Abstract
Background:
Few studies have examined respiratory dysfunction in patients with Becker muscular dystrophy (BMD).
Objective:
This study aimed to examine the characteristics of respiratory dysfunction in patients with BMD.
Methods:
The present retrospective study assessed respiratory parameters of adult BMD patients using medical records and compared these parameters with various patient characteristics to identify correlations. BMD patients aged 17 years and older who had been diagnosed genetically and/or pathologically were included in the analysis.
Results:
Of the source population of 133 patients, respiratory function was assessed in 85. Two of these patients had no symptoms, and eight had died. Mean % forced vital capacity (% FVC) was 94.2+/–21.7% (median, 96.1%; range, 5.1–134.1%). In 16 (19%) of the 85 patients, % FVC was <80%. Of these, seven were non-ambulant. Age, ambulation, and cardiac function did not significantly differ between patients with or without respiratory dysfunction, whereas age at onset was significantly lower in patients with respiratory dysfunction (7.7+/–4.7 years vs. 14.4+/–11.9 years; p = 0.001). One non-ambulant patient was a continuous NPPV user, and one patient had been recommended NPPV use but refused. Autopsy of one patient revealed that the diaphragm and intercostal muscles were less affected than proximal skeletal muscles.
Conclusion:
BMD patients are at risk of developing respiratory dysfunction due to dystrophic changes in respiratory muscles. Respiratory function should be carefully and periodically monitored in these patients.
Keywords
INTRODUCTION
Becker muscular dystrophy (BMD, OMIM 300376) is an X-linked recessive form of muscular dystrophy caused by mutations in the dystrophin (DMD) gene on chromosome Xp21.2 [1]. The DMD gene is the largest gene identified in humans and contains 79 exons. Mutations in this gene prevent the production of functional dystrophin protein [2]. Since the discovery of the DMD gene, efforts have been ongoing to develop effective strategies to treat Duchenne muscular dystrophy (DMD) and BMD.
DMD is associated with mutations that interrupt the open reading frame of the DMD gene, resulting in severe muscle weakness and, typically, death before the age of 30 years [3, 4]. In contrast, BMD is typically associated with mutations that maintain the open reading frame, producing an altered, but partially functional, dystrophin protein with no change in its C-terminal domain [3, 5]. BMD patients exhibit variable phenotypes, but are less severely affected than those with DMD, and have a much longer normal life expectancy [5].
Only a few studies have touched on respiratory dysfunction in BMD patients. We previously reported that 27% of BMD patients had respiratory dysfunction based on data from the national registry of Japanese patients with dystrophinopathy (Registry of Muscular Dystrophy; Remudy. http://www.remudy.jp/) [6]. In that study, among 35 BMD patients with respiratory dysfunction, one ambulant patient used non-invasive positive pressure ventilation (NPPV). However, no study to date has described the risk factors and background of respiratory dysfunction in this patient population. To this end, the present study aimed to investigate the clinical details of BMD patients by assessing their past and present respiratory parameters, and to identify factors that correlate with the severity of respiratory dysfunction.
PATIENTS AND METHODS
Aim and design
This study aimed to clarify whether BMD is a risk factor for respiratory dysfunction. We also assessed whether respiratory dysfunction correlates with phenotypes and/orgenotypes of BMD patients. This study was approved by the Ethics Committee of the National Center of Neurology and Psychiatry (NCNP).
Patients
We enlisted male patients with dystrophinopathy who visited the National Center Hospital of NCNP from July 1979 to September 2016. Patients who were ambulant at age 17 were defined as having BMD in this study [6]. Among these patients, those with BMD who were aged 17 years and older, and whose respiratory function was evaluated in the supine or sitting position, were selected for this study. Respiratory dysfunction was defined as % forced vital capacity (% FVC) <80%. Cardiac dysfunction was defined as the state in which patients required administration of cardiac protecting medication such as angiotensin converting enzyme inhibitors or beta-blockers, or had an ejection fraction <50%. None of the patients in this study were administered cardioprotective medication before they began to show signs of reduced cardiac function.
Study population
Medical records of genetically and/or pathologically confirmed BMD myopathy patients who underwent pulmonary function tests at the National Center Hospital of NCNP were retrospectively reviewed. We collected data on genetic diagnosis, cardiac function, and % FVC, as well as chest X-rays and/or CT scans, for analysis.
Data handling and analysis
Data were analyzed using descriptive statistics, and each variable was compared against age, respiratory dysfunction, cardiac dysfunction, and mutations. The t-test was used to compare means of the two groups. Data for the two study populations were analyzed using chi-square contingency table analysis. All analyses were performed using SPSS for Macintosh (Version 18; SPSS Inc., Chicago, IL).
RESULTS
General characteristics (Table 1)
At the end of September 2018, a total of 133 BMD patients who met the inclusion criteria were considered candidates. Among these, 85patients were evaluated using pulmonary function tests. Mean age at evaluation of % FVC was 38.7+/–12.4 years (range, 14–65; median, 38) and mean age at BMD onset was 13.18+/–9.5 years (range, 1–53; median, 9.5). Eighty-three patients were symptomatic (any skeletal muscle symptoms) and two were asymptomatic (no skeletal muscle symptoms) presenting only with hyperCKemia;60 (70.7%) patients were ambulant, while 25 (29.3%) were not; and mean age at loss of ambulation was 34.7+/–10.4 years (range, 18–54; median, 34.5). Eight patients had died due to sudden death (3), gastric cancer (1), acute abdomen (1), cardiopulmonary failure after choking (1), heart failure (1), and unknown cause (1). Patients who died of cardiopulmonary failure were autopsied.
General patient characteristics
Reduced respiratory function was observed in 20% of patients. No significant difference in age was found between patients with or without respiratory dysfunction. Two patients were asymptomatic and only had high CK.
Respiratory function
% FVC in BMD patients was 94.2+/–21.7 % (range, 5.1–134.2; median, 96.1) in the sitting position. When compared in the same patient, % FVC in the sitting position was significantly larger than that in the supine position (97.8+/–17.1 vs. 94.4+/–16.0; p < 0.001). The maximal difference between sitting and supine positions was 26.5% (sitting vs. supine: 111.1 vs. 84.6). There were no significant differences in % FVC by ambulation status (ambulant without assistance vs. ambulant with assistant vs. non-ambulant: 96.8+/–14.9 vs.101.1+/–21.8 vs.86.1+/–31.1) and cardiac dysfunction (with vs. without cardiac dysfunction: 90.3+/–25.0 vs. 98.6+/–16.8; p = 0.074). There was no significant correlation between age and % FVC (ρ= 0.005, p = 0.964).
Sixteen patients(19%) had a % FVC <80. Of these, 15 did not have lung and/or thoracic diseases that could affect their respiratory function in chest X-rays and/or chest CT scans at the first evaluation after presenting with respiratory dysfunction. One patient had not been evaluated radiologically. Of the 16 patients with % FVC <80, seven were non-ambulant and their symptom onset was significantly earlier than patients with normal respiratory function (with vs. without respiratory dysfunction: 7.7+/–4.7years vs. 14.4+/–11.9 years; p = 0.001). Two patients had respiratory failure; one patient required nocturnal NPPV and another patient who refused NPPV died. These patients were all early onset (before 10 years old), non-ambulant, had cardiomyopathy, and had no deletion or duplication mutations in multiplex ligation-dependent probe amplification (MLPA) or multiplex PCR (sequence examination were not performed).
When reviewing past data of included patients, % FVC decrements in several non-ambulant patients were greater than 20% during 10 years, whereas these decrements were not observed in any of the ambulant patients (Fig. 1A and 1B).
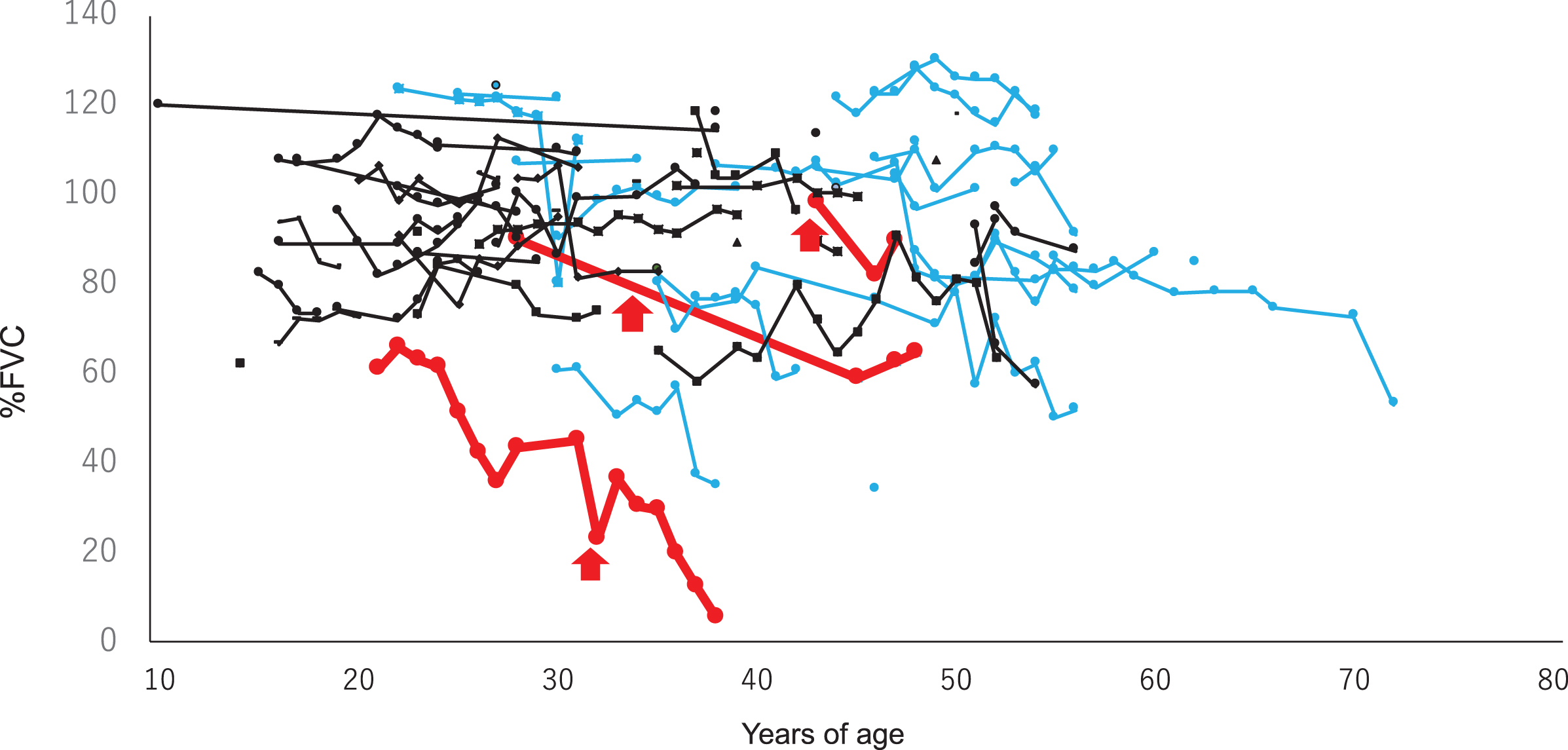
Serial changes in % FVC of ambulant (n = 61,
Genetic analysis (Table 3, Supplementary Table 1)
There were 57 (67.1%) patients with deletion mutations, 4 (4.7%) with duplication mutations, 4 (4.7%) with small mutations, 11 (11.8%) with no mutations by MLPA, and 9 (10.6%) with no mutations by multiplex PCR (Table 2). Dystrophinopathy was confirmed by immunohistochemical staining with muscle biopsy specimens for those with no mutations. Compared to patients with deletion mutations, those with micromutations and no mutations (as assessed by MLPA and multiplex PCR) had higher % FVC decrements and a higher incidence of respiratory dysfunction.
Genotyping and clinical characteristics
Patients carrying micromutations and patients without no deletion/duplication mutations had more severe % FVC decrements and a higher incidence of respiratory dysfunction. Note that patients who underwent no genetic examinations are excluded from this table.
Autopsy findings (Figs. 2, 3)
The autopsy record of a 72-year-old man with respiratory failure (final % FVC, 53%) was available for analysis. The patient noticed calf hypertrophy at age 10. He lost ambulation at age 45 and was diagnosed with BMD (Fig. 2A and 2B). Mini-multiplex Western blotting showed a deficiency of dystrophin protein, which supported the diagnosis of dystrophinopathy (Fig. 2J). He first visited our hospital at age 43, presenting with marked thigh atrophy and calf hypertrophy. His CK was 1862 IU/L. No obvious mutation was found by MLPA or sequencing of the DMD gene. Treatment for cardiomyopathy began at age 55, and respiratory function had deteriorated since age 61 (Fig. 3A). Chest CT at age 70 (two years before death) revealed moderate thinning of the diaphragm (Fig. 3B–3E). The patient died of cardiopulmonary failure as a result of refusing NPPV or any other form of resuscitation after choking at age 72. His autopsy revealed that almost all fibers were replaced by adipose tissue and fibrous tissue in the biceps brachii, quadriceps, and iliopsoas, whereas the diaphragm and inter costal is showed moderate to severe fiber size variation, endomysial fibrosis, and adipose tissue infiltration. Muscle fibers of the tongue, pharyngeal, and cricothyroid muscles were affected much less than respiratory muscles (Fig. 2C–2J).
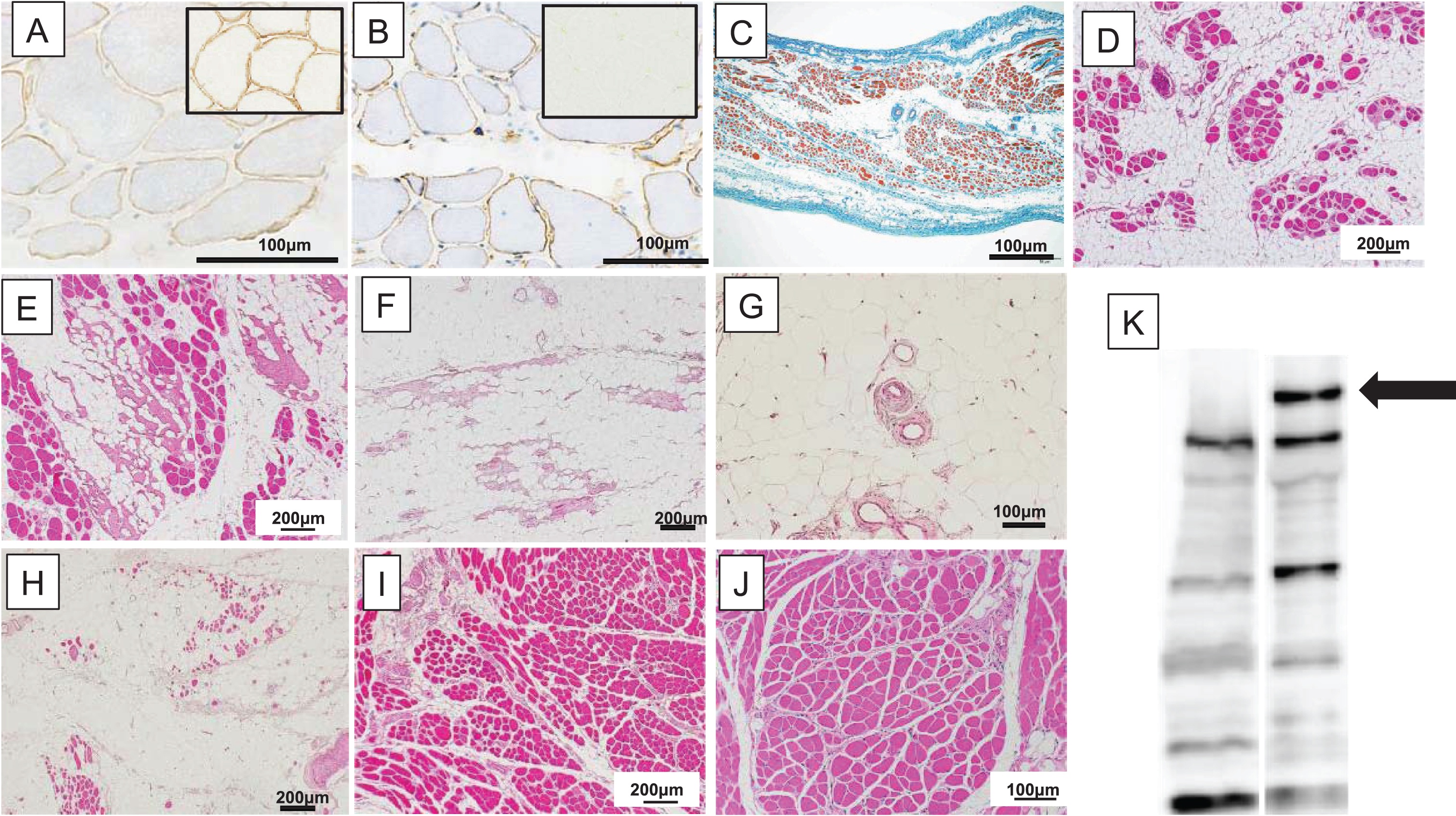
Autopsy findings in patients with respiratory disfunction. (A, B) Biopsy findings in a 43-year-old patient. (C–F) Autopsy findings at age 72 (paraffin-embedded). Immunostaining of (A) dys2 (1:100) showed faint and patchy staining of sarcolemma (controlsare shown in the inlets). (B) Up-regulation of sarcolemmalutrophin (DRP2 (1:100)), which is the autosomal homolog of dystrophin that is upregulated in dystrophinopathy sarcolemma, was observed. Autopsy findings revealed that loss of muscle fibers in (C, D) the diaphragm and (E) intercostalis were milder than in the (F) biceps brachii, (G) quadriceps, and (H) iliopsoas, whereas muscle fibers of (I) the tongue and pharyngeal (J) muscles were more spared compared to respiratory muscles. Mini-multiplex Western blot analysis [10] of biopsied muscle (left lane) showed a deficiency of dystrophin protein (arrow, Anti-dystrophin (Dy8/6C5) 1:50). Right lane is normal control.
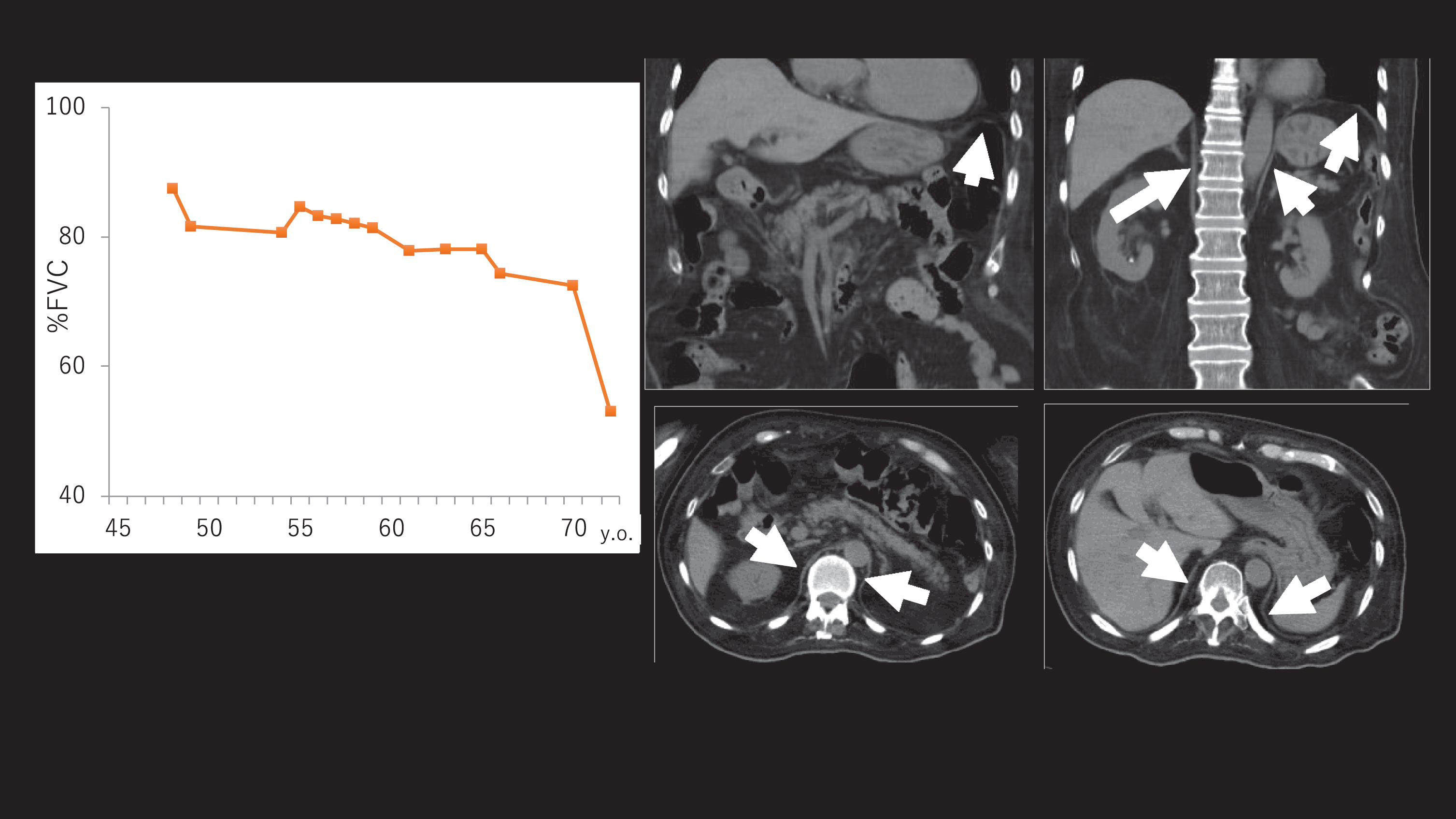
Clinical data of an autopsied patient. (A) Serial changes revealed that % FVC was markedly decreased after his 60’s. (B–E) Chest CT at age 70 (two years before death). (C, D) Coronal and (E, F) axial sections. The diaphragm was moderately atrophic (arrow).
DISCUSSION
The present study clarified that patients with BMD are at risk of respiratory dysfunction, even while they remain ambulant. We also found that these instances of respiratory dysfunction were caused by dystrophic changes in respiratory muscles, and not due to other pulmonary complications. Autopsy indicated that respiratory muscles were markedly involved compared to pharyngeal muscles and were spared compared to proximal muscles, reflecting the fact that respiratory failure occurred after the progression of proximal weakness.
Respiratory failure risk still remains ambiguous for patients with BMD, as ambulation, genotyping, and disease duration did not significantly correlate with respiratory dysfunction. Only a few studies have reported on respiratory failure in BMD patients. For instance, in one study, Magri et al. reported that 26 of 103 BMD patients showed a restrictive pattern in respiratory assessment, beginning at a mean age of 28.6+/–11.7 years. They also noted that among patients aged 35 years and older, 28% showed respiratory involvement and none needed ventilotherapy [7]. In another study, Nakamura et al. reported that among young BMD patients, six showed nocturnal hypercapnia and/or were NPPV users, and one of four NPPV users was ambulant. LVEF was not informative for predicting respiratory function. They also reported that BMD patients exhibited asymptomatic sleep hypercapnia without a decline in SpO2while their % FVC was still greater than 70% of predicted values [8]. In yet another study, one BMD patient harboring an exon 45–53del mutation used NIV at age 48, among a study population of 48 BMD patients [9].
Considering the reports above and our previous study using Remudy [6], some BMD patients apparently are at risk of respiratory dysfunction and require NPPV even while still ambulant. In the present study, we observed a worsening of respiratory function (from mild to severe respiratory failure) during 10 years, especially among non-ambulant patients (Fig. 1), but the reason for this is unclear. However, not all patients with early onset disease or non-ambulant patients exhibited severe respiratory dysfunction, suggesting that predictive factors of respiratory failure remain unclear. Yet, since non-ambulant patients tended to have smaller % VC, the severity of skeletal muscle symptoms may predict respiratory dysfunction. We also compared ambulation status and % FVC within each decade but found no significant differences among ambulation groups (data not shown).
The present study is the first to analyze autopsy findings from patients with BMD and offer a detailed description of respiratory muscle involvement. While the extent of involvement was milder than in proximal limb muscles, some respiratory muscle involvement was observed, with apparent dystrophic changes and muscle loss. These findings correlated with the patient’s clinical presentation.
This study has several limitations worth noting. First, the recruitment of patients from NCNP, which is highly specialized in muscle disease, is a potential source of selection bias. Indeed, NCNP patients may be more severely affected than the general BMD patient population. Therefore, our findings might not be generalizable. Second, our study population was small, and thus we may not have had sufficient power to detect significant differences. In addition, since respiratory function is measured in severely affected (i.e., non-ambulant) patients, it is possible that physicians might have omitted respiratory function tests in mildly affected BMD patients. A large scale, cross-sectional study could help identify key factors responsible for respiratory dysfunction as well as correlations between respiratory dysfunction and other risk factors. To address this issue, we have initiated a prospective observational study of BMD patients. Finally, we defined BMD clinically based on loss of independent ambulation at age 17 or older, and hence, our patients might have included those with dystrophinopathy phenotypes, which may be considered “mild DMD” or “intermediate dystrophinopathy” if an immunohistochemical or genetic definition is used. As these features are more common with “micromutations” (e.g., nonsense, missense, and frame shift mutations), it is not surprising that patients with such mutations have lower FVC.
Our findings suggest that BMD patients are at risk of respiratory dysfunction, although we fell short of identifying concrete risk factors. Respiratory function may deteriorate in these patients rapidly, such that they require ventilation support within years of onset of BMD. For this reason, we recommend that the respiratory function of BMD patients be measured yearly for those who are non-ambulatory or have a % FVC below 80%, and every few years (two or more based on clinical judgment) for those who are ambulatory with stable/normal FVC.
Footnotes
ACKNOWLEDGMENTS
This work was partly supported by Research on Intractable Diseases of Health and Labor Sciences Research Grants, Comprehensive Research on Disability Health and Welfare Grants, Research and Development Grants for Comprehensive Research for Persons with Disabilities and Practical Research Project for Rare/Intractable Diseases from Japan. Agency for Medical Research and Development, AMED (JP18dm0107103, YS), and Health and Labor Science Research Grants, and an Intramural Research Grant (29-3, 29-4, 30-8, 2-4, 2-6) for Neurological and Psychiatric Disorders from the NCNP.
