Abstract
Comparing results from jitter studies performed with voluntary and electrical activation is difficult to perform quantitatively, particularly in complex signals as seen in reinnervation. High jitter values in individual spikes in these multispike signals can be missed with both activation methods, which introduces a bias towards more normal values.
With voluntary activation, triggering on a spike from an abnormal end-plate in multispike potentials will overestimate individual jitter values and the number of abnormal jitter values.
With electrical stimulation, artefactually-increased jitter may be caused by subliminal stimulation, which causes uncertainty at the stimulation point. Electrical stimulation also may activate many axons, causing signal summation, with erroneous or impossible jitter estimation.
Awareness of such pitfalls can improve the correct performance and interpretation of jitter recordings. Quantitative comparisons of results can be made between studies in reinnervated muscle performed with the same activation method, but not between results obtained with different activation methods.
Highlights
Difficulties in expressing jitter in complex signals Jitter recordings with voluntary activation. How to express overall abnormality in recordings with complex signals Jitter recordings with electrical stimulation. How to express overall abnormality in recordings with complex signals
INTRODUCTION
In electrodiagnostic studies (EDX) the muscle is activated by either voluntary contraction or electrical stimulation. In some studies, both activation methods are used, e.g. when surface voluntary EMG is compared to the response to electrical stimulation [1], and in Single Fiber EMG (SFEMG). In the latter case each laboratory usually prefers one activation method, but may use the other one in certain situations. Several studies have compared the jitter obtained with the two methods in normal muscle [2–6].
In conditions with reinnervation, the signals are usually complex due to reorganization of the motor unit (MU), with grouping of muscle fibers. Jitter is usually also increased, particularly in the early phase of reinnervation, probably representing immature motor end-plates [5, 7–11].
In routine EMG recordings jitter is usually assessed subjectively as motor unit potential stability as a complement to other EMG parameters. Jitter provides information about the functional chronicity of reinnervation: Large jitter and blocking are seen in the early phase of reinnervation and recordings with less jitter are seen in later phases. In SFEMG the Fiber Density (FD) is an important parameter to demonstrate reinnervation. With concentric electrodes, however, as used in this study, FD cannot be measured; instead, parameters such as motor unit potential (MUP) amplitude, duration and polyphasicity provide evidence of reinnervation. In this study we measured jitter to assess neuromuscular transmission in reinnervated muscles.
It has been our general impression that in neurogenic conditions there may be a discrepancy between voluntary activity (e.g. integrated surface EMG) and evoked compound muscle action potential (CMAP) that is not seen in normal muscle. It has also been our impression that jitter is less abnormal with electrical stimulation than with voluntary activation in the same reinnervated muscle. We therefore undertook studies to compare the jitter measured with the two activation methods in reinnervated muscles. After preliminary studies, however, it became apparent that technical issues in such a comparison are presently unsurmountable and decided to describe these difficulties to assist others making similar attempts. Recordings from different types of neurogenic disease were used as examples of these difficulties; no attempt was made to stratify the material by chronicity or severity.
METHODS
Selected patients with any kind of neurogenic lesion and corresponding EMG abnormality were asked to participate in this study. Conventional electrodiagnostic studies were performed to characterize the neurogenic condition. Informed consent was then obtained from participating patients to have jitter studied with both voluntary activation and electrical stimulation. All studies were performed on KP.net equipment (Natus, USA) with a concentric needle electrode (AMBU® Neuroline, Denmark) and filter settings for jitter analysis of 1000 Hz-10 KHz. The study was in accord with the Helsinki Declaration of 1975 and approved by the ethics committee of the Faculdade de Medicina de São José do Rio Preto, São Paulo, Brazil, where the EMG studies were performed.
Stimulation for jitter analysis was performed at 10 Hz and intervals were measured between stimulus onset and peaks of accepted signals. Stimulus pulses of 0.1 msec duration were used at the lowest intensity that was demonstrably supraliminal for the signal being analyzed.
For voluntary jitter analysis, intervals were measured between the automatically-detected peak of the triggering spike and the peaks of the other accepted signals.
Quality control
The operator (JK) assessed and confirmed that stimulation was supraliminal for all the peaks that were accepted for analysis. Three of the authors (ES, JK and DS) then together evaluated the quality of the recorded signals via internet video communication. The clinical information of the patient was known only to the operator but information on the reviewed signal traces indicated if voluntary or stimulation activation had been used.
To be included for analysis individual spikes had to meet the following previously-published criteria [4]: The spike should have a fast rising slope without notches or shoulders. In superimpose mode, the rising phase of each of 10 consecutive traces should be parallel. A disturbance from other muscle fibers may be accepted if the disturbance occurred at an amplitude level less than half of the amplitude of the negative rising phase of the spike. The peaks should be separated by more than 300μsec to assure that there was no overlap between the spikes under study. Minimal background activity could be accepted, but high amplitude disturbances close to the spikes under study were excluded. These disturbances can cause a minor increase or decrease of the jitter value for the individual spikes, depending on the frequency content of the disturbances, their amplitude and time relation to the spike under study and the shape of the spike. The effect of these disturbances is less for the peak trigger algorithm than for the amplitude level method.
For voluntary signals: At least 20 traces had to be accepted for a jitter value to be measured. For each study, at least 15 jitter values should be obtained. For complex signals with 3 or more spikes, the triggering spike that gave the lowest summated jitter of the complex was chosen as the trigger (see below).
For stimulation signals: Supraliminal stimulus intensity must be verified by the operator during recording. At least 30 traces had to be accepted. For each study, at least 25 jitter values from individual spikes should be collected.
Jitter values exceeding 150μsec were truncated to 150μsec to avoid the effect of the velocity recovery function (VRF) produced by impulse blocking, which usually occurs when jitter is this great.
RESULTS
Thirty-five patients were included. The diagnosis at the time of the study was: radiculopathy (13 patients), motor neuron disease (10), chronic inflammatory demyelinating polyneuropathy (6), Guillain-Barré syndrome (3), polyneuropathy, organomegaly, endocrinopathy, monoclonal gammopathy, and skin changes (1), Charcot-Marie-Tooth disease type 2 (1) and chronic post-polio sequelae (1).
Five studies with too few data were excluded: 4 stimulation studies and 1 voluntary jitter study. In the 30 remaining paired studies, 621 voluntary and 905 stimulation spikes were analyzed. The tibialis anterior muscle was studied in 20, extensor digitorum in 6, and triceps in 4 patients. The mean age of these patients was 49.5±13.2 years, and 19 were male.
The mean jitter values among the studies were: 56.4±22.3μsec for voluntary and 40.2±16.2μsec (mean, SD) for stimulation jitter, respectively (p < 0.05). The stimulation values were on average 74.7±25.0% (range: 39.9–168.6%) of the voluntary values.
The difference in jitter measured by voluntary and stimulation activation varied considerably among patients (Fig. 1).
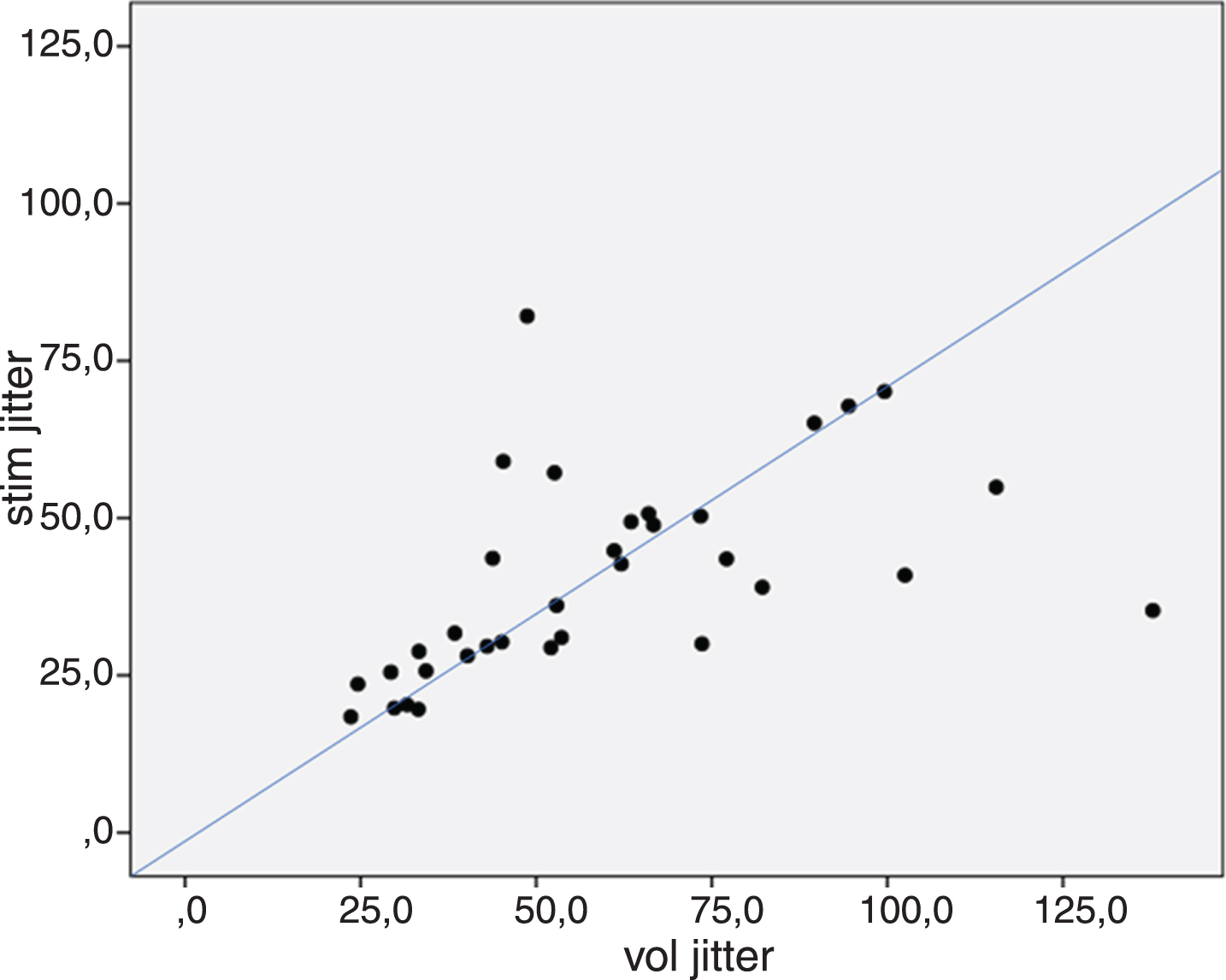
Comparison of jitter (MCD values) measured in the same reinnervated muscle with voluntary (vol jitter) vs stimulation activation (stim jitter). The line indicates the theoretical relationship between the 2 values, i.e., stimulation jitter should be 71% of the voluntary value.
DISCUSSION
With electrical stimulation the jitter is measured between the stimulus and a single muscle fiber, thus is produced by only one endplate, whereas two endplates produce the jitter measured with voluntary activation. If the endplates have similar and Gaussian-distributed jitter, the stimulated jitter is expected to be 1/√2 = 71% of the voluntary jitter values (Fig. 1). In previous comparisons in healthy controls [2, 6], the stimulation jitter was between 67 and 77% of the voluntary jitter value, except in the orbicularis oculi muscle, where the stimulation jitter was 55% of the voluntary value. In our material the range was much larger (the stimulation jitter was between 40% and 168% of the voluntary jitter), and because of the large scatter of data, there was no significant difference between the jitter measurements with the two different activation methods. As shown in Fig. 1, the discrepancy between voluntary and stimulated jitter seems to be unpredictable.
Why is this comparison so difficult to make in reinnervated signals? While it can be performed in normal muscles where we usually record signals from only 1–3 muscle fibers, perhaps the problem lies in the complex signals seen in reinnervation.
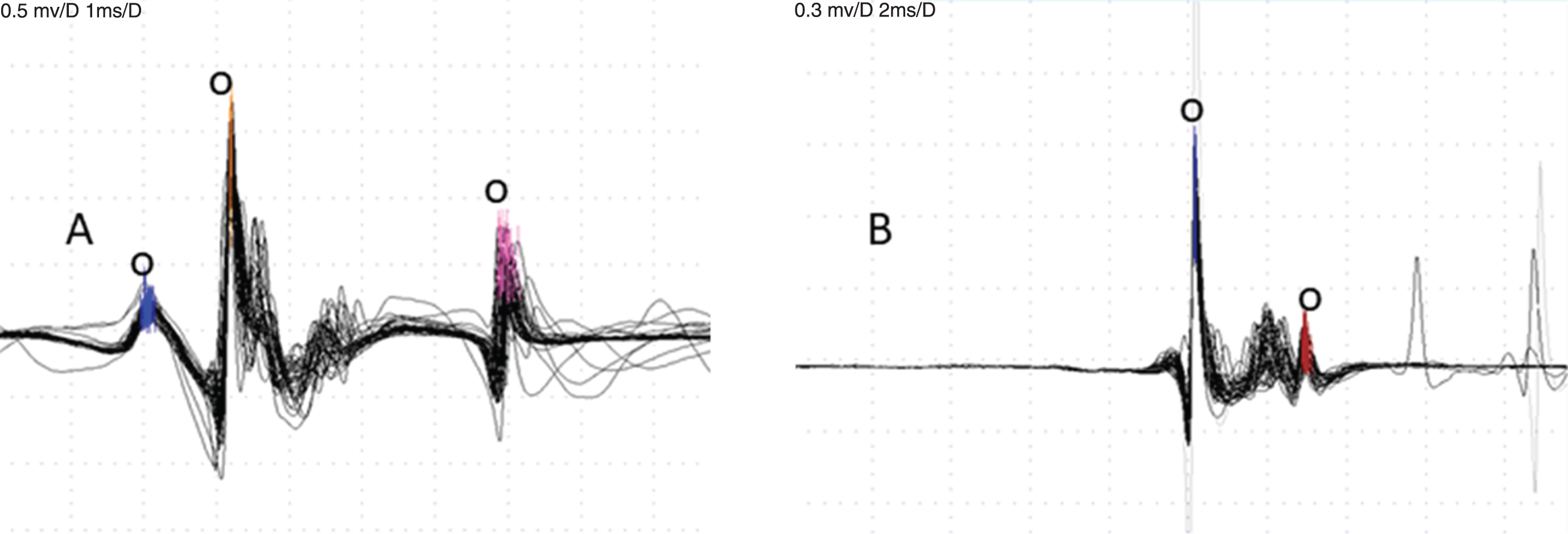
Voluntary CNE recordings. A – 3 spikes are accepted for analysis (small circles), the second is triggering. Jitter is 55 and 71μsec, respectively. B – the largest spike is triggering and one other spike is accepted (small circles). Jitter 31μsec.
Interpretation of results
Assessment of jitter in complex signals with voluntary activation
For voluntary jitter studies that measure the combined jitter of the triggering spike and the spike under study, interpretation is somewhat complicated in recordings with multiple spikes. In a 2-spike recording the jitter is the same no matter which spike is used as the trigger, and if the jitter is increased, we do not know if both end-plates are abnormal or only one. In a recording with 3 or more spikes, the jitter in the triggering spike will contribute to the calculated jitter between it and each of the other spikes. If the triggering spike has increased jitter, this will increase the jitter for all the other spikes, which would give quite erroneous results. Thus, it is important that the analysis in signals with multiple spikes be made using the “best” trigger, as determined by the minimal summated jitter value [12, 13]. This process is somewhat tedious but necessary in a study of complex signals. Some EMG equipment have a feature that finds the minimal summated jitter to facilitate the search for the best triggering spike.
This is a significant pitfall in analysis of complex signals. In this study, attention was given to finding the best trigger for each complex signal with increased jitter. This is not a problem in stimulation jitter studies, where each spike represents only a single end-plate.
In complex signals it is usually not possible to assess the jitter quantitatively for all spikes. Spikes with large jitter may not fulfill the quality criteria for acceptance (amplitude, rise-time, amount of jitter, amount of blocking) (Fig. 2). These signals show that some obviously abnormal spikes cannot be analyzed quantitatively. Thus, for voluntary recordings, many spikes escape analysis, which gives a clear bias towards normal spikes in complex signals; this is not seen in normal material or in myasthenia gravis, where signals usually have a simple shape. It should be noted that signals recorded with concentric electrodes, as here, are generally more complex than those recorded with the more selective SFEMG electrode. This is one of the reasons why it is impossible to obtain a correct picture of the overall jitter in such signals if only the calculated jitter values are considered. Other methods, such as jiggle analysis [14] have not been used to clarify this issue.
Assessment of jitter in complex signals with electrical stimulation
In electrical stimulation jitter studies, difficulties arise not only because each MU is complex, but also because axons from different MUs may be simultaneously stimulated (Fig. 3). As with voluntary activation studies, stimulation jitter estimations limited to accepted individual spikes may not reflect the overall abnormality in a recording, and signals containing obviously abnormal components may be called normal if only the calculated jitter values are considered.
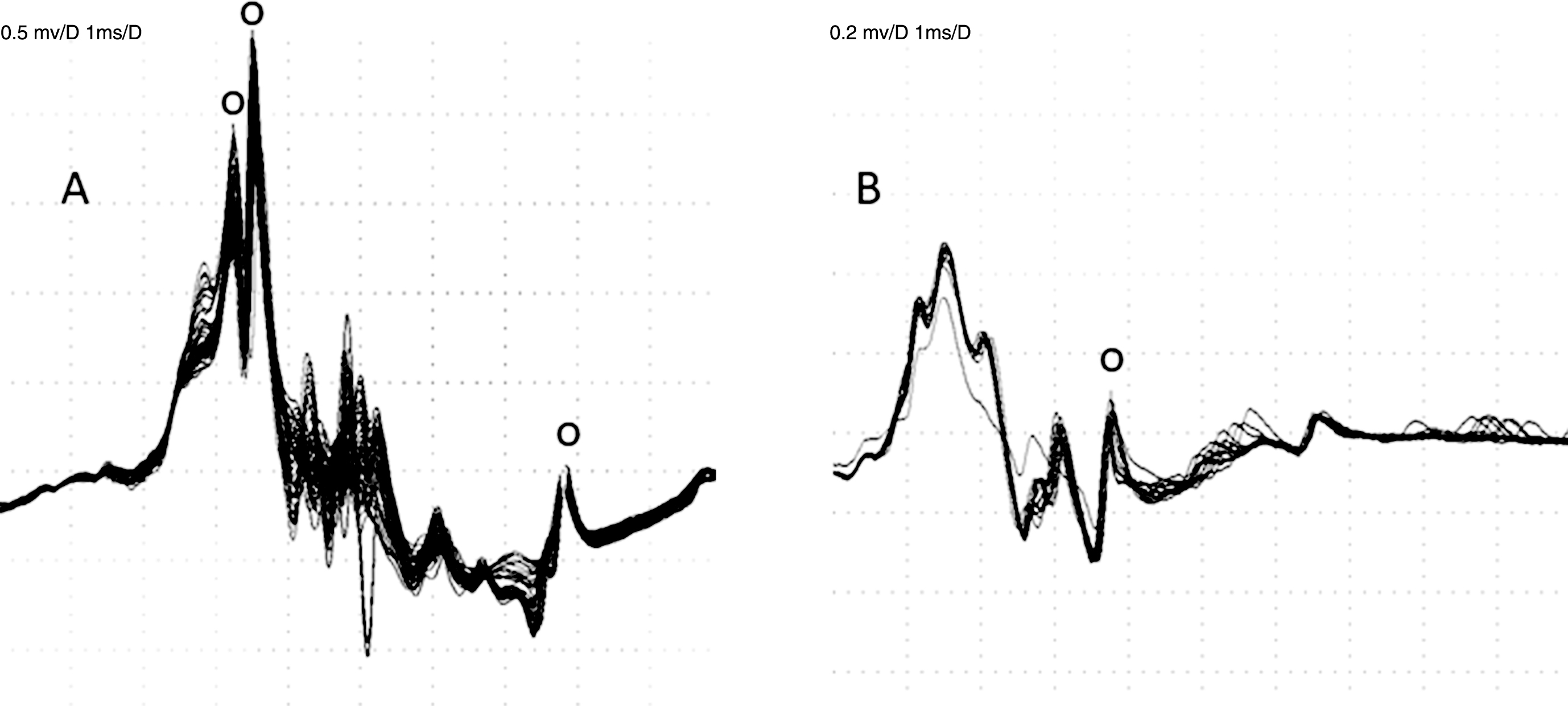
Stimulation jitter study. A – Jitter is 14, 24 and 14μsec, respectively, for the 3 spikes indicated by small circles. B – Only one spike has been accepted, with jitter of 38μsec (small circle). Some obviously abnormal spikes in both signals are too complex to fulfil acceptance criteria or occur too infrequently to be measured. These two recordings would be called normal based on the calculated jitter values.
The following reasons for increased jitter with stimulation have been more fully discussed in a separate publication [13].
Subliminal stimulation with jitter at the stimulation point [15, 16]. (This is an issue also for simple signals.) This possibility can be addressed by increasing and decreasing the stimulus intensity during signal acquisition. Spikes that appear or block together belong to the same axon (Fig. 4). Thus, if a group of 5 spikes behave together and 2 spikes have normal jitter, then we know that the stimulation is supraliminal and the increased jitter in the other 3 spikes is in the endplate. This procedure requires minimal changes in stimulation intensity if the excitability is very similar for adjacent axons. In a mixture of normal and abnormal spikes the most abnormal may evade analysis if their latencies overlap.
Another phenomenon is the axon reflex, which frequently occurs with intramuscular stimulation [17] and produces signals with a dual distribution of latencies when there is even minimal variation in stimulation intensity. Note that the axon reflex is most easily seen when the jitter is low.
The presence of F-responses that disturb the interval between stimuli and thus produce a VRF effect [18].
Different types of axons may be activated during voluntary (smallest MUs and smallest axons) and electrical stimulation (larger axons). In pathology, different excitability due to the underlying condition may play a role.
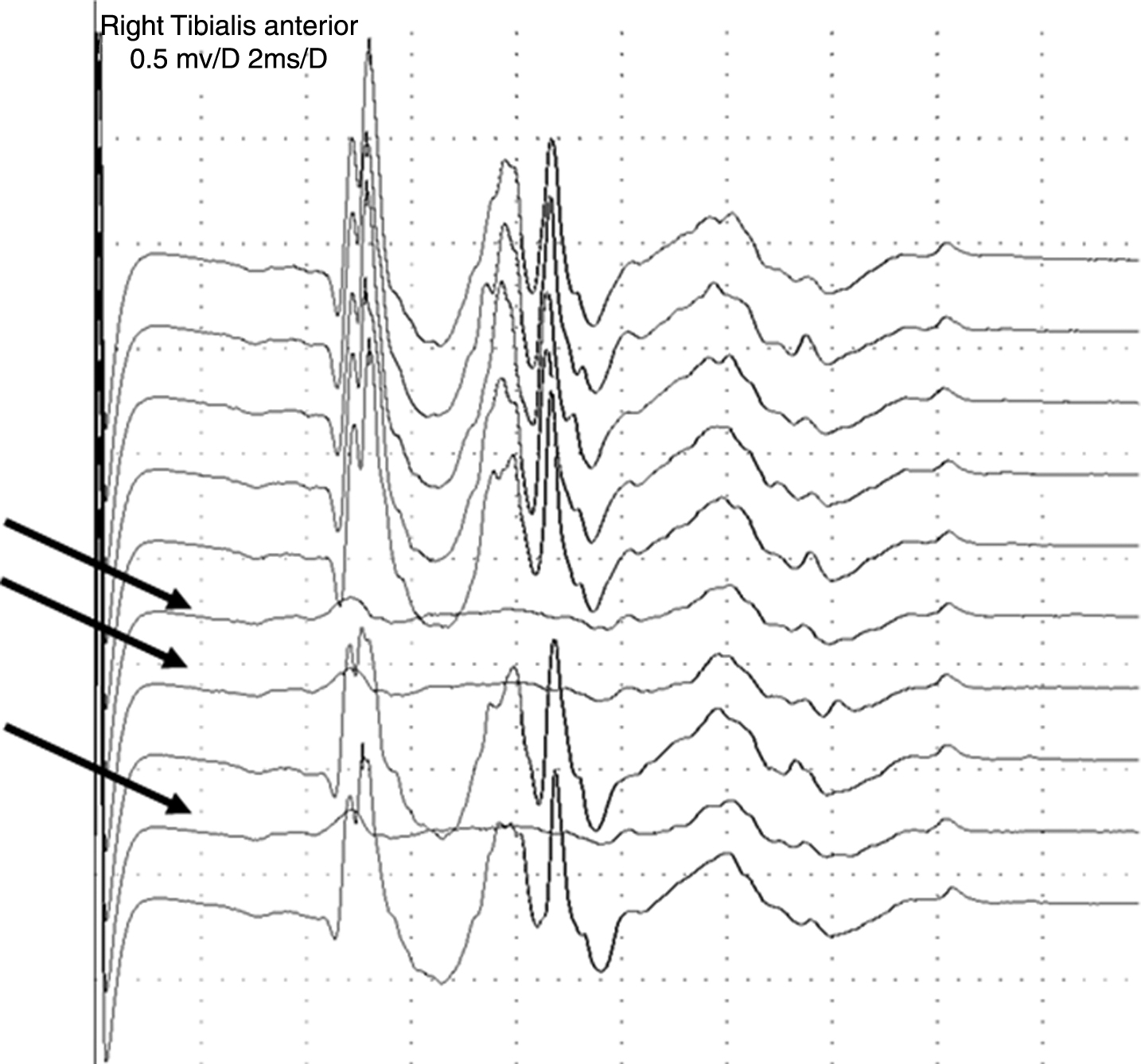
Stimulation jitter study in a radiculopathy with complex signals. The stimulus intensity is being varied. With decreasing stimulus intensity the complex signal with sharp spikes disappears in three traces (arrows), while activity from background fibers, seen as more diffuse signals, remain, indicating that they belong to other motor axons.
Summary
We conclude that even with optimal conditions for analysis, it may still be difficult to compare voluntary and stimulation jitter values from complex signals from reinnervated muscle using conventional jitter analysis. The same may also be true for complex signals from myopathic conditions. The issues of summation of jitter in 2 motor end-plates with voluntary contraction and liminal stimulation in electrical activation lead us to conclude that comparisons can be made between results obtained with the same activation method but not between studies performed with the other method. Other means of assessing MU instability, e.g. jiggle analysis [14], may be useful for comparing results between the two methods.
Footnotes
ACKNOWLEDGMENTS AND SOURCES OF SUPPORT
Funding: João Aris Kouyoumdjian was supported by a specific grant for SFEMG studies (2008/10082-69 and 2017/13262-4) from the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), Brazil. The other authors did not receive any specific grant from funding agencies in the public, commercial, or not-for profit sectors
Disclosures: The authors have no conflict of interest to report.
