small intestine and each colonic segment were defined on the CT scan. Total CTT in days was calculated using the following equation: (ROM(total number)+5)/10 [8].
The CT scans of all participants were visually inspected for presence of non-resorbed tablets in the GI tract to investigate whether affected gut motility can cause decreased absorption of orally ingested medication.
Colonic volume
Segmental and total colon volumes were defined on CT scans using dedicated software (PMOD Technologies, Z ü rich, Switzerland). Regions of interest (ROIs) were manually defined on each CT slice and fused, yielding 3D volumes of interest (VOIs) of the caecum, ascending, transverse, descending, and rectosigmoid colon (Fig. 1). The descending-to-rectosigmoid transition was defined at the axial level of the sacral promontory. Final absolute segmental volumes were calculated by subtracting gas volume, segmented in the VOIs at a –300 Hounsfield CT units (HU) threshold.
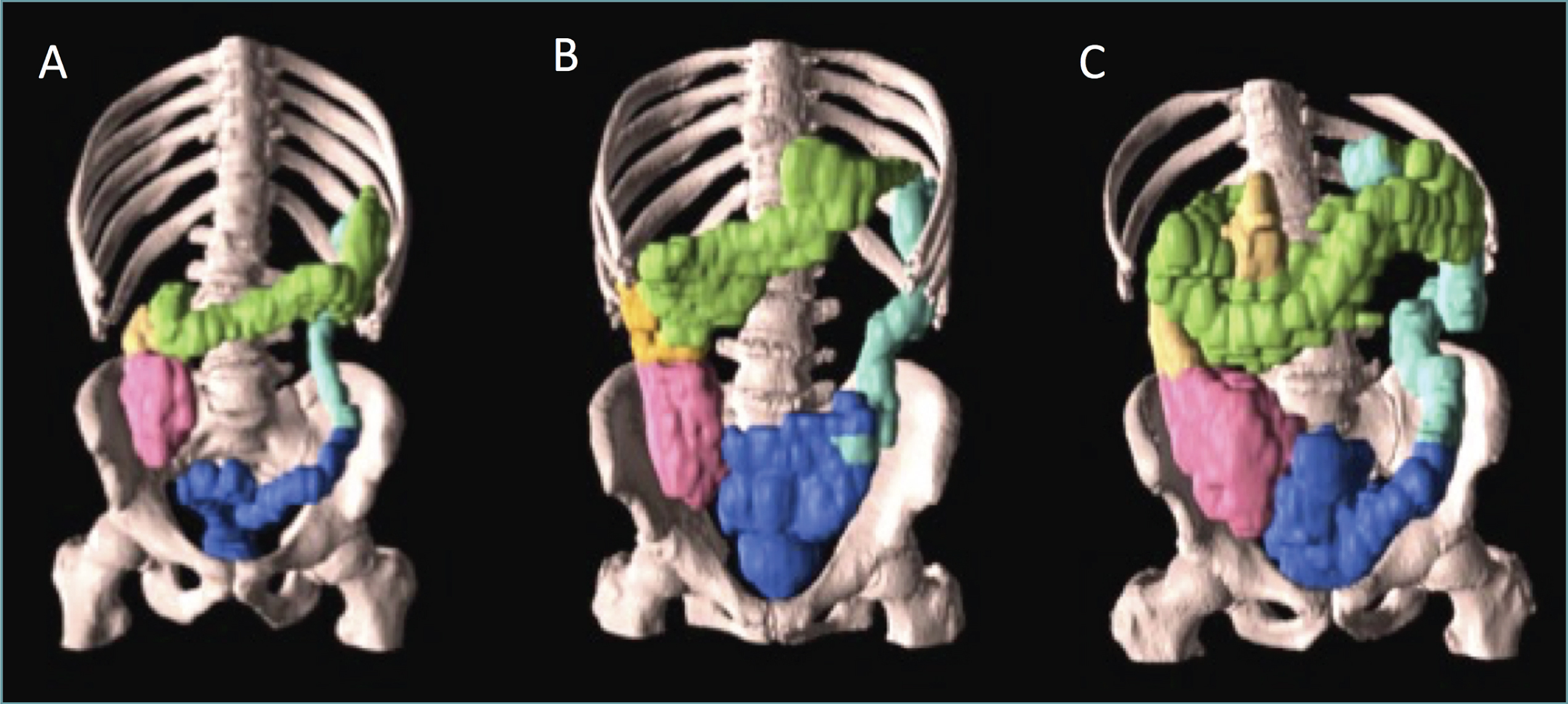
Colonic volume in a healthy control subject (A), a typical PD patient (B), and a PD patient with severely increased volume (C). Red indicates caecum, yellow ascending, green transverse, light blue descending, and dark blue recto-sigmoid part of the colon.
Gastric emptying
Gastric emptying time (GET) was scintigraphically evaluated after an overnight fast. Participants ingested a standardized solid meal of 120 mL egg white omelet containing 19–37 MBq 99mTC-labelled colloid, two slices of white bread, 30 g of jam, and 120 mL water in maximum 10 minutes [9]. Summed abdominal one-minute images were performed on a Picker Marconi Axis dual-head SPECT camera (Picker International, Cleveland, Ohio) in sitting position at 0, 30, 60, 90, 120, 150, and 180 minutes after meal ingestion and semi-automatically analysed using Hermes software (Hermes Medical, Stockholm, Sweden). Total counts in ROIs outlining the stomach were defined on each image, and the exponential half-emptying time (T1/2) was estimated from time-activity curves.
Questionnaires
Upper GI symptoms and presence and severity of constipation were evaluated using the ROME III nausea-vomiting and ROME III functional constipation questionnaires [10]. ROME III defines constipation as the presence of two or moresymptoms at least 25% of the time for >3 months with onset at least 6 months prior to testing (straining for defecation, lumpy/hard stools, sensation of incomplete evacuation, sensation of anorectal obstruction, manual maneuvers, and <3 bowel movements per week), loose stools rarely present without laxative, and insufficient criteria for irritable bowel syndrome (IBS). Of note, IBS is a diagnosis of exclusion, in which no well-defined disease entity can be identified as causative of constipation. Thus, we elected to exclude the IBS criterion in the ROME definition of constipation. Constipation were also diagnosed by the Cleveland constipation scoring system (CSS) [11], where a cut-off score >15 was used to define constipation. Finally, NMS symptoms in general, including constipation, were evaluated by the non-motor symptoms questionnaire (NMSQuest), defining constipation as <3 bowel movements per week and/or straining during the past month [12].
Clinical assessment
Motor function and clinical stage was evaluated in 30 of 32 PD patients using UPDRS part III [13] and the Hoehn and Yahr scale (H&Y) [14]. Cognitive function was assessed by the Mini Mental State Examination (MMSE) [15] and olfactory function with the Sniffin’ Sticks 16-item identification test (Burghart, Wedel, Germany) [16].
Statistics
Statistical analyses were performed using STATA 13 (College Station, TX: StataCorp LP) and Prism 6 (GraphPad Software, La Jolla, USA). Demographic and clinical data were tested using unpaired two-way t-tests or equivalent non-parametric tests as appropriate. Differences between groups in both segmental number of ROM and segmental volumes were analyzed by two-way ANOVA with post hoc multiple comparison correction. Multiple regression analyses were performed to evaluate associations between colonic volume and sex, weight, body mass index (BMI), body surface area (BSA), and lean body mass (LBM). Correlations between objective markers and individual questionnaire items (i.e. ordinal data) were tested using Spearman rank correlation. Total questionnaire scores was considered to approximate interval data and tested with Pearson correlation. Receiver-operating characteristic (ROC) analyses were performed to determine the optimal colonic transit and volume cut-off thresholds for separating PD patients from controls.
RESULTS
Demographic and clinical data are shown in Table 1. No between-group differences were seen in cognitive performance or in gastric emptying time (Fig. 2). The PD group displayed significantly decreased olfaction and significantly higher rates of constipation and NMS symptoms in general. Medication residues were identified in the small intestine of only two PD patients.
Demographic and clinical data of patients with Parkinson’s disease (PD) and healthy controls
Data given as mean±SD or median (range). *n = 24 PD and 16 controls. **n = 22 PD and 17 controls. Abbreviations: BMI = Body Mass Index, MDS-UPDRS = Movement Disorder Society-Unified Parkinson’s Disease Rating Scale, MMSE = Mini Mental State Examination, NMSQuest = Non-Motor Symptoms Questionnaire, CSS = Cleveland Constipation Scoring System.
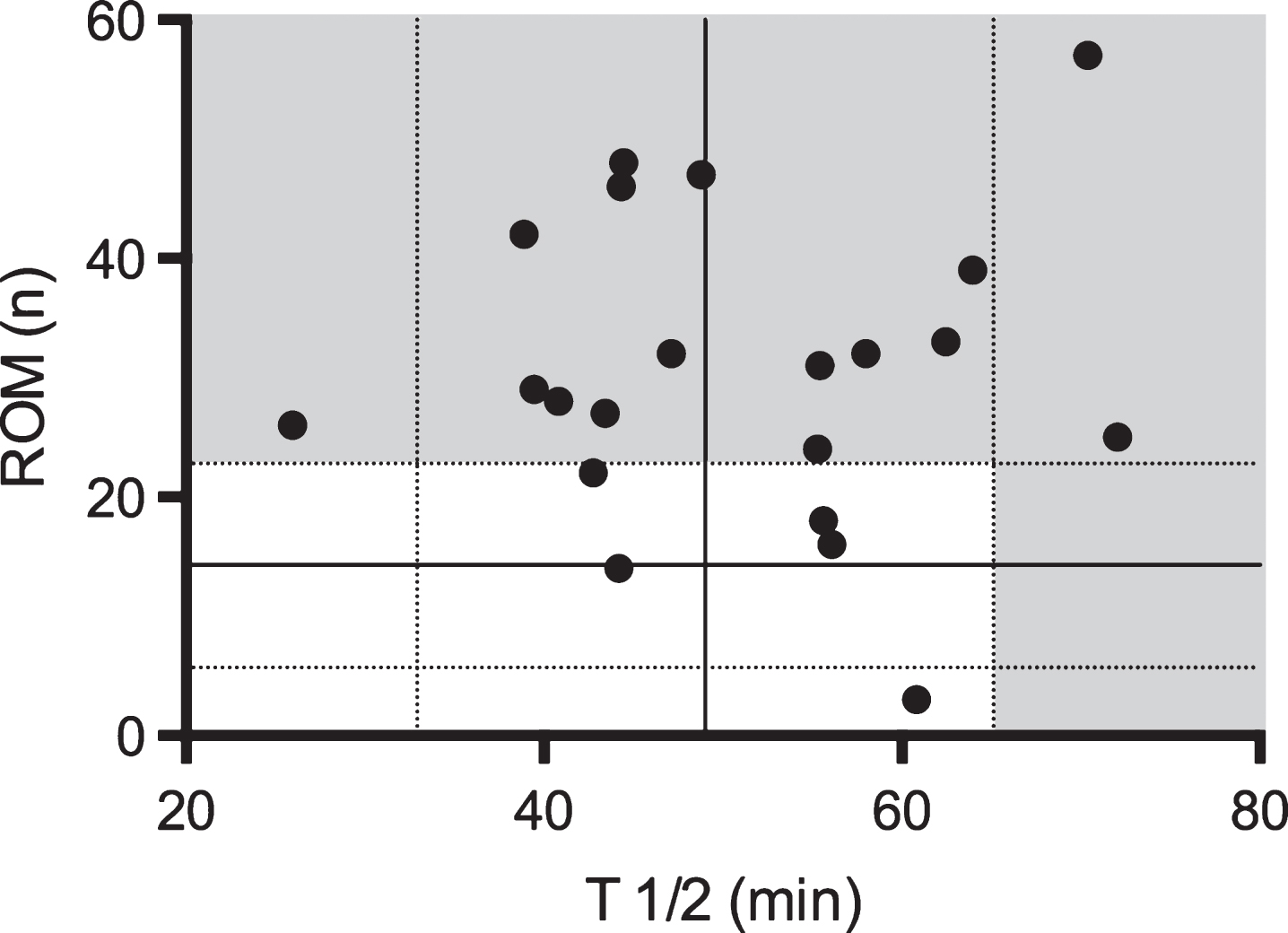
Plot of gastric emptying time given as T½ and number (n) of retained Radio Opaque Markers (ROM) in 21 PD patients, who underwent both examinations. Solid lines denote mean values and dashed lines +/–1SD for the control subjects. Gray areas indicate > 1SD from control mean.
Colonic transit time
Total number of retained ROM was significantly higher in the PD group (p < 0.0001; Fig. 3A) corresponding to significantly longer total CTT (Table 1). The difference was most significant in the transverse and rectosigmoid colon (p < 0.0001; Fig. 3B). Furthermore, ROMs were present in the small intestine of 5 PD patients, indicative of delayed oro-caecaltransit.
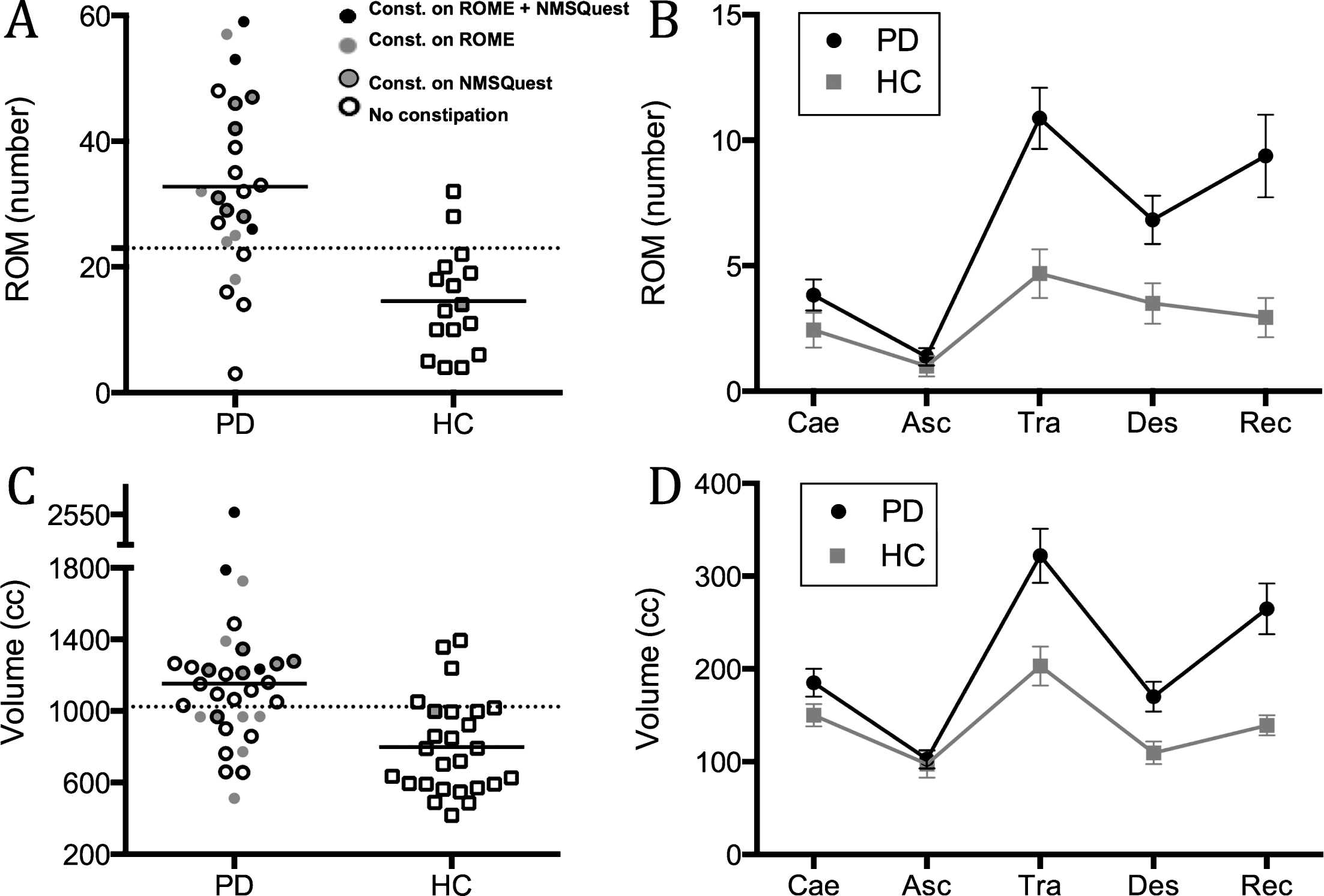
Based on ROC analyses, a cut-off of 23 ROM was optimal and yielded sensitivity of 79%, specificity of 88%, and a positive likelihood ratio (LR+) of 6.3 for separating the groups (Fig. 3A; dashed line).
Colonic volume
Representative 3D colon volumes are depicted in Fig. 1. Only sex was significantly associated with colonic volume, and volume data were therefore corrected for sex prior to further analyses.
Total colonic volume was significantly larger in the PD group (p < 0.0001; Fig. 3C). Significant between-group differences were seen in the transverse and rectosigmoid colon (p < 0.0001; Fig. 3D). ROC analyses yielded an optimal total volume threshold of 1024 cc translating into a sensitivity of 66%, specificity of 85%, and a positive likelihood ratio (LR+) of 4.3 for separating patients from controls (Fig. 3C; dashed line).
Correlations and questionnaires
No correlations were seen between UPDRS motor score and total number of ROM (p = 0.74) or total colonic volume (p = 0.16), or between disease duration and ROM (p = 0.30) or total volume (p = 0.0057). Also, gastric emptying time did not correlate with total number of ROM (p = 0.90; Fig. 2) or total colonic volume (p = 0.70). LEDD correlated positively with total volume (p = 0.0002) but not with total number of ROM (p = 0.079).
Positive correlations were seen between number of ROM and colonic volume in ascending (p = 0.037), transverse (p = 0.012), descending (p < 0.0001), rectosigmoid (p < 0.0001), and total colon (p < 0.0001) in the PD group and in transverse (p = 0.003), descending (p = 0.009), rectosigmoid (p = 0.040), and total colon (p = 0.0003) in the control group.
ROME III
According to ROME criteria, 32% of patients and 0% of controls were constipated. Each questionnaire item grades constancy of the symptom in question. Adding these to a total value, a positive correlation was seen with total colonic volume (p = 0.002) but not with total number of retained ROM. For question 9–15 values, i.e. questions specifically targeting colonic and rectoanal function, a significant correlation was seen with total volume (p = 0.0008), and a near-significant correlation to total ROM (p = 0.054). Question 9 (<3 bowel movements per week) correlated significantly with descending (p < 0.0001), rectosigmoid (p = 0.012), and total volume (p = 0.0010). Question 10 (hard stools) correlated with both total ROM (p = 0.031) and total volume (p = 0.0003), the latter highly driven by the rectosigmoid part (p = 0.0010). Question 11 (straining) correlated with total ROM (p = 0.013) and total volume (p = 0.0048), in both cases most distinct in the rectosigmoid segment. Correlations are graphically depicted in supplementary Figure 1.
NMSQuest
Constipation was present in 38% of PD patients and 6% of controls, using the NMSQuest definition. Importantly, the ROME and NMSQuest criteria showed highly discordant results in defining subjects as constipated or not (Fig. 3). The NMSQuest total score (including all NMS) correlated significantly with total colonic volume (p = 0.024). Question 5 (<3 bowel movements per week or straining) and 7 (incomplete emptying) each correlated with total colonic volume (p = 0.012 and p = 0.0067). None of the two questions correlated significantly with total ROM. Correlations for NMSQuest, CSS, and objective markers are shown in supplementary Figure 2.
CSS
Based on CSS criteria, constipation was seen in 3% of patients and 0% of controls. The CSS total score correlated with total ROM (p = 0.031) and total volume (p = 0.0006).
DISCUSSION
In the present study, 79% of early-stage PD patients had significantly increased CTT and 66% showed increased colonic volume. In colon sub-segments, virtually identical patterns of significantly increased volume and retained ROM were seen in the transverse and rectosigmoid colon. Applying three different questionnaires, 32%, 38%, and 3% of PD patients were defined as constipated, respectively.
Colonic transit time
To our knowledge this is the first study to investigate colonic volume using CT and correlate results with the widely used ROM technique as a measure of CTT. It may also be the first published data evaluating retained ROM on an abdominal CT scan, which allows precise definition of the specific number of ROM in each colonic segment. Given the highly variable colonic anatomy, this is considered an advantage in order to establish more exact estimations of segmental transit time as well as the ability to evaluate slow transit constipation vs. outlet constipation, and the presence of ROM in the small intestine.
The difference in CTT was more pronounced in the transverse, descending, and rectosigmoid segments of the colon, indicating that early stage PD patients exhibit a combination of slow transit and outlet constipation [17]. This observation is in line with previous results from transit time studies of PD patients. One study reported evenly distributed ROM throughout the colon and rectum and another study reported significantly increased rectosigmoid transit time in PD patients [18, 19]. However, these studies used plain x-ray methodology.
Colonic volume
In the present study, segmental volumes were significantly increased in the PD patients, specifically in transverse and rectosigmoid segments. This finding suggests combined rectosigmoid outlet obstruction caused by dystonia of the external anal sphincter and generalized slow transit time, probably as a result of damage to the enteric and parasympathetic nervous system. A previous study of opioid-induced constipation found pathologically increased volume also in the caecum and ascending colon, indicating presence of more universal slow CTT as compared to sphincter dystonia and decreased rectal emptying [20]. This supports the probable combination of outlet obstruction and slow transit time in PD patients in the present study.
Gastric emptying
In contrast to the high prevalence of delayed CTT in PD patients, no difference was seen in GET between the PD group and controls, implying that colonic dysfunction is more severe in early stage PD patients compared to gastric dysmotility (Fig. 2). Previous studies have demonstrated a colo-gastric reflex, i.e. decreased gastric emptying as a consequence of rectal distension [21, 22]. However, the present data does not support that the colo-gastric reflex is of clinical importance in early-stage PD.
Previously published GET data in PD patients employed differing methodologies and reported equivocal results, which was recently reviewed in detail [23]. Some studies used similar gold standard scintigraphic methods as the current study. Two studies reported significantly prolonged GET in PD patients, although the data showed wide ranges. The remaining studies found no significant difference between PD patients and controls. In contrast, studies using the alternative 13C-based breath test more often show delayed GET in PD patients. Since the 13C-breath tests rely not only upon mechanic gastric emptying, but also small intestinal absorption and liver metabolism, it remains controversial to what degree gastric emptying is delayed in early-stage PD.
Questionnaires
Concerning subjective constipation symptoms and their relationship to objective colonic dysfunction, four important observations emerged. First, objective colonic dysfunction was considerably more prevalent than subjective constipation, and thus will be markedly under-diagnosed by simply enquiring the patient about subjective symptoms. This discrepancy is in line with other non-motor PD symptoms such as hyposmia. Only 50% of PD patients report subjective hyposmia, whereas 80–90% exhibit objectively decreased olfaction on smell tests [24, 25]. Second, correlations between subjective symptoms and CTT were moderate (0.3 <r<0.5) and slightly stronger with colonic volume (r > 0.5). Third, objective colonic measures showed better correlation with straining and hard stools as compared to decreased defecation frequency. This implies that the most commonly used definition of constipation (<3 bowel movements per week) is not the best predictor of measurable colonic dysfunction, and more attention should be devoted to enquiring patients about straining and stool consistency. Fourth, the three different constipation questionnaires yielded different prevalence estimates of constipation, ranging from 3% to 38% in PD patients. Importantly, different constipation definitions did not categorize the same patients as being constipated (Fig. 3).
In summary, objective colonic dysfunction was more prevalent than subjective symptoms and correlations between the two were not convincing. This discrepancy is supported by a recent study of 29 highly selected PD patients, all of whom were constipated (ROME III criteria) and 78% displayed prolonged CTT [26]. In our study, only 32% were constipated using ROME criteria, but 79% still showed prolonged CTT.
Malabsorption
Previous studies reported non-absorbed tablets on CT scans as high-density material in the stomach [27]. In the present study, non-absorbed tablets were identified in the small intestine of only two PD patients. No residues were seen in the stomach. This negative finding goes against the commonly held notion that malabsorption of medication is a common problem in PD. However, CT scans may lack sensitivity to reliably detect partially (but insufficiently) resorbed tablets. Alternatively, lack of observed tablets could be explained by differences in tablet radiopacity [28].
Limitations
Some limitations need to be addressed. Colonic VOIs were defined manually on CT scans, potentially introducing user-dependent bias in volume estimation. However, our control data was in line with previously published healthy control data from a large scale colon volume study [29].
Five PD patients received laxatives during the examinations, which may have affected the results. Nevertheless, laxatives would result in a more rapid CTT and probably decreased colonic volume, and so bias the results toward the null. Standardized food and beverage intake were not applied, which may have affected the results. However, the purpose was to evaluate GI function under normal everyday conditions, and study-induced changes in eating habits or physical exercise would have compromised generalizability to the clinical setting.
All patients but one received anti-parkinsonian treatment during the study, potentially influencing gastrointestinal function. Indeed, our finding of a positive correlation between LEDD and colonic volume (but not CTT) suggests that dopaminergic medication may excacerbate constipation. The present results should be further verified in de novo untreated PD patients.
The CSS and ROME III questionnaires have not been validated in PD patients, and the summed ROME III item values have also not been validated for use as an overall constipation severity score.However, these values were used exploratively in the present study to test correlations between subjective symptoms and objective markers.
Conclusion
Significantly delayed CTT and increased volume were seen in early-to-moderate stage PD patients compared to controls. The difference was most distinct in the transverse and rectosigmoid colonic segments. The diagnosis of subjective constipation relied heavily upon which questionnaire was employed, and different questionnaires did not define the same patients as being constipated. Objectively measured colonic dysfunction was far more prevalent than symptom-based constipation. The subjective symptoms showed modest correlations with CTT, and stronger correlations with colonic volume. In conclusion, more research is needed to elucidate the relationship between objective colonic dysfunction and subjective constipation.
FINANCIAL DISCLOSURES/CONFLICTS OF INTEREST
None related to this study.
All other authors have no disclosures to declare.
Footnotes
ACKNOWLEDGMENTS
This work was supported by Parkinsonforeningen, Denmark
