Abstract
Background:
Several retrospective case series have suggested rituximab (RTX) might improve patients with refractory Myasthenia Gravis (MG).
Objective:
In this study, we aimed to evaluate prospectively the efficacy of RTX on muscle function in refractory generalized anti-acetylcholine receptor (AChR) MG patients.
Methods:
Enrolled patients received 1 g of RTX at day 0, day 14, and 6-month follow-up (M6). The primary endpoint was improvement of muscle function at 12-month (M12) based on myasthenic muscle score (MMS). Secondary endpoints were an improvement of the MG Foundation of America Postintervention Status (MGFA-PIS), respiratory forced vital capacity, occurrences of acute MG exacerbation and requirement of associated immunosuppressants and immunomodulatory agents.
Results:
Twelve patients were enrolled, and 11 completed the study. Only a single patient presented an improvement of at least 20 points on MMS at M12, although 2 patients displayed an increase of at least 18 points at M12. MGFA-PIS had improved in 55% of patients by M12. The clinical improvement was not associated with a reduction of immunosuppressant burden.
Conclusions:
These results provide data on the effect of RTX in patients with severe, refractory anti-AChR Abs generalized MG. Even though primary outcome was only reached in a single patient at M12, a beneficial effect of RTX on muscle function was seen in half of the patients at M12 and persisted in a third of patients at M18.
Keywords
INTRODUCTION
Myasthenia gravis (MG) is a rare autoimmune disorder of the neuromuscular junction, in most cases caused by antibodies directed towards the acetylcholine receptor (AChR) [1]. MG is clinically characterized by a fluctuating muscle weakness that can involve any skeletal muscle and ranges from fatigability to severe weakness. Glucocorticoids combined with first-line immunosuppressive agents (such as azathioprine or mycophenolate mofetil) are often required in patients with generalized MG [2]. Nonetheless, 5–10% of patients will develop refractory MG which fails to respond to otherwise adequate doses and duration of conventional immunosuppressive therapy [3–5]. Retrospective case series have shown that RTX may improve patients with refractory MG [1]. We report herein an open label prospective multicentre pilot phase II trial on the efficacy and safety of rituximab (RTX) in 12 patients with refractory generalized MG.
MATERIALS AND METHODS
Patients
Patients with generalized MG were eligible to participate in this study if they were considered refractory to conventional treatments. Patients were defined as having severe generalized MG at the time of enrolment based on the MG Foundation of America (MGFA) clinical classification [9] (clinical stage Class IVa, IVb or V), and were eligible if they fulfilled the following inclusion criteria: onset at age > 18 years; positive anti-AChR antibodies (Abs) (radioimmunoprecipitation, RIA kit, RSR Limited), and intolerance or inadequate response for > 12 months to prednisone (Pred) and at least 2 other immunosuppressive and/or immunomodulatory agents (
Exclusion criteria were ocular myasthenia, myasthenia associated with malignant thymoma or pregnancy; severe cardiac dysfunction (ejection fraction≤30% ), severe adverse reaction after mono-clonal antibody infusion, active infectious disease including bacterial or viral infection (such as human immunodeficiency virus or hepatitis B virus), anemia (haemoglobin level < 8 g/dl), neutropenia (absolute neutrophil count < 1500×103/μl), and immunoglobulin G and/or M < 5.0 or 0.40 mg/ml, respectively.
Patients were screened during a clinic visit or hospitalization and recruited after validation of eligibility criteria. The inclusion period covered 3 years beginning in January 2008. The last patient was enrolled in September 2010. Follow-up occurred over an 18-month period.
Ethics statement
Written informed consent was obtained from each study patient. The protocol was approved by the Pitié-Salpêtrière Hospital Ethics Committee (June 2007) and the French Health Products Safety Agency (Afssaps) (ref. Afssaps: A70399-45; EudraCT: 2006-005900-15; clinicalTrials.gov NCT00774462). The authors confirm that all ongoing and related trials for this drug/intervention are registered.
Treatments
Patients received two infusions of 1 g of RTX separated by 2 weeks’ intervals, followed by 1 g infusion 6 months after the day-14 (D14) injection. Over the study period, Pred dose increase up to 1 mg/kg/day, maintenance or change for another immunosuppressive drug, plasma exchange, and/or IVIg infusion was allowed.
Assessments
Study visits occurred at baseline (M0), day-7 (D7), D14, D21, 6-month (M6), M12, and M18 after the first RTX infusion. Muscle weakness was determined using the MGFA clinical classification and quantitative myasthenia gravis (QMG) score [6] and myasthenic muscle score (MMS) [7]. Health-related quality of life was scored using the Rand Medical Outcomes Study 36-Item Short-Form Health Survey (SF-36) at M0 and M12.
Respiratory muscle involvement was assessed by pulmonary function tests (PFT) measuring forced vital capacity (FVC expressed as % of predicted value) at M0 and M12. Anti-AChR Abs titers were determined using radioimmunoprecipitation (RIA kit, RSR Limited) at M0, M6, M12 and M18 (positivity threshold≥0.5 nmol/L).
Immunophenotyping of peripheral blood mononuclear cells was performed by flow cytometry. For peripheral blood B lymphocyte count, blood cells were stained with anti-CD19 (B4, mouse IgG1) fluorescein isothiocyanate labelled monoclonal Abs (Beckman Coulter, Miami, Florida, USA) with the use of Coulter PrepPlus and TQPrep cell preparation systems and were analyzed on an Epics XL flow cytometer (Beckman Coulter) using the System II and Expo 32 software. Absolute lymphocyte counts were determined using Flow-Count Fluorospheres (Beckman Coulter).
End points
The primary end point was defined as an improvement of at least 20-points on the MMS [8] at M12.
Secondary end points, measured at M12, were: an improvement on MGFA quantitative myasthenia gravis (QMG) score (decrease of at least 3-points [6]), an improvement on MGFA-PIS, an improvement of forced vital capacity (increase of at least 10% ), a decrease in the titer of anti-AChR Abs, an improvement on SF-36 score (+10 and +5 points for multi-item scales and the physical and mental components summaries, respectively, and a treatment improvement was a cessation of an immunosuppressant, IVIg and/or a≥50% decrease in Pred dose at M12 compared to M0. On the other hand, the addition of an immunosuppressant (not due to intolerance and/or side effects) and/or IVIg and/or≥50% increase in Pred dose was considered as treatment intensification and no change in treatment was considered as treatment stabilization.
Adverse events
The data safety and monitoring board monitored overall safety independently from the participating institutions.
Statistical analyses
No calculation of sample size was made for this pilot study. Based on the initial provision of including 12 patients, an improvement in muscle strength among six patients would have resulted in a success rate estimate > 25% (inferior limit of 90% confidence interval).
Descriptive statistics were displayed using mean, median and range, or n and percentage with 95% confidence interval.
RESULTS
Patient characteristics at baseline
Between January 2008 and September 2010, 12 patients were enrolled in the study (Fig. 1). Eleven patients completed the study: 8 women and 3 men, with a median (range) age of 44 (24–61) years at study entry (Table 1) and the median (range) disease duration prior to study entry was 13 (3–32) years. All patients had been previously treated for a median of 12 years and thymectomy had previously been performed in 8 patients. Immunomodulatory treatments received prior to RTX are detailed for each patient in Table 1. Before enrolment, patients had previously received a mean of 4 immunomodulatory treatments. The MGFA clinical status was IIIa for 2 patients (IVa during the preselection period), IVa for 4 patients and IVb for 6 patients. At baseline, median (range) FVC was 82 (33–98) %. At baseline, SF-36 scores were low, with a median (range) physical functioning norm based score of 38 (0–70), with a median (range) physical component summary score of 35 (5–61) and a median (range) mental component summary score of 38 (8–46), knowing that each item is scored on a 0 to 100 range.
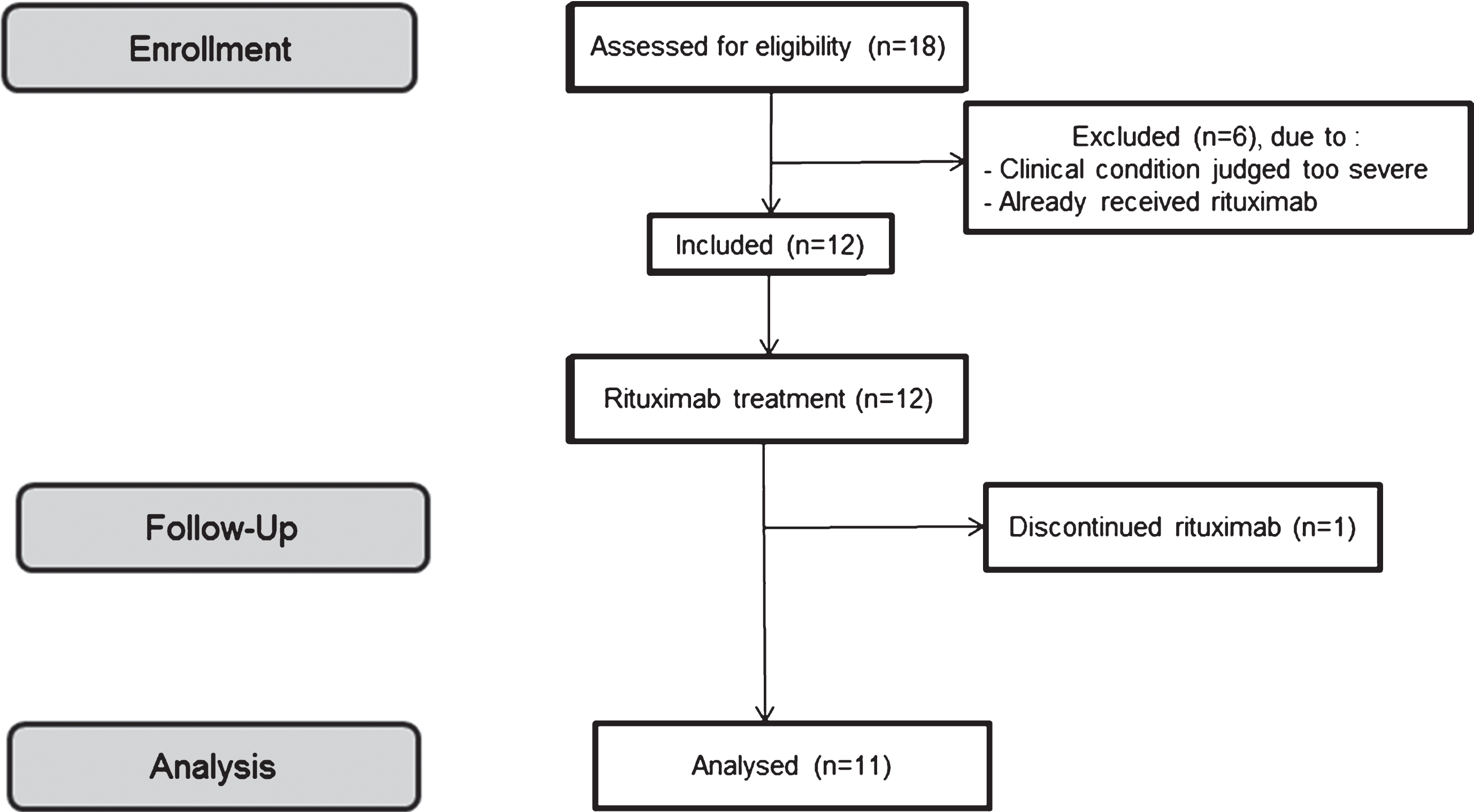
Flowchart.
Patients characteristics, muscular evaluation and treatment administration at baseline
Abbreviations: M = male; F = female; Y = yes; N = no; Pred = prednisone; IVIg = intravenous immunoglobulin (1 g/kg/month); PE = plasma exchange; AZA = azathioprine (2 g/kg/day); MMF = mycophenolate mofetil (2 g/day); CiC = cyclosporine (3–5 mg/kg/day); CYP = cyclophosphamide (2 mg/kg/day); Chl = chlorambucil; Tac = tacrolimus (0,1–0,2 mg/kg/day); NA = data not available.
Clinical evolution
Individual changes in MMS and QMG scores between baseline and M12 are depicted in Fig. 2 while the MGFA-PIS is depicted in Fig. 3. Only one patient (K) achieved a≥20 points increase in MMS between M0 and M12. The median (range) MMS remained unchanged from 67 (27–87) at baseline to 66 (27–95) at M12. Patients A and C had an increase in their MMS of 18.5 and 19, respectively, at M12. Five patients achieved a≥3 points decrease in QMG score between M0 and M12, including all three patients who had an improvement of≥18.5 on the MMS score. MGFA-PIS improved in 6 patients and worsened in 2 patients between inclusion and M12. At M12, mean (range) variation of FVC was –3 (–12 to +8) %. Significant worsening of FVC (≥10% ) was seen in only one patient at M12, although FVC remained over 60%. At M12, median (range) values for the SF-36 components remained stable at 40 (25–55) and 32 (24–51) for the physical and the mental component summary scores, respectively, whereas the physical functioning norm based score improved (median 50; range, 25–95).
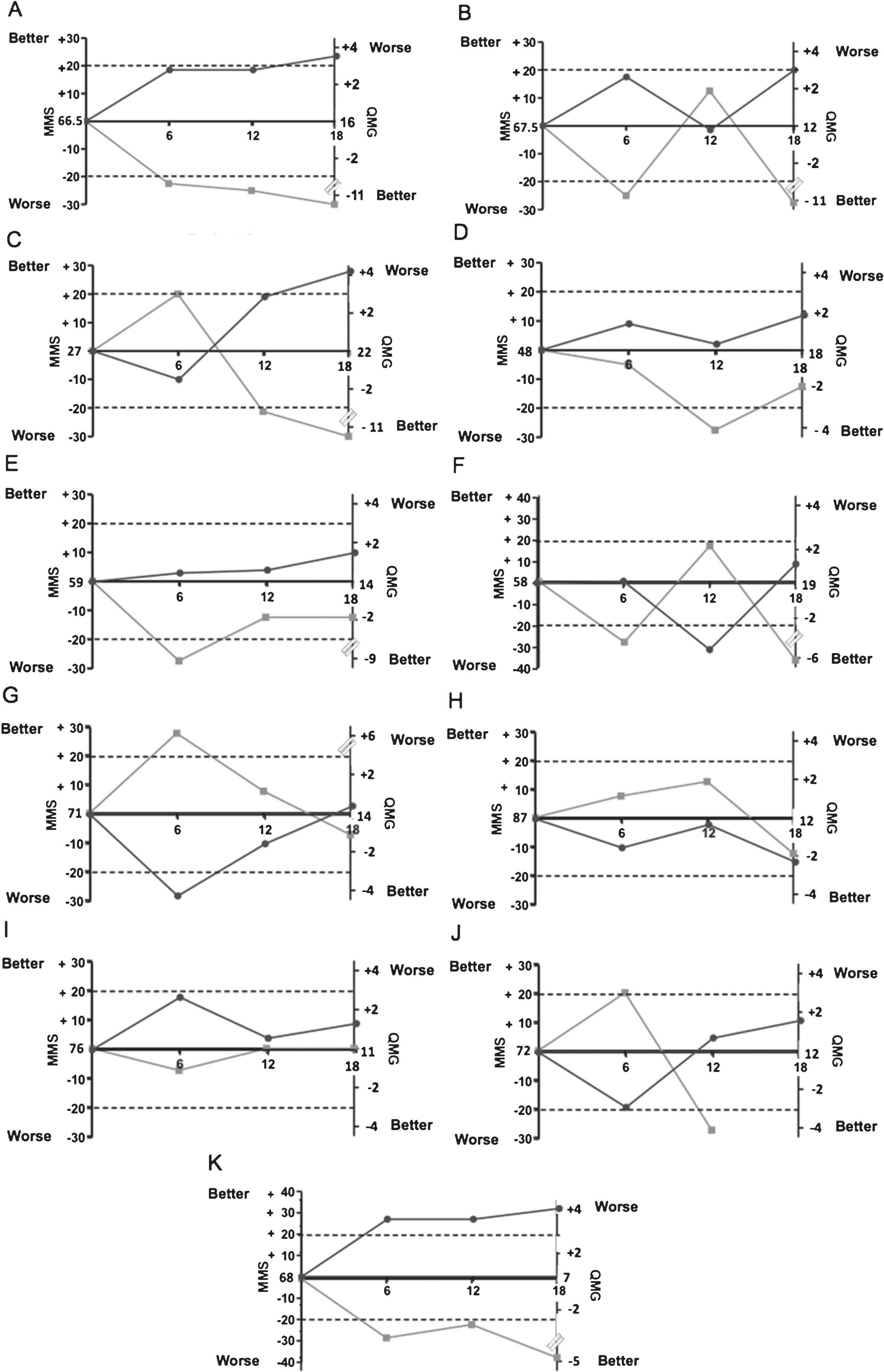
Muscle function evolution from baseline to 18-months. Myasthenic muscle score (MMS) (dark grey) and Quantitative myasthenia gravis (QMG) score (light grey) is represented for each patient who terminated the study. The dotted line represents the clinically significant variation of MMS (≥20-point change) and QMG score (≥3-point change).
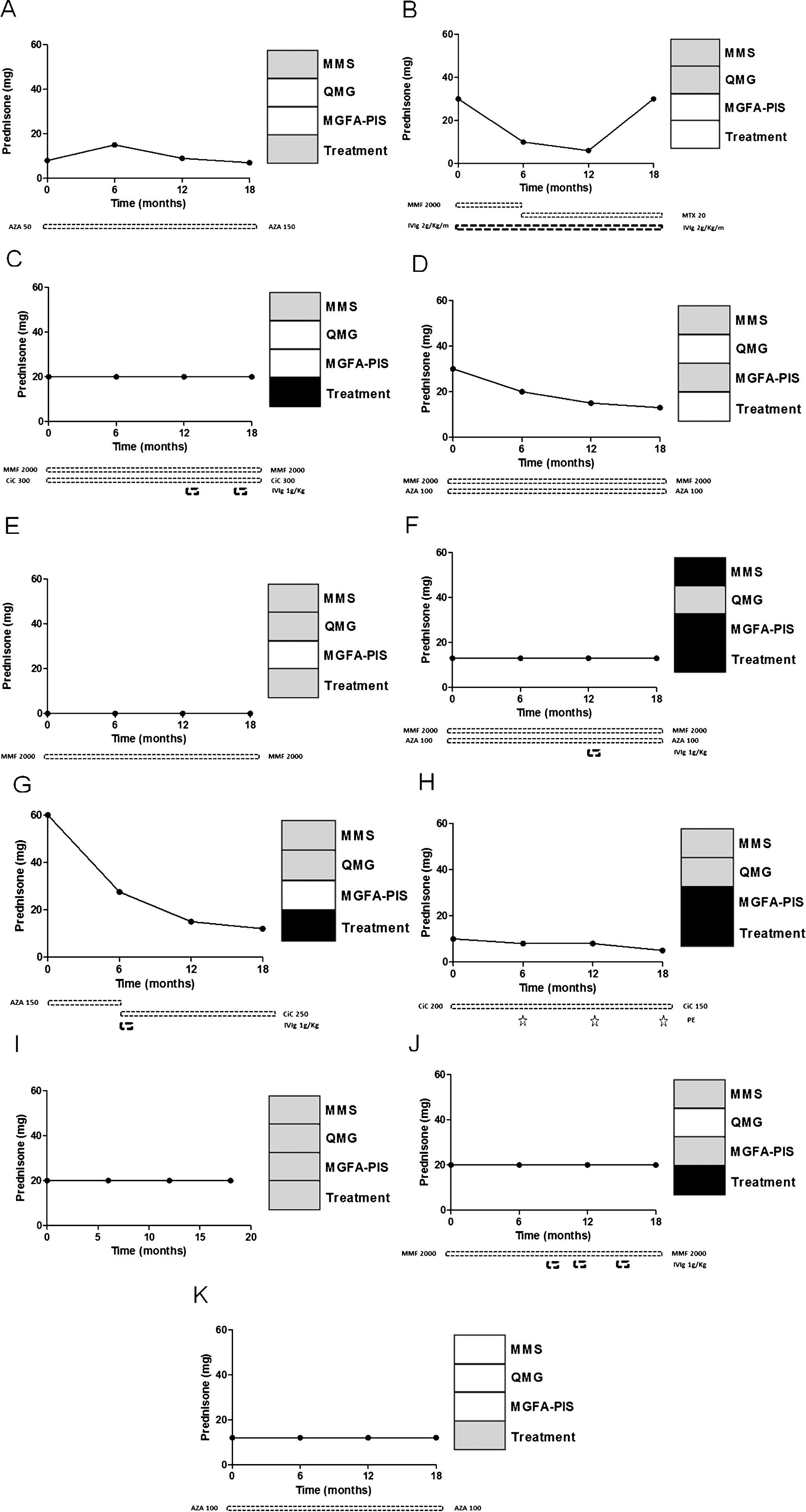
Immunosuppressive/immunomodulatory treatment outcome following rituximab. Prednisone dose (mg), immunosuppressive agents (mg) (AZA = azathioprine, MTX = methotrexate, MMF = mycophenolate mofetil, CiC = ciclosporine) and immunomodulatory treatment (IVIg = intravenous immunoglobulin, PE = plasma exchange) evolution following rituximab for 11 patients (patient A-K) who terminated the study. On the right side, 4 boxes represent the status at M12 (vs M0) concerning MMS, QMG score, MGFA-PIS, and treatment modification. Black represents worsening, grey represents no change, and white represents an improvement.
At M18, the patient who had achieved the primary endpoint maintained his improvement, and 3 other patients reached a 20 points increase in MMS (Fig. 2). Five patients achieved a≥3 points decrease in QMG score between M12 and M18, including all four patients who also had an improvement on MMS. Between M12 and M18, MGFA-PIS improved in four patients (including three patients who had an improvement on both MMS and QMG score) and worsened in two others.
Treatment modification
Following rituximab administration 5 patients (45% of 11 patients) received intensified treatment (4 patients required increase or initiation of immunosuppressant agents, 4 patients in need of IVIg, 1 patient required plasmapheresis) (Fig. 3). Only one patient had a myasthenic exacerbation requiring intensive care unit stay following an episode of gastroenteritis. Of note, the patient achieving primary endpoint at M12 had no change of prednisone and immunomodulatory treatment. In those achieving an increase of MMS≥20 points at M18, treatment was unchanged in two patients (A and B) and intensified with intermittent IVIg therapy in another (patient C).
Safety analysis
The patient who withdrew from the study at week 3 (after the second infusion of RTX) experienced peripheral oedema the day following each RTX infusion (D0 and D14) related to underlying severe cardiac failure secondary to severe radiation-induced coronary artery disease. This patient died 15 months later of cardiorespiratory arrest. No other patient left the study due to an adverse event and no other death occurred during the study period. Six infectious adverse events occurred during the study period, most of them were of mild to moderate severity (except the episode of gastroenteritis described above): 2 non-febrile flu-like syndromes, 1 viral gastroenteritis, 2 cutaneous infections and 1 herpes zoster infection. Of note, no severe adverse event occurred during RTX infusion and no malignancy was detected during the study.
Immunological analysis
Median (range) anti-AChR Abs titer did show some improvement from 49.15 (0.64–100) nmol/L at baseline to 30.85 (0.51–100) nmol/L at M12. However, individual anti-AChR Ab titer changes did not correlate well with clinical outcome (not shown). Flow cytometry analysis (data not shown) performed at D21 demonstrated CD19 + B-cell accounted for≤0.2% of total peripheral mononuclear cells in all patients but one. At M6, 3 patients had an increase of the CD19 + lymphocyte count of > 0.05%, though these changes were not correlated with significant muscle function decline.
DISCUSSION
Despite the efficacy of immunotherapy, a subset of MG patients remains refractory to multiple immunosuppressant treatments. Zaja et al. [10] first reported the successful use of RTX in a patient who developed MG after a human leukocyte antigen-matched bone marrow transplant for acute myelogenous leukemia. Several case reports and case series have since been published.
A large body of evidence supports a humoral immune-mediated mechanism underlying MG [11–14]. The pathogenic role of anti-AChR Abs has been demonstrated in animal models [15]. These Abs mediate a functional reduction in postsynaptic AChR activity by multiple mechanisms including complement-mediated destruction of the AChR, cross-linking of AChR resulting in enhanced turnover of these receptors, and allosteric interference of the ACh:AChR ligand receptor interaction [16]. It also has been shown that clinical improvement of MG after immune therapy is commonly associated with a decrease in the titer of anti-AChR Abs [17].
These last observations support the therapeutic rationale in favor of using RTX, a chimeric monoclonal antibody targeting the CD20 antigen, for the treatment of refractory anti-AChR Abs MG patients. The expression of the CD20 is restricted to the late pre-B-cell stage before differentiation to plasma cells (PCs). Therefore, RTX may act primarily by depleting the precursors of PCs via several putative mechanisms: antibody-dependent cellular cytotoxicity (through Fc
This prospective trial enrolled exclusively anti-AChR antibody positive refractory MG patients for RTX treatment. At M12, only a single patient achieved the primary endpoint of an improvement of at least 20 points on the MMS (patient 11), with 2 others approaching the primary endpoint (patients A and C), 5 patients achieved a decrease of at least 3 points on the QMG score and MGFA-PIS improved in 6 patients at M12. Interestingly, a total of 4 patients met the definition of improvement on both MMS and QMG score at M18.
To our knowledge, the use of RTX to treat refractory MG patients was reported in only two other prospective clinical trials [22, 23] (total
Anti-AChR Abs are present in approximately 90% of adult MG patients and are heterogeneous in several respects, namely light chain type, IgG subclass, binding site affinity, and AChR subunit specificity. Interestingly, RTX, which targets the PC precursors, depletes short-lived PCs while having little or no effect on the long-lived PCs subset [26]. One meta-analysis revealed that RTX was more effective in patients with anti-MuSK Abs MG than in patients with anti-AChR Abs MG [1]. It has been hypothesized that anti-MuSK Abs are IgG4, an IgG isotype almost exclusively produced by short-lived plasma cells [23], thus explaining the preferential efficacy of RTX reported in these patients. Furthermore, a trend toward an inverse correlation between the disease duration and the response rate to RTX has been reported [1]. These observations might contribute to the results seen in our patients since we exclusively had anti-AChR Abs MG patients with prolonged disease duration and multiple immunosuppressive and/or immunomodulatory treatment prior to RTX initiation. Response to RTX may be different in a shorter disease duration group of patients.
The duration of RTX therapy necessary to achieve and maintain disease remission has been unclear. Extend follow-up of RTX-responders would have been useful in assessing relapse rate which has been reported in the anti-AChR Abs MG subset in up to 56% at about 3 years after the last RTX infusion [27].
Finally, adverse events related to the RTX were mainly infections. Only one of the patients with gastroenteritis (no pathogen documented) needed hospitalization for hydration and transient observation in the intensive care unit for 24 hours. Since patients enrolled in the present study had concomitant immunosuppressants and/or prednisone, the role of RTX in the occurrence of infections cannot be properly evaluated without a control group.
In conclusion, this pilot, prospective, phase II study provides data on the effect of RTX in patients with severe, refractory anti-AChR Abs generalized MG. Less spectacular effects of RTX than previously reported were seen in our cohort of MG patients that may be related to our patients’ characteristics and reporting bias. Nevertheless, a beneficial effect of RTX on muscle function was seen at M18 in a third of patients with long-standing disease duration.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This work was supported by the French Ministry of Health (PHRC); and the Association Française contre les Myopathies (AFM). Rituximab was kindly provided by Roche. Dr. Landon-Cardinal is the recipient of Clinical Fellowship awards from the Université de Montréal Rheumatology Program - Abbvie Educational Grant and the Association des médecins rhumatologues du Québec - Visithan-Khy Educational Grant.
