Abstract
Background:
Spinal muscular atrophy (SMA) is a genetic neurodegenerative disorder with onset predominantly in infants and children. In recent years, newborn screening and three treatments, including gene replacement therapy (Onasemnogene abeparvovec-xioi), have become available in the United States, aiding in the diagnosis and treatment of children with SMA.
Objective:
To evaluate parents’ experiences with newborn screening and gene replacement therapy and to explore best practices for positive newborn screen disclosure and counseling of families.
Methods:
We conducted semi-structured interviews (n = 32) and online surveys (n = 79) of parents whose children were diagnosed with SMA (on newborn screening or symptomatically) and treated with gene replacement therapy.
Results:
Gene replacement therapy was most parents’ first treatment choice, although concerns regarding long term efficacy (65%) and safety (51%) were common. Information provided during the newborn screening disclosure was quite variable. Only 34% of parents reported the information provided was sufficient and expressed need for more information about treatment. Although many parents experienced denial of the diagnosis at initial disclosure, 94% were in favor of inclusion of SMA on newborn screening. Parents were almost universally anxious following diagnosis and over half remained anxious at the time of study participation with uncertainty of the future being a key concern. Many parents had difficulty processing information provided during their first clinic appointment due to its complexity and their emotional state at the time.
Conclusions:
Utilizing this data, we provide a recommendation for the information provided in newborn screening disclosure, propose adjustments to education and counseling during the first clinic visit, and bring awareness of parents’ mental health difficulties.
INTRODUCTION
Spinal muscular atrophy (SMA) is an autosomal recessive neurodegenerative disease that affects approximately 1 in 5,000 to 1 in 11,000 individuals. The carrier frequency for the common exon 7 deletion is 1–3%, based on ancestry [1–7]. Prior to the availability of treatment, SMA was the most common genetic cause of death in children under the age of two. Due to the high disease incidence and a common deletion accounting for approximately 95% of cases, SMA was suggested as an addition to the recommended uniform screening panel (RUSP) in 2008. However, this was denied due to the lack of pilot data showing feasibility to screen for SMA utilizing available technology and treatment options. From January 2016 to January 2017, a pilot study of over 3,800 newborns in New York City demonstrated feasibility of utilizing multiplex real time PCR on dried blood spot cards to detect homozygous deletions of SMN1 as a means of newborn screening (NBS) for SMA. The study also showed general population acceptance of the screening with 93% of parents opting in [2, 8]. In December 2016, Nusinersen became the first disease modifying treatment approved by the Food and Drug Administration (FDA). Based on these two changes, SMA was added to the RUSP in July 2018. A positive NBS for SMA is highly specific for disease status; however, confirmatory testing, including SMN2 copy number, is required prior to treatment [2, 9–12]. Newborn screening for SMA is available in 48 of the United States (accounting for 98% of infants born) as of January 2023 [13]. Since the addition of SMA to the RUSP, two additional therapies have been FDA approved –Onasemnogene abeparvovec-xioi in May 2019 and Risdiplam in August 2020.
Outside the United States, NBS for SMA is also available in regions in Taiwan, Germany, Belgium, Australia, Italy, Russia, Canada, and Japan. This accounts for approximately two percent of all newborns worldwide [14]. As of January 2023, Onasemnogene abeparvovec-xioi has been approved for use in 45 countries [15].
Previous studies show individuals and families with SMA, the general population, and expectant parents with no history of SMA all supported NBS for SMA even prior to approval of disease modifying therapies [16–20]. However, because both NBS and gene replacement therapy are recent advances, limited research exists assessing parental experiences surrounding these interventions. Experiences with Onasemnogene abeparvovec-xioi, rather than other treatment options, were of interest given it is the first FDA approved systemic gene replacement therapy and the first long acting treatment option for SMA. The goal of this study was to describe parents’ experiences with NBS and gene replacement therapy and to explore best practices for positive newborn screen disclosure and counseling of families.
MATERIALS AND METHODS
Participants
Parents of children diagnosed with SMA who received Onasemnogene abeparvovec-xioi were recruited between July 2021 and August 2022. Children could be diagnosed symptomatically or pre-symptomatically by NBS. Recruitment occurred through the Nationwide Children’s Hospital (NCH) Neuromuscular Clinic and/or CureSMA outreach efforts.
Study design
This was a two-part study, with a semi-structured interview and an online Redcap survey. Interviews and surveys were only offered in English. Information collected in both parts included parents’ experiences and reactions to the NBS process (if applicable), first neuromuscular clinic appointment, treatment decisions and administration experience, and outlook on their child’s diagnosis post-treatment. IRB approval for the study was obtained from Nationwide Children’s IRB. Consent was obtained verbally for interviews and online for surveys.
Interviews were offered to parents of patients cared for in the neuromuscular clinic at NCH. They were performed via phone or during clinic appointments. Separate interview guides were developed for parents of children diagnosed on NBS and those diagnosed symptomatically, with input from all senior authors.
Surveys were completed by families identified at NCH and through CureSMA. Responses utilized 5-point Likert scales, multiple choice and open ended questions. Eight questions were added to the survey after study initiation to solicit additional details of newborn screen disclosure and genetic counseling involvement in patient care. A minimum of 42 responses were analyzed for each question.
Data analysis
Thematic coding of verified interview transcripts was conducted by four study team members. Conventional content analysis using a constant comparative approach was used with subsets of interviews within each group. Sequential coding team discussions were used to determine consensus of themes and exemplar quotes. Coding was conducted separately for parents of children diagnosed symptomatically (Symptomatic group) and by NBS (NBS group). Survey results were analyzed via comparative statistics. Chi square analyses were utilized to investigate factors influencing parents’ responses to NBS disclosure and anxiety related to diagnosis at the time of survey completion.
RESULTS
Survey results
The survey was sent to 338 individuals and 79 complete responses were received; a response rate of 23%. Parents were mostly mothers (82%), white (84%), had a bachelor’s or master’s degree (51%), and considered themselves at least somewhat religious (72%). Twenty-nine (36.7%) had children diagnosed symptomatically and 50 had children diagnosed on NBS (63.3%). Six parents (7.6%) had multiple children with SMA, seventeen (22%) were aware of their carrier status of SMA prenatally, and eight obtained a prenatal diagnosis of SMA (10%). The full demographic information of the survey participants is provided in Table 1.
Demographics of survey participants
*Values may be larger than number of patients in the cohort due to use of multiple treatments in some patients.
Newborn screening
A total of fifty (63%) parents had children diagnosed on NBS. Thirty-eight (76%) reported being aware that their child was receiving NBS. Nine (18%) were aware that SMA was included on this screening, all of whom were either known carriers (n = 8) or had an older child with SMA (n = 1). Pediatricians most commonly disclosed the positive NBS result (71%, n = 30) followed by neurologists (12%, n = 5), genetics providers (12%, n = 5) or OBGYNs (5%, n = 2). Information provided in the disclosures included the name of the diagnosis for 95% (n = 40), symptoms of SMA for 60% (n = 25), existence of treatments for 57% (n = 24), and details of treatment options for 40% (n = 17).
Only 42% (n = 21) of parents felt that the NBS disclosure allowed them to understand the diagnosis. Twenty-eight (56%) felt the disclosure helped them understand the necessary next steps in their child’s healthcare.
Parents who were well informed about symptoms of SMA, treatment availability, and details of treatment options reported better understanding their child’s NBS result, diagnosis, and next steps required for their child’s medical care. These comparisons are summarized in Table 2. In addition, parents who received information in more of these categories reported better understanding of the NBS results (p = .004), diagnosis (p = .003), and next steps in care (p = .003). Disclosing provider (pediatrician vs non-pediatrician) did not impact the inclusion of diagnosis name (p = .49), symptoms of SMA (p = .55), existence of treatment (p = .92) or details of treatments (p = .43) in the NBS result call. Provider type did not impact parents’ understanding of the results (p = .09), diagnosis (p = .31) or next steps (p = .49). The majority of parents (88%) were anxious following the disclosure, regardless of amount of information provided (p = .16) or disclosing provider (p = .30). Despite this, parents were generally glad that their child had NBS for SMA (94%, n = 47) and felt that it positively impacted their child’s health (94%, n = 47).
Factors influencing parents’ response to NBS disclosure
*Due to low incidence of responses conveying a lack of anxiety at disclosure, ‘disagree’ and ‘neutral’ responses were combined into a single category.
Treatment with gene replacement therapy
The majority of parents in both groups (Symptomatic, N = 29 and NBS, N = 50) felt hopeful following discussion about available treatment options (80% Symptomatic; 94% NBS). Most felt that gene replacement therapy would improve their child’s symptoms (84% Symptomatic; 94% NBS), but understood that it is not a cure for the condition (72% Symptomatic; 62% NBS). Parents often had safety concerns related to gene replacement therapy (58% Symptomatic; 46% NBS). Although almost all felt grateful that their child received gene replacement therapy (97% Symptomatic; 98% NBS), the majority did worry about the possibility of its effectiveness “wearing off” (58% Symptomatic; 68% NBS).
Impact of diagnosis on parent and outlook on future
Parents from both groups (Symptomatic, N = 29 and NBS, N = 50) endorsed feeling scared (93% Symptomatic; 92% NBS) and anxious (100% Symptomatic; 96% NBS) following their child’s diagnosis of SMA. Although fewer reported these feelings at the time of study, the majority still expressed these emotions (62% Symptomatic, 50% NBS scared; 86% Symptomatic, 52% NBS anxious).
Parent age (p = .87), sex (p = .52), and education level (p = .44) did not impact current anxiety. Parents with children who were unable to sit (p = .002) or walk independently (p = .001) or who required use of a wheelchair (p = .002), feeding support (p = .03) or respiratory support (p = .03) were more likely to report anxiety at the time of survey. Parents in the NBS group were more likely to report anxiety if their child was younger (p = .04). These findings are summarized in Table 3.
Factors about child influencing parental anxiety at time of study
A small proportion of parents felt that the diagnosis of SMA made it difficult to bond with their child (13% Symptomatic; 14% NBS) and/or felt disconnected from their child (10% Symptomatic; 14% NBS). Twenty-one (42%) in the NBS group felt that ‘receiving the diagnosis so early’ prevented them from enjoying the newborn period with their child.Most agreed that the diagnosis of SMA was serious (65% Symptomatic; 66% NBS), but did not think that their child’s current health status was serious (24% Symptomatic; 14% NBS). When asked about their child’s future, more parents in the NBS group thought that their child would have a “normal life” (74%) compared to the Symptomatic group (38%), but similar proportions felt that their child would have a “meaningful life” (93% Symptomatic; 90% NBS) and felt hopeful for their child’s future (93% Symptomatic; 92% NBS).
Care and counseling related to SMA
Parents were happy with the clinical care their child receives for SMA (100% Symptomatic; 96% NBS) and were trusting of their child’s care team (100% Symptomatic; 96% NBS). Both groups understood the need for continued medical care related to their child’s SMA diagnosis following treatment (100% Symptomatic; 94% NBS).Regarding counseling following diagnosis, parents generally felt the amount of information provided for disease (79% Symptomatic; 84% NBS), testing (69% Symptomatic; 78% NBS), genetics (69% Symptomatic; 88% NBS), and treatment (83% Symptomatic; 92% NBS) was appropriate. Timing of information was mostly thought to be appropriate for disease, genetics, and treatment information (83–86% Symptomatic; 82–90% NBS) or later than desired (14–17% Symptomatic; 10–18% NBS). Regarding information about testing for SMA, 28% of parents in the Symptomatic group felt this came too late. Verbal communication was felt to be most useful for all topics (79-86% Symptomatic; 76-88% NBS), but a large proportion also agreed that written (55–69% Symptomatic; 66–76%) or visual (52–62% Symptomatic; 40–58% NBS) formats would be helpful. More parents in the symptomatic group (97%, n = 28) utilized the internet to learn about SMA than in the NBS group (78%, n = 39) and more often found it to be useful across all topics (62–73% Symptomatic; 44–56% NBS).Genetic counseling was more commonly received by parents in the NBS group (72%, n = 36) than the Symptomatic group (59%, n = 17). This usually occurred at an appointment immediately following diagnosis (58%, n = 21) or at a separate appointment prior to gene replacement therapy (28%, n = 10). Most parents felt that meeting with a genetic counselor immediately following diagnosis would be the most beneficial time (77%, n = 44). Genetic counseling was seen as most useful in assisting in the understanding of diagnosis (82% Symptomatic; 83% NBS), and risk for themselves (76% Symptomatic; 92% NBS) or family members (88% Symptomatic; 83% NBS) to be carriers of SMA. To a lesser extent, parents felt it was helpful in understanding treatments (59% Symptomatic; 64% NBS).
Thematic analysis of qualitative interviews
Thirty-two parents (of twenty-six children) completed the interview process. Twenty-five (78.1%) were mothers and seven (21.9%) were fathers. Of these, eighteen (56.3%) were parents of children diagnosed on NBS and thirteen (40.6%) were parents of children diagnosed symptomatically. Nineteen had children (59.4%) with 2 copies of SMN2, ten (31.3%) with 3 copies, and three (9.4%) with four copies. One parent had two children with SMA, one in the NBS group and one in the symptomatic group, and both experiences were discussed during the interview. Two parents in the NBS group had a previous child who had died of SMA, but were interviewed with respect to their living child. One couple in the Symptomatic group were identified as carriers for SMA prenatally. Full demographic information for the cohort studied is provided in Table 4.
Demographics of interview participants
*Values may be larger than number of patients in the cohort due to use of multiple treatments in some patients.
Receiving symptomatic diagnosis (symptomatic group)
Parents of symptomatic children described a lengthy diagnostic process and had to fight for appropriate referrals and testing. A variety of factors led to delays in diagnosis including lack of provider familiarity with SMA, wait times for specialty appointments, and receiving incorrect diagnoses prior to their SMA diagnosis.
And I was like, well, can we test for SMA?...I feel like he’s got all the symptoms . . . And he said, ‘oh, I don’t think that’s necessary . . . kids with SMA, they are they’re way sicker than him. They struggle to breathe. They can’t hold their head up’ . . . He goes, ‘I would be very surprised if he has SMA.’ And he was, I mean, very adamant that it wasn’t that. So I was like okay. I guess that’s a sigh of relief, it’s not that. (Symptomatic, Mother)
Receiving newborn screening diagnosis (NBS group)
Excluding one, all parents were aware that their child had NBS completed and recall the sample being collected. None understood the types of diseases included on the screening. Only parents with prior children diagnosed with SMA were aware that SMA was included on the screening.
We just knew that there was a general . . . newborn screening that they did at the hospital. But we didn’t know that they were screening for anything like that. We had no idea. (NBS, Mother)
Parents received the positive NBS result from a variety of providers, most commonly pediatricians. The amount and type of information disclosed was quite variable. Parents generally wished more information had been provided in the initial phone call, with many specifically desiring more discussion about treatment.
She said that we got some results back that may be concerning, and that they wanted me to take her immediately the next day to get tested again to be sure that it was positive or not, but they didn’t tell me what it was because they didn’t want me to worry. But that made me worry more because I had no idea what was going on with her. They just said it was really urgent and life threatening, so that scared me. (NBS, Mother)
And I think you got to just be able to just say, hey, you know, here are some options that are available . . . Here’s what our next steps will be. Here’s how you can start working towards what we’re hopeful for. You know, there was none of that . . . I’m sure if they were to reflect on that, they would have said they would agree. But, you know, certainly offer hope, offer solutions, not just like a reign of terror kind of thing. (NBS, Father)In some cases, the information provided to families was outdated or inaccurate, often leaving parents with a bleak impression of their child’s future.I do remember her saying . . . that children who normally are born with this, either they can’t walk, they can’t crawl, they can’t eat. Most of them are wheelchair bound. And she said some of them are like a vegetable. And I remember her saying that . . . I broke down...you can’t tell a parent that. (NBS, Mother)
Parents’ opinions on NBS for SMA (Both groups)
Counseling for parents following SMA diagnosis (Both groups)
Treatment with gene replacement therapy (both groups)
Impact of diagnosis on parent and outlook on the future (both groups)
DISCUSSION
In this cohort, gene replacement therapy was parents’ first choice for treatment, similar to the findings of Deng et al. performed in the United States [21]. Parents voiced similar rationale for this choice: one time dosing, less invasive administration route, and perceived better outcomes compared to the other treatment options [21]. Despite this, about half of survey respondents reported safety concerns with the therapy, including its novelty, side effects, young age at treatment, and concerns about the procedure.
After receiving gene replacement therapy, parents in the NBS group experienced anxiety while awaiting signs that the therapy was working, leading to increased watchfulness of their child. This lessened with the emergence of normal motor milestones. Parents in the Symptomatic group were more quickly reassured by improvements seen in their children’s motor function following treatment.
A theme of cautious hope emerged regarding parents’ views of their child’s diagnosis and future following gene replacement therapy. Lack of long term outcome data and uncertainty in their child’s future health were the most common concerns voiced by parents. This finding was similar to the U.S. study and a similar study conducted in Australia, where the majority of parents (65%) had concerns related to the treatment longevity [21, 22].
Newborn screening for SMA was highly supported in our cohort, with 94% of parents glad that their child was screened. Many parents cited the importance of early diagnosis and treatment in best outcomes for their child. These findings were consistent with both the Australian and U.S. studies, where 100% and 94.4% of the parents were satisfied with the program, respectively [21, 22]. Additionally, a major theme in the Symptomatic group was the significant delays experienced in obtaining their child’s SMA diagnosis, which has also been shown in prior studies, highlighting the need for NBS for efficient diagnosis and treatment [23, 24].
In both our cohort and the Australian cohort, only 76% of parents were aware that their child had NBS. Additionally, most were unaware of the types of conditions that were screened [22]. Lack of parental knowledge and education about NBS is an issue that has been highlighted in other studies and has been associated with increased shock and confusion for parents receiving a positive screen [25–27].
Only 34% of parents felt the information provided in the NBS disclosure was sufficient, a smaller proportion compared to the Australian study, where 59% felt the information provided was acceptable [22]. Insufficient depth of information in disclosure and perception that providers are uninformed about the diagnosis they are disclosing have been described previously for other NBS conditions [26–30]. Parents participating in interviews specifically wanted more information about treatments during the disclosure; an important finding given only 40% were given details of treatment options. In our study, parents receiving more information in the NBS disclosure reported better understanding of the NBS result, diagnosis, and next steps in care.
Parents in the NBS group commonly reported denial and disbelief in their child’s diagnosis due to lack of visible symptoms. This, coupled with inaccuracies in information provided during initial disclosure, led to inflated hope that the NBS was a false positive or that their child may be an unaffected carrier. The hope for false positive in our study was also demonstrated in the Australian study, where 48% of parents hoped for a false positive and reported disbelief that their healthy appearing child could have SMA [22]. Shock and disbelief have also been commonly reported in parents receiving a positive NBS for other diagnoses [26, 29–31].
Following NBS disclosure, most parents felt anxious and scared, similar to the findings in the Australian study where 83% reported anxiety [22]. Anxiety immediately following NBS disclosure was not relieved by information provided in the call or type of provider giving the information. Disclosing providers should anticipate that anxiety in this time period is universal. Until this phone call, parents see their child as healthy. This call shatters that perception. Our goal should not be to alleviate these emotions immediately, but rather to support families as they grapple with this transition.
While many parents reported anxiety reduced with time, over half were still anxious at the time of taking the survey. We found that parents with more symptomatic children (with lower motor function or need for respiratory or feeding support) were more likely to report ongoing anxiety. Additionally, parents with children diagnosed on NBS were more likely to report anxiety if their child was younger, likely due to them still adjusting to the diagnosis and awaiting normal milestones to develop.
Regarding clinical follow up and counseling after positive NBS for SMA, two main themes emerged. First, parents felt overwhelmed at the first clinic appointment. They struggled to process the amount of information provided while still in a state of shock. These sentiments were shared by 41% of parents in the Australian cohort who noted emotional response to the diagnosis and complexity of the information being presented as barriers to comprehension [22]. Parents have reported similar feeling regarding initial clinic appointments following positive NBS for metabolic disorders [26]. Second, parents took notice of providers’ compassion at this first appointment. They appreciated providers taking time to thoroughly explain the aspects of their child’s diagnosis and allowing them space for their feelings during the appointment. These findings were similar to a study focused on NBS results disclosure for cystic fibrosis and congenital hypothyroidism [27]. While the majority of new SMA diagnoses in the United States will be on NBS, these themes were consistent in the Symptomatic group. This is relevant for counseling families of children with variants not detectable by NBS and in areas where NBS is not available.
Genetic counseling was more commonly provided in the NBS group (75%). Those who received genetic counseling reported it was useful in understanding the diagnosis, carrier risks for themselves and family members, and treatments. Most (77%) felt that meeting with a genetic counselor immediately following diagnosis would be most beneficial.
Parents felt receiving information verbally was most useful for understanding of disease, testing, genetics, and treatment, but the majority felt that written or visual information would also be helpful. Parents in the prior U.S. study also felt additional formats of information related to treatment options were helpful [21]. Additionally, parental interest in written summary materials has been described in reference to other conditions on the NBS [27].
Limitations of this study include that interviews and surveys were only offered to parents of children who received gene replacement therapy. Although in our experience at NCH, every family with a child under the age of two years chose to pursue gene replacement (only one of which could not receive it due to elevated AAV9 antibodies), responses to questions regarding treatment choice are limited by the participant inclusion criteria. Additionally, in the survey, responses to questions are dependent on participant interpretation. This is especially true in the more open-ended questions, including those related to education preferences following diagnosis. Finally, interviews and surveys were only offered in English so we are unable to comment on whether parents who do not read and/or speak English would have differing experiences with these processes.
CONCLUSIONS AND RECOMMENDATIONS
This study identified three key aspects of the parental experience to target for improved communication.
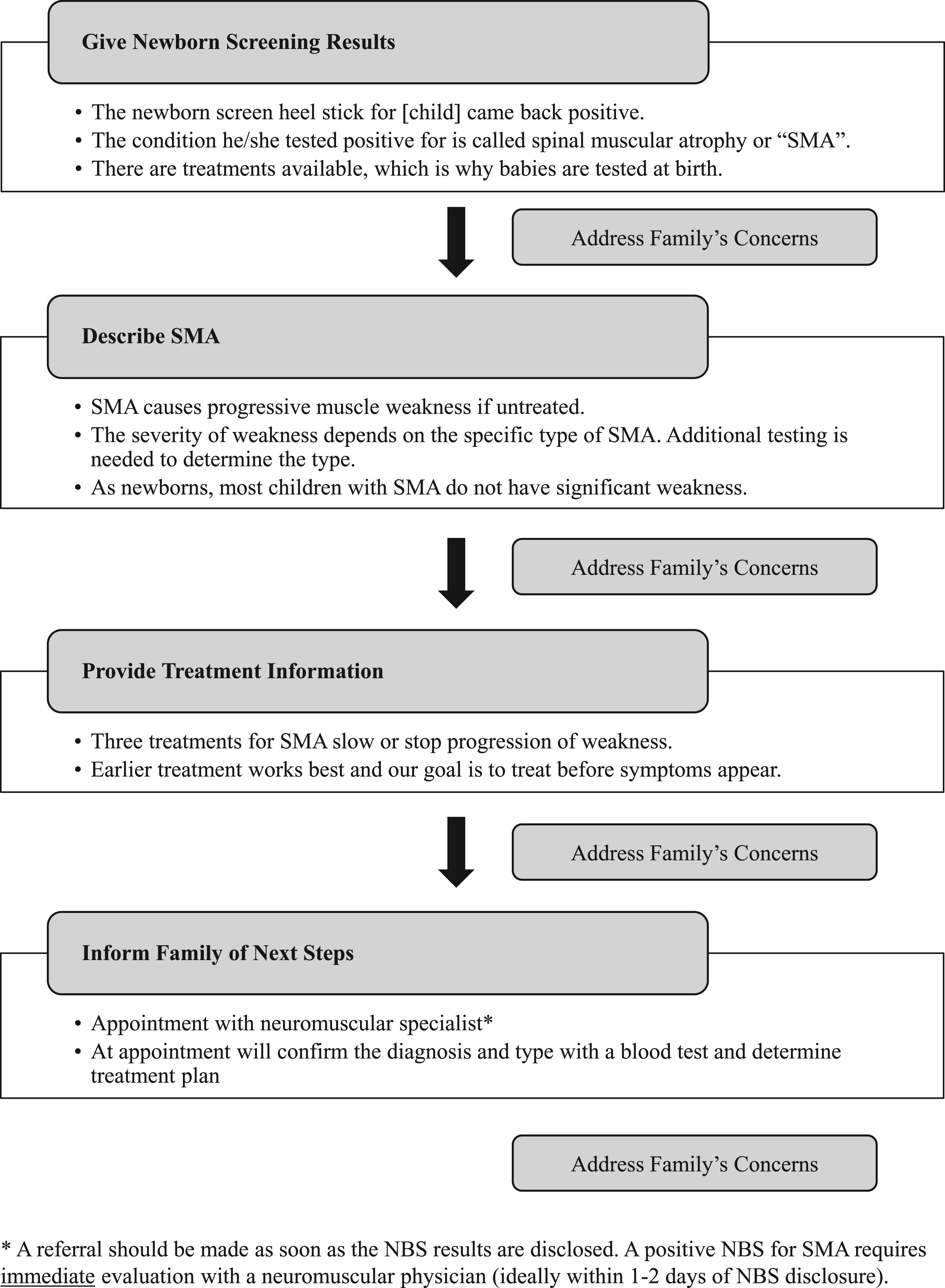
Suggested content for disclosure of a positive NBS for SMA.
Footnotes
ACKNOWLEDGMENTS
We would like to thank CureSMA for their assistance in distributing study recruitment information to their members and all of the parents who took the time to share their experiences.
FUNDING
This work was supported by Award Number UL1TR002733 from the National Center for Advancing Translational Sciences and the National Society of Genetic Counselors Pediatric and Clinical Special Interest Group. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Advancing Translational Sciences or the National Institutes of Health.
AUTHOR CONTRIBUTIONS
Contributed to study conception and design: A.M., M.W., A.C., K.V., N.H.
Conducted participant interviews: A.M.
Analyzed and interpreted data: A.M., M.W., A.C., K.V., N.H., A.H., A.D., P.P, A.W.
Drafted and reviewed the final publication: A.M., M.W., A.C., K.V., N.H., A.H., A.D., P.P, A.W.
CONFLICTS OF INTEREST
Dr. Waldrop has received clinical trial support from Sarepta therapeutics and Novartis Gene Therapies and has received consulting fees from Sarepta therapeutics. Dr. Connolly has served on advisory boards for Biohaven, Scholar Rock, Sarepta Therapeutics, Edgewise, Avexis, and Genentech-Roche. She receives clinical trial support from Novartis Gene Therapies, Biohaven and Scholar Rock. The remaining authors have no relevant disclosures.
DATA AVAILABILITY
The data supporting the findings of this study are available within the article and/or its supplementary material.
