Abstract
The introduction of numerous therapeutic advancements in the management of myasthenia gravis (MG) may add difficulties in clinical decision-making, especially when no recommendations tailored to the local context are available. For this reason, the Saudi Arabia Neuromuscular and Electrodiagnostic Medicine (SANEM) chapter of the Saudi Neurology Society launched an initiative to discuss and agree on issues related to the management of MG in the Gulf Cooperation Council (GCC) region. An expert panel from all GCC countries (Saudi Arabia, United Arab Emirates, Bahrain, Kuwait, Qatar, and Oman) was formed to develop practical recommendations using the Delphi method to facilitate the management approach of MG and enhance patient outcomes.
Plain language summary
Myasthenia Gravis (MG) is a condition that affects how muscles work. With so many new treatments arising, the condition is becoming more complex to manage. This makes it challenging for doctors to choose the best care. To address this, the Saudi Neurology Society’s SANEM (Saudi Arabia Neuromuscular and Electrodiagnostic Medicine) chapter gathered experts from all GCC countries to create practical recommendations for MG using a structured method called the Delphi method. These recommendations make it easier for doctors to choose the best treatment for patients with MG.
Keywords
Introduction
Myasthenia Gravis (MG) is an autoimmune-mediated disease affecting the neuromuscular junction (NMJ), characterized by fatigable muscle weakness, which occurs when autoantibodies attack acetylcholine receptors (anti-AChRs) at NMJs or one of its anchoring units such as muscle-specific kinase or LRP4 units. 1 It has a global prevalence estimated at 12.4/100,000 population. 2 This immune process leads to the degradation of the postsynaptic membrane at the NMJ, resulting in distinct symptoms of MG. 3 Clinical symptoms of MG commonly include weakness in the eyes, throat, respiratory muscles, or throughout the body. 1 The most common symptoms in MG are ocular symptoms in 50% of cases, followed by respiratory problems and weakness which are observed in approximately 30%. The latter symptoms can be severe enough to precipitate an MG crisis and require mechanical ventilation due to weakness in the diaphragm and other respiratory muscles. 4 Severe bulbar weakness, characterized by dysphagia and weakness of the palate—often preceding respiratory muscle weakness—may necessitate nasogastric tubes for medication administration and adequate nutrition. 5 As such, MG also imposes a considerable burden on mental and social well-being, resulting in substantial implications for the overall quality of life and imposing financial burdens on both affected individuals and the broader society. Studies consistently show that severe muscle symptoms and disability are associated with depression, anxiety, and lower physical scores of health-related quality of life (HRQoL). 6 Recent research has revealed significant correlations between disease severity groups, HRQoL, and disability profiles in patients with MG. 7 Depression and anxiety were found in 36% and 33% of patients with MG, respectively. 4 Fatigue, also a prevalent symptom in MG, is associated with increased disease severity, higher rates of depression, and a decreased overall quality of life. 8
There is currently an absence of academic literature that delineates the best practices for MG care in the Gulf Cooperation Council (GCC) region. Guidance based on the clinical experience of specialists, contemporary therapeutic advancements, and local needs is needed. The development of MG guidelines tailored to the GCC region is particularly necessary to support local clinical practice and healthcare systems. In many GCC countries, hospital formularies and insurance providers require the availability of locally developed guidelines to facilitate medication registration, ensure regional availability, and account for country-specific cost considerations in both open and closed formularies. Therefore, the Saudi Arabia Neuromuscular and Electrodiagnostic Medicine (SANEM) chapter assumed responsibility for coordinating the formulation of expert opinions concerning MG care in the GCC, engaging specialists from all member nations (Saudi Arabia, Bahrain, UAE, Kuwait, Oman, Qatar) to collaboratively formulate recommendations using a modified Delphi method.
Methodology
We gathered information about MG and its management by carrying out a literature review in December 2023 by two specialized neuromuscular physicians. We conducted a search of PubMed to identify key clinical trials and guidelines on the management of MG. The search strategy included a combination of Medical Subject Headings (MeSH) and free-text terms related to MG (e.g., Myasthenia Gravis [MeSH] OR “myasthenia gravis”[tiab]) and management strategies (e.g., treatment, therapy, and guidelines). The survey for the voting consensus process before the expert panel meeting was developed. We used the Delphi modified method requiring a minimum agreement threshold of 70% among experts to adopt a statement. 9 Topics covered in the survey included epidemiology, diagnosis, outcome measures, management, definitions, and challenges. The survey was completed by 23 experts from all GCC countries (Saudi Arabia, United Arab Emirates, Bahrain, Kuwait, Qatar, and Oman) before attending the meeting on January 12th, 2024. Using the two-round modified Delphi method, topics related to items where no consensus was achieved were discussed and debated before carrying out a second round of voting and finalizing statements.
Results
Overview of agreed-on consensus criteria
A consensus process was conducted to establish expert agreement on key aspects of MG. During the advisory board meeting, experts discussed its epidemiology, diagnosis, and monitoring, along with symptomatic and maintenance therapy, IVIG and PLEX, thymectomy, myasthenic crisis, and refractory MG. They also addressed considerations for special populations and strategies for managing insufficient treatment response and side effects. A total of 37 statements were evaluated through two rounds of voting, resulting in 24 statements achieving consensus in either the first or second round, with three requiring reformulations before reaching an agreement (see Table 1). Three statements failed to reach consensus after 2 rounds and 10 statements were omitted due to insufficient evidence, with the expectation that further data would allow advisors to make more definitive judgments in their regard in the future.
Consensus outcomes from the advisory board meeting on myasthenia gravis.

Values have been rounded to the nearest whole number for clarity.
GCC, Gulf Cooperation Council; IVIg, intravenous immunoglobulin; MG, myasthenia gravis; MGFA, Myasthenia Gravis Foundation of America; PLEX, plasma replacement.
Additional questions were also included to assess expert preferences on
Preferred initial therapy choices for generalized MG (GMG), as illustrated in Figure 1.
The ranking of key treatment considerations for managing MG, as shown in Table 3.
The top selected therapies for GMG, as illustrated in Figure 4.
Factors influencing the decision for thymectomy, as shown in Table 3.
Conditions that define the optimal timing for thymectomy in eligible patients, as demonstrated in Table 3.
Expert feedback extended beyond the provided statements and questions; additional suggestions during the meeting were captured and integrated into the relevant sections of the manuscript.
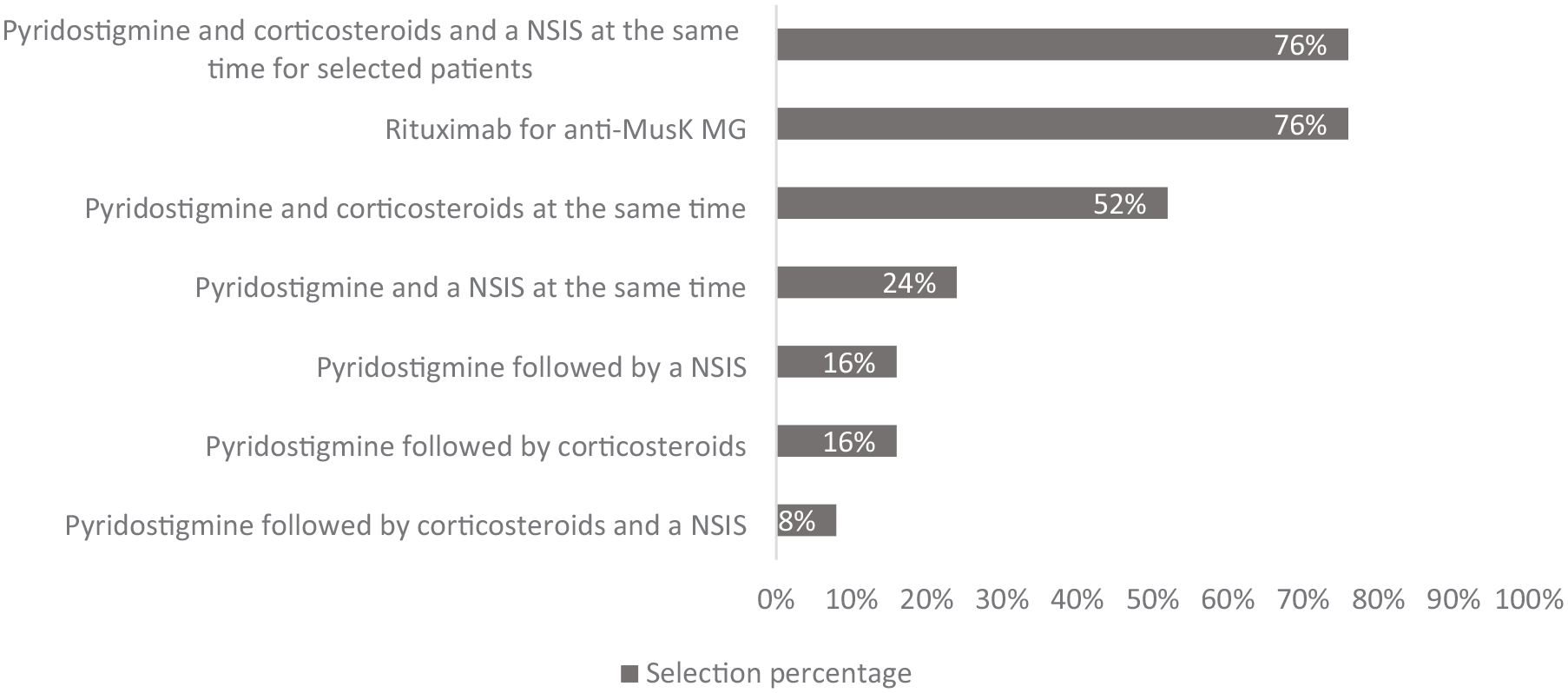
Preferred initial therapy choices for generalized myasthenia gravis: advisor selections.
Framing expert insights in light of the literature
Epidemiology of MG
MG exhibits a global incidence rate estimated at 0.3–2.8/100,000 population. 10 The current mortality rate associated with MG ranges from 5% to 9%, with an in-hospital mortality rate of 2.2% and a mortality rate of 4.7% during myasthenic crises. Notably, age and respiratory failure serve as significant predictors of mortality, with slightly higher rates observed in males (14%) compared to females (11%). Among MG patients, respiratory tract diseases, such as pneumonia and influenza, emerge as the primary underlying or contributing causes of death. It is important to highlight that MG presents at two distinct age peaks. The first peak occurs between the ages of 40 and 50, predominantly affecting women. The second peak occurs between the ages of 60 and 80 and affects men and women equally. 2
Epidemiological studies conducted worldwide consistently show a rising incidence and prevalence of MG, particularly among individuals aged 50 and older with a 1.5-fold increase in incidence and those 65 and older with a more pronounced 2.3-fold increase between 1987 and 2006.11,12 This surge in incidence could potentially be due to enhanced disease recognition and diagnostic tools, age-related immunological differences, or environmental exposures. 12 Moreover, the increase in the number of neurologists and neuromuscular specialists along with better data collection and registries could also account for this surge.
In a Japanese epidemiological study, the ocular form of MG was more prevalent in the infantile-onset group (80.6% of cases) when compared to Europe and North America (14%–30%). Similarly, a high frequency of ocular MG was observed in Chinese populations, indicating potential differences in immunological mechanisms. 12 The late-onset peak of MG is less prominent in Japanese, Indian, and Chinese patients compared to Caucasians. Asian populations also exhibit higher rates of anti-muscle-specific kinase (anti-MuSK) Ab-positive MG compared to Caucasians. Although MG can affect individuals of all ages, it is commonly referred to as “a disease of young women and old men.” 12 Late-onset MG has been increasing in Western and Asian countries over the past 30 years, potentially due to improved recognition and diagnostic tests. However, this increase may also suggest distinct immunological factors in the elderly or the involvement of environmental factors. 12 MG can also manifest during pregnancy for the first time, with variable effects on the disease. Exacerbation is observed in 41% of patients, remission in 29%, and no change in 30%. Moreover, women often report worsening symptoms before their menstrual period, coinciding with lower progesterone levels. 12 Factors such as gender, age, educational attainment, disease progression, immunosuppressive drug usage, acceptance of the illness, anxiety levels, and perceived social support have shown clear associations with a diminished quality of life among individuals diagnosed with MG. 6 Older patients with MG often present with multiple coexisting medical conditions and may have reduced tolerance to potent immunosuppressive medications which are commonly used in younger MG patients. 5
There have been limited studies regarding the epidemiology of MG in Saudi Arabia and the GCC region. Currently, there are no specific or updated statistics on the number of affected individuals in the region. Real-life statistics would help the GCC medical community and decision-makers understand MG, its prevalence, and its economic burden in the region. Some available data provides insights into the epidemiology of the disease in this region. A study conducted at King Fahad Hospital of the University in Khobar between 1991 and 1995 reported a prevalence rate of 0.56/100,000 individuals annually of MG in Saudi Arabia. 13 For other countries such as the UAE, there is still no available data about the incidence or prevalence of MG. 14 Despite the lack of national and regional statistics, our experts reached a consensus that the incidence of MG in the GCC region is within the worldwide incidence range (with 85% agreement) based on estimations from their clinical experience and based on multiple centers’ experience. Additionally, 66% of advisors reported actively following up on 10–59 patients with MG in their clinics, while the rest reported following up on at least 60 patients.
Diagnosis
First and foremost, the diagnosis of MG relies on patient assessment in the clinic. Identifying symptoms as delineated in the Japanese diagnostic criteria may facilitate the evaluation approach for the attending physician; these are (1) eye movement disorder (ptosis, diplopia), (2) facial muscle weakness, (3) dysarthria, (4) dysphagia, (5) chewing problems, (6) neck flexion and/or extension weakness, (7) limb muscle weakness, and (8) dyspnea. 15 At least one of these clinical features should be present to fulfill the “symptom” criteria of diagnosis. 15 Undoubtedly, the pattern of symptoms is exertion dependent and characterized by easy fatiguability. 15 Serological, electrophysiological, and pharmacological tests are used to confirm the diagnosis of MG in symptomatic patients (see Figure 2). 1 Similarly, the German guidelines establish MG diagnosis based on history and physical exam and use testing for confirmation purposes. 16

MG diagnostic pathway recommended by our experts.
The presence of anti-AChR antibodies confirms the diagnosis in the majority of GMG and approximately half of ocular MG cases. 17 anti-MuSK antibodies are detected in 8%–10% of cases with generalized disease.17,18 It has been reported that 30%–40% of AchR-negative patients are anti-MuSk positive. 19 Anti-low-density lipoprotein receptor-related protein 4 (anti-LRP4) antibodies are detected in as low as 2% and as high as 50% of seronegative cases for anti-AChR and anti-MuSK.17,20 Furthermore, anti-striated muscle antibodies are positive in approximately 30% of MG cases, particularly in those with thymoma.1,21,22 Although not essential for the diagnosis of MG, these auto-antibodies can serve as an adjunct marker whereby their presence raises suspicion for thymoma, particularly in patients less than 40 years of age. 23 It is also important to bear in mind the potential conversion of ocular MG (OMG) into GMG which could influence diagnosis. In fact, while previous literature estimated that close to 80% of OMG cases that generalize convert within the first year and up to 90% do so within the first 3 years, evidence from newer studies present varying results; a population-based study reviewing medical records of MG patients in Olmsted county, Minnesota between 1990 and 2017 found that 51% of MG patients presented with OMG and 55% of them generalized. 24 A total of 72% of these cases converted within the first 2 years and 94% within the first 5 years. 24
Electrodiagnostic testing such as repetitive nerve stimulation (RNS) and single-fiber electromyography (SFEMG) are valid laboratory studies to confirm MG diagnosis in the appropriate clinical context. 25 RNS test is widely available in the GCC region and is less technically demanding than SFEMG, which favors its use as an initial test. RNS involves repetitively stimulating nerves in a target muscle and measuring compound muscle action potential (CMAP) amplitudes. 26 The resulting CMAP is the sum of multiple muscle fibers’ action potential. 26 Proximal muscles demonstrate the highest diagnostic yield, but small distal extremities are favorable initial convenient sites to test. 26 A decrement of >10% detected between the first and fourth CMAP indicates abnormality. 26 SFEMG is not always available and may be less specific despite its superior sensitivity compared to RNS. 27 The latter electrophysiological test measures the difference in the onset of an action potential between muscle fibers innervated by one motor neuron, or the difference for the one repetitively stimulated single fiber. The difference in measured values is referred to as “jitter.” An increase in the latter beyond the reference value for age in the specific muscle tested is used to detect abnormality. 28 The Association of British Neurologists considers neurophysiology to be possibly useful in seronegative patients with suspected MG and gives RNS precedence over SFEMG. 29 Imaging (CT or MRI) helps in detecting thymic abnormalities, but it is important to bear in mind that MRI is more accurate than CT in distinguishing thymoma from thymic hyperplasia. 30 Even if the clinical presentation is typical for MG, serological testing for antibody subtypes is mandated due to its influence on management decisions. However, it is important to take into consideration the possibility of over-diagnosing MG based solely on a positive serological test, especially for atypical clinical presentations. Evidence from previous research shows it is best to avoid over-relying on serological and electrodiagnostic tests at the expense of clinical examination given that patients may be falsely diagnosed with MG due to positive results from seropositivity and electrodiagnostic tests. 31
To grade the severity of the disease once MG diagnosis is established, most of our experts use the Myasthenia Gravis Foundation of America (MGFA) clinical classification. As such, mild disease corresponds to MGFA class I while moderate and severe disease correspond to MGFA classes III and IV, respectively. 32 Alternatively, it is noteworthy to mention that the most recent German guidelines now propose classifying the disease based on “activity” by considering the severity/persistence of symptoms, MGFA, and exacerbations while on treatment. 16
Figure 3 illustrates the most selected investigations by our experts.

Choosing investigations based on our experts’ opinions.
Routine assessment
It is agreed by our experts that routine assessments are necessary to evaluate the severity and impact of MG on the quality of life. Hence, all patients with MG undergoing treatment need to be regularly evaluated, particularly if they have significant symptoms. In fact, quality of life disturbances may sometimes precede clinical discernibility. Additionally, routine assessments can be easily carried out—for example, facilitated through online communication between the coordinator and the patient before the scheduled visit—and that should further encourage clinicians to regularly evaluate their patients.
Outcome measures
While the selection of the outcome measure should be at the discretion of the treating physician, it is important to take into consideration factors such as ease of administration, time to completion, need for trained staff, interrater variability, and applicability to the different MG subtypes. 33 The majority of our experts would use MG activities of daily living (MG-ADL; see Table 2). MG-ADL is a patient-related outcome focusing on the functional consequences of muscle weakness (ocular, oropharyngeal, respiratory, and extremities). 34 Conveniently, it requires no training and no equipment, and can be completed within a few minutes The second most commonly selected scale by our experts is the quantitative MG (QMG) score, which is a physician-rated outcome originally developed to study the relationship between antibody titers and symptom severity (ocular, nonfacial, and facial/oropharyngeal).35,36
Selected outcomes measures by our experts.

MG, myasthenia gravis; MG-ADL, MG activities of daily living; MG-QoL15, Myasthenia Gravis-Quality of Life 15; MGC score, Myasthenia Gravis Composite score; QMG, quantitative MG.
In addition to the other outcome measures outlined in Table 2, there are additional measures suggested by our experts; these include bedside segmental strength assessment, myasthenia gravis manual muscle testing score (MG MMT), and myasthenia gravis index MG II. Therefore, clinicians are highly encouraged to capitalize on this wide variety of outcome measures to choose and combine from as they deem beneficial. Indeed, gathering patient-related information will likely enhance treatment selection and management approaches to optimize clinical outcomes and improve patients’ quality of life. 33 Additionally, in the context of clinical practice within the GCC region, experts emphasized the importance of employing scales that have undergone rigorous Arabic translation and validation. This approach enhances linguistic appropriateness for Arabic-speaking patients and enhances the reliability and validity of assessments. Two of our experts have already published validated Arabic scales for—MG-ADL and MGQOL—that are available for use.37,38
Management
The current landscape of MG treatment and management is evolving with a focus on early diagnosis, advanced therapeutic options, and enhanced patient care. Additionally, over the last few decades, there has been considerable exploration of potential treatment strategies for MG.
Corticosteroids
Supported by the existing literature and the clinical experience of our experts, corticosteroids remain the cornerstone of therapy for GMG besides initial symptomatic treatment with acetylcholinesterase inhibitors (e.g., pyridostigmine) in most patients (see Table 1). This is due to their efficacy and relatively rapid onset of action (within several weeks of treatment), as well as their cost and availability benefits. 39 They are also effective in managing OMG although further evidence is warranted to prove their role in preventing later generalization. 40 In the case of OMG, it was agreed that corticosteroids should be considered as treatment for patients who remain symptomatic despite pyridostigmine therapy. However, caution is warranted when initiating corticosteroid treatment, as it may initially worsen MG symptoms. 41 Additionally, long-term use should be approached carefully due to the increased risk of many side effects; these include osteoporosis, adrenal suppression, Cushing syndrome, hyperglycemia, glaucoma, psychiatric disorders, and cardiovascular events.41,42 As such, our experts find it beneficial to carry out periodic checks of body weight, BMI, bone density, and HbA1c for patients on long-term corticosteroids.
Nonsteroidal immunosuppressants
Considering the limitations associated with corticosteroid use, there is a need to continuously explore the effectiveness of steroid-sparing agents and targeted therapies in the treatment of MG.
Recently, the German guidelines differentiated highly active MG from less active when considering the nonsteroidal immunosuppressants (NSIS). 16 Furthermore, the guidelines showed that early aggressive treatment might lead to better long-term outcomes. 16 Taking these updates into consideration, there was a consensus among our experts that early severe disease requires more aggressive treatment. The group agreed to use MGFA of IIIb or more in the presence of bulbar or respiratory symptoms.
When opting for NSIS, there was a consensus that all generalized and OMG cases should initially receive corticosteroids until the selected NSIS exerts its therapeutic effect (see Table 1). Our experts agreed that NSIS should be used for all GMG as adjuvant therapy with corticosteroids (see selected therapies in Figure 4). In case of insufficient response or intolerable side effects to a certain immunosuppressive medication, it is recommended to consider other disease-modifying therapies or chronic IVIG as maintenance therapy as acceptable alternatives based on our experts’ clinical input (see Table 1) and the published literature.43–46

Our experts’ selection of top therapies complementing symptomatic treatment for different MG severities.
While the above therapeutic management decisions are proposed, consideration of recommendations from other international guidelines remains essential; it is noteworthy that the Association of British Neurologists deems it acceptable to consider NSIS only if the patient fails to achieve remission with corticosteroids alone unless the patient has a significant bulbar or respiratory weakness that does not rapidly resolve with corticosteroids. 29 The Association recommends considering early initiation of NSIS for patients with diabetes mellitus, osteoporosis, and ischemic heart disease. 29 The international consensus guidance broadens options and highlights that acceptable NSIS to be used in MG include azathioprine, cyclosporine, mycophenolate mofetil, methotrexate, and tacrolimus. 46
Adding azathioprine to prednisone is associated with a lower needed maintenance dose, a longer remission period, and fewer side effects. 47 Our experts consider it a favorable NSIS option given its cumulative evidence of benefits and encouraging safety profile in women of childbearing age, while other options such as mycophenolate mofetil can be contraindicated in pregnancy. 48 Azathioprine is also the first-line NSIS adopted by the Association of British Neurologists. 29 There is also a parallel inclination toward the utilization of this agent in the international consensus guidance. 46 The German guidelines also recommend first considering glucocorticoids and/or azathioprine in addition to symptomatic treatment for mild-to-moderate MG with any of the following serologic statuses: anti-AChR Ab-positive MG, anti-LRP4 Ab-positive MG, or seronegative generalized/juvenile MG. 16 If azathioprine is ineffective or cannot be used, the German guidelines deem it acceptable to consider mycophenolate mofetil, cyclosporin, or methotrexate. 16 This is in alignment with our experts’ votes as shown in Figure 4. Conversely, this is not the case in the Japanese guidelines because azathioprine was only recently approved for use in Japan and is still not commonly used. 15 Besides azathioprine, mycophenolate mofetil also garnered the majority of votes from experts (see Figure 4). However, it is important to note that despite its widespread use, randomized controlled trial (RCT) evidence regarding its efficacy remains inconclusive; it is believed that it is widely prescribed for MG primarily based on clinical expertise, as it did not demonstrate superiority over placebo in an international phase III trial. 49 Nevertheless, it is noteworthy that a recent study in 2024 compared mycophenolate mofetil to azathioprine and found both compounds to have similar efficacy with no significant difference in clinical outcomes. 50
As for calcineurin inhibitors, the effectiveness of cyclosporine in MG has been previously established in small trials.51,52 Tacrolimus hasn’t yet shown statistically significant steroid-sparing effects as per a recent systematic review, although it is generally safe and effective. 53 Another systematic review stated that this agent “might” have steroid-sparing benefits, but larger randomized controlled trials are needed for conclusive results. 54 Despite that, calcineurin inhibitors are the only nonsteroidal immunosuppressants used for MG in Japan. 15 The renowned antimetabolite, methotrexate, has demonstrated a steroid-sparing ability in a recent randomized open-labeled trial in a small group of participants; patients in the prednisone + methotrexate group required a significantly lower daily steroid dose in comparison to those in the prednisone-only group. 55 For mild-to-moderate MG, calcineurin inhibitors (Tacrolimus and Cyclosporine A) are among the selected therapies by our experts (see Figure 4).
Rituximab, an anti-CD20 monoclonal immunoglobulin, showed satisfactory results in terms of clinical benefits for patients with anti-MuSK Ab-positive MG in a recent multicenter blinded prospective review carried out over 10 years. 56 Additionally, the RINOMAX randomized controlled trial published in 2022 found rituximab to be a safe and effective treatment option for patients with recent-onset GMG symptoms (for a maximum of 12 months). 57 This is not the case for anti-AChR Ab-positive MG, whereby rituximab did not yield similar positive outcomes. This is based on a recent trial, the BEATMG study, which showed it was unlikely to have a clinically meaningful steroid-sparing effect from the use of rituximab in anti-AChR Ab-positive MG. 58 Besides anti-MuSK positivity, other factors increasing the likelihood of treatment success with rituximab include younger age at disease onset as well as milder disease. 59
While there was no consensus during the meeting among our experts to recommend CD20 antibody depletion therapy (rituximab) as a first-line disease-modifying intervention for highly active anti-AChR Ab-positive, anti-LRP4 Ab-positive, seronegative, or refractory MG—even when accounting for potential cost-effectiveness—it was suggested that its use may be appropriate in selected clinical scenarios. In such cases, the decision to initiate rituximab therapy should be individualized and made on a case-by-case basis, considering patient-specific factors and disease characteristics. Moreover, there was official consensus on the use of rituximab as a first-line agent for anti-MuSK Ab-positive MG (see Figures 1 and 4).
Newer approaches involve the use of specific monoclonal antibodies, commonly known as “biologicals,” to modulate the immune system and address the condition. 59 These include neonatal Fc receptor (FcRn) inhibitors (Efgartigimod, Rozanolixizumab) and C5 inhibitors (Eculizumab, Ravulizumab, Zilucoplan).60,61 Our experts recommend their use for different classes of MG as delineated in Figure 4.
FcRn inhibitors operate by blocking the binding of the FcRn to Immunoglobulin G (IgG), thereby impeding its protective function against catabolism. 62 For instance, a randomized phase III trial showed that the odds ratio of being an MG-ADL responder (⩾2 point improvement for at least 4 weeks) for a patient on Efgartigimod compared to one who is on a placebo is 4.95. 63
C5 inhibitors operate by blocking the activation of terminal complement by targeting C5 and disrupting C5a and C5b and as a consequence preventing the formation of the terminal complement component or the membrane attack complex (C5b-9). 64 A phase III trial in 2017 followed by an open-label extension in 2019 demonstrated rapid and sustained improvement with Eculizumab in MG-ADL and QMG scores in MG patients with poor symptom control despite prior therapy with two immunosuppressive agents or one immunosuppressive agent and chronic therapy with either IVIG or PLEX.65,66 Recently, Ravulizumab has been shown to induce rapid and maintained improvement in patients with anti-AChR antibody-positive (anti-AChR Ab-positive MG) GMG.67,68 Zilucoplan also showed a favorable efficacy and safety profile in a phase III trial published in 2023. 69 It is noteworthy to mention that Zilucoplan demonstrates a dual mechanism of action; it prevents the cleavage of C5 and competitively inhibits the formation of C5b6.70–72 Moreover, in alignment with the scientific literature, experts of the advisory board meeting did not reach a consensus regarding the use of complement inhibitors in patients with seronegative or anti-LRP-4 Ab-positive MG due to lack of clinical trials. 61 As for anti-MuSK MG, it is important to bear in mind that while complement inhibitors cannot be used because MuSK antibodies are of the IgG4 subclass which are unable to activate complement,73,74 FcRn inhibitors can be considered as a second-line option for this subpopulation of MG patients. This is because the latter monoclonal antibodies can accelerate the degradation of circulating IgG from all subclasses, including IgG4.75–77
Refractory MG
Despite all the previously mentioned therapeutic advancements, about 10%–15% of GMG patients still fail to respond to immunosuppressants, necessitating intravenous immunoglobulin (IVIg) or plasma replacement (PLEX).2,78
The selection criteria included the following, as suggested by Mantegazza and Antozzi 78 : (1) “Failure to respond adequately to conventional therapies,” (2) “Inability to reduce immunosuppressive therapy without clinical relapse or a need for ongoing rescue therapy such as IVIG or PLEX,” (3) “Severe or intolerable adverse effects from immunosuppressive therapy,” (4) “Comorbid conditions that restrict the use of conventional therapies,” and (5) “Frequent Myasthenic crises even while on therapy.”
Our experts agreed on the following criteria for “Refractory MG” (see Table 1):
- Frequent myasthenic crises even while on therapy.
- Failure to respond adequately to conventional immunosuppressive therapies.
For steroid-sparing immunosuppressants, the case is considered well controlled as long as they are clinically stable while on treatment (despite an inability to reduce immunosuppressive therapy without clinical relapse or a need for ongoing rescue therapy such as IVIG or PLEX). As such, in contrast to the criteria proposed by Mantegazza and Antozzi, our experts disagreed with the inclusion of “inability to reduce immunosuppressive therapy without clinical relapse or a need for ongoing rescue therapy such as IVIG or PLEX,” “severe or intolerable adverse effects from immunosuppressive therapy,” and “comorbid conditions that restrict the use of conventional therapies” as defining criteria of refractory MG. 78 There were different opinions regarding these descriptions, and they did not reach the agreement threshold (⩾70%). This is because the latter criteria were considered by many experts to reflect treatment-limiting factors rather than criteria of true refractoriness, which should describe disease biology and response per se to treatment. Thus, in case of confusion regarding a possible “refractory MG” case, the general neurologist is advised to refer the patient to a more specialized neuromuscular neurology physician. It is also important to recognize severe cases or MG crises demanding immediate treatment due to life-threatening complications like impaired swallowing or respiratory failure. 1 As for the duration criteria, our experts discussed that it may be appropriate to consider “failure to respond adequately to conventional immunosuppressive therapies” after 6 months from initiation of treatment. Additionally, failure to reduce steroids to minimize their side effects can also be considered treatment-refractory following the same time frame.
Experts’ ranking of treatment considerations
For patients with mild-to-moderate disease activity, there was no consensus regarding the hierarchy of considerations when determining the appropriate treatment for GMG. Our experts believe there is no single parameter that would be the most appropriate consideration for treatment selection; it is a rather multifactorial decision. In regard to the decision to recommend thymectomy, age, thymoma, and seropositivity status (see Table 2) are factors agreed upon as attributes that affect the choice of therapy. Other factors such as disease severity, response and tolerance to treatment, cost and accessibility, international guidelines, comorbidities, and patient preferences received varying prioritization ranks among experts (see Table 3). Pregnancy planning is also an important treatment consideration agreed upon by experts (see Table 1).
Advisors’ considerations and rankings for therapeutic management and thymectomy decision-making.

MG, myasthenia gravis.
For patients with highly active disease, which is defined as MGFA IIIb, and for those who show an incomplete or partial response after 6 months of treatment with corticosteroids and an NSIS, the discussion led experts to consider it acceptable to initiate more aggressive therapies when appropriate, including C5 inhibitors, FcRn inhibitors, or rituximab (see Figure 4). During the meeting, experts also discussed the importance of closely monitoring patients with active disease using clinical assessment and MG-ADL every 3 months. It was suggested that by 6 months, if there is incomplete improvement or intolerable side effects, escalation of therapy could be considered. For patients with stable disease, follow-up every 6 months was considered reasonable.
Myasthenic crisis
The experts agreed on the below definition of myasthenic crisis (see Table 1): A complication of MG characterized by worsening muscle weakness resulting in impending respiratory failure that could require ventilation
A myasthenic crisis may be preceded by a trigger such as a respiratory tract infection or may be untriggered as in the case of 30%–40% of patients. 79 Together with the use of PLEX/IVIG—which are the mainstay of treatment—it is important to manage the airway of the patient to eliminate any identifiable triggers and maintain close observation of the patient’s clinical status in the intensive care unit.46,79 Although both PLEX and IVIG show similar efficacy in the management of myasthenic crises, some guidelines may prefer PLEX due to its faster onset of action. 46 While corticosteroids offer added benefits in symptom management when combined with PLEX/IVIG (see Box 1), initiating them immediately in the emergency department should be avoided in the beginning and started a few days later, as they could potentially exacerbate muscle weakness and lead to further decompensation. 80 Those at higher risk of myasthenic crisis may have a history of previous episodes, severe disease, positive anti-MuSK, oropharyngeal weakness, and thymoma. 79 It is also important to consider the contraindications and comorbidities before choosing PLEX or IVIG. For instance, sepsis is a contraindication to the use of PLEX, while renal failure, hypercoagulability, and hypersensitivity to immunoglobulins preclude the use of IVIG. 46
Consensus among experts on issues regarding IVIG and PLEX utilization.

IVIG, intravenous immunoglobulin; MG, myasthenia gravis; PLEX, plasma replacement.
Thymectomy
Thymectomy is needed for any patient with thymoma whenever possible. In nonthymomatous generalized MG, thymectomy is recommended primarily for anti-acetylcholine receptor antibody (anti-AChR Ab)-positive patients.29,46,81
The MGTX trial compared clinical outcomes as measured by the QMG score for patients who received extended transsternal thymectomy with alternate-day prednisone versus those who only received prednisone. 82 Patients included in the trial had elevated anti-AchR titers and were diagnosed with MG for less than 5 years. By the end of 3 years of the trial, patients who received thymectomy displayed lower QMG scores, lower required doses of prednisone, fewer hospitalizations, and less frequent need to switch to azathioprine. 82 Thymus hyperplasia and thymoma are usually associated with an anti-AChR Ab-positive MG subtype rather than the other disease subtypes. 83 However, if the patient is not clinically stable and is experiencing moderate-to-severe symptoms, thymectomy should be postponed until MG is deemed well-controlled unless there is a malignant thymoma requiring thymectomy as soon as possible—even if MG is not under optimal control.29,46,81 It is noteworthy that thymus hyperplasia may rarely be associated with anti-MuSK and anti-LRP4 Ab-positive MG, although some studies found near-normal thymuses in patients with these disease subtypes and there is still no evidence to indicate thymectomy for them.46,84–86 The international consensus guidance allows for the consideration of thymectomy in anti-AChR Ab-positive GMG in case of incomplete response to immunosuppressive therapy or if the latter causes intolerable side effects. 46 Eligibility criteria for nonthymomatous patients usually entail an age between 18 and 65 years 82 and other guidelines such as those of the Association of British Neurologists recommend age less than 45 years. 29
The top three considerations that would influence the decision of experts to recommend thymectomy are seropositivy status (anti-AchR Ab-positive MG), presence or absence of thymoma, and age group (see Table 3). Once a patient is deemed eligible, experts recommend that thymectomy should be considered as early as possible following diagnosis, provided that the patient’s clinical condition remains stable. While our experts abide by the recommendations of currently existing guidelines regarding the timing and indication for thymectomy in MG patients, they also suggest that some patients might benefit from thymectomy regardless of their serological status or age (i.e., those with seronegative profiles, outside the 18–65 years age range, or show partial response to current immunosuppressive or immunomodulatory treatments). This decision, owever, should be individualized and made on a case-by-case basis upon consultation with a specialized neuromuscular physician. It is important to note that our local experts consider this option reasonable and feasible because the thymectomy procedure is usually covered and available for all citizens within the GCC healthcare system and is currently considered quite safe as compared to the past, and generally only requires a short hospital stay with no intensive care needed.
Special populations
Pregnant patients with MG
There is no agreement that pregnancy should be discouraged in patients with MG. As such, women of childbearing age with MG are not discouraged from becoming pregnant; however, planning the pregnancy based on the immunotherapy with the treating physician may be needed on an individual basis (see Table 1). It is important to involve a multidisciplinary team of specialists before, during, and postpartum to plan pregnancy and ensure the disease is stable well before gestation. Similarly, the German guidelines recommend a consultation at a specialized MG center in case of planned or actual pregnancy. 16 It is thus advised that nonspecialists refer any pregnant patient with MG to an expert facility. It is agreed that taking such preventative measures improves clinical outcomes for both the mother and the fetus. Moreover, while monitoring is crucial, patients with well-controlled disease before pregnancy are generally expected to remain stable while pregnant. 46
Elderly and juvenile patients with MG
Our experts discussed issues concerning the customization and adjustment of management approaches to suit particular patient demographics such as the elderly and juvenile populations. Experts agreed that management would be largely unchanged in comparison to that of the general population (see Table 1). However, it is important to note that symptoms, such as ptosis, diplopia, and facial muscle problems may be harder to detect in the population of the elderly. 87 Considering the high prevalence of obesity, hypertension, and diabetes mellitus in the GCC region—including Saudi Arabia, where approximately 22.2% of adults 60 years and older are obese, 55.4% are diabetic, and 49.1% are affected by hypertension 88 —our experts emphasized during the meeting the importance of minimizing the chronic use of corticosteroids in the management of MG, especially for the elderly patient subpopulation. This is because chronic steroid use is associated with adverse metabolic effects previously discussed, including weight gain, glucose intolerance, and exacerbation of comorbid conditions. It is also noteworthy that although we generally manage these patients similarly to the general population when it comes to the need for thymectomy, it is important to bear in mind that there remains insufficient evidence regarding thymectomy in these groups when unresponsive to treatment. Moreover, our experts advise referring MG patients eligible for thymectomy or fulfilling the criteria for refractory MG (difficult-to-treat) to a specialized center.
Conclusion
Although there are many therapeutic innovations in MG management to profit from as a treating physician, this can also make decisions more confusing and challenging. The absence of guidance tailored to the local context calls for the creation of recommendations intended for clinical practice in the GCC. For this purpose, this manuscript not only gives an overview of the current state of issues related to MG but also aids regional neurologists in refining their clinical decision-making using the local clinical expertise of GCC specialists. We aim to update our recommendations as needed and as clinical advances arise in the future.
