Abstract
Objective
Evaluate the quality and cost-effectiveness of economic evaluations of newborn screening (NBS) for Spinal Muscular Atrophy (SMA).
Methods
A systematic review was conducted following Cochrane Handbook guidelines and PRISMA-S checklist. From 146 identified papers, 22 were screened for full-text, and 5 were included. Studies were evaluated for quality of reporting and transparency using the CHEERs and QHES checklists. Data was extracted to inform the review.
Results
Four economic evaluations on NBS for SMA with high reporting quality were identified. Each study employed a cost-utility analysis with similar model structures, using a decision tree for screening and a Markov model for treatment outcomes. They each compared NBS with treatment vs clinical diagnosis (no screening) with treatment. Although treatment protocols of each study varied due to differences in the strategies considered and availability of treatment. All studies included a societal perspective in their analysis and considered a lifetime horizon ranging from 30 months to 100 years. Early NBS with treatment was found to be more cost-effective than late treatment in all studies with ICER values ranging from £−117,541 to $714,000 per QALY. The wide range of ICER values are due to assumptions of long-term outcomes which are still largely unknown.
Conclusion
NBS with treatment was found to be cost-effective by all studies when compared to no NBS and late treatment. Although there is uncertainty around long term outcomes. Future research should focus on collecting long-term efficacy and safety data and evaluating the cost-effectiveness of pre-symptomatic treatment.
Keywords
Introduction
Spinal muscular atrophy (SMA) is a rare genetic neurodegenerative disease characterized by progressive muscle weakness, respiratory failure, and, in the most severe cases, death. Homozygous deletion of the survival motor neuron (SMN1) gene causes approximately 96% of cases, with the remaining 4% caused by a pathogenic SMN1 missense or nonsense mutation. 1 The clinical SMA phenotypes are classified into four groups. Type 1 is the most severe. Without intervention, children with type 1 SMA never acquire the ability to sit independently and most die before the age of 2 years. 2 Children with type 2 SMA develop symptoms between 6 to 18 months, are unable to walk unassisted and live to adolescence or adulthood. 2 Children with type 3 SMA will develop the ability to walk unaided but will eventually lose this motor function. 2 The least severe type 4 SMA may exhibit symptom onset approximately between the ages of 20–30 years old. 2 SMN2 is a homologous paralog to SMN1 with 5 nucleotide differences, one of which impacts splicing of pre-mRNA resulting in full length survival motor neuron (SMN) protein transcript only about 15% of the time. 3 An increase in SMN2 copies will increase functional SMN protein, predictive of a milder phenotype.2,3
Several disease-modifying treatments (DMT) are now available to improve symptoms. Nusinersen, 4 onasemnogene abeparvovec, 5 and risdiplam 6 have shown efficacy in clinical trials, with ongoing studies to assess long-term efficacy and safety. The most critical factor in predicting treatment success is the patient's age at the initiation of treatment. 7 Current evidence indicates that SMA patients treated pre-symptomatically or soon after symptoms occur result in improved treatment efficacy. 7 Newborn screening (NBS) for SMA with a polymerase chain reaction (PCR) can reliably detect homozygous SMN1 deletion 8 to help identify patients who would benefit from DMTs. This approach will however miss the 4% of patients who have heterozygous mutations.
Studying rare diseases such as SMA is challenging. SMA is a heterogeneous population of different types affected by SMN2 copy numbers. The limited evidence may include only some of the subtypes, and current clinical trials are still ongoing to collect long-term efficacy and safety data. Most of these trials also lack a true comparison group. This increases uncertainty when creating economic evaluations with a need for more data. In addition, due to the high costs of DMT (onasemnogene abeparvovec is $2.1 million USD), cost-effectiveness studies on post-symptomatic patients resulted in very high incremental cost-effectiveness ratios (ICER), and therefore, were not cost-effective at the standard willingness to pay threshold of $50,000 per QALY. 9 Economic evaluations are conducted to assess the value for money of healthcare interventions and to guide resource allocation decisions. These evaluations benefit healthcare providers, policymakers, and patients by informing decisions on the most efficient use of limited healthcare resources. It is hypothesized that there may be reduced healthcare costs resulting from improved efficacy with pre-symptomatic treatment with DMT. 9 This systematic review aims to evaluate the quality of the studies and the cost-effectiveness of current economic evaluations of NBS for SMA.
Methods
To conduct this focused systematic review, a review of the existing literature was performed. The Cochrane Handbook for Systematic Reviews of Interventions was followed to ensure adherence to reporting guidelines. 10
Literature search methods
An experienced external librarian from the Institute of Health Economics conducted the literature search in May 2023. Given the expected limited number of papers on NBS for SMA, a date restriction was not used. The search included randomized control trials, observational studies, modelling studies, HTAs, or systematic reviews, and included the following databases and registers: Ovid MEDLINE (1946-present), Embase (1974-present), Cochrane Library, HTA Database, NHS Economic Evaluation Database, Repec, Clinicaltrials, Google Scholar, and Google advanced. A detailed search strategy is shown in Appendix 1.
Selection of studies
To determine if the identified articles matched the eligibility criteria, two researchers (CP, AP) each independently screened the titles and abstracts of the identified articles. The eligibility criteria for full-text inclusion were complete economic evaluations (cost-effectiveness, cost–utility, cost–benefit, cost-consequence or cost minimization). They could include RCTs, observational studies, modelling studies, HTAs, or systematic reviews. Papers were also required to be written in English, conducted in North America, Europe, Asia or Australia, and in either community or inpatient settings. These regions were selected as they have relatively well-developed healthcare systems, established newborn screening programs, and access to disease-modifying treatments for SMA. The economic evaluations from these regions are more likely to be comparable to the Canadian population. The interventions or comparators considered were NBS for SMA, no NBS for SMA, and early or late treatment. The full inclusion and exclusion criteria can be found in Table 1. The two reviewers compared their findings through meeting discussions to decide which full-text papers should be included. Excel was used to organize abstract titles as part of article review.
Inclusion and exclusion criteria.

SMA: Spinal muscular atrophy; SMN2 Survival motor neuron; QALYs: Quality adjusted life years.
Next, reviewers assessed the quality of the economic evaluations using the updated Consolidated Health Economic Evaluation Reporting Standards (CHEERS) 11 and the Quality of Health Economic Studies (QHES) checklists. 12 These checklists were used to evaluate each included paper to assess the quality of reporting and transparency of the economic evaluations. Each reviewer completed the checklists independently, marking each item with a yes or no response. The QHES checklist assigned quality scores based on predetermined weights for each question. Any discrepancies were addressed in a meeting until a consensus was reached. The CHEERS and QHES checklists can be found in Appendix 2.
Consultation
Clinicians were selected for consultation based on their expertise in SMA research and treatment. They were identified and included during the grant application process due to their extensive knowledge and experience in this field. Given the complexity of the topic, expert consultation was considered essential to enhance the accuracy of our review. The experts were consulted at various stages of our review while developing research questions, refining search strategies, and interpreting findings. Their insights helped ensure that our review encompassed the most up-to-date evidence, considered methodological nuances, and addressed potential biases.
Data extraction
In line with established guidelines by Wijnen et al., 13 we systematically extracted data from studies that met our eligibility criteria (n = 5).14–18 Our comprehensive data collection checklist covered various elements from each included paper. This included study details, such as the primary author, publication year, funding sources, publication type, study design, research setting, patient characteristics, intervention, comparator, eligibility criteria, and perspective. Additionally, we retrieved information relevant to economic evaluations, including the type of economic evaluation, analytical approach, time horizon, discount rates, inflation rates, cost categories, and the reference year for costing. Data on outcomes and effects, their respective sources, underlying assumptions, incremental cost-effectiveness ratios (ICERs), sensitivity analyses, and conclusions drawn by the study authors were also extracted. This systematic approach facilitated a structured and comprehensive evaluation of the studies included. The guidelines used for data extraction can be found in Appendix 3.
Results
Quantity of research available
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses literature search extension checklist (PRISMA-S) 19 was followed to ensure appropriate steps were taken to include and exclude papers, see Figure 1. The table of study selections can be found in Appendix 4. A total of 146 were identified, 22 underwent full-text screening, and 5 papers were included in this systematic review. Details on the studies included are in Table 2. All five included papers were economic evaluations on NBS for SMA that looked at health outcomes and cost. The first paper published in 2020 by Jalali et al. was modelled in the U.S. jurisdiction. 14 Shih et al. published a paper in 2021 15 looking at NBS for SMA in Australia and then published a second paper in 2022 16 assessing NBS of SMA combined with SCID. The input parameters for SMA and the model for SMA were the same in both papers. Therefore, the two papers were assessed together as one study when using CHEERS and QHES and the SCID portion was excluded as it is outside the scope of this systematic review. Velikanova et al. published a paper in 2022 modelling NBS of SMA in The Netherlands. 17 Lastly, Weidlich et al. published a paper in 2023 modelling NBS of SMA in England. 18 The five included papers are assessed as four studies in this systematic review to avoid double counting the second paper published by Shih et al. 16
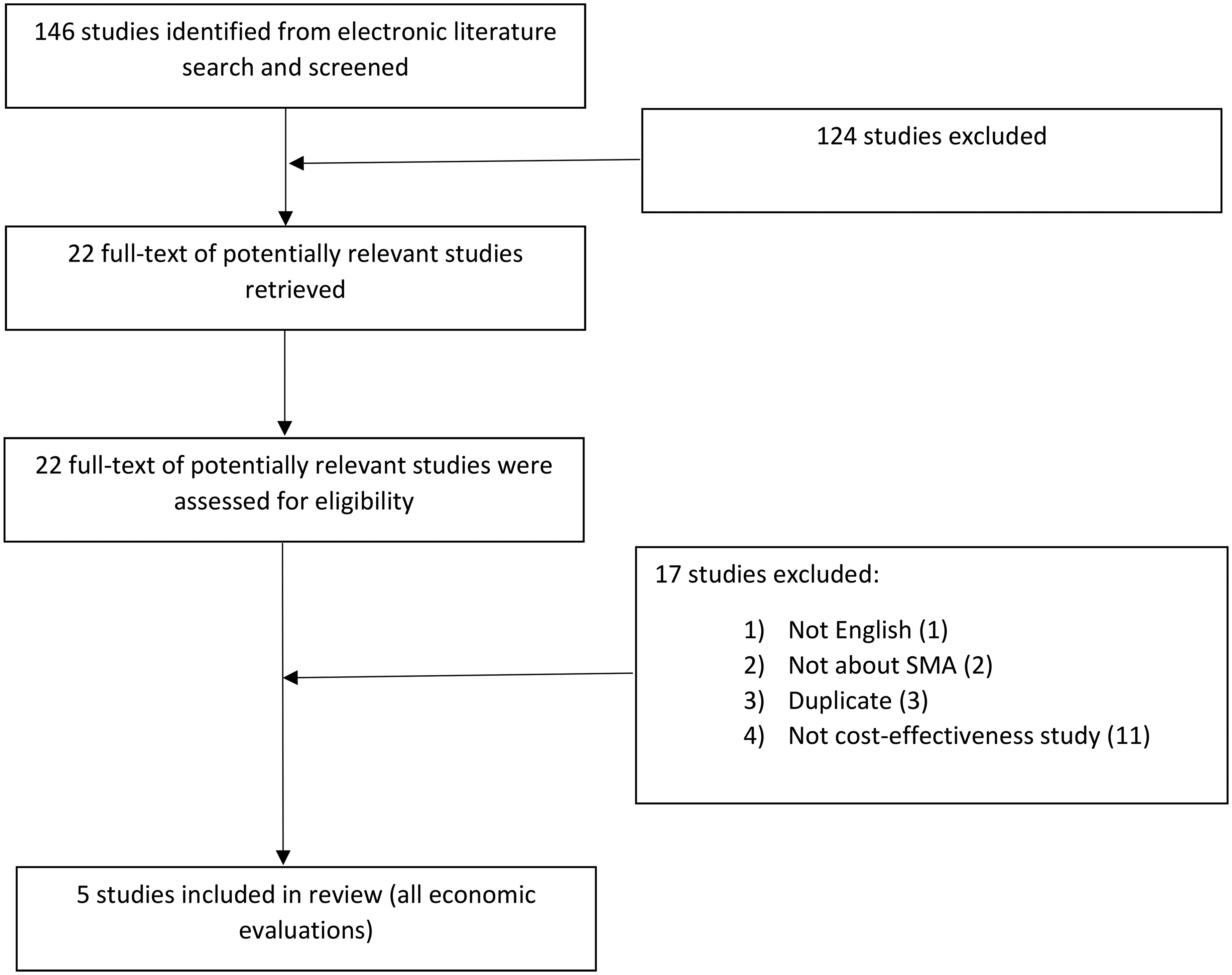
PRISMA diagram for selection of included studies. 19
Details on the included studies.
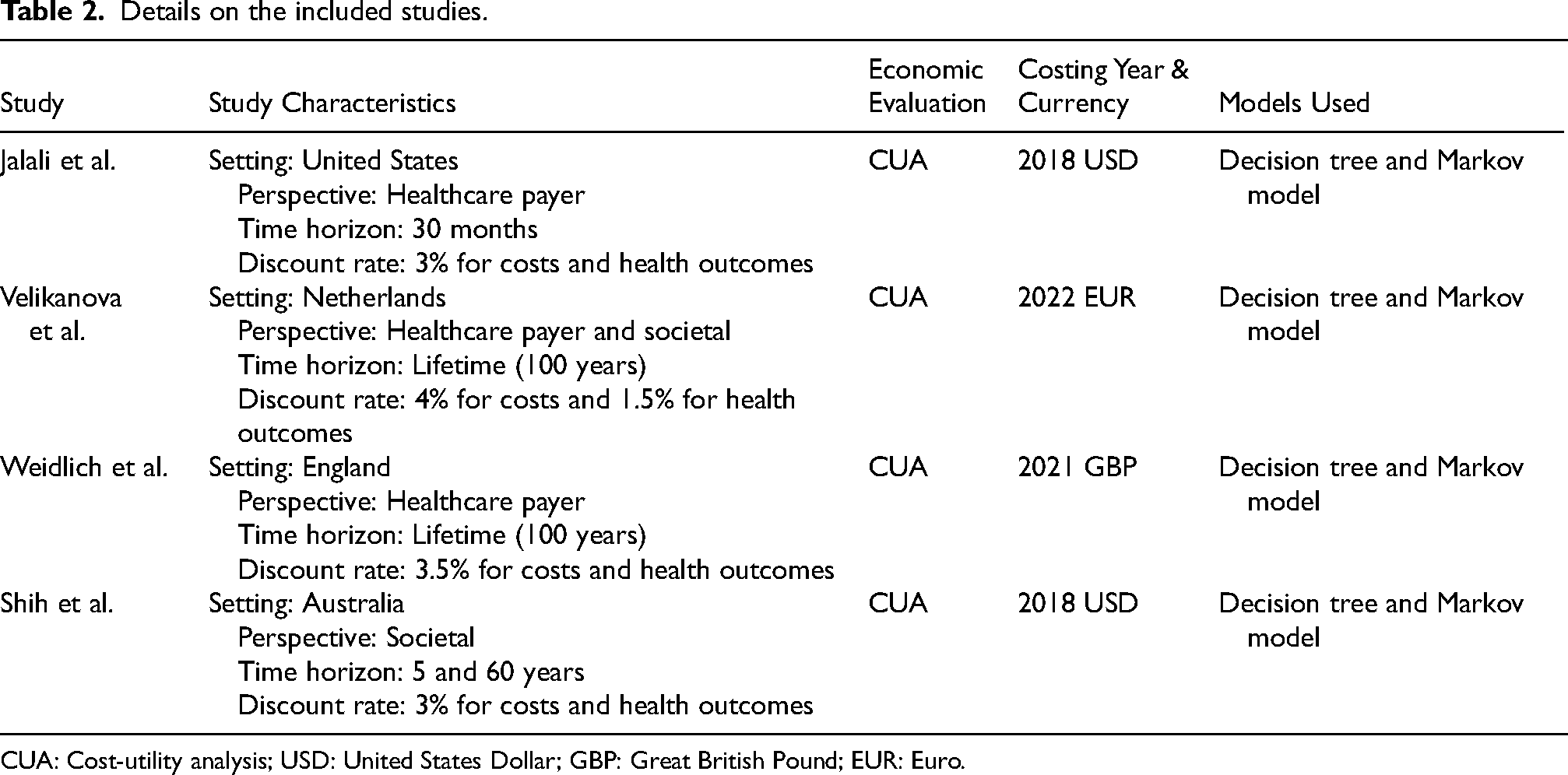
CUA: Cost-utility analysis; USD: United States Dollar; GBP: Great British Pound; EUR: Euro.
Reporting and quality assessment
As reported, we used the CHEERS checklists to assess the quality of reporting in the selected economic evaluations, with responses recorded as ‘yes’ or ‘no.’ Each study met most CHEERS and QHES checklist requirements, with only a few minor variances across the four included studies. When analyzing the papers according to the CHEERS criteria, we found that none explicitly had a health economic analysis plan, and none described the distributional effects of their findings, i.e., how the effects are distributed among various people, or the adjustments made to take priority populations into account. Additionally, only the Shih15,16 study included their approach to engagement with patients and others affected by the study, while none reported the effects of engagement. Otherwise, CHEERS criteria were satisfied by all papers. On the QHES checklist, three of the studies earned 89.5,14–18 while the remaining paper scored 81.5 14 out of 100. All papers lost partial marks for not justifying how they determined their time horizon, discount rate, and perspective of analysis. The paper that scored 81.5 also lost marks for not providing a diagram of its model structure. 14 All articles, however, met the remaining QHES requirements and received a score of 75 or higher, classifying them as high quality by the QHES instrument. In summary, the selected economic evaluations met most of the CHEERS and QHES checklist criteria, indicating a high overall quality, though there were notable areas for potential improvement. The purpose of the checklists is to assess the quality of reporting of the economic evaluations and does not assess the quality of the research. These results suggest that all included papers had high reporting quality. However, the absence of details about the model structure in Jalali et al. made it more difficult to assess that study and illustrates the importance of including information about model structure when reporting model based economic evaluations. This also poses an argument for whether a more significant emphasis should be placed on a model structure in checklists to assess reporting quality.
Setting and population and screening
The studies included in this systematic review assessed the cost-effectiveness of NBS for SMA. The main population of interest was live births per year in the corresponding setting for each paper. The population of infants who didn’t receive NBS was compared with infants who received NBS for SMA. Jalali et al. focused on SMA Type 1 infants in the United States. 14 The remaining papers followed infants with SMA Type 1, 2, and 3.14–18 Shih et al. also differentiated SMA patients between those with SMN2 2 and 3 copies in Australia.15,16 Velikanova et al. 17 and Weidlich et al. 18 further assessed SMA patients with SMN2 2, 3, and 4 copies from The Netherlands and England, respectively.
Screening and treatment
Screening programs in each paper were broadly consistent in including all live newborns. Reporting on the screening method between all the included studies varied in detail. Velikanova reported screening in more detail, outlining the use of polymerase chain reaction (PCR) genotyping assay performed on a dried blood spot (DBS). 17 Shih et al. included retesting due to non-amplification of the initial blood sample. 15
Treatment protocols between each study varied due to differences in the strategy considered in the model and availability in each setting at the time of publication. Each study compared NBS with treatment (nusinersen, onasemnogene abeparvoc, risdiplam, or supportive care) vs clinical diagnosis (no screening) with treatment. Table 3 outlines the intervention, comparator, and treatment plans in each study. Table 4 details the clinical trials used to inform treatment efficacy.
Treatment protocols for included studies.

NBS: Newborn screening.
Clinical diagnosis means there was no newborn screening.
Treatment included percentages of patients treated with nusinersen, onsemnogene abeparvoc and risdiplam.
Clinical trials used to inform the study.

NBS: Newborn screening; SMA: Spinal Muscular Strophy; SMN: Survival Motor Neuron.
Types of economic evaluation
All included studies were cost-utility analyses (CUAs). Three studies were from the healthcare payer perspective.15–18 These three studies also included the societal perspective and accounted for caregiver's productivity loss as an indirect cost.15–18 Screening and treatment were modelled across a lifetime time horizon in all studies. Jalali et al.'s analysis was from the societal perspective but adjusted the administration cost of nusinersen from the private payer perspective. 14 Additionally, Jalali et al. defined the lifetime horizon as 30 months because long-term survival rates for Type 1 SMA treated with nusinersen were unavailable. 14 Other studies used 60–100 years as the lifetime horizon.15–18
Model structure
The screening process was modelled with a decision tree, and the long-term treatment outcomes were modelled with a Markov model in three of the studies.15–18 Jalali et al. stated using a Markov model, but a model structure diagram was not published, so the model structure could not be confirmed. 14 The papers that used a decision tree and Markov model provided similar model structure diagrams.15–18 Weidlich et al. based their model structure on the previously published Velikanova et al. model structure.17,18 Shih et al. initially published the 2021 SMA model structure, then published the 2022 model structure that combined SMA with SCID NBS. The way that SMA was modelled in the 2022 publication followed the same pathways as the 2021 publication, so it was counted as one study for this systematic review.15,16
Input parameters
All papers used total live births in their corresponding jurisdiction and applied SMA rates and costs from other countries’ literature when unavailable for their own. For example, Velikanova et al. used SMA rates published in Belgium and Germany and applied them to their Netherlands context. 17 Shih et al. used the US listing price of gene therapy because it was unavailable in Australia.15,16
Transition probabilities for the Markov model were based on clinical trials for SMA drugs and were extrapolated to assess long-term outcomes from all the papers15–18 except one. 14 Utility values for each health state in the Markov Model were based on literature, most of which were on SMA patients and/or their caregivers. Three studies were similar in how they defined health states and used utility values for each health state to calculate the QALYs.15–18 The Permanent Ventilation Assistance (PVA) health state for three of the studies was based on different literature and consistently found that it is equivalent to 0,15–18 the same as death. Velikanova et al. and Weidlich et al. used one reference to inform the utility of each health state.17,18 The utility value for the non-sitting health state was solely based on a conference abstract by Thompson et al., which made it more difficult to assess the robustness. 20 Shih et al. used multiple references to inform the utility values for the non-sitting and sitting without support health states.15,16 Furthermore, Shih et al. referenced literature using results from SMA patient-reported outcome measures (PROMs) and the youth version of the EQ-5D to inform other health state utility values.15,16 Jalali et al. used LY (life years) and QALY data from asthma and Duchenne muscular dystrophy patients as a proxy for estimating long-term health outcomes for SMA patients. 14
Direct healthcare costs were broadly consistent across studies, but indirect costs varied. For instance, Weidlich et al. included caregiver-related costs, transportation expenses, and loss of productivity for patients. 18 Jalali et al. considered indirect costs related to permanent ventilation assistance (PVA) and age-specific annual patient costs. 14 In contrast, Shih et al. included indirect costs of informal care and parental loss of productivity.15,16
Sensitivity analysis
Probabilistic sensitivity analyses (PSA), which consider probability distributions of model parameters to assess uncertainty, were conducted in all the included papers.14–18 PSAs provide a comprehensive understanding of the uncertainty in model outcomes by simultaneously varying all parameters according to their probability distributions. In contrast, deterministic sensitivity analyses (DSA), which vary one model parameter at a time, were conducted in three of the studies.15–18 DSA helps identify the parameters that have the most significant impact on the results.
Study findings
Jalali et al. conclude that NBS with nusinersen treatment was preferred over clinical diagnosis and no treatment and clinical diagnosis and nusinersen treatment, but only cost-effective with a WTP of over $300,000 and $500,000 per event-free LY, respectively, due to the high cost of treatment and the rarity of SMA. 14 The other papers found NBS with early treatment cost saving and, therefore, cost-effective compared to clinically diagnosed SMA and treatment.15–18 Shih et al. compared scenarios where patients with SMA used nusinersen vs onasemnogene abeparvovec vs supportive care and found that NBS and early treatment with gene therapy was dominant over clinical diagnosis with nusinersen treatment.15,16 Velikanova et al. and Weidlich et al. found that NBS with treatment was dominant over clinical diagnosis and treatment.17,18 Table 5 summarizes the ICERS, cost, health outcomes, and thresholds for each included paper.
Summarizes the ICERS, costs, health outcomes and thresholds for each paper.
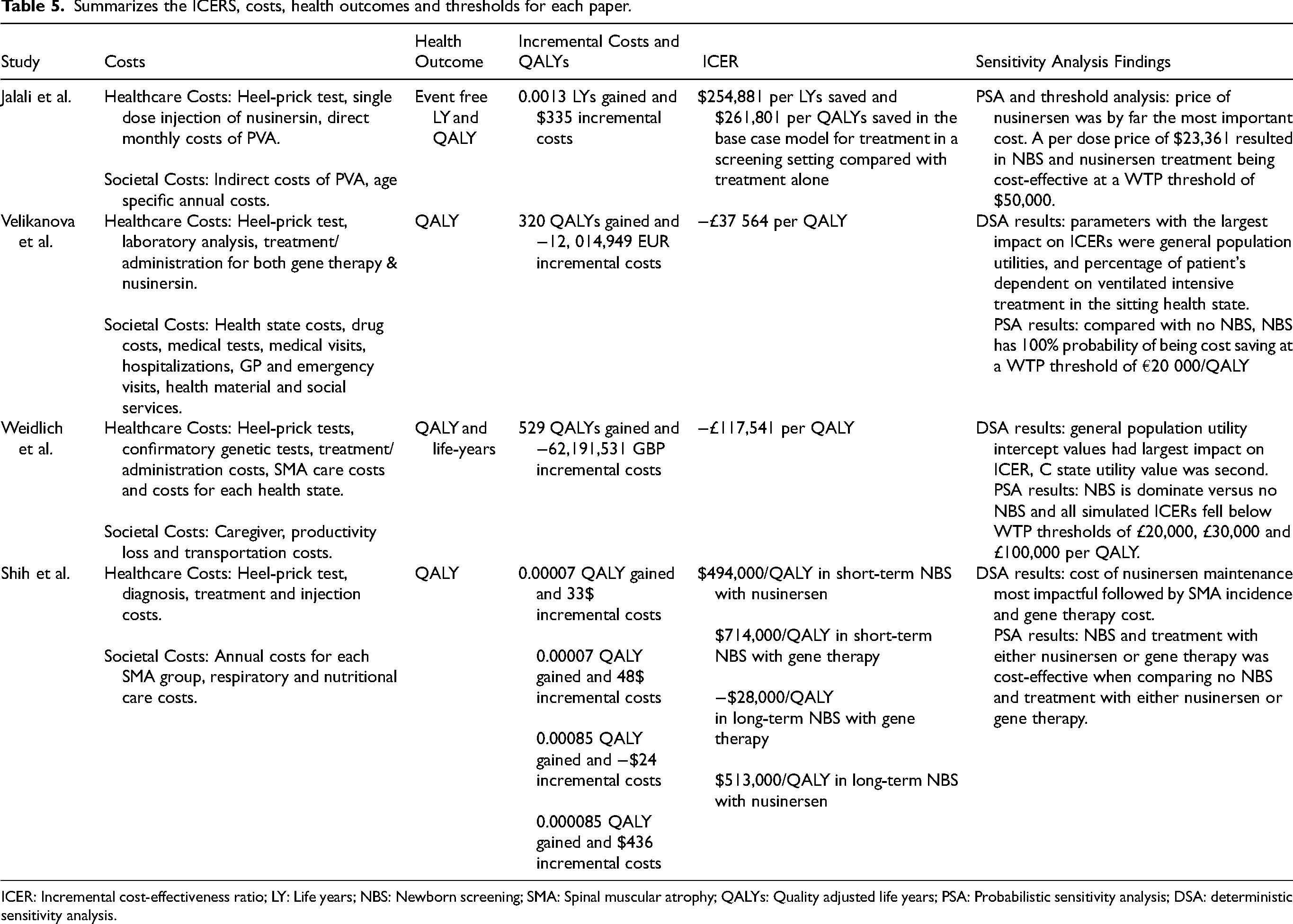
ICER: Incremental cost-effectiveness ratio; LY: Life years; NBS: Newborn screening; SMA: Spinal muscular atrophy; QALYs: Quality adjusted life years; PSA: Probabilistic sensitivity analysis; DSA: deterministic sensitivity analysis.
Where PSA was conducted, NBS with treatment was cost-effective in 70% to 100% of the simulations. In the Shih et al. study, DSA showed that the parameter that had the most significant impact on the ICER is the cost of nusinersen maintenance injections.15,16 Velikanova and Weidlich et al. found in the DSA that the general population utility value had the most significant impact on the ICER, but it wasn’t explicit whether the cost of drugs was included in the DSA and this item was not shown in the tornado diagrams.17,18
All of the included studies modelled NBS tools that cannot detect SMA caused by a point mutation, which occurs in about 5% of SMA incidences.14–18 However, the false-negative rates due to point mutations varied across the selected studies: Shih reported a 5% rate, Weidlich reported a 4% rate, and Velikanova reported a 1% rate, while Jalali did not define a specific rate.14–18 This inability to detect point mutations is a limitation of current screening tests. 18
Discussion
To focus on economic evaluations of NBS for SMA, we limited the studies included to cost-effectiveness models that assessed health outcomes and cost. We found five published papers in total,14–18 though two were counted as one because the same input parameters and model were used.15,16 All papers had high reporting quality when assessed against the CHEERS and QHES tools. One paper found that NBS followed by treatment for SMA was cost-effective compared to clinical diagnosis followed by treatment with a high WTP. 14 Other papers found that NBS with treatment is cost-saving compared to clinical diagnosis and treatment.15–18 A meta-analysis was not conducted for this systematic review. Notwithstanding the limited methodological support for performing meta-analyses of economic evaluations, each included study varied in the time horizon, treatment strategy, intervention, and comparators, and combining the results to develop a meta-analysis would be inappropriate.
Based on the results of all four included studies, NBS with early treatment was more cost-effective with onasemnogene abeparvovec compared to clinical diagnosis and late treatment (onasemnogene abeparvovec, nusinersen, risdiplam, or best supportive care).14–18 Jalali et al. focused on treatment with nusinersen and concluded that a high WTP was required for NBS with nusinersen treatment to be cost-effective. 14 Shih et al. concluded that NBS with onasemnogene abeparvovec treatment is cost-saving compared to clinical diagnosis and treatment with nusinersen.15,16 Shih et al. also found in the sensitivity analysis that the cost of maintenance nusinersen had the most significant impact on the ICER. One dose of onasemnogene abeparvovec is costly, at approximately $2.1 million USD per patient, but Shih et al. found that providing maintenance injections of nusinersen for the patient's lifetime was more expensive.15,16 Velikanova et al. varied the percentage of patients treated with either nusinersen or onasemnogene abeparvovec by increments of 25%. They found that scenarios where onasemnogene abeparvovec was used in a higher percentage of patients the ICER values were less than in scenarios where more patients were treated with nusinersen. 17 Weidlich et al. created a treatment pattern informed by expert opinion that would resemble a patient's clinical experience in England. 18 Weidlich et al. found NBS and early treatment (where nearly 93% of the patients were given onasemnogene abeparvovec) cost-saving compared to clinical diagnosis and later treatment (where patients may take onasemnogene abeparvovec, nusinersen, risdiplam, or best supportive care depending on the SMA type). 18 Although the studies found onasemnogene abeparvovec to be the dominate treatment for SMA compared to nusinersen, this is largely due to the one-time cost nature of onasemnogen abeparvovec. The cost of nusinersen maintenance doses over a life-time time horizon led to higher ICER values. This is an assumption that is based on unknown long-term efficacy of onasemnogene abeparvovec and if efficacy does wain over time, this assumption may not hold. None of the included studies modelled a need for subsequent treatment with an alternate therapy, and it is not clear how frequently, if at all, this may happen in clinical practice.
Some of the studies were limited to short-term efficacy data for pre-symptomatically treated SMA patients due to limited clinical trial results at the time of their publication. Shih et al. and Velikanova et al. used the NURTURE clinical trial (nusinersen) to inform treatment effectiveness for pre-symptomatically diagnosed patients and assumed the efficacy was equal between onasemnogene abeparvovec and nusinersen due to the lack of data on onasemnogene abeparvovec.15–17 Jalali et al. focused on treatment with nusinersen and were able to use short-term data from the NURTURE trial. 14 Weidlich et al. was the most informed paper on short-term efficacy data for pre-symptomatically detected SMA patients, the later publication data meaning more data from clinical trials were available for all three different types of drugs to treat SMA. They used short-term efficacy data for pre-symptomatically treated SMA patients from the NURTURE, RAINBOWFISH (risdiplam), and SPR1NT (onasemnogene abeparvovec). 18 Although Shih et al., Velikanova et al., and Weidlich et al. varied in treatment plans and short-term efficacy data used, they all found NBS with early treatment (mostly with onasemnogene abeparvovec) cost-saving compared to clinical diagnosis and later treatment which strengthens this conclusion.15–18
The need for more data on the long-term efficacy of SMA treatment limited all the studies. Long-term treatment efficacy data is still being collected and has yet to be available during this systematic review. All the included papers assumed that patients who gained a motor function and were on SMA drug treatment would maintain that motor function until death.14–18 Three of the studies extrapolated existing data on survival for each health state.15–18 Authors using survival curves to extrapolate data did not explicitly state statistical methods used to verify results. Jalali et al. estimated long-term outcomes using asthma and Duchenne health outcomes as proxy data for SMA. 14 More research and results on long-term health outcomes from treatment are required to confirm these papers’ results.
Studies show that SMA causes a significant health-related quality of life (HRQoL) burden on patients and their caregivers. 21 However, current published utility values for SMA patients and their caregivers are of moderate quality and, therefore, should be fully justified and tested in sensitivity analysis when used for economic evaluations. 21 Each included study was hampered by the absence of high-quality utility data. Jalali et al. used LY and QALY values from asthma and Duchenne patients as a proxy for SMA due to the lack of published utility values for pediatric SMA patients. 14 However, utility weights were only applied to SMA patients when they reached adulthood. This study focused on Type 1 SMA patients where symptoms typically initiate in childhood, and the time horizon for this study was 30 months. The authors didn’t justify this method, and it was not tested in the sensitivity analysis. 14 Velikanova et al. and Weidlich et al. used similar literature to inform their utility values.17,18 A conference abstract was used to inform the Not-sitting health state utility value. 20 Conference abstracts do not fully publish their methodology, which limits the reliability of their findings. 21 The nusinersen technology appraisal submitted to NICE informed the sitting unassisted health state utility value. 22 NICE concluded that the utility values lacked face value and were highly uncertain. 21 In addition, the PVA utility value in the Velikanova et al. study referenced a paper that did not publish utility values 23 and so the Velikanova et al. PVA utility value could not be validated. The Weidlich et al. and Shih et al. studies applied the same utility value in their models. 18 Shih et al. referenced similar literature as Velikanova et al. and Weidlich et al. to inform utility values. Shih et al. also used additional literature with different methodologies (such as the EuroQol-5 Dimension 5 Level (EQ-5D-5L), and Patient Reported Outcome Measure Information System Preference measure (PROPr)).15,16 However, EQ-5D-5L and PROPr have shown to have poor agreement, with PROPr producing significantly lower utility scores compared to EQ-5D-5L. It has been argued they should not be used interchangeably in SMA economic evaluations. 24 The three studies that included utility values in the DSA found that the utility value for one health state could alter the ICER by tens of thousands of dollars in the upper and lower bounds.15–18 This shows that using the appropriate utility values in NBS for SMA economic evaluations is significant. Coupled with the lack of age-appropriate and validated preference-based measures for pediatric SMA patients and their caregivers and limited utility estimations for SMA patients undergoing treatment, more research in this area is needed to model SMA in cost-effectiveness studies better. 21
Another consideration for the CUA of NBS for SMA is the variability in SMA incidence across different populations. The studies in our analysis used an incidence range between 1:10,000 and 1:11,000, while other literature indicates that SMA incidence can vary from 1:7000 to 1:27,000 depending on the geographic region.25,26 This variability may impact the cost-effectiveness of NBS programs and should be included in sensitivity analysis. Additionally, all of the models in our analysis assumed an immediate choice of a single treatment. However, in clinical practice, bridge therapy is sometimes necessary due to elevated antibodies against the AAV9 vector. This scenario was highlighted in a recent American study, where nusinersen or risdiplam were used before treatment with OA. 27 The inclusion of bridge therapy could impact CUA results and should be considered in future models.
The results of this review carry important implications for policymakers and other stakeholders in the healthcare field. For example, the potential for cost savings with early treatment identified by NBS could influence decisions on resource allocation or the implementation of screening programs. The geographic context of these studies highlights the need for more localized cost-effectiveness analyses that address regional healthcare priorities and constraints.
Conclusion
SMA is a rare genetic disorder that is inherently challenging to study due to unavailable evidence on the long-term efficacy of treatment and high-quality utility data on patients undergoing treatment and their caregivers. This increases uncertainty in economic evaluations that must use proxies or extrapolate short-term data to estimate long-term effects. However, clinical trials for SMA drugs have shown significant improvement in patients’ quality of life, especially when provided pre-symptomatically, and DMTs are approved for treatment. 9 Therefore, conducting economic evaluations on NBS for SMA is important. The included studies in this review were of good quality in their reporting according to CHEERS and QHES. They showed there is a benefit for patients to be screened to receive treatment earlier compared to later treatment.
Based on the studies included in this review, NBS for SMA can be recommended as a cost-effective use of resources, and in certain circumstances may be cost saving. Despite different settings, comparators, input parameters, modelling, and the high cost of treatment, all included studies found that NBS with early treatment improves patient health outcomes and is considered cost-effective when compared to no NBS and late treatment. However more data is needed on the long-term efficacy of treatments, the need for follow-on treatments if initial treatment efficacy wanes, and on the long-term safety and side effects of treatment.
Supplemental Material
sj-pdf-1-jnd-10.1177_22143602251336862 - Supplemental material for Systematic review for economic evaluations on newborn screening for spinal muscular atrophy
Supplemental material, sj-pdf-1-jnd-10.1177_22143602251336862 for Systematic review for economic evaluations on newborn screening for spinal muscular atrophy by Alexander C Pace, Corrina Poon, Pranesh Chakraborty, Maryam Oskoui, Hugh McMillan, Alex Mackenzie and Jeff Round in Journal of Neuromuscular Diseases
Footnotes
Acknowledgements
CP and AP independently screened the titles and abstracts of identified articles, completed data extraction and checked the quality of the studies. Any disputes were resolved by JR. PC, HM, MO and AM provided consultation on developing research questions, refining search strategies, and interpreting findings. Manuscript writing was completed by CP and AP, with edits made by MO, HM and JR.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by Muscular Dystrophy Canada together with Novartis Pharmaceuticals.
Conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Hugh McMillan has been a consultant and received honararia from Novartis Gene Therapies Inc, Hoffman La-Roche Ltd and Pfizer. He has participated in clinical trials with Roche, PTC Therapeutics, Biogen, DYNE, PepGen, ReveraGen, Catabasis, Novartis, and Sarepta. The institution of Dr Oskoui has received research support from Roche Genetech, Novartis, Santhera, Muscular Dystrophy Canada, Canadian Institutes of Health Research. Pranesh Chakraborty has received research support from Biogen, Biomarin, Takeda, Cambrooke, Vitaflo, Nutricia, NFDC, CIHR and CF foundation. He has also served on advisory boards for Sanofi, Ultragenyx, Waters and Horizon. Jeff Round and his employer (The Institute of Health Economics) received no additional funding directly related to this work. Alex Pace has no disclosures to report. Corrina Poon has no disclosures to report. Alex MacKenzie has no disclosures to report.
Data availability
No data, models or methodology were proprietary in this review, all papers can be found publicly.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
