Abstract
BACKGROUND:
The thousands of chemicals and compounds found in cigarette smoke, including many free radicals and oxidants, can cause oxidative damage to DNA either directly or by producing reactive oxygen species in cultured cells. This study was carried out to analyze the urinary 8-Oxoguanine DNA Glycosylase (OGG1) and 8-Hydroxy-2’ –Deoxyguanosine (8-OHdG) among cigarette and non-cigarette smokers in South-West, Nigeria.
METHODS:
Urine samples were collected from 250 cigarette smokers and 200 non-cigarette smokers who live in south-west, Nigeria. Questionnaire was administered first to both cigarette and non-cigarette smokers prior to specimen collection. About 10mls of urine samples were collected from each of the subjects and was used to carry out Oxoguanine Glycosylase and 8-Hydroxyl-2-deoxyguanosine using spectrophotometer at 450 nm wavelength.
RESULTS:
There was a significant (
CONCLUSION:
On the basis of this study, it has been observed and established that indulging in cigarette smoking can result in oxidative stress and DNA damage.
Keywords
Introduction
The most significant risk factor for certain malignancies is universally accepted to be cigarette smoke [1]. Numerous oxidants and free radicals that cause oxidative damage are present in cigarette smoke along with thousands of other chemicals, heavy metals, and compounds. In cultured cells, cigarette smoke damages DNA by oxidative stress either directly or by producing reactive oxygen species [1]. It has been reported that exposure to cigarette smoke significantly reduces the regenerative potential of adipose derived stem cells [2], which consequently leads to deranged morphology of red blood cells (RBC), which results in reduced oxygen carrying capacity of the blood [3]. Cigarettes are lit and inhaled into the mouth and lungs, typically via a cellulose acetate filter [4]. The primary cause of smoking-related disorders is nicotine addiction, which causes a person to repeatedly inhale a number of toxicants present in cigarette smoke, such as nitrosamines, polycyclic aromatic hydrocarbons, volatile organic compounds, and numerous toxic heavy metals. Since DNA damage appears to be the main underlying cause of cancer [5], it has been determined that these substances are the most significant known carcinogens [6]. Included in this list of carcinogens are acrolein, formaldehyde, acrylonitrile, 1,3-butadiene, acetaldehyde, isoprene, aromatic amines, arsenic, benzene, benzo(a) pyrene, beryllium, cadmium, chromium, ethylene oxide, nickel polonium-210, polycyclic aromatic hydrocarbons (PAH) [7]. Over 7,000 compounds have been found in cigarette smoke, at least 250 of which are known to have particular toxicological qualities, 69 of which are known to cause cancer and are hazardous [8].
While specific DNA repair systems effectively repair the majority of DNA damage brought on by reactive oxygen species, chemicals, radiation, and other harmful stimuli, persistence and accumulation of DNA damage can happen depending on the extent of the damage and the ability of DNA repair mechanisms [9]. Double-strand breaks (DSBs), one of several forms of DNA damage, are the most severe and challenging to precisely repair [10]. Genomic instability is caused by mutagenesis and chromosomal rearrangements brought on by accumulated DNA damage [11]. The most often found and researched DNA lesion in nuclear and mitochondrial DNA is 8-hydroxyl-2-deoxyguanosine (8-OHdG), an oxidized nucleoside of DNA [1]. 8-OHdG is secreted in the urine as a result of DNA repair. Numerous studies have suggested that urine 8-OHdG may be a risk factor for diabetes, cancer, and atherosclerosis in addition to being a biomarker of broad cellular oxidative stress [12]. Because it can be quantitated with great sensitivity, 8-hydroxyl-2-deoxyguanosine (8-OHdG), a prominent by-product of hydroxyl radical attack on DNA, is regarded as a valid biomarker for oxidative damage [13]. Additionally, multiple studies demonstrate that 8-OHdG is an important biomarker related to carcinogenesis [14]. A cell uses a variety of procedures known as DNA repair to detect and repair damage to the DNA molecules that encode its genome [1]. 8-oxyguanine glycosylase, is one of the most prevalent DNA repair indicators [9]. OGG1 is a glycosylase with two distinct functions that can both cleave the mutagenic lesion’s glycosidic bond and break the DNA strand [15]. Numerous variables, such as the kind of cell, the cell’s age, and the extracellular environment, affect the speed of DNA repair. A cell that has sustained significant DNA damage or that is unable to repair DNA damage successfully can enter one of three states: senescence, an irreversible condition of dormancy; apoptosis, or programmed cell death; or uncontrolled cell proliferation, which can result in the growth of a malignant tumor [16]. For a cell’s genome to be intact and, consequently, for that cell to operate normally, DNA repair is essential. Numerous genes that were first implicated in determining lifespan are now known to play a role in DNA damage repair and protection [17]. This study was conducted in South-West, Nigeria, to examine urinary OGG1 and 8-OHdG levels in cigarette and non-cigarette smokers.
Materials and methods
Study area
This study was carried out in the south-west states of Nigeria, including Ondo, Ekiti, Osun, Oyo, Ogun, and Lagos State. Although there are several different Yoruba dialects spoken by the locals, South Western Nigeria is known to be the geopolitically zone in Nigeria with the highest level of educational advancement. The weather is the same throughout the year in all of the south-west states of Nigeria.
Study population
This study was done on a population of 250 people who exclusively smoke cigarettes and 200 people who have never smoked cigarettes or any other kind of tobacco before. All the six south-west states of Nigeria were used to find study participants for this investigation.
Inclusion criteria
The following criteria were met to include subjects in this study: Male and female participants in this study
had to have smoked cigarettes for at least five years and ranged in age from 25 to
65 years and above. Those who agreed
to take part in the study. As
non-smokers, men and women of similar ages who are neither tobacco users or smokers
and have no history of renal disorders, were enlisted.
Exclusion criteria
The following conditions precluded participants from participating in this study: At the time of sample
collection, subjects who had not smoked cigarettes for up to five (5) years were not
included in the study. Subjects that
had a history of any type of renal illness. Subjects who declined to participate
and subjects who are younger than 25
years old.
Questionnaire
Each participant completed a well-structured questionnaire that focused on their biographical information and sociodemographic traits.
Ethical consideration
The Ethics and Research Committees of the Ministries of Health of Ado Ekiti, Ekiti State; Akure, Ondo State; Osogbo, Osun State; and Ibadan, Oyo State with the approval no MOH/EKHREC/EA/P/11; OW/380/VOL.CX/150; OSHREC/PRS/569T/181 and AD13/479/4046B respectively, provided the protocol for this study.
Specimen collection
Prior to the collection of the specimens, a questionnaire was first given to smokers and non-smokers. Early in the morning, a fresh urine sample of 10 ml was collected from cigarette smokers (test) in motor parks, tricycle parks, motorcycle (Okada) parks, as well as from non-smokers in their various homes, and transported to the departments of medical laboratory science, Afe Babalola University, Ado Ekiti; Department of Medical Laboratory Science at Achievers University, Owo, and Department of Biomedical Laboratory Science, College of Medicine, University of Ibadan where the samples were processed and analyzed. Each subject’s urine sample was stored in sterile Eppendorf tubes and kept at -20°C, where it was utilized for the OGG1 and 8-OHdG tests at the Medical Laboratory Science department of the Faculty of Basic Medical Sciences at the University of Lagos in Lagos State, Nigeria.
1. 8-OHdG determination
Test principle: This kit’s microtiter plate has been pre-coated with an 8-OHdG-specific antibody. A biotin-conjugated antibody specific for 8-OHdG was then added to the appropriate microtiter plate wells containing standards and samples. Then each microplate well received an addition of Avidin conjugated to Horseradish Peroxidase (HRP) and was incubated. Only the wells containing 8-OHdG, biotin-conjugated antibody, and enzyme-conjugated Avidin showed a change in color after the TMB substrate solution was added. Sulphuric acid solution was added to stop the enzyme-substrate reaction, and the color change was measured spectrophotometrically at a wavelength of 450 nm+10 nm. In order to determine the concentration of 8-OHdG in the samples, the O.D. of the samples was compared to the standard curve.
Procedure
The diluted standard, blank, and sample wells were identified. A total of 7 standard wells and 1 blank well were prepared. The relevant wells were filled with dilutions of the standard, blank, and samples totaling around 100 L each before the plate sealer was applied. After that, it was incubated at 37°C for two hours. Each well received roughly 100 mL of detection reagent B working solution after the liquid had been withdrawn but not washed away. After the wells were sealed with the Plate Sealer, they were incubated for 30 minutes at 37°C. As in the fourth stage, the aspiration/wash procedure was done five times. The wells were then filled with approximately 90 liters of substrate solution, capped with fresh plate sealer, and incubated for 20 minutes at 37°C (and protected from light). The substrate solution was added, which caused the liquid to turn blue. About 50 liters of stop solution were added to each well of this combination, turning it yellow. The liquid was then blended by tapping the plate’s sides. Gently tapping the Plate produced thorough mixing. The bubbles on the liquid’s surface as well as fingerprints and water drops on the plate’s bottom were eliminated. The plate was read using a spectrophotometer at 450 nm wavelength [18].
OGG1 determination
Test principle: An OGG1-specific antibody was pre-coated on the microtiter plate included in this kit. An OGG1-specific biotin-conjugated antibody was applied to the appropriate microtiter plate wells before standards and samples. Following that, each microplate well received a dose of Avidin conjugated to Horseradish Peroxidase (HRP), which was then incubated. Only the wells containing the OGG1 biotin-conjugated antibody and enzyme-conjugated Avidin changed color after the addition of the TMB substrate solution. Sulphuric acid solution was added to stop the enzyme-substrate reaction, and the color change was determined spectrophotometrically at a wavelength of 450 nm+10 nm. The O.D. of the samples was then compared to the standard curve to determine the concentration of OGG1 in the samples.
Procedure
The wells for the diluted standard, blank, and sample were identified. A total of seven standard wells and one blank well were prepared. The proper wells were filled with dilutions of the standard, blank, and samples totaling around 100 L each before the plate sealer was applied. Then, it was incubated at 37°C for two hours. Each well’s liquid was drained (not washed). Each well received roughly 100 L of the Detection Reagent B working solution. Following plate sealing, the wells were incubated for 30 minutes at 37°C. As in the fourth stage, the aspiration/wash procedure was done five times. Each well received roughly 90 L of substrate solution. The wells were sealed with fresh Plate Sealer, and they were then heated to 37°C for 20 minutes (and protected from light). The substrate solution was added, which caused the liquid to turn blue. Each well received approximately 50 mL of Stop Solution, which caused the liquid to turn yellow. The liquid was blended by tapping the plate’s edges. Gently tapping the Plate produced thorough mixing. The bubbles on the liquid’s surface as well as fingerprints and water drops on the plate’s bottom were eliminated. The plate was read using a spectrophotometer at 450 nm wavelength [19].
Statistical analysis
All of the data were analyzed using the statistical program Statistical Analysis for
Social Sciences (SPSS) version 24. To compare the means of the various analytes at
Results
Table 1. About 450 subjects were recruited for this study, out of which 250 were cigarette smokers while 200 were non-cigarette smokers. Out of the 450 subjects recruited, 385 were males consisting of 223 of male cigarette smokers, and 162 of non-cigarette smokers. Whereas 65 females were recruited, out of which 27 were cigarette smokers and 38 were non-cigarette smokers.
Frequency of gender among the cigarette and non-cigarette smokers
Frequency of gender among the cigarette and non-cigarette smokers
Table 2. Among the 250 cigarette smokers, 25–34 years age group recorded the highest prevalence (39%) of smokers followed by 35–44 years (38%), 45–54 years (11%), 55–64 years (7%) and 65 years and above (4.8%). Among the 200 non–cigarette smokers, 25–34 years age group recorded the highest prevalence of non–smokers (62%), followed by 35–44 years (25%), 45–54 years (7%), 55–64 years (3%) and 65 years and above (3%).
Frequency of age groups of cigarette and non-cigarette smokers
Table 3. Out of the 250 cigarette smokers recruited for this study, 80(32%) usually smoke 6–10 cigarette sticks in a day which was the highest followed by 68(27%) usually smoke≤5 cigarette sticks in a day; 58(23%) usually smoke 11–15 cigarette sticks in a day; 27(11%) usually smoke 16–20 cigarette sticks in a day; 8(3%) usually smoke≥30 cigarette sticks in a day; 5(2%) usually smoke 21–25 cigarette sticks in a day while 4(2%) usually smoke 26–30 cigarette sticks in a day being the lowest.
Frequency of number of cigarette sticks smoked per day among cigarette smokers
Table 4. There was a significant
(
Relationship between 8-OHdG and OGG1 parameters of cigarette and non-cigarette smokers.
*Significant at
Table 5. Urine 8-OHdG of male and female (344.64±93.67 vs 355.71±65.10) and OGG1 (219.50±59.15 vs 230.71±63.04) measured levels of cigarette smokers were slightly reduced in male when compared to female.
Comparison of measured parameters (8-OHdG and OGG1) between male and female sex group of cigarette smokers.
*Significant at
Table 6. the measured Urinary 8-OHdG of male and female (219.99±66.83 vs 239.04±74.04) level of non-cigarette smokers were found to be reduced in male when compared to female while the urinary OGG1 (98.83±59.33 vs 90.23±39.61) level were found to be slightly raised in male when compared to female.
Comparison of measured Urinary 8-OHdG and OGG1 parameters between male and female sex group of non-cigarette smokers
*Significant at
OGG1 and 8-OHdG were positively and significantly correlated (0.248*, 0.00) (Table 7.0).
Correlation among measured 8-OHdG and OGG1 of cigarette smokers
*Correlation is significant at the 0.05 level (2-tailed).
Table 8. Positive correlation was seen between 8-OHdG vs OGG1.
Correlation among measured 8-OHdG and OGG1 of non-cigarette smokers
*Correlation is significant at the 0.05 level (2-tailed).
Figure 1. There was a significant increase in the biochemical analysis of OGG1 and 8-OHdG among the cigarette smokers when compared with that of non-cigarette smokers.
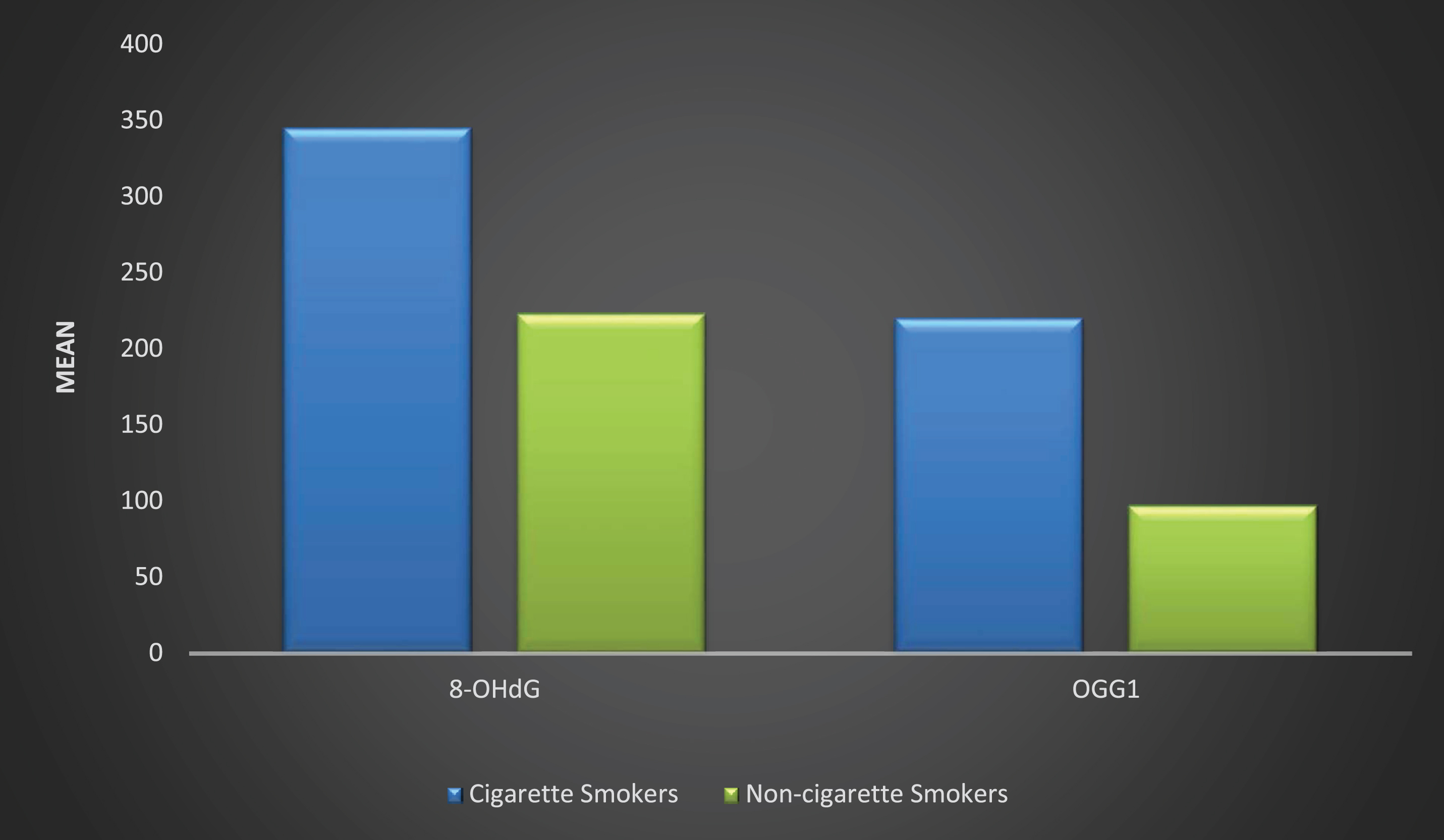
Relationship of urinary biochemical analysis of OGG1 and 8-OHdG between cigarette and non-cigarette smokers.
A total of 450 subjects from south west Nigeria were recruited for this study, out of which 250 were cigarette smokers while 200 were non-cigarette smokers. About 385 male subjects were recruited, of which 223 were the cigarette smokers while 162 were non-cigarette smokers. The numbers of female subjects recruited for this study were 65 of which 27 were cigarette smokers while 38 were non-cigarette smokers. This observation is in agreement with WHO [20] that reported that numbers of males involved in cigarette smoking doubles that of females in south-west Nigeria. The ages of subjects ranged from 25 to 65 years and above. Among the 250 cigarette smokers recruited, subjects between the ages of 25–34 years old recorded the highest prevalence (39%) of smokers followed by 35–44years (38%), 45–54years (11%), 55–64 years (7%) and the least prevalence recorded for this group were subjects with ages of 65years and above. This finding is in agreement with Fact Sheet [21] that smoking prevalence is highest among young adults between the ages of 20 to 35 years and continues to be lowest among adults that are above 60 years. This trend shows that majority of the subjects started smoking at a very younger age and they became addicted to it. This can also be caused as a result of peer group influence. The number of cigarette sticks a cigarette smoker usually smoke per day varies widely with 80(32%) of the subjects smoking 6-10 cigarette sticks in a day which was the highest followed by 68(27%) smoking≤5 cigarette sticks in a day; 58(23%) smoking 11-15 cigarette sticks in a day; 27(11%) smoking 16-20 cigarette sticks in a day; 8(3%) smoking≥30 cigarette sticks in a day; 5(2%) smoking 21–25 cigarette sticks in a day while 4(2%) usually smoke 26–30 cigarette sticks in a day being the lowest. This finding is in agreement with a study carried out by Inyang et al. [22] where it was observed that subjects who are involved in smoking 6–10 cigarette sticks per day were 38(32.2%) and subjects involved in smoking 11–15 cigarette stick per day were 32(27.1%). According to Arcavi and Benowitz [23], the adverse effects of cigarette smoke on the body system depends on the number of years an individual has been smoking and the number of cigarettes sticks an individual can consume in a day.
8-OHdG is an oxidized derivative of deoxyguanosine, the urinary excretion of 8-oxoGua is an important biomarker of oxidative stress and may predict cancer risk and diseases such as cancers that are associated with ongoing oxidative stress, inflammation, and tissue turnover. [24].
Urinary 8-Hydroxyl-2-deoxyguanosine (8-OHdG) in cigarette smokers was highly significant
(
DNA repair is a collection of processes by which a cell identifies and corrects damage to
the DNA molecules that encode its genome [27].
Urinary OGG1 is a good biomarker which plays an important role in repairing oxidative DNA
damage induced by chemical agents, such as tobacco. The urinary OGG1 level was significantly
(
Conclusion
On the basis of this study, it has been observed and established that indulging in cigarette smoking can result in oxidative stress and DNA damage.
Conflict of interest
The authors have no conflict of interest to report.
