Abstract
Background
Respiratory impairment in neuromuscular disorders (NMDs) is generally assessed using forced vital capacity (FVC). Any improvement in FVC trajectory will delay ventilatory support; however, the change required for patients to perceive a noticeable clinical benefit, the clinically meaningful threshold (CMT), has not been defined in NMDs.
Objective
To derive the within-person and between-group CMTs for FVC (% predicted) in patients with late-onset Pompe disease (LOPD).
Methods
This analysis leverages data from the Phase 3 COMET trial (NCT02782741, registered 25 May 2016), which assessed the efficacy of avalglucosidase alfa (AVA) versus alglucosidase alfa (ALG) on upright FVC (% predicted) in LOPD. Anchor- and distribution-based methods were used to estimate the within-person and between-group CMTs for FVC at Weeks 49 and 97.
Results
COMET enrolled 99 participants aged ≥18 years (52% male; mean age 48.0 years). The within-person CMT for absolute change in FVC expressed as % predicted was estimated as 3.0% [95% confidence interval (CI) 2.3, 3.8]. The proportion of patients with a meaningful increase in FVC was higher in the AVA versus ALG group across the CI of the estimated CMT (odds ratios: 2.3–2.6; nominal p-values: 0.026–0.058). The between-group CMT, needed to evaluate differences between treatment groups, was estimated as 2.1% predicted [95% CI 1.1, 3.1].
Conclusions
We identified a narrow range of within-person and between-group CMTs for upright FVC (% predicted) in LOPD. Post hoc application of these thresholds to COMET showed that a greater proportion of patients in the AVA group had clinically meaningful improvement in FVC versus ALG. These findings may aid in interpretation of data from studies in other NMDs.
Keywords
Introduction
Neuromuscular disorders (NMDs) are characterized by an array of functional limitations and symptoms affecting either skeletal muscle or the peripheral nervous system, often with the involvement of the respiratory system.1,2 Thus, assessment of respiratory function is routinely performed, especially for patients with progressive disorders that may eventually lead to chronic hypercapnic respiratory failure.2,3 A common parameter used to assess respiratory impairment in NMDs is forced vital capacity (FVC) 2 , which has been widely used as an outcome measure in clinical trials and research in NMDs.4,5 Although any change in FVC trajectory is important as it would delay the need for ventilatory support 6,7, the magnitude of change required for the patient to perceive benefit remains uncertain due to an absence of established clinically meaningful thresholds (CMTs) in NMDs. In addition, thresholds to interpret differences in FVC between treatment groups during interventional studies are lacking.
Pompe disease is a rare NMD caused by deficiency of the lysosomal enzyme α-1,4-glucosidase that, if untreated, results in irreversible damage to skeletal and cardiac muscles causing physical impairment and respiratory insufficiency.8–10 The late onset form of Pompe disease (LOPD) is characterized by progressive weakness of proximal and axial muscles, with respiratory distress mainly due to diaphragmatic weakness. 11 Enzyme replacement therapy (ERT) with alglucosidase alfa (ALG) was approved for the treatment of Pompe disease in 2006.8,12 Avalglucosidase alfa (AVA) is a next generation ERT designed for increased cellular uptake and glycogen clearance 13 , and was recently approved in the United States in 2021 and Europe in 2022.14,15 The Phase 3 COMET trial (NCT02782741) evaluated the relative efficacy and safety of these two ERTs on respiratory function, motor end points, and patient-reported outcomes (PROs) in treatment-naïve patients with LOPD. 16 The COMET trial met its primary end point of non-inferiority of AVA compared with ALG at Week 49, with a 2.43% (95% confidence interval [CI]: [-0.13, 4.99]; p = 0.0074, non-inferiority margin 1.1%) greater increase from baseline in FVC (% predicted) with AVA versus ALG; however, superiority was not reached (p = 0.063). 16 These clinical data provide an opportunity to derive CMTs for FVC.
Understanding meaningful change for outcome measures allows different stakeholders, such as patients, healthcare providers and payers, and regulatory agencies, to make informed decisions on treatment effects that can be considered clinically meaningful. Interest in clinical meaningfulness began in the 1980s when the concept of minimal clinically important difference was introduced to determine the smallest amount of change that patients perceive as beneficial. 17 The connection between clinically important differences and patient perception provides an additional evaluation of clinical trial end points, which are traditionally evaluated on statistical significance for disease-based clinical metrics alone.18,19 This is particularly relevant for randomized clinical trials in which patient populations are limited, and thus achieving statistical superiority is challenging.
CMTs are used to aid the interpretation of treatment effects in two ways: (1) the within-person CMT is the threshold that needs to be achieved for a given patient to perceive clinical benefit; and (2) the between-group CMT is the threshold that should be used to evaluate differences between groups of patients receiving differing therapies. 20
The present analysis leverages the robust data set obtained in the COMET trial to derive the within-person and between-group CMTs for FVC (% predicted) in LOPD based on patients’ report of noticeable benefit, and thus help address the knowledge gap in NMDs. Subsequently, the derived CMTs for FVC (% predicted) can be applied to data collected in the COMET trial. This approach will inform interpretation of clinical trial results in Pompe disease (e.g., COMET) and may assist in the interpretation of similar studies in other NMDs.
Materials and methods
Patients
Data from the randomized, double-blind, Phase 3 COMET trial 16 , which evaluated the safety and efficacy of AVA (n = 51) compared with ALG (n = 49) in treatment-naïve patients with LOPD, were analyzed. The trial included two treatment periods, a 49-week primary analysis period, and an open-label extended treatment period, in which patients allocated to the ALG treatment group in the primary analysis period were switched to AVA treatment. 16
Complete eligibility criteria and study design have been published previously 16 Briefly, relevant inclusion criteria from COMET included a confirmed diagnosis of Pompe disease (representative of LOPD patients), absence of prior Pompe-specific treatment, baseline FVC in the upright position from 30 to 85% predicted, and ability to walk at least 40 meters without stopping or using an ambulation-assistance device. 16 This analysis only included patients enrolled in COMET aged ≥18 years. This study was performed with institutional review board approval at all sites. 16 All procedures were completed in accord with the ethical standards of the Independent Ethics Committees or Institutional Review Boards of the institution where data were collected.
Assessment of change in FVC (% predicted)
Respiratory function was the primary efficacy end point of the COMET trial, assessed as the change in FVC (% predicted) from baseline to Week 49; these results have been previously reported. 16
PRO instruments
Several PRO measures were collected in adult participants in the COMET trial, of which a number were considered as anchors to derive the within-person and between-group CMTs for FVC (% predicted). A summary of each PRO anchor is shown in Table 1. Three generic PRO anchor measures were considered, including the 12-item Short-Form (Version 2) Health survey (SF-12v2; norm-based score range 0–100 with higher scores indicating better health-related quality of life) 21,22, the EQ-5D-5L mobility and usual activities dimensions (five-point scale for each dimension) 23 , and Patients’ Global Impression of Change (PGIC) assessments (seven-point scale with higher scores indicating a better impression of change).24,25 Disease-specific PRO anchors included the Pompe Disease Impact Scale (PDIS) difficulty performing activities domain (score range from 0 ‘not at all difficult’ to 4 ‘extremely difficult’) 26,27 and Pompe Disease Symptom Scale (PDSS) shortness of breath domain (score range from 0 ‘none’ to 10 ‘as bad as I can imagine’).26,27
Definitions of meaningful change for each PRO anchor.
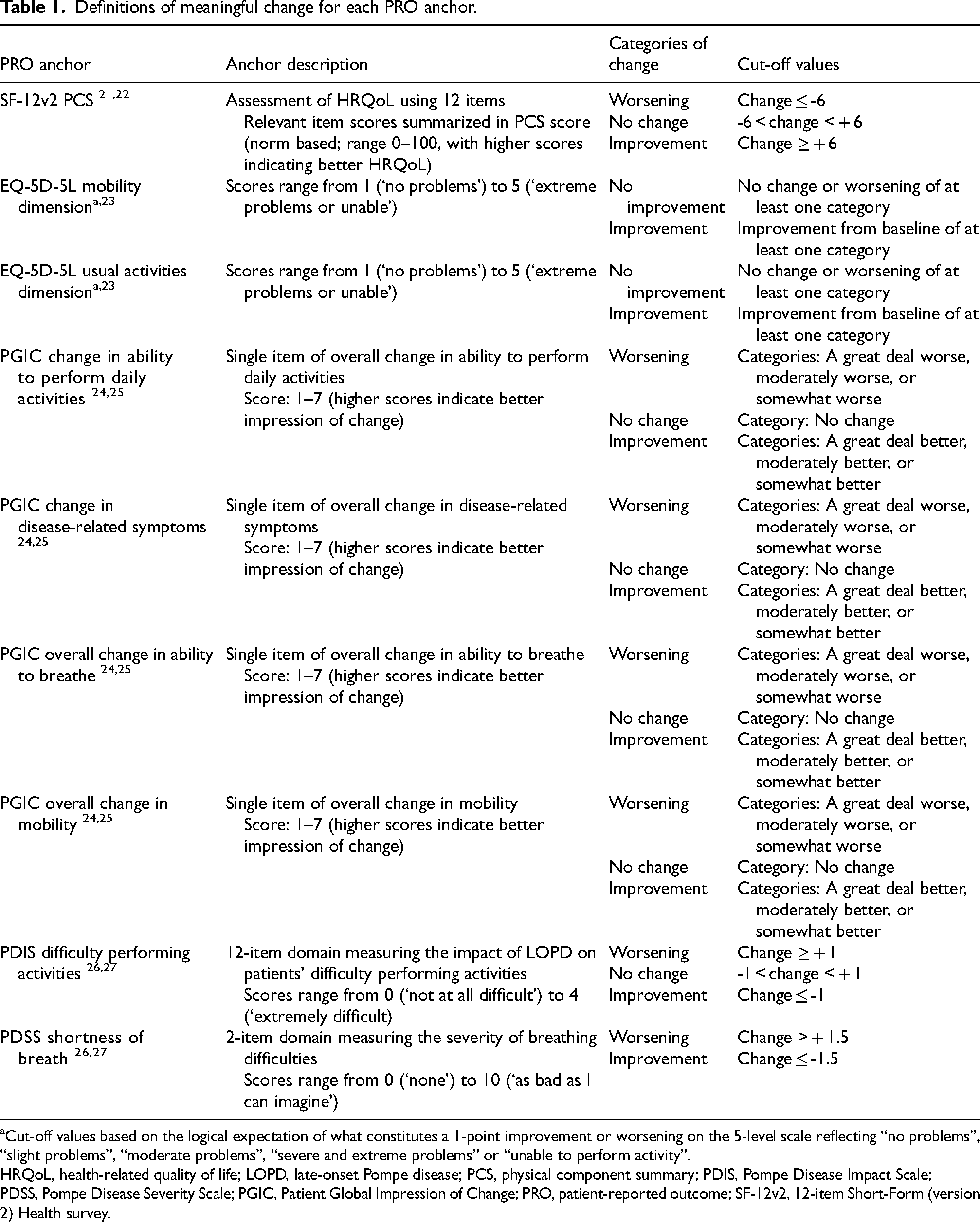
Cut-off values based on the logical expectation of what constitutes a 1-point improvement or worsening on the 5-level scale reflecting “no problems”, “slight problems”, “moderate problems”, “severe and extreme problems” or “unable to perform activity”.
HRQoL, health-related quality of life; LOPD, late-onset Pompe disease; PCS, physical component summary; PDIS, Pompe Disease Impact Scale; PDSS, Pompe Disease Severity Scale; PGIC, Patient Global Impression of Change; PRO, patient-reported outcome; SF-12v2, 12-item Short-Form (version 2) Health survey.
Within-person CMTs
Within-person CMTs for upright FVC (% predicted) were derived using distribution- and anchor-based methods.20,28 A distribution-based approach is a statistical method used to establish what amount of change in the measure of interest could be explained by the natural variability of the outcome values. Accordingly, it should be applied to values at a cross-sectional point, usually baseline data. An anchor-based method is a statistical approach used to determine clinically meaningful thresholds or differences by comparing changes in an “anchor” measure to changes in the measure of interest. The anchor measure should be simple to understand and the level of change that would be considered clinically meaningful in the relevant population should be established. The changes in the measure of interest are assessed for each anchor measure. Using multiple anchors is recommended to increase the reliability of the threshold estimates, and the results are then triangulated to obtain one single estimate.
The distribution-based method used was the standard error of measurement (SEM); a reliability coefficient of 0.93 was used based on the available literature 29 , as it was not possible to perform test-retest analyses on COMET study data. For anchor-based analyses, the PROs used as anchors in this study were selected given their presumed association to respiratory function. The response categories were considered as “improvement”, “no change” and “worsening” for each anchor and are depicted in Table 1. The appropriateness of each anchor was assessed using Spearman’s correlation (SF-12v2, PDIS, PDSS) or polyserial correlation (PGIC and EQ-5D-5L dimensions) for the relationship between the change in upright FVC (% predicted) and each anchor at Week 49 of the primary analysis period and Week 97 of the open-label extension period. Correlations ≥0.3 are considered desirable, though lower values can be justified in certain cases. 18 Since LOPD is a rare disorder limiting the size of the study population, anchors with positive or negative correlations exceeding 0.25 were considered in the estimation of the within-person change threshold. 18
Within-person change thresholds were calculated using descriptive statistics for absolute change in FVC expressed as % predicted from baseline to Week 49 or Week 97 for each anchor level (see Table 1 for categories). Values were presented on a forest plot to identify if there was overlap and convergence across anchors and time points (Week 49 and Week 97). The triangulation was performed using the correlation-weighted average approach 30 : a weighted average and 95% CI were calculated for both timepoints, in which within-person estimates from each anchor were weighted by their correlation. Within-person CMT estimates based on the relative change in FVC, expressed as % change, are presented in the Supplementary Material.
Anchor-based methods were supplemented by empirical cumulative distribution function (eCDF) curves. The eCDF curves separate patients into groups based on PRO responses (e.g., improved, no change, and worsened) and plots the change from baseline in upright FVC (% predicted) on the x-axis and the cumulative proportion of patients within each group experiencing up to that level of change on the y-axis.20,28
Between-group CMTs
To evaluate treatment group differences, between-group CMTs were estimated using anchor-based estimates and more specifically using the difference in mean change in FVC (% predicted) from baseline to Week 49 or Week 97 between patients reporting “any level of improvement” and those reporting “no change” (see Table 1). Data analysis revealed that in most cases the mean change from baseline in FVC (% predicted) in patients with one category improvement was numerically lower than that for “no change” (See Supplementary Table 2). Thus, to be conservative, improvement was defined as any improvement. The same anchors and the same appropriateness criteria were applied. A weighted average and the corresponding 95% CI were also calculated.
Meaningful change in FVC (% predicted) in the COMET trial
The derived within-person CMT was applied post hoc in an analysis of data from COMET; patients with a meaningful change in FVC (% predicted) were defined by the derived within-person CMT at Week 49. The proportion of patients with a meaningful increase in FVC (% predicted) between AVA and ALG were compared using a logistic regression model adjusted for baseline upright FVC (% predicted), age at baseline, and sex; odds ratios (ORs) and 95% CI were provided, along with the nominal p-value. The corresponding eCDFs were generated by treatment arm. Patients with missing data for change from baseline were considered as patients with no meaningful FVC (% predicted) increase.
The derived between-group CMT was also compared with the observed group difference in the COMET trial to determine if the difference between AVA and ALG on FVC (% predicted) was clinically meaningful at a group level.
All analyses were performed using SAS (version 9.4).
Results
Study population
The modified intention-to-treat (mITT) population included 100 participants from COMET, of whom 99 were aged ≥18 years. Baseline demographics and characteristics of the adult participants were representative of the general population with LOPD (Table 2). The mean ± standard deviation (SD) age of COMET participants was 48.0 ± 13.9 years (range 19–78 years) with similar distribution between male and female. Most participants were white (94.9%) and enrolled in Europe (51.5%). The mean ± SD age at onset of first symptoms of Pompe disease was 35.5 ± 16.2 years and the baseline mean upright FVC was 61.8 ± 13.3% predicted.
Baseline characteristics and PRO scores (mITT adult population).
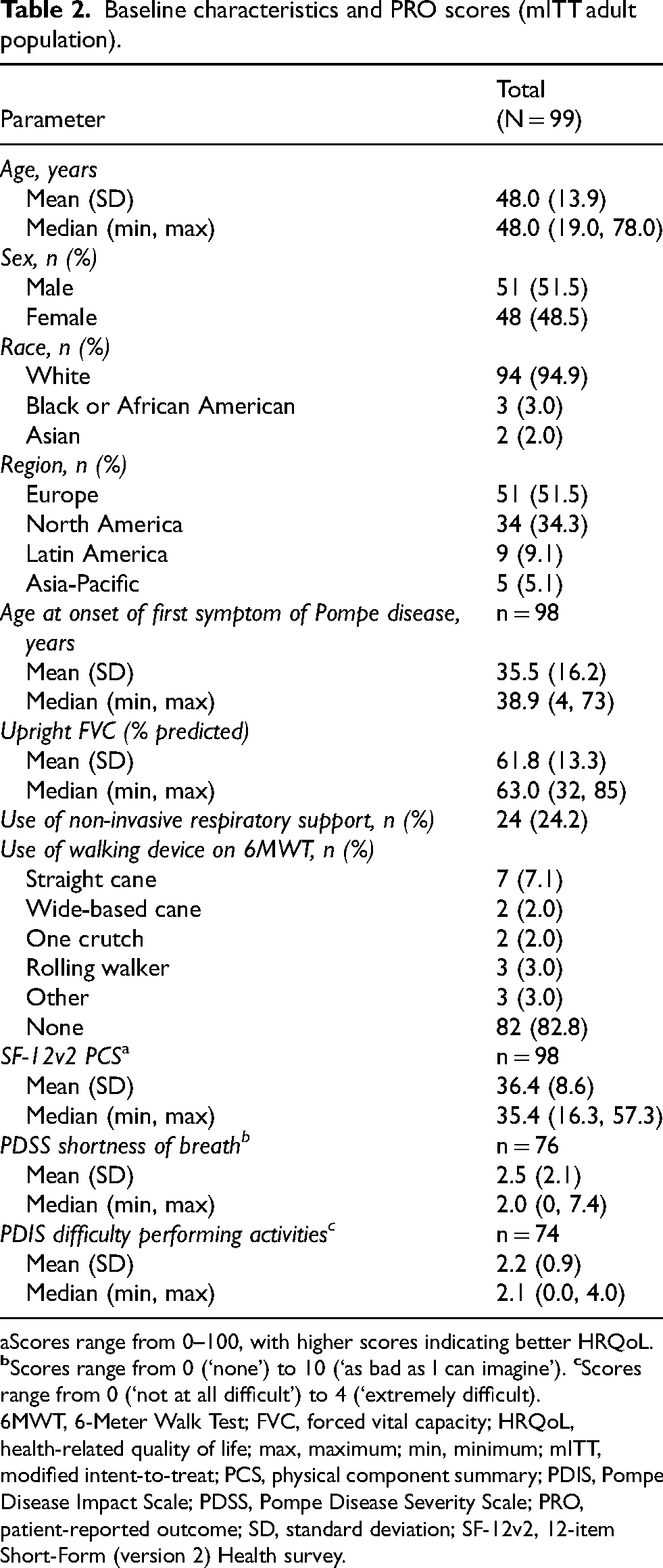
Scores range from 0–100, with higher scores indicating better HRQoL.
6MWT, 6-Meter Walk Test; FVC, forced vital capacity; HRQoL, health-related quality of life; max, maximum; min, minimum; mITT, modified intent-to-treat; PCS, physical component summary; PDIS, Pompe Disease Impact Scale; PDSS, Pompe Disease Severity Scale; PRO, patient-reported outcome; SD, standard deviation; SF-12v2, 12-item Short-Form (version 2) Health survey.
Within-person CMT
The distribution-based method yielded an estimated CMT for FVC of 3.5% predicted using the SEM, computed using the baseline SD of FVC (SD = 13.3) for all 99 patients without regard to their subsequent treatment arm.
For anchor-based estimates, the correlation coefficients between the various PROs and change in FVC (% predicted) observed at Week 49 and Week 97 are presented in Table 3. Values in bold reflect anchors that exceeded the aforementioned 0.25 threshold. Correlations at Week 49 ranged from 0.02 to 0.28, and at Week 97 ranged from 0.01 to 0.42. Anchors used to derive the CMT for FVC at Week 49 included the SF-12v2 PCS, PGIC change in the ability to perform daily activities and change in disease-related symptoms assessments, and PDIS difficulty performing activities domain. At Week 97, the PGIC change in the ability to perform daily activities, change in disease-related symptoms, and overall change in ability to breathe assessments were used as anchors. Correlations for relative change data are presented in Supplementary Table 1.
Correlation between change in anchors and absolute change in upright FVC (% predicted) at Week 49 and Week 97.
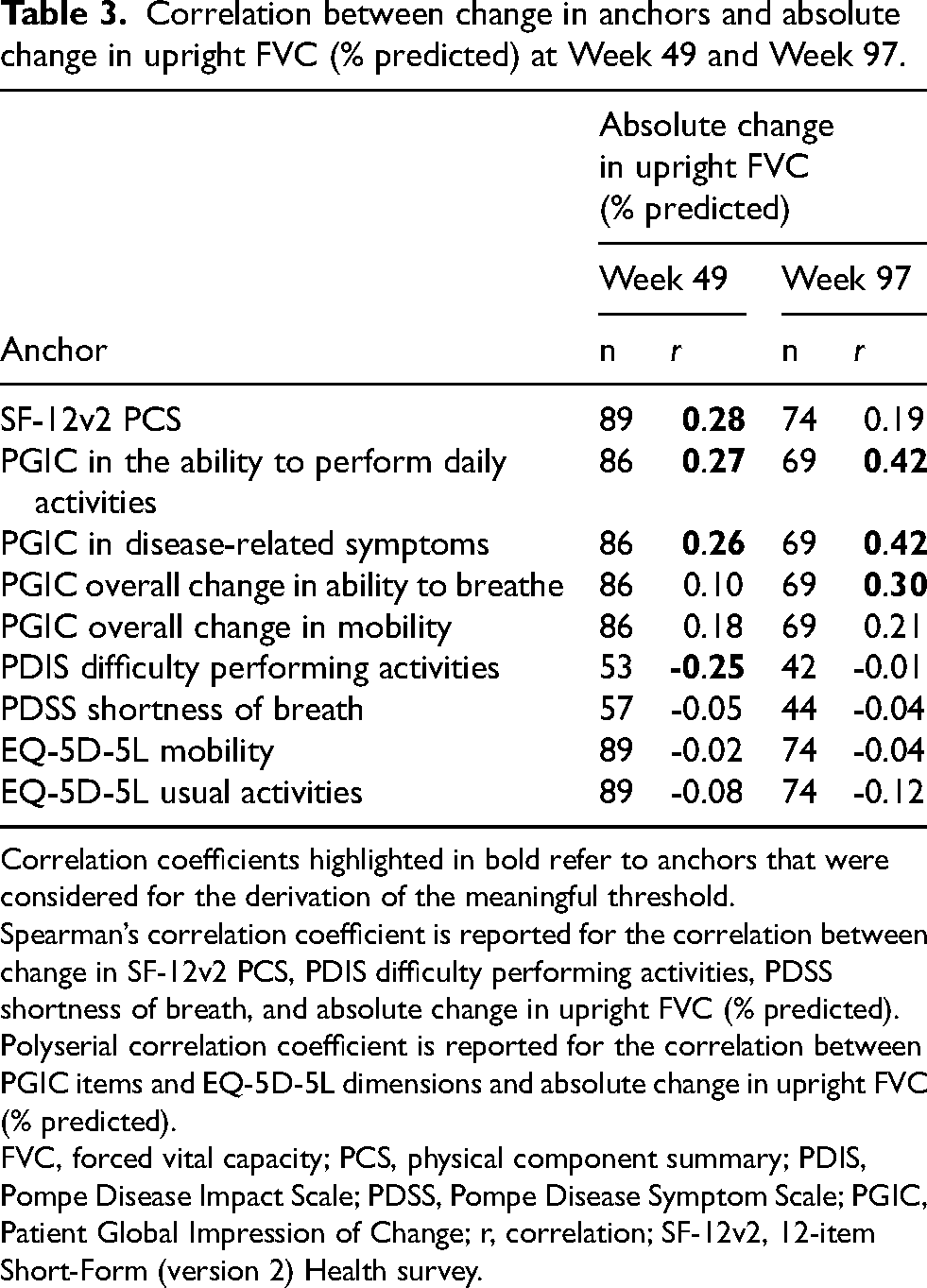
Correlation coefficients highlighted in bold refer to anchors that were considered for the derivation of the meaningful threshold.
Spearman’s correlation coefficient is reported for the correlation between change in SF-12v2 PCS, PDIS difficulty performing activities, PDSS shortness of breath, and absolute change in upright FVC (% predicted). Polyserial correlation coefficient is reported for the correlation between PGIC items and EQ-5D-5L dimensions and absolute change in upright FVC (% predicted).
FVC, forced vital capacity; PCS, physical component summary; PDIS, Pompe Disease Impact Scale; PDSS, Pompe Disease Symptom Scale; PGIC, Patient Global Impression of Change; r, correlation; SF-12v2, 12-item Short-Form (version 2) Health survey.
The absolute change in FVC expressed as % predicted across the anchor outcome groups is shown in Supplementary Table 2. The majority of patients showed no change in SF-12v2, PDIS, and PDSS measures, with less than 10% of patients reporting symptomatic worsening. In contrast, improvement of symptoms (small, moderate, or large) was noted for 49–69% of patients on PGIC anchors across time points, with fewer individuals (7–23%) reporting worsening of their symptoms. For most anchors/PROs, a graded response in FVC (% predicted) was noted, whereby an increase in FVC was observed in patients categorized as having improvements in the anchor score. Conversely, a decrease was observed in patients having a worsening in the anchor score. Data for relative change in FVC expressed as % change across the anchor groups are shown in Supplementary Table 3.
To inform a recommended threshold for absolute change in upright FVC (% predicted), all CMT estimates from appropriate anchors were triangulated (Figure 1A). A narrow range of CMTs were noted at both Week 49 and Week 97 across anchor-based estimates (2.2–3.9% and 2.6–3.0%, respectively) (Figure 1A). The correlation-weighted estimate for absolute change in upright FVC was 3.0% predicted [95% CI 2.3, 3.8] at Week 49 and 2.9% predicted [95% CI 2.6, 3.1] at Week 97. The eCDF curves show a clear separation between patients experiencing worsening and those experiencing improvement on the anchors across the full range for change in FVC (% predicted) at Week 49 (Supplementary Figure 1). Similar analysis of the relative change in FVC expressed as % change yielded a CMT of 4.9% [95% CI 3.5, 6.2] at Week 49 and 4.2% [95% CI 3.5, 4.8] at Week 97 (Supplementary Figure 2A); eCDF curves for relative change in FVC expressed as % change at Week 49 are shown in Supplementary Figure 3.
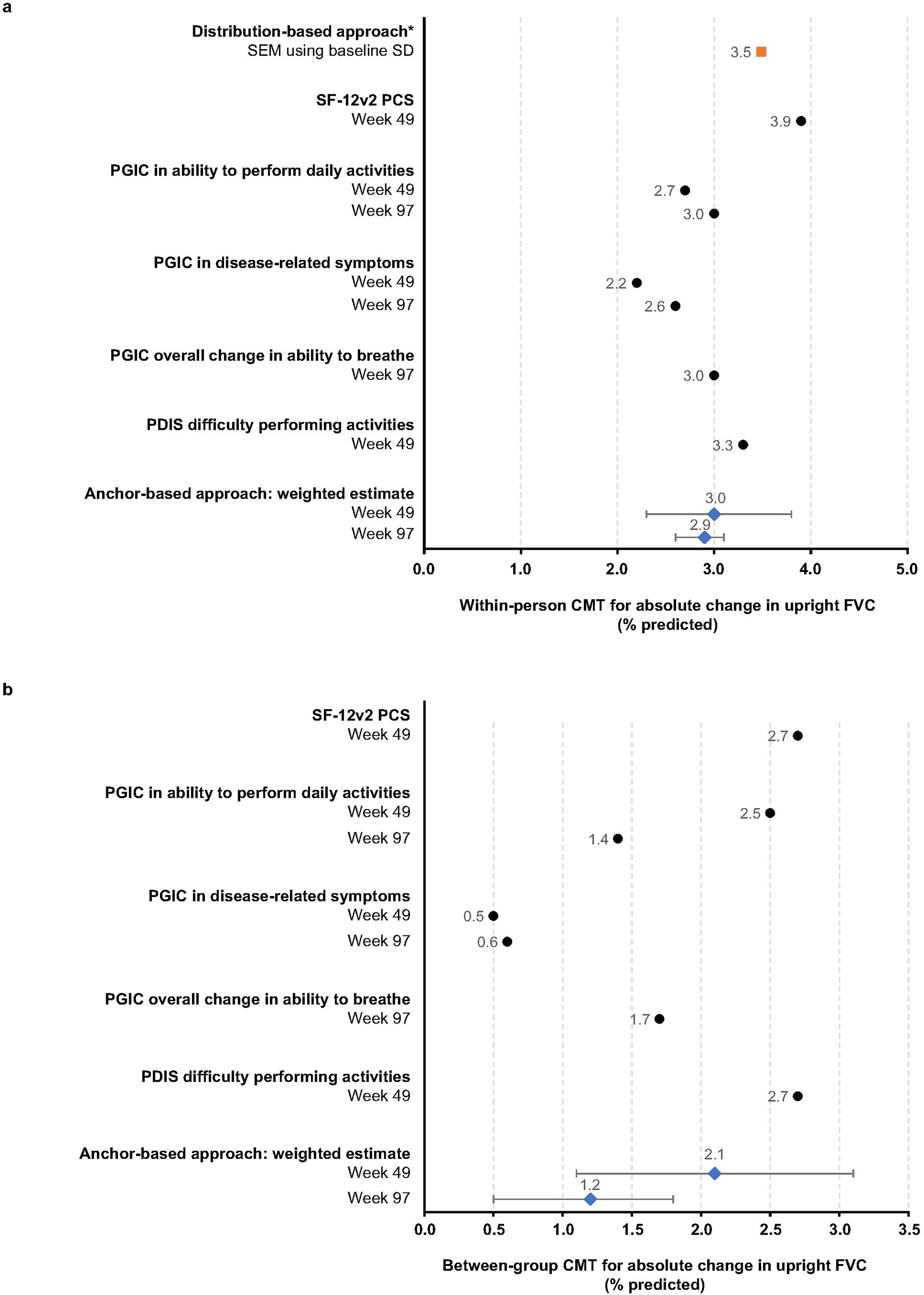
Results of the a. within-person and b. between-group CMT estimates for absolute change in FVC (% predicted): anchor-based analyses and distribution-based analyses. *Estimates from the distribution-based approach were not included in the weighted estimate for the within-person CMT. Distribution-based estimate is represented as an orange square. Anchor-based individual estimates are represented as black circles. Correlation-weighted estimates are represented in blue diamonds along with their 95% CI and considered only those anchors with r ≥ 0.25 at Week 49 or Week 97. CI, confidence interval; CMT, clinically meaningful threshold; FVC, forced vital capacity; PCS, physical component score; PDIS, Pompe Disease Impact Scale; PGIC, Patient Global Impression of Change; r, correlation; SD, standard deviation; SEM, standard error of the mean; SF-12v2, 12-item Short-Form (version 2) Health survey
Between-group CMTs
The complete range of estimates and the suggested between-group CMTs at Weeks 49 and 97 are shown in Figure 1B. The correlation-weighted average anchor-based CMT for the between-group absolute change in FVC at Week 49 was 2.1% predicted [95% CI 1.1, 3.1]. Estimates were slightly lower at Week 97 (1.2% predicted [95% CI 0.5, 1.8]) with overlapping confidence intervals. For relative change, the correlation-weighted average % difference in FVC at Week 49 was 3.5% [95% CI 1.6, 5.4] and at Week 97 was 1.9% [95% CI 0.4, 3.5] (Supplementary Figure 2B).
Post hoc application to the COMET trial
The derived within-person CMT was used in a post hoc analysis of COMET data to determine the proportion of patients that achieved a meaningful increase in FVC (% predicted) in the AVA and ALG groups at Week 49. Data are shown in Figure 2 across the full range of the derived within-person CMTs (2.2 to 3.9% predicted). At the 3% CMT, a greater number of patients in the AVA group showed an increase in upright FVC (% predicted) from baseline to Week 49 compared with those in the ALG group (OR 2.3; nominal p = 0.058). Data spanning the CI of the estimated CMT from 2% to 4% predicted showed similar ORs (2.6 and 2.4, respectively), with nominal p-values ranging from 0.026 to 0.045. The eCDF curve for within-person change in upright FVC (% predicted) shows clear separation between groups at each level of change from baseline to Week 49, with fewer patients treated with AVA demonstrating worsening of FVC (% predicted) compared with those treated with ALG (Figure 3).
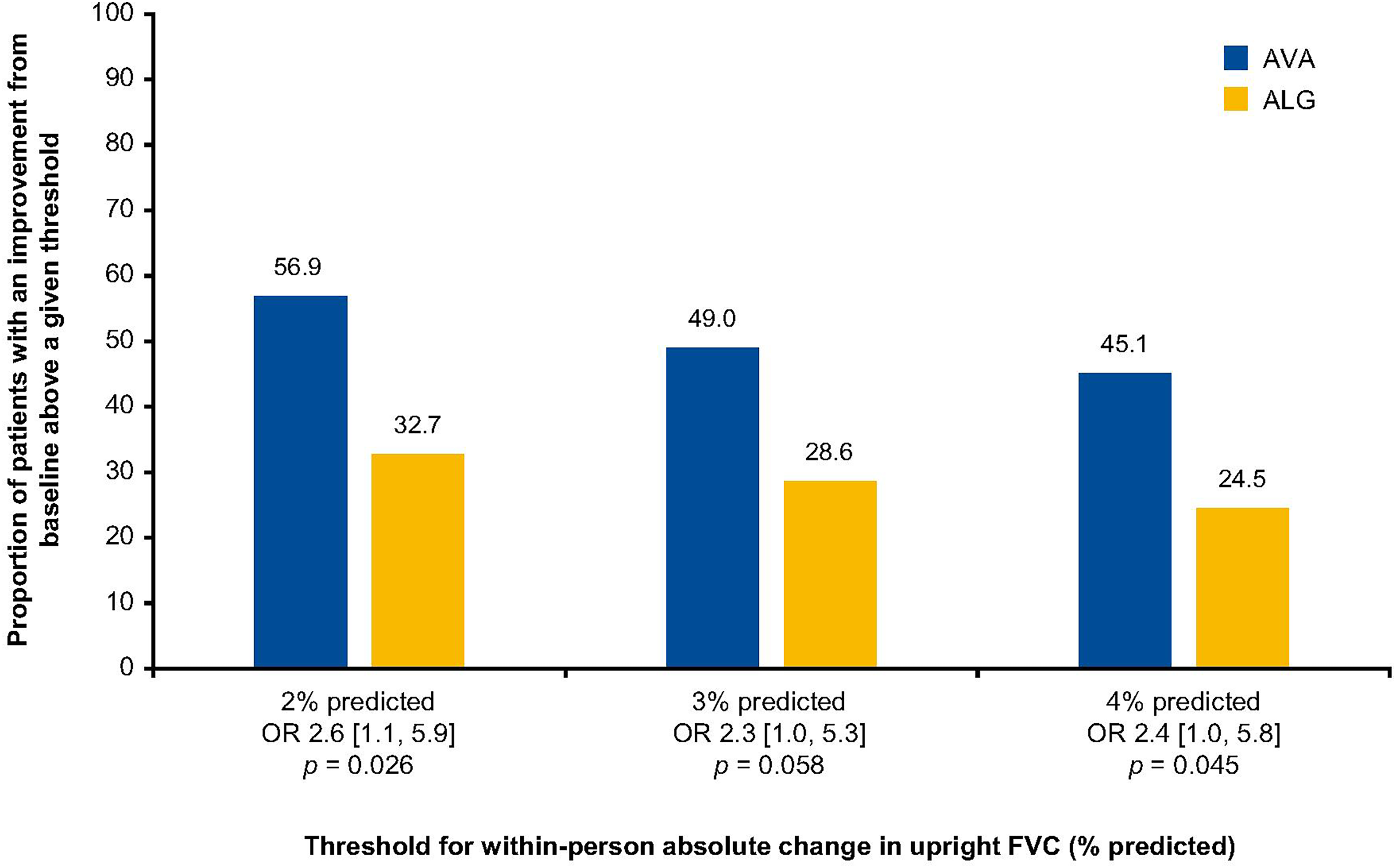
Patients with a meaningful change from baseline to Week 49 in upright FVC (% predicted). OR [95% CI] are presented. OR values larger than 1.0 represent better odds for AVA compared with ALG. ALG, alglucosidase alfa; AVA, avalglucosidase alfa; CI, confidence interval; FVC, forced vital capacity; OR, odds ratio
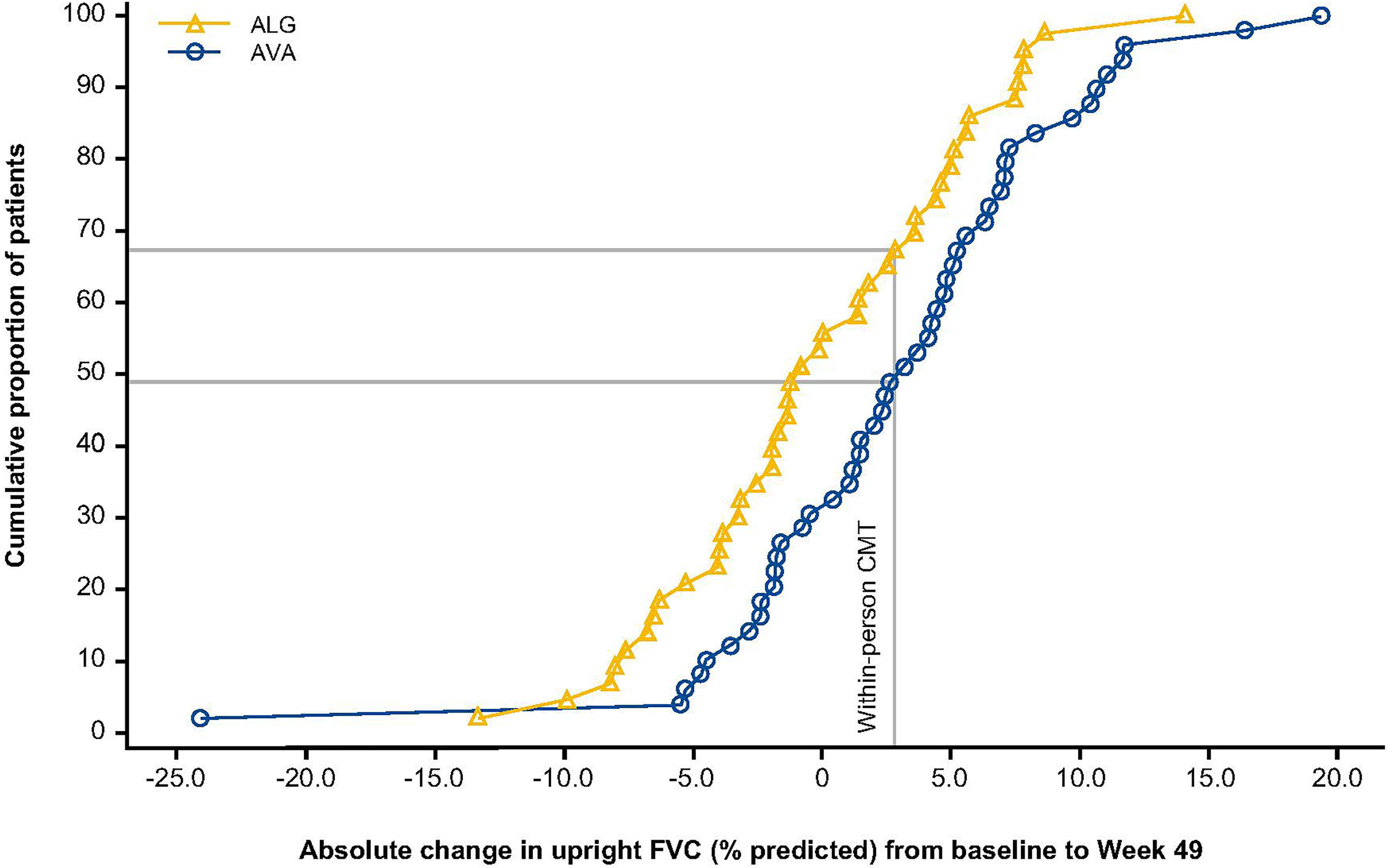
Empirical cumulative distribution function of absolute change in upright FVC (% predicted) from baseline to Week 49 by treatment groups. Only patients with non-missing change from baseline are included in this graph. ALG, alglucosidase alfa; AVA, avalglucosidase alfa; CMT, clinically meaningful threshold; FVC, forced vital capacity
The results of the analyses using the clinically meaningful relative change in FVC expressed as % change to compare patients treated with AVA versus ALG are shown in Supplementary Figure 4. At the 5% CMT, a greater number of patients in the AVA versus ALG group showed a clinically meaningful relative increase in upright FVC (% change) at Week 49 (OR 2.5 [95% CI 1.1, 5.9]; nominal p = 0.032). Corresponding eCDF curves for relative change in FVC expressed as % change are shown in Supplementary Figure 5.
The observed between-group difference in FVC (% predicted) between the AVA and ALG groups in COMET of 2.43% [95% CI -0.13, 4.99] at Week 49 16 , resides within the derived between-group CMT interval (0.5 to 2.7%) and is above the point estimate of 2.1%, suggesting a clinically meaningful difference for the patient between the two treatments.
Discussion
The present study expands the knowledge of CMTs for upright FVC (% predicted), based on the patient perspective, by deriving threshold values in a sample of patients with LOPD, with potential for extrapolation to other progressive NMDs with respiratory system involvement such as muscular dystrophies and primary myopathies. While any improvement in FVC (% predicted), including stabilization, is important and will be likely to delay the onset of respiratory outcomes (e.g., respiratory failure, need for invasive ventilation, or need for ventilatory support) 6,7, it remains important to quantify whether the improvements in respiratory function observed in clinical trials are associated with patient perception of a clinical benefit.
One important goal common for clinical trials is determining if the effect of the experimental treatment is significantly better than that of the control treatment. However, statistical significance does not, by itself, indicate whether the detected effect corresponds to a clinically meaningful treatment effect for patients. It is thus important to identify CMTs for the population of interest and apply them to clinical trial findings as a complement to statistical interpretations of the results.
We estimated within-person and between-group CMTs using both anchor- and distribution-based methods; multiple anchors were applied at two trial time points, in accordance with methodological guidelines.20,28 The preference or prioritization of the within-person or between-group CMT depends on the points of view, motivations, and interests of the different stakeholders. Namely, the US FDA routinely requests clinically meaningful within-person thresholds to supplement end points derived from group-level data (e.g., mean change from baseline). 20 Conversely, the European Medicines Agency commonly utilizes between-group estimates to contextualize statistically significant differences observed between treatment groups. 31
The estimates for within-person CMT derived in this study were closely aligned across multiple analyses. The results indicate that patients will likely perceive a clinical benefit when the absolute improvement in upright FVC is in the range between 2.2 to 3.9% predicted. The observed range for within-person improvement in upright FVC (% predicted) from our study is in line with estimates from patients with lung restriction due to idiopathic pulmonary fibrosis. 29 In addition, while our estimates for the between-group CMT differed slightly (approximately 1 to 2% predicted) from those derived in a recently published study, it likely reflects differences in the anchors used and the baseline characteristics of the respective study populations. 32 Nevertheless, the confidence intervals for the between-group CMTs were largely overlapping reinforcing their applicability to clinical studies to evaluate differences between treatment groups. In the pivotal Phase 3 COMET trial, the observed difference in FVC of 2.43% predicted between AVA and ALG treatment was within the derived between-groups CMT range reported in the present analysis (0.5 to 2.7% predicted), confirming the presence of clinically meaningful benefits with AVA treatment. 16
An additional post hoc analysis focused on the proportion of patients who experienced a meaningful improvement (i.e., increase) in FVC (% predicted) over the 49-week treatment period. Maximizing the rate of meaningful increases in FVC (% predicted) is an important treatment goal; however, in a progressive disease such as LOPD, clinically meaningful improvement could also be defined as the prevention of meaningful worsening of PRO anchor measures or reducing the percentage of patients who experience a meaningful decrease in FVC (% predicted). There were far fewer patients in the COMET trial who reported worsening on the PRO anchor measures, which makes it difficult to estimate the threshold for meaningful FVC worsening with confidence. However, the percentage of patients with any worsening of FVC (% predicted) was smaller in the AVA group than the ALG group (shown in Figure 3). These considerations highlight the importance of considering both types of improvement, achievement of a meaningful increase and prevention of meaningful worsening in FVC (% predicted), when evaluating treatment effectiveness.
Application of the CMTs for FVC derived in the present study to certain other NMDs may be justified since these diseases share a common pathophysiology for development of respiratory failure. For instance, sleep-related hypoventilation is the precursor of diurnal respiratory failure in NMDs and manifests at a similar extent of lung restriction across different NMDs.6,7,33 In addition, the need for mechanical ventilatory support in NMDs occurs at similar values of vital capacity as with Pompe disease. 7 Given the aforementioned similarities between LOPD and other NMDs, it may be justifiable to extrapolate the results from this study to a more general NMD context that can be confirmed by future studies.
The present study has several strengths. Despite the rareness of LOPD, 99 adult patients were enrolled in COMET allowing for robust analyses and conclusions. In addition, multiple anchors were used to derive within-person and between-group CMT estimates for upright FVC, similar to those anchors used in a study reporting within-person change thresholds in patients with idiopathic pulmonary fibrosis. 29 The estimates obtained were also largely consistent across various anchors and time points, thus providing further confidence in the findings of our study.
There are additional factors that should be considered as potential limitations when evaluating these results. Firstly, weak correlations were observed in this study between PROs and the change in upright FVC (% predicted), likely due to the limited population size available in this rare disease and the heterogeneity of disease manifestations across patients. Consequently, the threshold for correlation coefficients was lowered to 0.25 rather than the cut-off of 0.30 suggested by Revicki et al. [2008]. 18 Secondly, between-group clinically meaningful changes are generally derived by comparison between individuals who report being “a little better” (improvement) or “a little worse” (worsening) and those exhibiting “no change”. As LOPD is a rare disease with limited population size, all individuals with any change on the anchor were included. While this approach was successful, it should be noted that the estimated CMT should be considered a conservative high value, potentially overestimating the minimally important improvement threshold. 18 Lastly, in the present study, the responder analysis of COMET FVC data was hindered by lack of prior published within-person CMTs in other populations of patients with NMDs, including LOPD. Accordingly, the derivation of a within-person CMT and subsequent responder analysis had to be obtained from the same study. To avoid bias, the focus was thus on the 49-week double-blind portion of the COMET study.
In conclusion, this analysis successfully identified a narrow range of within-person and between-group CMTs for upright FVC (% predicted) in LOPD. When applying the within-person thresholds to COMET trial data, we observed a greater proportion of patients treated with AVA had clinically meaningful improvements in respiratory function compared with those receiving ALG therapy. These estimates will aid healthcare providers caring for patients with LOPD in assessing the clinical significance of: 1) a longitudinal change in FVC in a given patient, by using the within-person CMT and 2) an observed difference in lung function outcomes across clinical trial patient groups by using the between-group CMT. Given the similar pathophysiology of respiratory failure in LOPD and other NMDs, this study’s findings may aid in interpretation of data obtained from interventional studies in other neuromuscular conditions.
Supplemental Material
sj-docx-1-jnd-10.1177_22143602251332829 - Supplemental material for Defining clinically meaningful thresholds for forced vital capacity in patients with neuromuscular disorders: Lessons learned from the COMET study in Pompe disease
Supplemental material, sj-docx-1-jnd-10.1177_22143602251332829 for Defining clinically meaningful thresholds for forced vital capacity in patients with neuromuscular disorders: Lessons learned from the COMET study in Pompe disease by Kenneth I Berger, Cristina Ivanescu, Jérôme Msihid, Magali Periquet, Alaa Hamed, Kristina an Haack, Tianyue Zhou, Nadine van der Beek, Matthias Boentert, Ruth Pulikottil-Jacob and Laurence Pollissard in Journal of Neuromuscular Diseases
Footnotes
Acknowledgments
The authors would like to thank all the patients, their families, and the investigators and staff from participating sites involved in this trial. The authors would also like to thank Christine Taniou (Aixial company), funded by Sanofi, for the programming of the statistical analyses; Christina Daskalopoulou and Sara Bestea (IQVIA), funded by Sanofi, for their contributions to the drafting of this manuscript; and Pranshu Roy (Sanofi) for consolidating comments during manuscript development. Medical writing and editorial support, under the direction of the authors, was also provided by Amy Watkins, PhD, and Kerry Guest, PhD, of Ashfield MedComms, an Inizio company, and funded by Sanofi in accordance with Good Publication Practice guidelines.
The authors would like to acknowledge Giulio Flore, MSc, (IQVIA) for his contributions to the analyses discussed in the manuscript before his death on November 4, 2022.
Author contributions
All authors were involved in drafting the work or revising it critically for important intellectual content, and in final approval of the version to be published. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Sanofi.
Conflict of Interest
Data availability
Qualified researchers may request access to patient-level data and related trial documents including the trial protocol with any amendments, statistical analysis plan, and dataset specifications. Further details on Sanofi’s data sharing criteria, eligible studies, and process for requesting access can be found at: ![]() .
.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
