Abstract
Pompe disease is a rare, metabolic, autosomal recessive disorder. Early diagnosis is critical for progressive Pompe disease as delays can significantly alter the clinical course of the disease. Diagnostic modalities, including dried blood spot testing and genetic testing, are available and are effective for diagnosing patients with late-onset Pompe disease (LOPD). However, clinicians face numerous clinical challenges related to the diagnosis of the disease. Two expert group committee meetings, involving 11 experts from the United Arab Emirates, Kuwait, the Kingdom of Saudi Arabia, and Oman, were convened in October 2019 and November 2020 respectively to develop a uniform diagnostic algorithm for the diagnosis of pediatric and adult LOPD in the Arabian Peninsula region. During the first meeting, the specialty-specific clinical presentation of LOPD was defined. During the second meeting, a diagnostic algorithm was developed after a thorough validation of clinical presentation or symptoms, which was performed with the aid of existing literature and expert judgement. A consensus was reached on the diagnostic algorithm for field specialists, such as neurologists, rheumatologists, general practitioners/internal medicine specialists, orthopedic specialists, and pulmonologists. This specialty-specific diagnostic referral algorithm for pediatric and adult LOPD will guide clinicians in the differential diagnosis of LOPD.
HIGHLIGHTS
In the Arabian Peninsula region, clinicians face numerous clinical challenges related to the diagnosis of late-onset Pompe disease (LOPD). Suggestive symptoms of LOPD include limb– girdle muscles weakness, unexplained hyperCKemia, and respiratory insufficiency. Considering the high consanguinity rate in the region, physicians are encouraged to set a low threshold for LOPD screening in the appropriate clinical setting. Dried blood spot testing for GAA activity should be performed first in the appropriate clinical setting, followed by confirmatory genetic testing.
ABBREVIATIONS
Alanine aminotransferase
Aspartate aminotransferase
Basilar artery
Creatine kinase
Dried blood spot testing
Duchenne muscular dystrophy
Electromyography
Forced vital capacity
Acid alpha-glucosidase
General practitioners/internal medicine specialists
Kingdom of Saudi Arabia
Lactate dehydrogenase
Late-onset Pompe disease
Limb–girdle muscle weakness
Myasthenia gravis
Newborn screening
Neuromuscular disorders
Randomized-controlled trials
Sleep-disordered breathing
Spinal muscular atrophy
Positive vacuoles
INTRODUCTION
Pompe disease is a lysosomal storage disorder characterized by a deficiency in the lysosomal enzyme acid alpha-glucosidase (GAA) due to mutations in the GAA gene [1]. This deficiency leads to the accumulation of glycogen in multiple organs, specifically in the skeletal and respiratory muscles [1]. The estimated incidence of Pompe disease is reported to be 1 in 40,000 live births [2, 3]. Late-onset Pompe disease (LOPD) is a progressive muscle disorder affecting individuals of varying ages, particularly from early childhood (above 12 months of age) to late adulthood [4, 5]. The estimated global incidence of LOPD is 1 in 57,000 people [6]. Two prospective trials conducted in Taiwan (N = 344,056) and Austria (N = 34,736) in the context of newborn screening (NBS) depicted the high incidence of LOPD in Taiwan (1 in 26,466 live births) [7] and Austria (1 in 8684 live births) [8]. These results indicated that LOPD is underestimated or underdiagnosed in countries without national screening programs. Underdiagnosis in countries which lack national screening program constitutes the most significant barrier to the management of LOPD. Therefore, an effective and early screening strategy is essential to overcome underdiagnosis in countries without national screening programs. It is noteworthy that early diagnosis can lead to early initiation of treatment, which may improve outcomes in LOPD patients [9].
In the Middle East and North Africa (MENA) region, consanguineous marriages are culturally favored and are considered traditional in most of the communities [10]. A study conducted on 3212 Saudi families showed that the overall rate of consanguinity was 57.7% with a high frequency of first-cousin marriages (28.4%) [11]. Even a cross-sectional study that was carried out in a Tehrani population (n = 93) demonstrated the high degree of consanguinity (43%) in this region and that the incidence of LOPD (n = 3) was reported in families with consanguinity [12]. Therefore, consanguinity could be one of the determinants of the prevalence of LOPD in the MENA region [10]. However, there is a lack of awareness of LOPD among healthcare professionals (HCPs) in the MENA region, which is a significant challenge that may lead to a delay in diagnosis, missed diagnosis, and delay in treatment [9].
The important signs and symptoms that occur in 80% of patients with LOPD include musculoskeletal and respiratory symptoms [5, 13]. The gastrointestinal symptoms that are commonly observed in LOPD patients include constipation, diarrhea, poor weight gain, and hepatomegaly [5, 13]. The other clinical manifestations are ptosis, brain aneurysms, arrhythmias, facial muscle weakness, dysphagia, dysarthria, a rigid spine, and macroglossia [13]. The symptoms of LOPD may mimic that of other diseases, including unspecified myopathy, limb–girdle muscular dystrophy, and inflammatory myopathy [9, 14–24]. This broad clinical spectrum of manifestations that mimic symptoms of other diseases is a significant barrier that hinders initial diagnosis and thereby causes diagnostic delays. Therefore, differential diagnoses play a crucial role in the accurate diagnosis of LOPD.
Due to the broad clinical spectrum of presenting symptoms, which are of a wide range of complexity, there is a necessity for a high degree of suspicion for the accurate diagnosis of LOPD [9]. The diagnostic tests that are performed as a part of clinical evaluation in suspected individuals include: (1) measurement of creatine kinase, which alters from normal (60–305 IU/L) to 15 times the upper limit of the normal value; (2) measurement of lactate dehydrogenase, alanine transaminase, and aspartate transaminase, which are frequently increased; (3) assessment of forced vital capacity (FVC), which is reduced in the majority of LOPD patients; (4) electromyography (EMG), in which suggestive signs of LOPD include myopathic EMG, specifically in proximal muscles, myotonic discharges without clinical myotonia, particularly in paraspinal muscles, fibrillation potentials, positive sharp waves, and complex repetitive discharges; and (5) dried blood spot testing (DBS), in which GAA activity is reduced [9].
A positive DBS testing should be followed by a confirmatory test for the diagnosis of LOPD [9]. The array of confirmatory tests encompasses (1) lymphocyte GAA activity, in which GAA is assayed in purified lymphocyte preparations; (2) measurement of GAA activity in skin fibroblast cultures, in which GAA is assayed in cultured fibroblasts from skin biopsy; (3) molecular genetic testing (DBS-extracted DNA) or gene sequencing for detecting variations that include targeted mutational analysis, deletion/duplication analysis, and full sequence analysis; and (4) muscle tissue biopsy with or without GAA enzyme analysis [9]. Pathogenic mutations can be detected through genetic testing, and in muscle biopsy, vacuoles that stain positively for glycogen with glycogen accumulation in the lysosomes and cytosol are observed during the advanced stages of the disease [9]. Although several novel diagnostic strategies are available for diagnosing LOPD, an appropriate diagnostic criterion, including DBS testing of lymphocyte GAA activity and skin fibroblast GAA activity, for clinicians is still lacking in the Arabian Peninsula region.
Overall, under-recognition, missed diagnosis, delayed diagnosis, lack of appropriate diagnostic facilities, and lack of awareness about LOPD among HCPs and families of LOPD patients are the unmet clinical challenges that are widespread across this region. Additionally, LOPD diagnosis is often challenging due to the complex and broad spectrum of clinical symptoms, which overlap with other diseases [9, 14].
With this background, the unmet clinical challenges of diagnostic clues for LOPD patients were examined, particularly from the perspective of the Arabian Peninsula region. The present manuscript aims to establish diagnostic clues for clinicians across the Arabian Peninsula region to better diagnose LOPD patients, that is pediatric LOPD patients and adult LOPD patients, by developing a diagnostic algorithm for different specialists, including neurologists, pulmonologists, rheumatologists, orthopedic specialists, and general practitioners/internal medicine specialists (GPs/IMs).
METHODS
Two expert group committee meetings involving 11 experts with expertise in the department of neurology from the United Arab Emirates, Kuwait, the Kingdom of Saudi Arabia (KSA), and Oman, were convened in October 2019 and November 2020. The committee aimed to develop a consensus-based diagnostic algorithm for pediatric and adult LOPD that would aid clinicians in the early diagnosis of LOPD in the Arabian Peninsula region.
During the first meeting, the clinical presentation and recommendations for the diagnosis of pediatric LOPD were stratified according to specialty, and a clear demarcation of symptoms for different specialists, including neurologists, pulmonologists, GPs/IMs, and rheumatologists, was developed. A similar stratification was developed for the diagnosis of adult LOPD, in addition to a clear-cut symptom demarcation for different specialists, including neurologists, pulmonologists, GPs/IMs, and orthopedics.
After the development of diagnostic criteria for pediatric and adult LOPD, a literature search was performed using PubMed for randomized control trials to identify the prevalence of clinical symptoms in patients with LOPD. The search terms included late-onset Pompe disease and clinical symptoms associated with LOPD.
During the second meeting, all the experts reviewed and validated the evidence and critically discussed the applicability of the literature for LOPD in the Arabian Peninsula region.
A diagnostic algorithm was developed after validation of clinical presentation or symptoms, which was performed with the aid of existing literature and expert judgement.
RESULTS
Development of a consensus-based diagnostic algorithm
The proposed pediatric and adult LOPD clinical presentation and recommendations for different specialties were developed during the expert group committee meetings and are presented in Fig. 1 and Fig. 2, respectively. The literature supporting clinical presentation and recommendations for pediatric and adult LOPD, which was reviewed and validated by experts, is depicted in Table 1 and Table 2, respectively. Finally, a consensus-based diagnostic algorithm was developed for field specialists based on the clinical experiences of experts and a summary of evidence (Fig. 3 and Fig. 4).
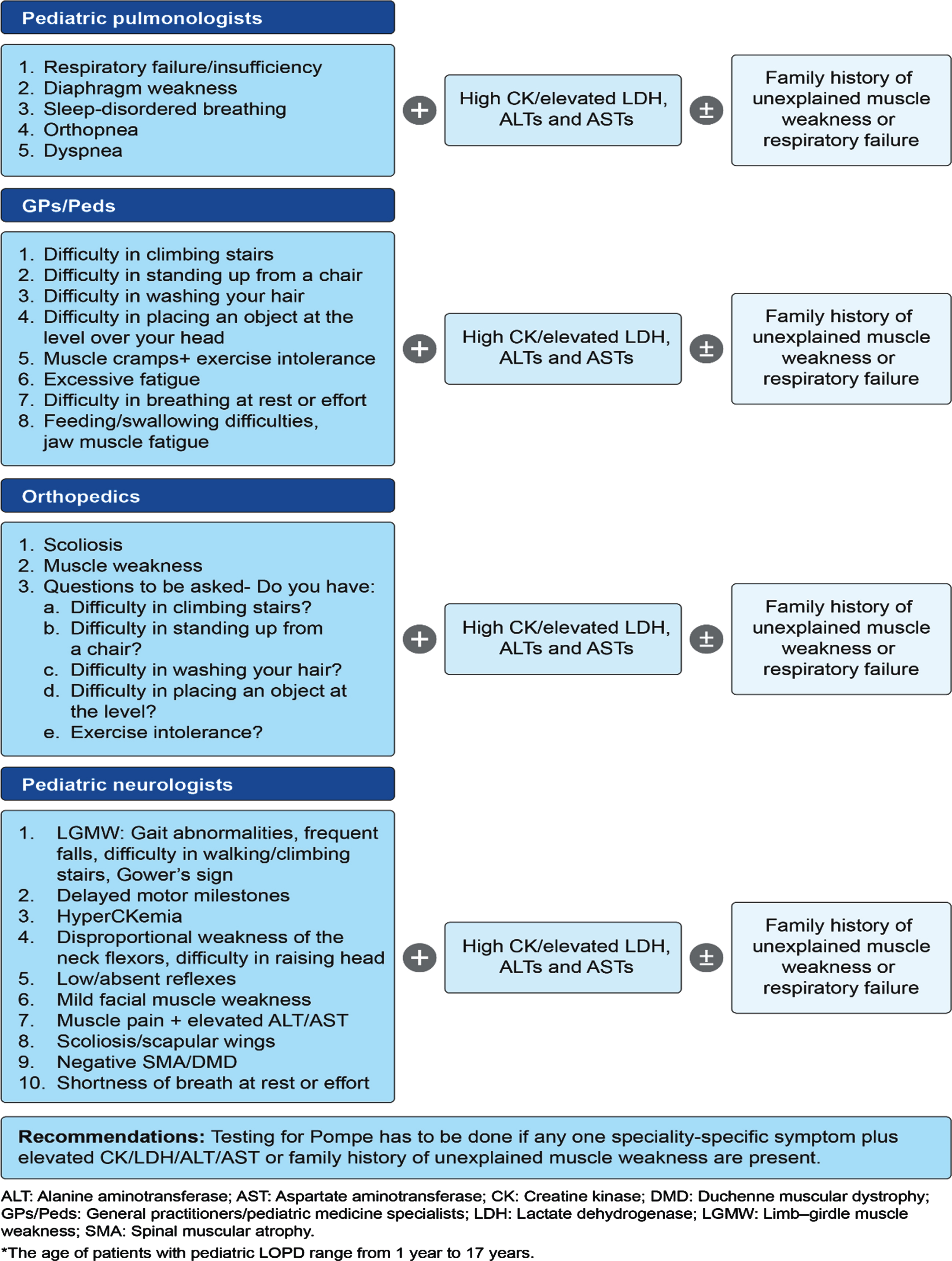
Proposed pediatric LOPD* clinical presentation and recommendations by experts during the first Expert Group Committee Meeting.
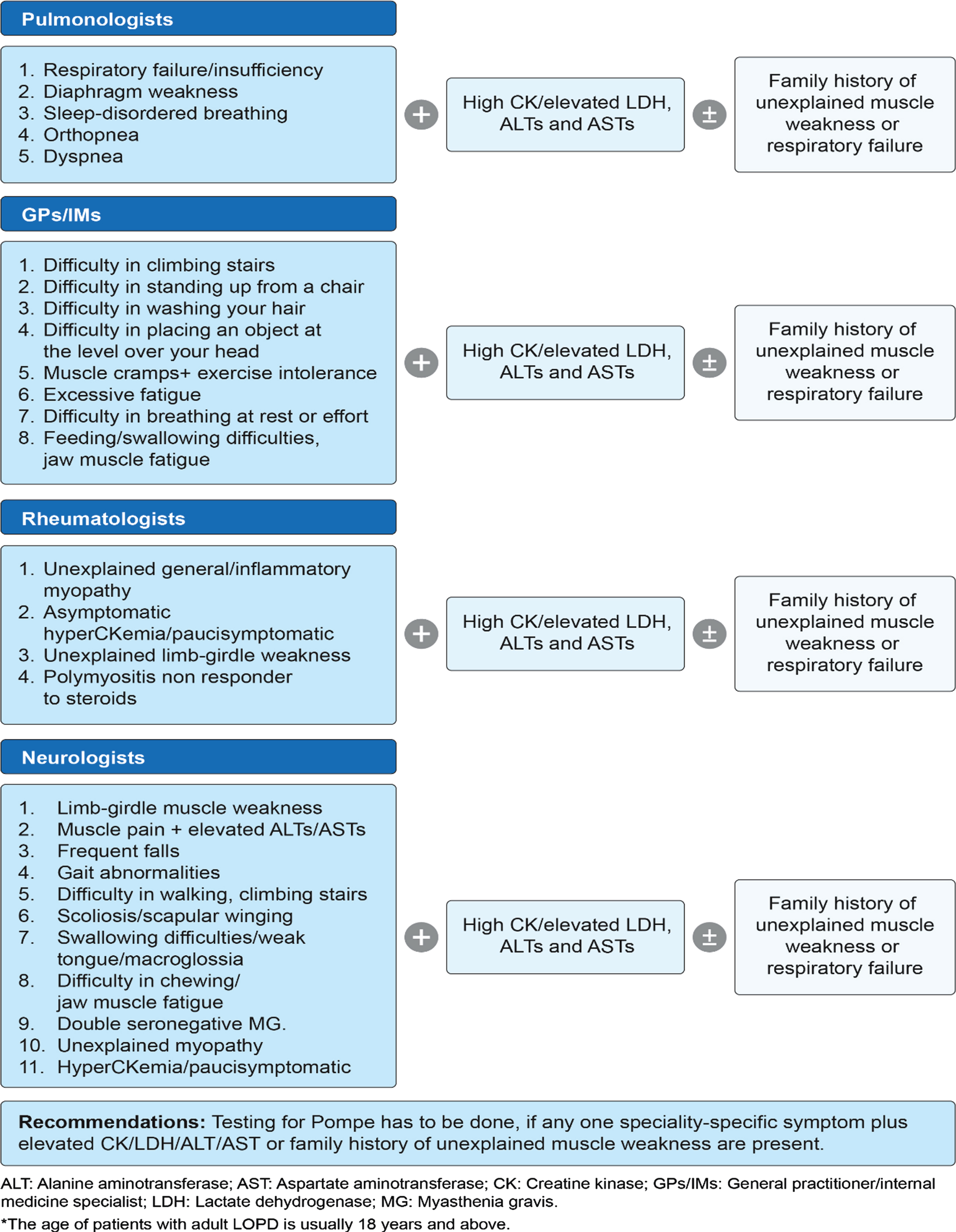
Proposed adult LOPD* clinical presentation and recommendations by experts during the first expert group committee meeting.
Clinical evidence rationalizing the importance of clinical presentation and recommendations for each specialty for the diagnosis of pediatric LOPD
*The clinical presentation and recommendations for the internal medicine are the same for the diagnosis of pediatric and adult LOPD and thus combined clinical validation by existing literature was performed for pediatric and adult LOPD symptoms. ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; CK: Creatine kinase; DMD: Duchenne muscular dystrophy; LGMW: Limb–girdle muscle weakness; LOPD: Late-onset Pompe disease; SMA: Spinal muscular atrophy.
Clinical evidence rationalizing the importance of clinical presentation and recommendations for each specialty for the diagnosis of adult LOPD
ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; CK: Creatine kinase; MG: Myasthenia gravis; RCTs: Randomized-controlled trials.
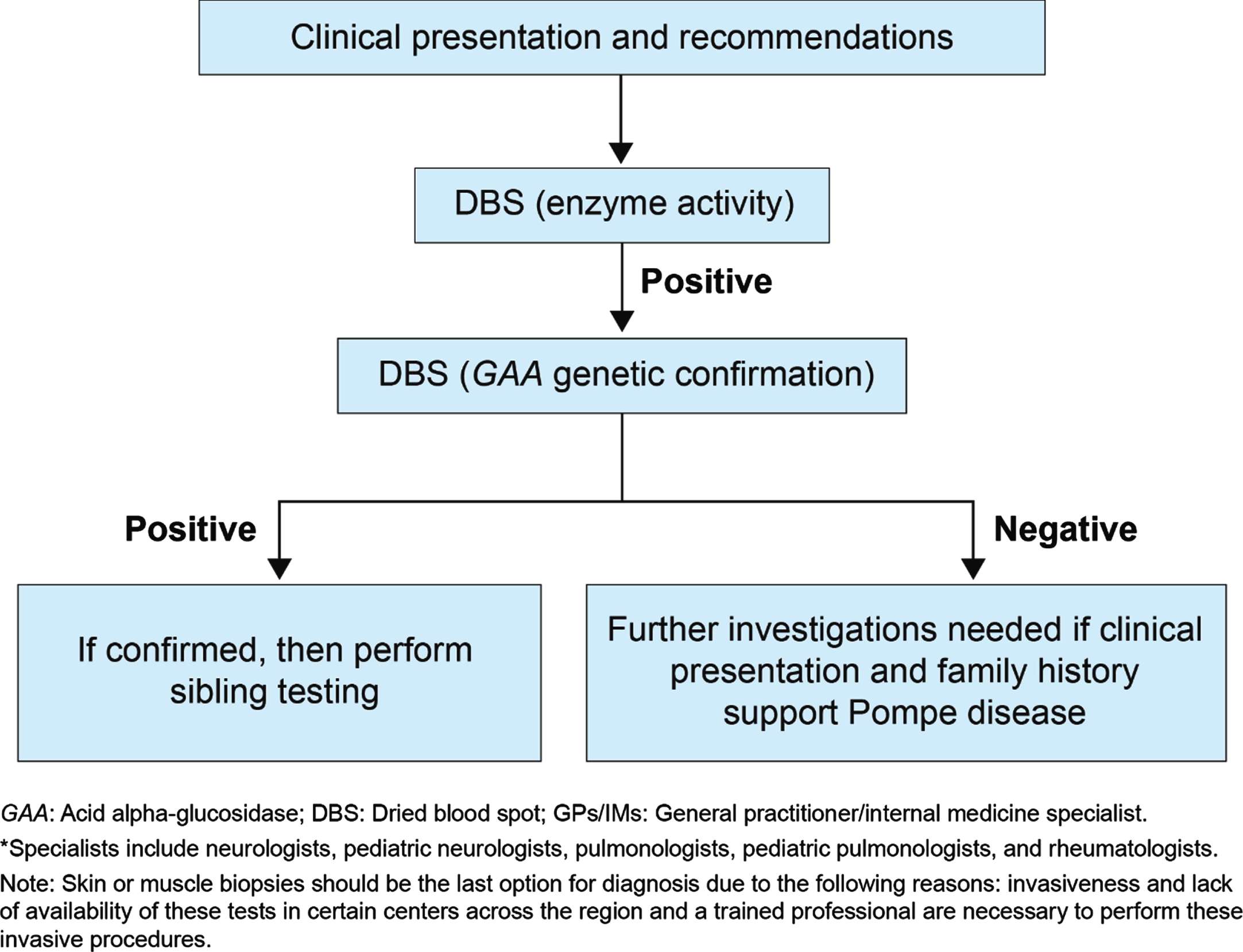
Proposed diagnostic algorithm for all specialists* except GPs/IMs/Peds /orthopedics.
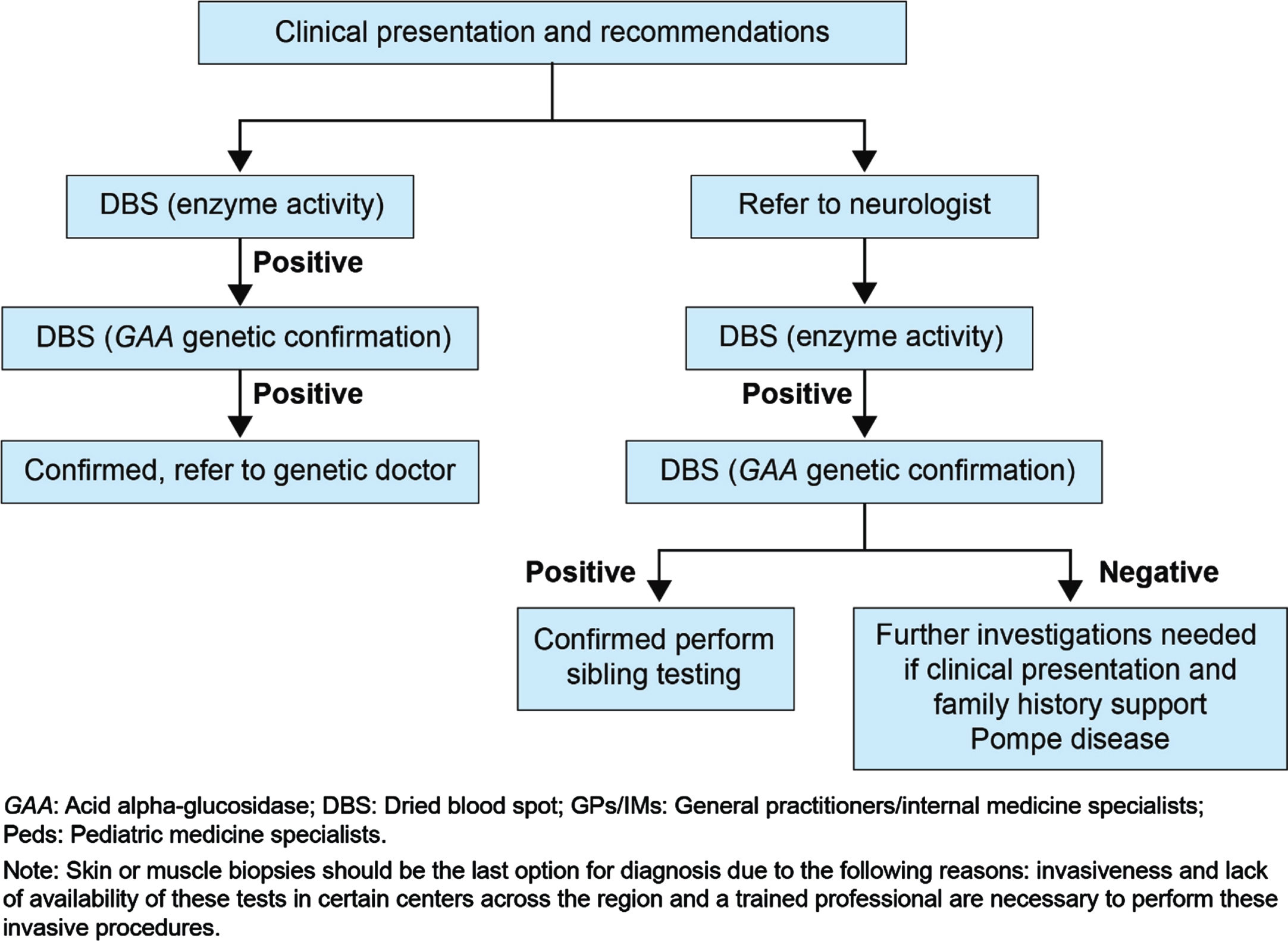
Proposed diagnostic algorithm for GPs/IMs/ Peds/ orthopedics.
DISCUSSION
Late-onset Pompe disease is a progressive and debilitating disorder. Delays in diagnosis and treatment have negative consequences on a patient’s morbidity and mortality. Therefore, an appropriate diagnostic approach is essential for the early and accurate diagnosis of LOPD. Although international [14] and regional guidelines [9] are available for the diagnosis of LOPD, clinicians in the Arabian Peninsula region face numerous diagnostic challenges. The goal of this manuscript is to provide solutions to the diagnostic challenges of LOPD, thereby facilitating early and accurate diagnosis.
Late-onset Pompe disease is characterized by an insidious onset, a slowly progressive course, and clinical variability between patients, wherein symptoms may start at any stage of life, ranging from early childhood to adulthood [4, 5]. The majority of the symptoms of LOPD can mimic other medical conditions, thus creating the biggest challenge for clinicians from various specialties for accurate diagnosis of the disease. In addition, the clinical diagnostic clues of LOPD for various specialties play a crucial role in diagnosing LOPD. The current manuscript emphasizes that no symptom should be overlooked by clinicians as underdiagnosis and a delay in diagnosis may lead to fatal consequences and early referral to neurologists may enhance accurate diagnosis of LOPD. These specialty-specific diagnostic algorithms of pediatric and adult LOPD for field specialists, such as neurologists, rheumatologists, GPs/IMs, orthopedic specialists, and pulmonologists, based on clinical judgement of experts and summary of the evidence may facilitate the early and accurate diagnosis of the disease.
Consensus-based diagnostic approach for pediatric neurologists and neurologists
Neurologists and pediatric neurologists should suspect LOPD in patients whose clinical manifestation is an ‘unexplained’ limb–girdle muscle weakness [31] as well as asymptomatic hyperCKemia [27]. Scoliosis is prevalent in LOPD patients and should not be left unnoticed while suspecting pediatric and adult LOPD [35]. Respiratory problems such as dyspnea and respiratory distress should not be overlooked as these are the most common symptoms of pediatric LOPD and 50% of pediatric LOPD patients require respiratory assistance [27]. Patients with pediatric LOPD present with frequent falls [34], have difficulty in walking, and require wheelchair assistance [32]. Therefore, these symptoms should not be overlooked by pediatric neurologists while suspecting LOPD. Tongue abnormalities on brain MRI are common in adult LOPD patients compared to other neuromuscular disorders (NMDs). Particular attention to the tongue while reviewing brain MRIs may be a crucial clue for the diagnosis of muscle weakness in adult LOPD patients [45]. Dolichoectasia of the basilar artery (BA) is the most striking cerebrovascular finding in adult LOPD patients as Pompe disease is associated with BA dilation, elongation, and elevated bifurcation height of the BA, resulting in cerebrovascular complications [46]. Therefore, neurologists should recommend a computer tomography angiography or a magnetic resonance angiography in adult LOPD patients for early detection of cerebrovascular malformations as these could lead to life-threatening events such as subarachnoid hemorrhage or brainstem compression [33].
Consensus-based diagnostic approach for pulmonologists and pediatric pulmonologists
Pulmonologists play a key role in diagnosing and managing Pompe patients as Pompe disease can heavily impact respiratory function. Pompe disease weakens the diaphragm muscles, including expiratory muscles, inspiratory muscles, and upper airway muscles. Diaphragm weakness initially manifests during sleep, leading to sleep-disordered breathing (SDB). Weakening of the diaphragm muscles leads to respiratory insufficiency, hypercapnia, shallow breathing, orthopnea, impaired cough, impaired speech, impaired swallowing, increased risk of atelectasis, and chest infections. Respiratory muscle weakness causes respiratory dysfunction [65]. Approximately, 60% of LOPD patients have a mild reduction (< 80% predicted) and 30% –40% have a moderate reduction in vital capacity (< 60% predicted) [14], indicating the progressive loss of pulmonary function. While assessing the pulmonary function in infants, certain clinical factors, such as daytime energy level and the degree of fatigability (i.e. the ability to feed without developing increased work of breathing) should be additionally assessed as the evaluation of pulmonary function in infants is technically difficult [14]. Weakness of the diaphragm can be an early and major finding in Pompe disease, and a reduction in vital capacity, sleep hypoventilation, and respiratory failure often develops following respiratory dysfunction. Pulmonologists should look for the aforementioned symptoms for the accurate diagnosis of LOPD.
Consensus-based diagnostic approach for rheumatologists
Untreated LOPD results in significant disability, including impairment in muscle structure and function; deterioration in mobility, respiration, and activities; and handicap in social participation (e.g. work, study, and recreation) [66]. The clinical clues that can help rheumatologists distinguish LOPD from autoimmune myopathy include [67]: Myotonic discharges on EMG. The diaphragm tends to be more severely involved than other skeletal muscles in Pompe; therefore, a low FVC should raise suspicion for Pompe disease. Muscle biopsies from patients with Pompe disease usually reveal periodic acid-Schiff (PAS)-positive vacuoles and do not have inflammatory infiltrates as seen in patients with autoimmune myopathy. Pompe patients do not respond to steroid treatment, which is the treatment strategy for inflammatory myopathy.
Consensus-based diagnostic approach for general practitioners/internal medicine specialists
General practitioners/internal medicine specialists should not overlook respiratory symptoms because SDB is highly prevalent in pediatric and adult LOPD and may comprise alveolar hypoventilation or obstructive sleep apnea or both [53]. Fatigue is a common and disabling problem in patients with early and advanced stages of Pompe disease [60]. The follow-up of patients with LOPD should focus on respiratory and limb–girdle muscle function, the capacity to perform daily activities, and the presentation of fatigue and pain [62]. Overall, GPs/IMs should look for LOPD in patients whose clinical manifestation is an “unexplained” limb–girdle muscular weakness even without vacuolar myopathy in muscle biopsy, respiratory symptoms, hyperCKemia, and fatigue [53, 62].
Consensus-based diagnostic approach for orthopedics
Untreated pediatric LOPD results in significant disability, including impairment in muscle structure and function; deterioration in mobility, respiration, and activities; and handicap in social participation (e.g. work, study, and recreation) [66]. Therefore, muscle weakness and difficulty in walking should not be overlooked by orthopedics while suspecting pediatric LOPD. Scoliosis poses a significant burden in pediatric LOPD patients and thus the presence of this symptom in suspected pediatric LOPD patients should be considered an important sign of LOPD and require a confirmatory test for pediatric LOPD [35].
CONCLUSION
The heterogeneous clinical presentation, slow progression, and similarity of LOPD to other NMDs can create a diagnostic challenge for clinicians. Therefore, there is a need for increased awareness of LOPD among clinicians. In clinical practice, there appears to be inconsistency in the patterns of patient evaluation and a lack of standardized diagnostic testing, specifically in the Arabian Peninsula region. Although guidelines are available that aid in the diagnosis of LOPD, several challenges are faced by clinicians for the early and accurate diagnosis of LOPD. Moreover, the phenotype of LOPD is often difficult to distinguish from that of other genetic and acquired NMDs. The goal of this manuscript was to provide solutions to the diagnostic challenges of LOPD and to develop a consensus-based simplified screening algorithm for different specialties to facilitate timely and accurate diagnosis in all patients who may have LOPD. This consensus-based simplified screening algorithm, which is supported by evidence, will guide clinicians in the differential diagnosis of LOPD. It will also facilitate direct referral, which may assist in the early and accurate diagnosis of LOPD.
Footnotes
ACKNOWLEDGMENTS
BioQuest Solutions has provided the editorial support for the preparation of this review manuscript.
FUNDING
The funding for logistics and editorial support for the development of this consensus manuscript was provided by Sanofi Genzyme.
AUTHOR CONTRIBUTIONS
All authors reviewed, provided critical revisions, and approved the final manuscript.
POTENTIAL CONFLICTS OF INTEREST
The authors declare no conflict of interest.
DECLARATION OF INTERESTS
All the authors have received personal fees in the form of honoraria for attending two Sanofi Genzyme advisory board meetings, however, they were not paid for writing the manuscript.
