Abstract
Introduction
Radiofrequency ablation (RFA) is an important and minimal-invasive treatment option for hepatic tumors and metastases [1–7]. It belongs to the energy-based ablation modalities and relies on proper needle electrode placement into the tumor via an open, laparoscopic or percutaneous approach. The electrode delivers a high frequency alternating current into the surrounding tissue resulting in tissue heating. As tissue temperature exceeds 55°C, cell death occurs leading to coagulation necrosis surrounding the probe. The intended ablation zone should include the entire tumor volume including a safety margin of 5 to 10 mm to ensure complete ablation.
Benign periablational enhancement (BPE) can be visualized immediately after thermal ablation as hyperemic rim surrounding the ablated tissue on CT or MRI follow-up imaging [8]. Pathologic examinations showed that BPE is an inflammatory response to thermal ablation with initial reactive hyperaemia and subsequent fibrosis/giant cell reaction [9]. This induced hyperaemia may physically cause significant heat sink effects in the proximity of the ablation zone may limiting the final extent of the thermal destruction.
Heat sink effects are a major problem in thermal ablation procedures as they can cause incomplete tumor ablation and thus limit curative treatment approaches. They are also accused to account for the high post-ablation recurrence rates reported of up to 36.5 percent [4, 10–14].
Hepatic RFA has been successfully combined with arterial and portal-venous blood flow reduction/interruption resulting in improved patient survival and reduced tumor recurrence [15–17].
Data on the type of blood supply of the benign periablational enhancement zone, whether it is an arterial, portal-venous or mixed supply, is insufficient. Therefore, the aim of this study was to dynamically characterize the type of blood supply of the periablational enhancement zone immediately after hepatic radiofrequency ablation (RFA) using continuous CT-perfusion imaging.
Materials and methods
We utilized an in-vivo porcine liver model - a detailed description of the applied experimental setup has previously been published [18]. All principles of laboratory animal care according to the guidelines of the European Societies of Laboratory Animal Sciences were followed throughout the entire experiments. The regional office for health and social welfare approved the study.
In-vivo porcine liver model
The study was performed in 5 healthy domestic pigs aged 148±16 days with an average body weight of 69 kg±2 kg. Intrahepatic multipolar RFA was performed using an open approach under general anesthesia. Three internally cooled bipolar electrodes (CelonProSurge 150-T30, Olympus Surgical Technologies Europe, Hamburg, Germany) and a corresponding generator (CelonPower System, Olympus Winter & Ibe GmbH, Teltow, Germany) were utilized. The probes were introduced into the liver strictly parallel with a constant distance of 15 mm or 20 mm using CT-guidance. Every lesion in this study was ablated with a power of 90 W and a target energy application of 50 kJ. The probe’s tips were placed centrally in one of the four liver lobes. Every ablation was performed during physiological liver perfusion.
Perfusion CT
Immediately after every RF-ablation, perfusion CT (pCT) of the liver was acquired. We used an 80-slice MDCT scanner (Aquilion PRIME, Toshiba Medical Systems, Otawara, Japan) and the following CT parameters: 100 kV, 200 mA, 0.35 s rotation time, mean mAs = 108. Before acquisition, 40 ml of Iomeprol in a concentration of 400 mg/ml were automatically injected with an injection speed of 5 ml/s (Imeron® 400, Bracco Imaging, Milan, Italy). In total, a post-ablation pCT consisted of 40 single and consecutive scans following a standardized time protocol. Shuttle mode for constant bidirectional CT table movement was used to cover a craniocaudal length of 16 cm.
Evaluation and analysis
The acquired pCT datasets were loaded into the 4D CT Body Perfusion tool of the Vitrea®-Software (Vital Images, Minnetonka, USA). In order to generate time-density-curves of the arterial and the portal-venous perfusion, circular ROIs were drawn in the abdominal aorta and in the central portal vein. A freehand ROI encompassing the complete benign periablational enhancement zone surrounding a single ablation zone was drawn to generate a time-density-curve of the ablation’s rim perfusion. Time to peak perfusion of all three resulting curves was recorded in order to calculate the time interval between arterial and BPE-peak as well as the interval between portal-venous and BPE-peak (Fig. 1). Furthermore, the maximum diameter of the periablational enhancement zone in each ablation was documented.
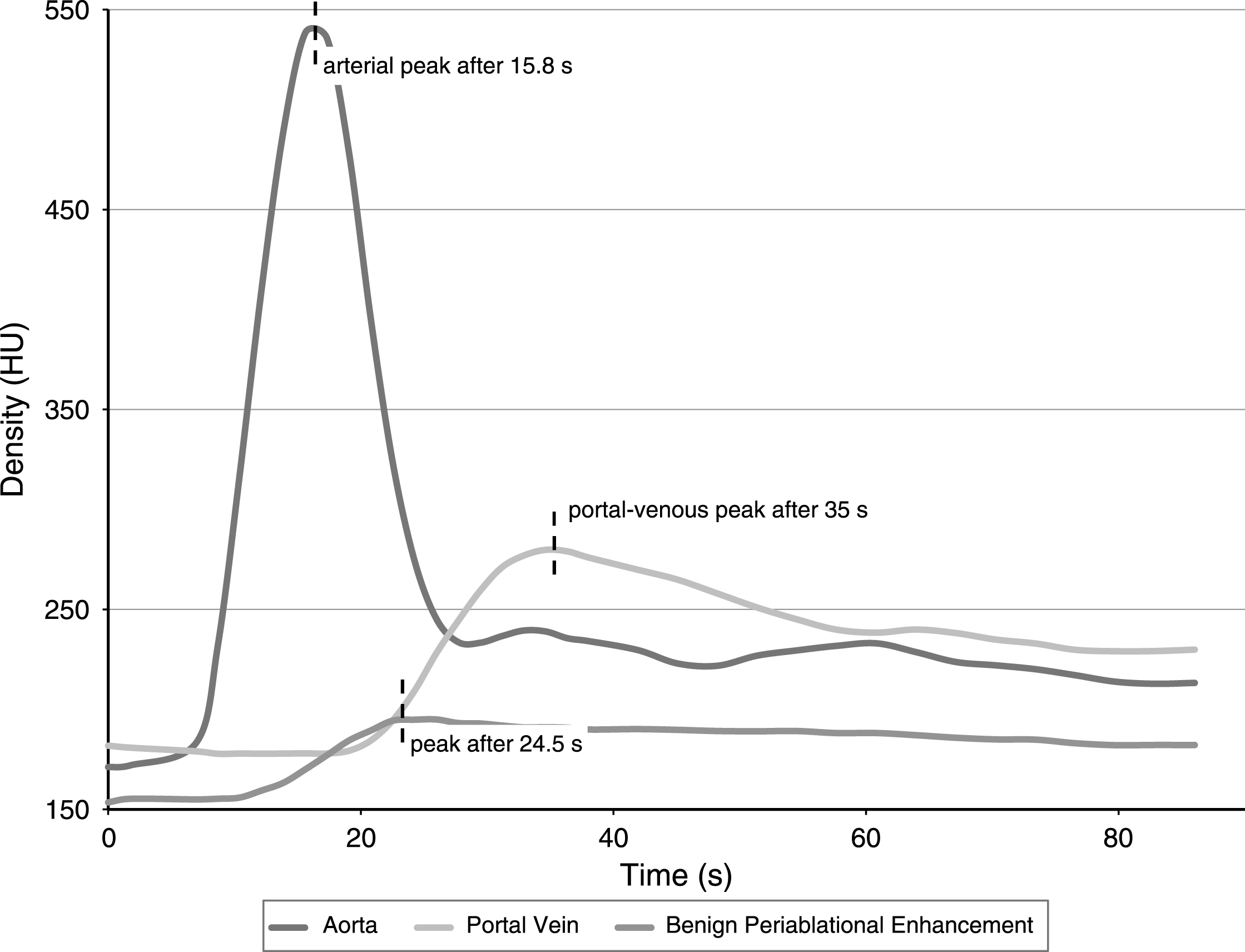
Exemplary time-density-curve for one single ablation zone: The peak of the benign periablational enhancement occurred 8.7 seconds after the peak perfusion in the abdominal aorta and 10.5 seconds before the peak perfusion in the central portal vein.
SPSS was used for data analysis (IBM Corp. Released 2013. IBM SPSS Statistics for Macintosh, Version 22.0. Armonk, NY: IBM Corp.). Descriptive statistical analysis included the calculation of mean values and corresponding standard deviations.
We conducted ten RF-ablations in 5 animals. Benign periablational enhancement could be detected in eight ablations on immediate post-ablation pCT as hyperdense and circular rim surrounding the hypodense ablation zone (Fig. 2). The remaining two ablations showed diffuse intra-lesional hemorrhage on pCT from that a periablational enhancement zone could not explicitly be differentiated. Thus, these two lesions were excluded from further analysis.
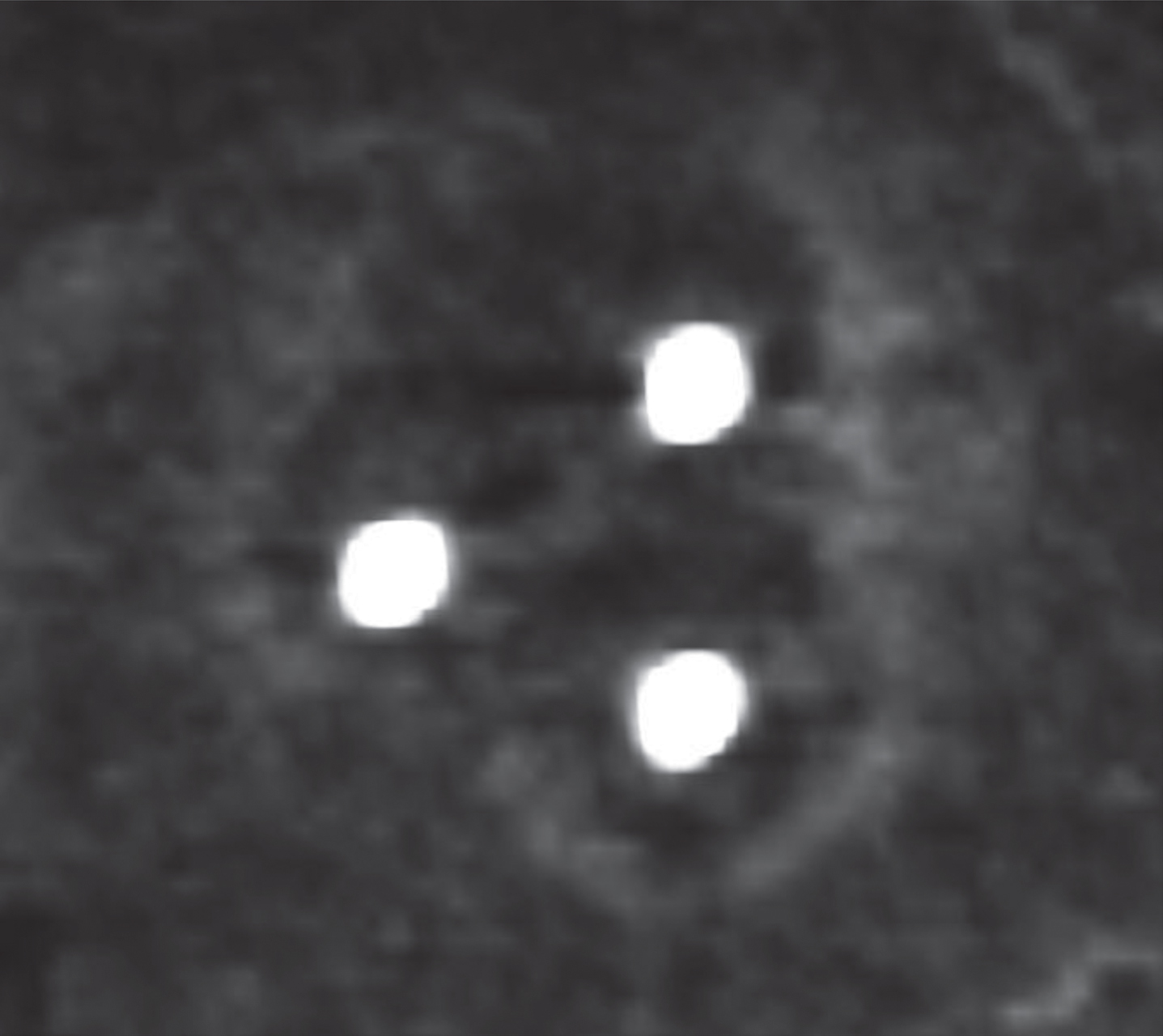
Immediate post-ablation perfusion CT shows the benign periablational enhancement surrounding the hypodense ablation zone.
Benign periablational enhancement rim showed a mean maximum diameter of 4.3±0.9 mm (n = 8 ablations). Figure 1 exemplarily depicts a time-density-curve for one ablation zone. In all eight ablations, the generated time-density-curves showed that benign periablational enhancement could be visualized after arterial perfusion and before portal-venous perfusion started. After intravenous contrast injection, the peak aortal density was observed after 18.7±5.3 s (mean±standard deviation), the peak BPE density after 26.8±5.0 s and the peak portal density after 43.7±8.0 s (N = 8). The peak of the benign periablational enhancement arose in mean 8.1±1.6 s after the peak perfusion in the abdominal aorta and 16.9±5.5 s before the peak perfusion in the central portal vein. We did not observe any ablation zone that showed peak perfusion of the periablational enhancement zone after portal-venous perfusion.
The applied study design allowed for the investigation of the benign periablational enhancement’s type of blood supply using immediate postablational perfusion CT. Our results suggest an arterial and not a portalvenous hyperperfusion of the periablational rim following multipolar RFA of the liver.
Benign peripheral enhancement can be visualized on contrast-enhanced ultrasonographic, CT- or MR-imaging immediately after thermal ablation procedures such as RFA, laser ablation and microwave ablation [9]. It is caused by vasodilation in the context of an instant inflammatory response to thermal damage resulting in reactive hyperaemia [8, 19]. This reaction can raise significant heat sink effects in the proximity of the ablation zone already during thermal ablation, although this hypothesis has never been proven and is certainly hard to verify. Peri-procedural heat sink effects are blamed for the high local recurrence rates after thermal ablation procedures [12–14, 21]. To avoid or reduce these heat sink effects thermal ablation procedures have been successfully combined with transcatheter arterial chemoembolization and selective balloon occlusion of hepatic vessels [15–17, 23]. Our results support the approach of temporarily interrupting the arterial blood flow during thermal procedures of the liver in order to avoid reduced heat distribution in the proximity of the targeted tumor volume.
This study has certain limitations. Although the results of this study were performed using a porcine liver model, we think that our results can be transferred to humans, since porcine animal models have broadly and successfully been used in translational research [24–26]. The use of intravenous agents for general anesthesia denotes a further limitation of this study as they may have altered hepatic perfusion parameters. We used ketamine and xylazine for general anesthesia. Both agents are widely used in animal experiments and have proven to marginally influence hepatic perfusion and metabolism [27–29].
In conclusion, benign periablational enhancement after RFA is a result of arterial and not portalvenous hyperperfusion. It is a potential source of periablational cooling effects that could lead to incomplete tumor ablation, early relapse and limitations in achievable lesion size. Thus, interrupting the arterial blood flow using balloon occlusion or transarterial chemoembolization during RFA is a promising approach in order to reduce heat sink effects especially in the boundary area when ablating larger tumor volumes.
