Abstract
Background:
Refractory generalized myasthenia gravis (GMG) remains a substantial therapeutic challenge. Telitacicept, a recombinant human B-lymphocyte stimulator receptor-antibody fusion protein, holds promise for interrupting the immunopathology of this condition.
Objectives:
This study retrospectively assessed the effectiveness and safety of telitacicept in patients with refractory GMG.
Design:
A single-center retrospective study.
Methods:
Patients with refractory GMG receiving telitacicept (160 mg/week or biweekly) from January to September in 2023 were included. We assessed effectiveness using Myasthenia Gravis Foundation of America post-intervention status (MGFA-PIS), myasthenia gravis treatment status and intensity (MGSTI), quantitative myasthenia gravis (QMG), and MG-activity of daily living (ADL) scores, alongside reductions in prednisone dosage at 3- and 6-month intervals. Safety profiles were also evaluated.
Results:
Sixteen patients with MGFA class II–V refractory GMG were included, with eight females and eight males. All patients were followed up for at least 3 months, and 11 patients reached 6 months follow-up. At the 3-month evaluation, 75% (12/16) demonstrated clinical improvement with MGFA-PIS. One patient achieved pharmacological remission, two attained minimal manifestation status, and nine showed functional improvement; three remained unchanged, and one deteriorated. By the 6-month visit, 90.1% (10/11) sustained significant symptomatic improvement. MGSTI scores and prednisone dosages significantly reduced at both follow-ups (p < 0.05). MG-ADL and QMG scores showed marked improvement at 6 months (p < 0.05). The treatment was well tolerated, with no severe adverse events such as allergy or infection reported.
Conclusion:
Our exploratory investigation suggests that telitacicept is a feasible and well-tolerated add-on therapy for refractory GMG, offering valuable clinical evidence for this novel treatment option.
Keywords
Introduction
Myasthenia gravis (MG) manifests as an acquired autoimmune disorder, typified by the production of antibodies, predominantly anti-acetylcholine receptor (AChR) or muscle-specific kinase (MuSK), that compromise the neuromuscular junction. This leads to hallmark symptoms of skeletal muscle weakness and fatigability. 1 While corticosteroids and traditional immunosuppressants have yielded positive outcomes in a majority of MG cases, achieving remission often necessitates sustained and tailored pharmacotherapy. Recent strides in understanding MG’s immunopathogenesis have catalyzed the advent of more targeted and potent therapeutic options.2–5 Refractory generalized myasthenia gravis (GMG), which affects an estimated 10–15% of all individuals with MG, presents a formidable clinical challenge. 6 This subset of patients exhibits resistance to standard corticosteroid treatments and other immunomodulatory therapies, or they suffer from intolerable side effects. Additionally, maintenance therapy may require interventions such as intravenous immunoglobulin (IVIG) or plasma exchange (PE), intensifying both the disease burden and healthcare utilization.7,8 The clinical trajectory for these patients is often marred by exacerbations, hospital admissions, and episodes of respiratory failure requiring mechanical ventilation, compromising their quality of life and daily functioning. 9 Given the significant clinical and socio-economic implications of refractory GMG, there exists a pressing need for innovative therapies that are both efficacious and well tolerated.
Recent advancements in the therapeutic landscape for MG have highlighted the significance of targeting autoreactive B-cells, pivotal in the disease’s pathogenesis. These cells interact with follicular helper T cells, promoting the survival of memory B lymphocytes and their differentiation into plasma blasts or plasma cells that secrete pathogenic antibodies.10–14 This understanding has shifted the focus toward monoclonal antibodies against B cells as a potential treatment strategy. In confronting refractory GMG, the search for effective therapies includes a broad spectrum of options, such as chimeric antigen receptor T-cell therapy 15 and hematopoietic stem cell transplantation. 16 These interventions are evaluated against a comprehensive set of criteria encompassing efficacy, safety, accessibility, sustainability, and cost-effectiveness. Rituximab (RTX) has been explored as a potential intervention for MG, with its use showing promise in certain subsets of patients. While RTX has shown efficacy in MuSK antibody-positive MG, due to the specific nature of MuSK as an IgG4 antibody, the results in AChR antibody-positive MG, which involves IgG1 and IgG3 antibodies, have been more conflicting, and clinical practice varies widely.17–19 This variability underscores the need for alternative B-cell targeting strategies.
Research is increasingly focusing on the role of cytokines and chemokines in B and T cell-mediated pathologies in MG. B-cell activating factor (BAFF), crucial for B cell survival and maturation, and a proliferation-inducing ligand (APRIL), which supports cell growth and B cell homeostasis in conjunction with BAFF, are of particular interest. 20 While belimumab, a BAFF inhibitor, showed promise in systemic lupus erythematosus, 21 its efficacy in MG remains unproven, 22 highlighting the complexity of targeting these pathways in MG. Elevated levels of APRIL in MG suggest its potential as a therapeutic target, especially in specific MG subtypes, underscoring the need for continued investigation into these targeted therapies. 23
Telitacicept, a fusion protein combining transmembrane activator and calcium modulator and cyclophilin ligand interactor (TACI) with an immunoglobulin framework, represents a novel therapeutic approach by concurrently targeting BLyS (BAFF) and APRIL. 20 This blockade inhibits the proliferation and maturation of B and T lymphocytes, pathways implicated in the pathogenesis of various autoimmune conditions. 24 Approved in China since March 2021 for Systemic lupus erythematosus (SLE) treatment, telitacicept has shown promise across a spectrum of autoimmune disorders, including IgG4-related disease, 25 rheumatoid arthritis, 26 and neuromyelitis optica spectrum disorders. 27 The encouraging preliminary outcomes of an ongoing phase II clinical trial of telitacicept in GMG (unpublished data) have led to the initiation of a phase III Food and Drug Administration-approved study. Recent case report of refractory GMG patients responding to telitacicept further underscores its potential benefit. 28 Pharmacokinetic studies have provided valuable insights into the behavior of telitacicept within the body post-administration. Notably, it has been observed that the telitacicept complex can remain detectable for up to 71 days following a single dose, indicating its prolonged presence in the systemic circulation. 29 This extended detectability underscores the drug’s metabolic process, with a half-life of nearly 2 weeks, necessitating roughly five half-lives for complete clearance from the body. While the detection of telitacicept at such a late stage does not directly correlate to ongoing therapeutic effectiveness, this pharmacokinetic profile suggests a potential for sustained efficacy beyond the immediate treatment period. Importantly, the long-term impact of telitacicept, attributable to its dual inhibition of APRIL/BAFF and suppression of long-lived plasma cells (LLPC), 30 posits a rationale for evaluating the timing of treatment cessation, especially in light of observed clinical improvements.
In light of telitacicept’s demonstrated efficacy across a range of autoimmune diseases, including MG, its potential therapeutic role in refractory GMG warrants further investigation. This study aims to retrospectively assess the clinical outcomes associated with telitacicept as an add-on treatment in patients with refractory GMG.
Methods
Study design and patients
In this retrospective analysis, we reviewed the data of telitacicept as an add-on treatment in patients with refractory GMG at the Department of Neurology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology from January to September in 2023. In our study, the inclusion criteria were as follows: (1) confirmed diagnosis of refractory GMG, classified as classes II–V according to the Myasthenia Gravis Foundation of America (MGFA) criteria8,19; (2) history of inadequate response to at least two immunosuppressive therapies, an inability to tolerate the toxicities associated with such treatments, a tendency for relapse upon tapering or cessation of steroids, or a persistent requirement for IVIG or PE; and (3) patients who had received telitacicept as an add-on treatment. In this study, an inability to tolerate the toxicities of immunosuppressive therapies encompasses adverse reactions such as severe osteoporosis, avascular necrosis of the femoral head, renal insufficiency, and diabetes mellitus, as well as contraindications related to pre-existing comorbidities. Exclusion criteria were stringently set to omit those with severe acute infections preceding treatment onset, women who were pregnant or lactating, patients with known hypersensitivity to biological agents, and individuals with significant hepatic dysfunction.
Treatment, follow-up, and assessment
Patients were administered subcutaneous telitacicept at a dose of 160 mg, on either a weekly or biweekly schedule, dictated by disease severity and individual economic considerations. Concomitant immunosuppressive therapies, comprising steroids, and immunosuppressants, were continued as part of the treatment regimen.
Patients were followed regularly every 3 months, with a combination of outpatient visits and telephone follow-ups for assessment. Evaluations were conducted at baseline (defined as the last assessment prior to telitacicept initiation), and subsequently after the treatment. Comprehensive data encompassing demographic details, clinical presentation, age at onset, disease duration, MGFA classification, antibody profiles, MG-related activities of daily living (MG-ADL) scores, quantitative myasthenia gravis (QMG) scores, incidence of myasthenia crises, coexisting conditions, and current immunosuppressive therapies were meticulously documented.
Clinical outcomes were appraised at the 3- and 6-month mark post-treatment initiation, utilizing the MGFA post-intervention status (MGFA-PIS) to categorize responses as improved, unchanged, worsened, or exacerbated, with mortality also being recorded. The ‘minimal manifestation status (MMS) or better’ category encapsulated the spectrum from MMS, pharmacologic remission (PR), to complete stable response. MMS was defined as a clinical state where patients exhibit no symptoms or functional limitations from MG, yet demonstrate some weakness upon examination of certain muscles. 31 Additionally, the myasthenia gravis treatment status and intensity (MGSTI) score served as an overarching outcome metric. 32 The assessment also included a detailed evaluation of the MGFA-PIS, MG-ADL, QMG scores, and reductions in the daily dosage of prednisone to evaluate telitacicept’s therapeutic effectiveness. Notably, QMG scores were obtained 3–4 h post the last pyridostigmine dose to mitigate the potential confounding effects of anticholinesterase agents.
All adverse events encountered during the treatment phase were systematically recorded and classified per the Common Terminology Criteria for Adverse Events version 5.0, encompassing infectious, non-infectious, and hypersensitivity reactions.
Statistical analysis
Statistical analysis and graphical illustration were executed using SPSS version 27.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism version 9.2.0 (GraphPad Software, San Diego, CA, USA). Demographic and clinical characteristics of the patients were summarized using descriptive statistics. Categorical variables and frequency counts were presented as frequencies and percentages. Continuous variables were presented either as means with standard deviations or as medians with interquartile ranges (IQR), contingent upon the distribution normality. For longitudinal analysis across multiple time points, one-way repeated measures analysis of variance (ANOVA) was employed for normally distributed datasets, while the Friedman test was utilized for non-parametric variables. p Values less than 0.05 denoted statistical significance.
Results
Baseline characteristics of patients
In this retrospective analysis of refractory GMG, we included 16 patients, with eight males and eight females. The mean age at disease onset was 50 ± 17.25 years, spanning from 15 to 69 years old, while the mean disease duration prior to treatment was 27.56 ± 26.04 months. Detailed demographic and clinical characteristics are delineated in Table 1.
Baseline demographic and clinical characteristics of patients with refractory generalized myasthenia gravis.

AchR-Ab, anti-acetylcholine receptor antibody; CKD, chronic kidney disease; DM, diabetes mellitus; F, female; HTN, hypertension; M, male; M, months; MGFA, Myasthenia Gravis Foundation of America; MuSK-Ab, muscle-specific tyrosine kinase; NA, not available; SSA, Sjogren’s disease; WHO, World Health Organization; Y, years.
A majority of patients, comprising 10 patients (62.5%), were categorized under MGFA class II, with the remaining six (37.5%) falling between classes III and V. Notably, five individuals (31.3%) had a history of myasthenic crisis. Serological testing revealed that 14 patients (87.5%) were positive for AChR antibodies, with half of these also testing positive for Titin antibodies. Two other patients presented with positive Titin antibody and MuSK antibody, respectively. Thymomas were identified in six patients, all of whom underwent thymectomy. Histopathological examination, following the World Health Organization (WHO) thymoma classification, revealed a range of types, including B1 (one case), combined B1 and B2 (one case), B2 (two cases), and combined B2 and B3 (one case), with one classification undisclosed. Of particular note was a patient with type B2 thymoma, whose disease extended to the chest wall, lung, and diaphragm. Intriguingly, two individuals underwent thymectomy during the course of telitacicept treatment; both experienced no postoperative myasthenic crisis and reported a gradual alleviation of symptoms. There were 11 individuals with baseline steroid doses at 20 mg/day or higher (Table 2).
Treatment and effectiveness assessment information for each individual.

AZA, azathioprine; CTX, cyclophosphamide; FK506, tacrolimus; IL-6R, interleukin-6 receptor antagonists; IVIG, intravenous immune globulin; MG-ADL, myasthenia gravis activity of daily living; MGSTI, myasthenia gravis treatment status and intensity; MMF, mycophenolate mofetil; MTX, methotrexate; NA, not available; PE, plasmapheresis; Pri, prednisolone; QMG, quantitative myasthenia gravis; RTX, rituximab; TAC, tacrolimus.
Within them, 12 patients (75.0%) presented with various comorbidities. Hypertension and diabetes mellitus were the most prevalent, each affecting five individuals (31.3%). Thyroid disease was noted in three cases (18.8%), and chronic kidney disease in two (12.5%). Additional conditions included myositis, peripheral neuropathy, hyperuricemia, and lacunar infarction, each identified in one patient. A subset of these comorbidities aligns with components of metabolic syndrome, particularly hypertension, diabetes mellitus, chronic kidney disease, and hyperuricemia. These are often associated with glucocorticoid and tacrolimus therapy. Conversely, the occurrence of other autoimmune disorders such as thyroid disease, myositis, and peripheral neuropathy may be intricately linked with MG as the underlying primary condition.
Treatment pattern
In our study, immunotherapy usage patterns were diverse, with 14 patients having used more than two immunosuppressive therapies, and some having received up to six different agents, in addition to undergoing thymectomy [Figure 1(a) and Table 2]. Glucocorticoid was ubiquitously utilized across most patients (15 cases, 93.8%), followed by tacrolimus in 11 cases (68.8%), and mycophenolate mofetil in 6 cases (37.5%). IVIG was administered in three cases (18.8%), PE and RTX each in two cases (11.1%). A minority of patients were also treated with interleukin-6 receptor (IL-6R) antagonists, methotrexate, cyclophosphamide, and azathioprine [Figure 1(b)].

Immunotherapy in refractory generalized myasthenia gravis. (a) Types of immunosuppressive agents previously administered to patients with refractory generalized myasthenia gravis. (b) Prevalence and distribution of immunosuppressive agents used prior to the study commencement.
The reasons for incorporating telitacicept as an add-on treatment included inadequate immunotherapy response (nine cases, 56.3%), adverse drug reactions or intolerance (eight cases, 50.0%), and disease relapse subsequent to glucocorticoid tapering or cessation (three cases, 18.8%). Throughout the follow-up, all patients continued telitacicept treatment with no alteration in dosages of concomitant medications, excluding glucocorticoid. Notably, two individuals discontinued telitacicept upon attaining substantial therapeutic benefits.
Effectiveness of telitacicept
All participants had completed a 3-month clinical follow-up, with 11 patients extending to a 6-month review. Notably, five patients had not reached the 6-month follow-up mark and were thus assessed with only 3 months of medication history to date. The clinical outcome post-telitacicept administration was evaluated via MGFA-PIS [Figure 2(a)]. At the initial 3-month follow-up, 75% (12 out of 16) manifested clinical improvement; one patient (6.25%) achieved PR, two (12.5%) attained MMS, and nine (56.3%) were noted to have functional improvement according to MGFA-PIS. Conversely, one individual experienced worsening symptoms necessitating an escalated prednisone dosage. The 6-month visit underscored sustained enhancements in 90.1% (10 of 11) of patients, with one (9.1%) maintaining PR, five (45.5%) at MMS, and four (36.4%) demonstrating continued improvement in MGFA-PIS. During the follow-up, amelioration in muscle weakness was distributed across various muscle groups, with extraocular and bulbar muscles showing slower rates of improvement compared to others [Figure 2(b)].
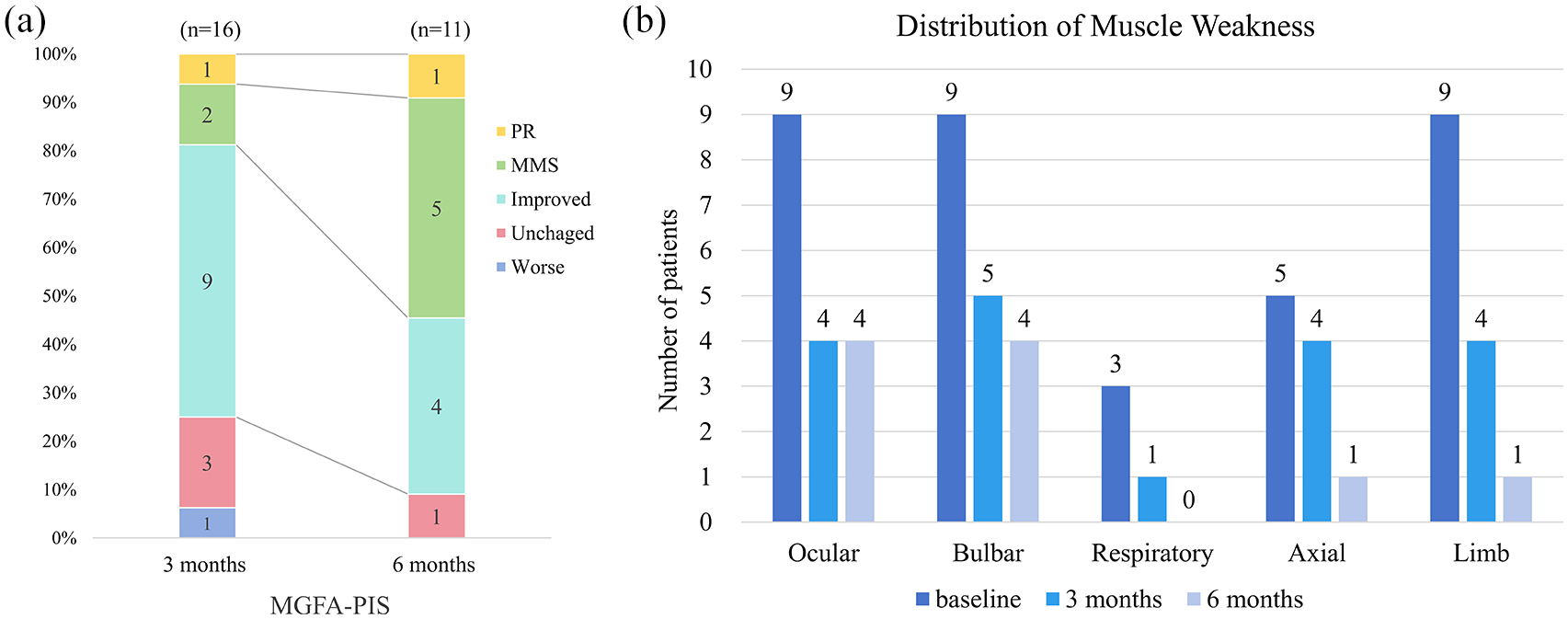
Assessment of clinical status using MGFA-PIS post-telitacicept treatment. (a) Improvement in clinical symptoms was observed in 12 out of 16 patients (75%) at the 3-month follow-up mark. Persistent and significant symptom improvement was noted in 10 of 11 patients (90.1%) at the 6-month follow-up, with six patients achieving symptom remission to MMS or PR status. (b) Improvement in muscular weakness was noted across all muscle groups to varying degrees. Notably, extraocular and bulbar muscles exhibited a slower rate of improvement compared to other muscle groups.
As shown in Figure 3(a), the baseline median MGSTI score was 4 (IQR 4–6), with most patients (75%) at a level 4 severity and the remainder (25%) at level 6. Post-treatment evaluations revealed a substantial decrease in MGSTI scores to a median of 2.5 (IQR 1–5) at the 3-month mark (p < 0.001, N = 16), which further reduced to a median of 2 (IQR 2–4) by the 6-month visit (p < 0.001, N = 11), with 63.6% (7/11) of patients achieving an MGSTI level of 2. Correspondingly, there was a significant reduction in the mean daily prednisone dose from 23.53 ± 10.08 mg at baseline to 15.63 ± 8.97 mg at 3 months (p < 0.001), which was sustained at 6 months with a median reduction of 38.6% compared to baseline (p < 0.001) [Figure 3(b)].

Clinical outcomes following telitacicept treatment in refractory generalized myasthenia gravis. (a) There was a significant decline in median MGSTI scores at the 3-month follow-up (p < 0.05) and at the 6-month visit (p < 0.05). (b) A reduction in daily prednisone dosage was observed in nine patients (56.3%) at the 3-month follow-up (p < 0.001). By the 6-month evaluation, median prednisone dosage had decreased by 38.6% compared to baseline (p < 0.001). (c and d) Baseline mean QMG and MG-ADL scores were 14.44 ± 6.87 and 5.50 ± 2.92, respectively, which significantly decreased to 8.67 ± 4.06 and 1.22 ± 2.28 at the 6-month follow-up (p < 0.05).
The treatment period also witnessed notable improvements in clinical scales reflective of quality of life: mean QMG and MG-ADL scores at baseline were 14.44 ± 6.87 and 5.50 ± 2.92, respectively. These scores showed a statistically significant reduction to 8.67 ± 4.06 for QMG and 1.22 ± 2.28 for MG-ADL by the 6-month follow-up (one-way ANOVA p < 0.05), indicating a marked enhancement in patient functionality and symptom burden [Figure 3(c) and (d)].
Individualized dosing regimen for telitacicept
The physicians implemented an individualized dosing regimen for telitacicept, tailored to the varied severity of disease manifestation and the economic considerations of the patients. A quarter of the patients (25%) received a weekly dose of 160 mg, while the majority (75%) were administered telitacicept biweekly at the same dosage. At the 3-month assessment, both dosing schedules resulted in symptom improvement in 50% of the respective patient groups, with a median MG-ADL score reduction of 3 (IQR 0–8) in the biweekly group and 4 (IQR 0–8) in the weekly group.
Notably, five patients ceased telitacicept therapy due to achieving significant clinical improvement, with four attaining MMS and one achieving PR. In these five patients, the mean reduction in prednisone dosage at 3 months was 12.00 ± 7.58 mg, with a substantial cumulative decrease of 16.26 ± 8.54 mg at 6 months, reflecting a 53.3% reduction from baseline dosage. Intriguingly, despite discontinuing telitacicept at 3 months, patients 10 and 15 exhibited sustained effectiveness and a continued decrease in prednisone requirement for at least 6 months. Conversely, only one patient discontinued telitacicept due to inadequate response after 3 months of treatment.
Safety profiles
All patients reported no allergic reactions, infections, or serious adverse events attributable to the medication over the course of treatment and subsequent follow-up. Three patients (18.8%) encountered transient pain and swelling at the injection site, symptoms which resolved spontaneously within a few days. There was a single instance of a mild herpes zoster virus infection that was managed effectively and resolved within a fortnight. There were no deaths related to adverse events.
Discussion
In our study, the cohort of GMG patients displayed features consistent with the known clinical spectrum of the disease,9,33 including a heightened risk of myasthenic crises, frequent hospitalizations, a predominance of AChR antibodies, a significant incidence of thymomas (most commonly classified as WHO B2), and a comprehensive history of utilizing broad-spectrum immunosuppressive treatments. The introduction of telitacicept was associated with a favorable clinical response. A majority of patients experienced symptomatic improvement within the initial 3 months, a benefit which not only persisted but expanded by the 6-month mark. This therapeutic response was mirrored by substantive enhancements in standardized MG assessments, substantiating telitacicept’s potential as a significant add-on treatment for refractory GMG.
The MGSTI score, combining MGFA-PIS with immunosuppressant dosages, offers a nuanced measure of treatment impact. 32 In our study, we observed a substantial reduction in the daily prednisone dosage following the administration of telitacicept, beginning as early as 3 months. This aligns with evidence suggesting a gradual increase in the serum TACI–BLyS complex, peaking around 1-month post-treatment, 29 correlating with the clinical improvements seen at the 3-month mark. Remarkably, the significant reduction in both the QMG and MG-ADL scores at 6 months suggest a more protracted therapeutic effect of telitacicept. These findings collectively underscore the potential of telitacicept as an efficacious treatment for refractory GMG.
Within our cohort, two patients who had previously not responded to RTX exhibited a favorable response to telitacicept. This observation underscores telitacicept’s potential effectiveness in cases where conventional B-cell depletion strategies may falter, suggesting its role as a promising therapeutic option for refractory GMG. RTX targets CD20+ B cells, including autoreactive B cells and plasmablasts, but spares LLPCs, 34 possibly accounting for the incomplete responses in some MG cases. 35 Moreover, following RTX administration, a surge in serum BLyS levels may contribute to the persistence and activity of self-reactive plasma cells. 34 The fluctuating serum levels of BLyS and APRIL in response to treatment, as observed in neuromyelitis optica spectrum disorders (NMOSD) trial, underscore the significance of targeting both BLyS and APRIL in managing autoimmune conditions. 27 Our investigation into telitacicept, a novel therapeutic agent that binds to and neutralizes the activity of both BLyS and APRIL, suggests it may be an effective treatment for refractory GMG. By inhibiting the proliferation of plasma cells and mature B cells, telitacicept holds promise as a double-targeted therapy, potentially addressing the clinical challenges posed by refractory GMG.
In addressing the evolving therapeutic landscape of MG, telitacicept emerges as a highly promising agent. Its unique mechanism of action, targeting both BLyS and APRIL, 20 positions telitacicept not only as a potential game-changer for patients with treatment-resistant MG but also suggests its utility as an early intervention in severe cases, especially those intolerant to steroids due to pre-existing conditions. This speculative yet optimistic perspective underscores the need for rigorous evidence from forthcoming randomized controlled trials to validate telitacicept’s effectiveness and safety profile across these patient populations. Moreover, the distinct effectiveness of telitacicept, as opposed to the negative outcomes observed with belimumab, highlights the critical role of targeting the B cell maturation pathway and the survival of LLPCs in MG’s pathogenesis.36,37 The failure of agents like belimumab, which lack the dual inhibition on BLyS and APRIL, illustrates the necessity of a more comprehensive approach in disrupting the pathological B cell feedback loops. Elevated APRIL levels in MG patients further emphasize the potential of telitacicept in addressing the underlying mechanisms of the disease effectively. 23 As we venture into this new era of MG treatment, the introduction of telitacicept heralds a promising avenue for addressing the complex interplay of immunological factors in MG, meriting further exploration in clinical settings to fully ascertain its therapeutic potential and positioning within the broader spectrum of MG management strategies.
In our findings, telitacicept’s effectiveness was pronounced across various subgroups, underscoring its potential as a tailored therapeutic option for refractory GMG. Notably, its benefit extended to a patient with anti-MuSK antibodies, showcasing a significant reduction in the MGSTI score. Conversely, in a patient harboring anti-Titin antibodies, telitacicept did not significantly alleviate symptoms such as ptosis and limb weakness. Its effectiveness was also observed in two adolescent females who had not responded to standard immunosuppressants, indicating its potential utility across a broad age spectrum. It’s imperative to contextualize these findings within the broader treatment regimen. The administration of telitacicept was concomitant with foundational therapies, including glucocorticoids and immunosuppressants, suggesting a synergistic effect rather than an isolated impact. Such a combination could be pivotal for the observed sustained clinical benefits, even after cessation of telitacicept, as evidenced by the patients who maintained improvement at the 6-month mark following a 3-month treatment course. This enduring effect might be attributed to the pharmacodynamics of the BLyS–telitacicept complex, which exhibits prolonged formation and elimination times, allowing for the continuation of therapeutic effects well beyond the active dosing period. 29
In light of the significant effectiveness observed with telitacicept in our study, the decision to discontinue medication despite these promising results warrants further discussion. Clinical experience with telitacicept, primarily in the context of treating SLE and dry syndrome, suggests a typical treatment duration ranging from 6 to 12 months.38,39 However, the application of telitacicept in MG is relatively novel, with limited clinical evidence currently available. To date, only a few case reports have documented its use in MG, with treatment durations extending up to approximately 7 months.28,40 Given the scarcity of data, particularly concerning long-term safety beyond 6 months, a cautious approach to treatment duration is advised in clinical practice. Notably, the discontinuation of telitacicept after demonstrating significant effectiveness within a 6-month window aligns with a strategy to minimize adverse reactions, while capitalizing on the drug’s potential for enduring impact. This approach is further justified by telitacicept’s mechanism of action, which includes the dual inhibition of APRIL/BAFF and suppression of LLPC, potentially contributing to its lasting therapeutic effects even after cessation of treatment. 30 Consequently, while our study presents compelling evidence of telitacicept’s effectiveness in the management of MG, the cautious discontinuation reflects a prudent balance between maximizing clinical benefits and minimizing safety risks, underscoring the need for additional research to establish its optimal duration of use and long-term safety profile in this patient population.
The safety profile of telitacicept, as observed in this study, adds to the growing evidence of its potential as a safe treatment modality for refractory GMG. The trial period passed without any serious adverse events, suggesting that telitacicept is well tolerated among patients. Notably, minor injection site reactions were infrequent and self-limiting. This safety assurance is particularly relevant given the concerns associated with long-term B cell depletion therapies. 41 Telitacicept, by contrast, may offer a targeted approach that minimizes the risk of such extended immunosuppression, thereby potentially reducing the likelihood of adverse outcomes related to immune system compromise. As such, our findings reinforce the profile of telitacicept as a viable and safe therapeutic option in managing refractory GMG.
The retrospective nature of this study, being conducted at a single center, presents inherent limitations that may affect the generalizability of our findings. The modest sample size of 16 individuals, while reflective of the rarity of refractory GMG, further constrains the breadth of data and limits the applicability of our results across diverse patient populations. Notably, our cohort included only one patient with MuSK antibody-positive MG and no seronegative patients, which restricts the extrapolation of our findings to these important subgroups of MG. These factors, combined with the differences in antibody profiles, age ranges, and disease duration within our study group, limit the comparability of telitacicept’s effectiveness with other immunotherapeutic agents such as RTX or eculizumab. Additionally, the study’s design did not allow for the determination of the most effective dosage and duration of telitacicept treatment. The absence of baseline measurements for immune biomarkers in several patients precludes a detailed analysis of immunological changes correlating with clinical improvements. Furthermore, variations in dosing effectiveness and the cost-effectiveness of telitacicept necessitate further investigation. The follow-up period’s duration was insufficient to evaluate the long-term impact and sustainability of telitacicept as a treatment option comprehensively. Considering the nascent stage of telitacicept’s application in treating GMG, a comprehensive analysis of its cost-effectiveness remains challenging. The scarcity of published data on telitacicept’s cost-effectiveness precludes a detailed discussion within the scope of our current study. Due to the exploratory nature of this study and the rarity of telitacicept use in MG, we did not conduct a formal sample size calculation. This limitation points to the necessity for future studies with detailed sample size calculations to robustly evaluate telitacicept’s efficacy.
Conclusion
This exploratory investigation provides insights into the use of telitacicept for refractory GMG, underscoring its feasibility and tolerability in a clinical setting. Our findings contribute valuable clinical evidence supporting the potential of telitacicept as a promising therapeutic option for managing refractory GMG. The results pave the way for more extensive research, advocating for the execution of further multicenter randomized controlled trials with long-term follow-up to validate the efficacy and safety of telitacicept.
