Abstract
BACKGROUND: The albumin-bilirubin (ALBI) score is a novel indicator of liver function. Some studies showed that the ALBI score was a predictive marker for the prognosis and efficacy of drug therapy in malignancies. We aimed to assess the predicted role of ALBI score in the sensitivity to therapy with trastuzumab in patients with human epidermal growth factor receptor 2 (HER2) positive breast cancer (BC). The clinical data of 226 HER2-positive BC patients at the Harbin Medical University Cancer Hospital from January 2017 and December 2018 were retrospectively collected. The ALBI score was calculated with serum albumin and bilirubin before diagnosis. The associations between ALBI score and trastuzumab resistance were analyzed by logistic regression analyses. The patients with trastuzumab resistance had higher ALBI scores compared with the patients without trastuzumab resistance. Moreover, there were weak correlations between the ALBI score and lymph node status (
Introduction
Breast cancer (BC) is the most frequent malignant tumor and also causes the majority of cancer-related deaths among women [1]. BC can be classified into several subtypes based on molecular characteristics; about 15–20% are subtyped as human epidermal growth factor receptor 2 (HER2) positive BC [2]. HER2-positive BC is more likely to have aggressive behavior and shorter overall survival (OS) before the launch of trastuzumab (a humanized anti-HER2 monoclonal antibody) [3, 4]. Although trastuzumab has been successful in improving outcomes, approximately 15% of women diagnosed with early HER2-overexpressing BC show primary resistance to trastuzumab [5, 6, 7]. Unfortunately, HER2-positive BC patients with primary trastuzumab resistance (PTR) have a short survival benefit, which accounts for a high rate of death [8]. Therefore, it is significant to find clinically useful biomarkers that can identify which of the patients are resistant to trastuzumab, which may be helpful to improve the survival of BC patients.
Pre-treatment liver function and pre-existing liver disease have been linked to the efficacy of chemotherapy and survival outcomes in cancer patients [9, 10]. The tools to evaluate liver function and the severity of liver disease include the model of end-stage liver disease (MELD), fibrosis-4 index (FIB-4), aspartate aminotransferase to platelet count ratio index (ARPI), platelet-albumin-bilirubin (PALBI) score, and albumin-bilirubin (ALBI) score [11, 12, 13]. Recently, the ALBI score has been widely used to predict the prognosis and the efficacy of drug therapy in various solid malignancies other than hepatocellular carcinomas, such as lung cancer [14], gastric cancer [15, 16], and colorectal cancer [17, 18]. Matsukane et al. reported that a high ALBI score at pre-treatment had shorter progression-free survival (PFS) and OS in patients with non-small cell lung cancer treated with immune checkpoint inhibitors [19]. Similarly, another study found that the high ALBI score group was associated with the failure of postoperative adjuvant S-1 (an oral fluoropyrimidine) monotherapy and caused shorter disease-free survival [16]. The ALBI score was initially developed to measure liver dysfunction in patients with hepatocellular carcinoma (HCC) [20]. The ALBI score is easy to calculate and more objective than other methods because it is only calculated with serum albumin and bilirubin [20]. To the best of our knowledge, there is no report showing the impact of the ALBI score on the resistance to trastuzumab treatment in HER2-positive BC. Therefore, the ALBI score is an attractive candidate as a new predictive indicator of the sensitivity to therapy with trastuzumab in patients with HER2-positive BC.
This research aimed to analyze whether the ALBI score predicted the resistance to trastuzumab treatment in patients with HER2-positive BC.
Materials and methods
Patient characteristics and specimens
HER2 positivity was defined as immunohistochemistry (IHC) 3+, or IHC (2+) and fluorescence in situ hybridization (FISH) positivity. The inclusion criteria for this study were as follows: (1) female patients aged at diagnosis were no less than 18 years; (2) all patients were pathologically confirmed to have HER2-positive BC; (3) the patients had complete clinical and follow-up data; and (4) patients had undergone surgery. Exclusion criteria included a history of malignancy, incomplete clinical data, and loss of follow-up. Follow-up was conducted through outpatient tracking and telephone. The last follow-up date was October 31, 2022, and the median follow-up time was 36 months.
This study was approved by the Institutional Ethics Review Board of Harbin Medical University Cancer Hospital (KY2022-10). Because of the retrospective nature of the research, informed consent was waived.
Treatment method
All patients received trastuzumab until disease progression or intolerable adverse reactions. Trastuzumab was given with a loading dose of 8 mg per kilogram of body weight, followed by 6 mg per kilogram every 3 weeks. Patients were treated with neoadjuvant chemotherapy or adjuvant chemotherapy, mainly including anthracycline and/or taxane regimens.
Data collection
Medical records were reviewed to collect the patients’ age at baseline, medical history, menstrual status, laboratory test results, and pathologic results such as tumor size, lymph node metastasis, histopathological type, proliferation index (Ki-67) expression, estrogen receptor (ER) status, and progesterone receptor (PR) status. ER, PR, and Ki-67 expressions were determined using IHC. ER and PR were evaluated as positive if there were at least 1% positive invasive tumor nuclei in the samples. Pre-treatment blood tests, including serum concentrations of albumin and bilirubin, were performed within a week before treatment. The ALBI score was calculated as follows: [(log
Study endpoint
PTR was the primary endpoint and was defined as a recurrence detected during adjuvant trastuzumab or within 12 months of their last (neo) adjuvant trastuzumab [22]. The secondary efficacy endpoint was PFS, defined as the time from the time of diagnosis to the first occurrence of any event, including local, regional, or distant recurrence of ipsilateral or contralateral breast cancer.
Statistical analysis
The descriptive statistics were reported as means with SDs or medians (interquartile range) for continuous variables, and percentages of the number for categorical variables. Student’s
Results
A total of 226 patients with HER2-positive BC who received treatment with trastuzumab were reviewed in this study. The median follow-up was 30 months (3 to 77 months). Patients ranged in age from 27 to 73, with a median of 51 years. In total, 103 (45.6%) patients were pre-menopausal, and 123 (54.4%) were post-menopausal. However, 30/226 (13.3%) patients exhibited PTR. We identified the optimal cut-off value of the ALBI score for PTR by ROC curve analysis. According to ROC curve analysis, the optimal cut-off value for the ALBI score was
The optimized cut-off value was determined for the ALBI score using ROC curve analysis.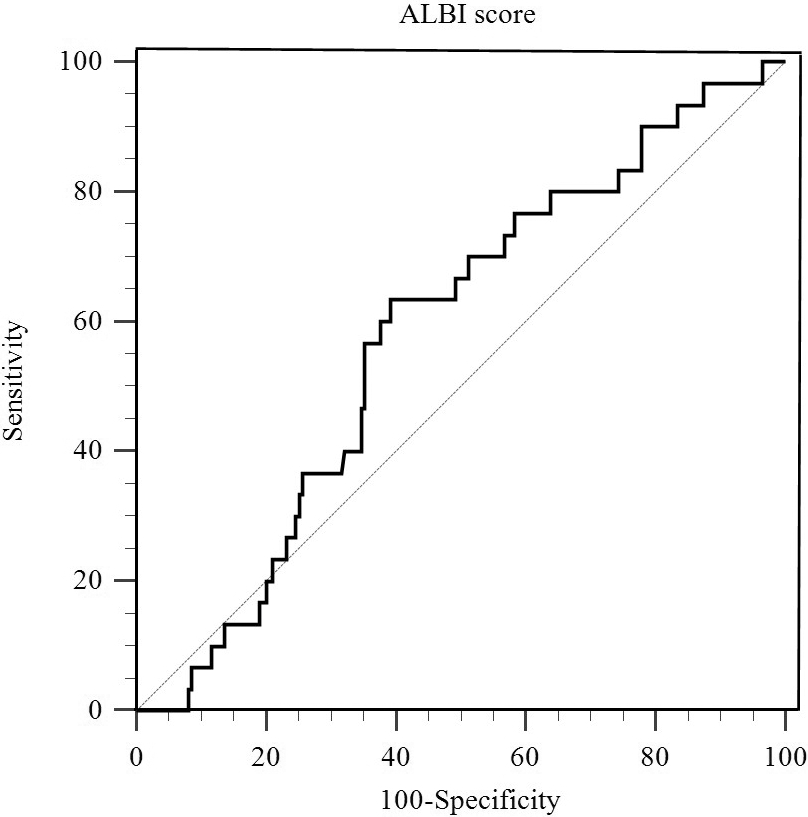
Detailed distributions of patients with HER2-positive BC according to PTR status are summarized in Table 1. The patients with PTR had higher ALBI score levels compared with the patients without PTR. There were significant differences in alanine aminotransferase (ALT), ALBI score (categorical variable), and lymph node status between the two groups. BMI (
Baseline characteristics of breast cancer patients according to the primary trastuzumab resistance status
PTR, primary trastuzumab resistance; BMI, body mass index; ALT, alanine aminotransferase; AST, aspartate aminotransferase;
Baseline clinicopathological parameters of breast cancer patients according to ALBI score levels
ALBI, albumin-bilirubin; BMI, body mass index; ALT, alanine aminotransferase; AST, aspartate aminotransferase;
The median follow-up was 36 months (interquartile range, 3–77 months), with the 3-year PFS being 81.4%. Overall, 42 (18.6%) events occurred with recurrence among 226 patients during the follow-up. There were 25 (11.1%) patients with distant metastasis: nine patients with metastases in the lung, five patients with metastases in the liver, and eleven patients with metastases in the bone. A log-rank test was applied to the Kaplan-Meier analysis to assess the prediction capacity of the ALBI score. Patients with low ALBI scores had better PFS than those with high ALBI scores (log-rank
The predictors of primary trastuzumab resistance in breast cancer patients
BMI, body mass index; ALT, alanine aminotransferase; eGFR, estimating glomerular filtration rate; ALBI, albumin-bilirubin; OR, odds ratio; CI, confidence interval.
Progression-free survival curves of subgroups divided by high and low ALBI scores.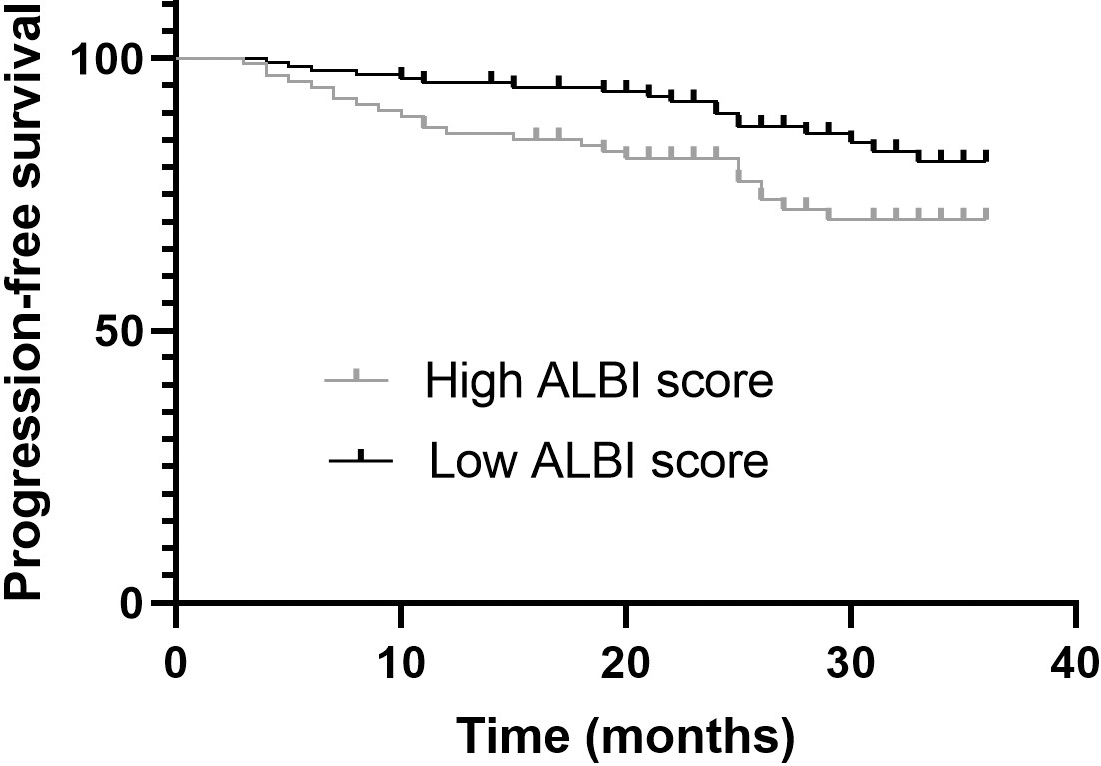
Univariate and multivariate logistic regression analyses were used to study the association between PTR and ALBI scores (Table 3). Univariate analyses revealed that the ALBI score and lymph node status had a significant correlation with PTR. BMI (
The predictors of progression-free survival in breast cancer patients
BMI, body mass index; ALT, alanine aminotransferase; eGFR, estimating glomerular filtration rate; ALBI, albumin-bilirubin; HR, hazard ratio; CI, confidence interval.
The statistical power was calculated by G*Power software when the ALBI score was used to predict trastuzumab resistance. The power of the study is 0.944 when the given significance level is 0.05, odd ratio is 2.87, and the sample size is 226.
In this study, we retrospectively investigated the impact of ALBI score on the sensitivity to therapy with trastuzumab and the prognosis in patients with HER2-positive BC. We found that a higher ALBI score was identified as an independent predictive factor for PTR and PFS in the univariate and multivariate analyses. These results indicate that the ALBI score may help us to distinguish patients with HER2-positive BC who will benefit from trastuzumab treatment.
The incidence of PTR was higher in the high ALBI score group than in the low ALBI score group in our study. This finding is similar to the results of the previous study report [16, 19]. Matsukane et al. reported that a high ALBI score at pre-treatment had a significantly shorter PFS and OS in patients with non-small cell lung cancer treated with immune checkpoint inhibitors [19]. Similarly, another study found that the high ALBI score group was associated with the failure of postoperative adjuvant S-1 (an oral fluoropyrimidine) monotherapy and caused shorter disease-free survival [16]. These findings confirm the same result: a high ALBI score was associated with poor response to therapy and a shorter PFS.
Furthermore, we have limited information on how a high ALBI score confers an increased risk of trastuzumab resistance. However, there are some possible explanations. Previous evidence has shown that altering drug transport (increasing drug efflux or decreasing drug influx) is the main reason for drug resistance [23, 24]. First, we know the main organ that metabolizes drugs is the liver. According to the ALBI score, a mathematical model for the assessment of liver function, a high score indicates liver dysfunction [25]. However, the hepatotoxic effects of trastuzumab have been reported in previous studies [26, 27, 28]. Because the liver is involved in drug metabolism, patients with liver dysfunction are more susceptible to the toxic effects of chemotherapy agents [9]. Then, the ALBI score is a combination of albumin and bilirubin values [20]. It has been shown that low albumin levels can increase trastuzumab clearance in cancer patients [29]. Albumin deficiency can increase the free drug concentration, thereby reducing its half-life and increasing its clearance, which generally causes cancer cells to have less access to the drug [30]. Impairment of liver metabolic and excretory function, as indicated by a high ALBI score, may have decreased the tolerability of drug therapy [16]. For these reasons, it is possible to explain why high ALBI scores are associated with trastuzumab sensitivity and prognosis in patients with HER2-positive BC.
However, several limitations should be mentioned in this study. First, this study is a retrospective analysis of HER2-positive BC patients who came from a single center. Second, the study population comprised only Asian participants. More studies on the multi-race population are needed. Third, the mechanisms of ALBI score involved in trastuzumab resistance were not explored, and further research is needed.
Conclusions
In conclusion, the ALBI score may serve as a potential marker of trastuzumab resistance in HER2-positive BC.
Author contributions
Conception: W-j H, J-r Y, and LZ.
Interpretation or analysis of data: W-j H, J-r Y, LZ, WW, M-l Z, and XW.
Preparation of the manuscript: WW and W-j H.
Revision for important intellectual content: R-t W and W-j H.
Supervision: R-t W.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-230077.
sj-docx-1-cbm-10.3233_CBM-230077.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-230077.docx
Footnotes
Acknowledgments
This work was supported by grants from the Natural Science Foundation of Heilongjiang (LH2021H075).
