Abstract
Bladder cancer is a neoplasm originated from bladder epithelial cells. The therapy for bladder cancer is so far not satisfactory. In this study, we examined the effects of Cordyceps militaris hot water extracts containing cordycepin on human bladder cells. Cordyceps militaris hot water extracts containing cordycepin was used to treat human T24 bladder carcinoma cells, and we found that Cordyceps militaris hot water extracts containing cordycepin decreased T24 cell survival in a dose-dependent manner, which was seemingly mediated by activation of A3 adenosine receptor and the subsequent inactivation of Akt pathways, resulting in increases in cleaved Caspase-3 and apoptosis. Overexpression of A3 adenosine receptor in T24 cells mimicked the effects of Cordyceps militaris hot water extracts, while A3 adenosine receptor depletion abolished the effects of Cordyceps militaris hot water extracts containing cordycepin. Together, these data suggest that Cordyceps militaris hot water extracts containing cordycepin may be a promising treatment for bladder cancer via A3 adenosine receptor activation.
Introduction
Adenosine receptor plays a key role in the regulation of cell survival and cell death. Four distinct subtypes of adenosine receptors have been identified in human, and they are A1, A2A, A2B, and A3. 1 Activation of A3 adenosine receptor (A3AR) has been shown to inhibit cancer growth,2 –5 which has been explained by different mechanism. Activation of A3AR induces suppression of downstream protein kinase, Akt, in some types of cancers.5,6 Inhibition of Akt leads to the inactivation of Lef/Tcf and β-catenin-responsive c-myc and cyclin D1, which in turn suppresses the cell survival. 7 In line with these findings, functional A3AR has been shown to inhibit Akt to enhance susceptibility of cancer cells to apoptosis. 8
Bladder cancer is a malignant tumor with high prevalence. Although patients with bladder cancer can be treated with a combination of surgery and chemotherapy, some cases appear to be very difficult to be cured. Moreover, tumor resistance against chemotherapy has been frequently found with undetermined mechanisms.9 –24
Cordycepin is the major bioactive component of Cordyceps militaris and has been reported to have anti-cancer, anti-oxidant, and anti-inflammatory effects.25,26 However, the molecular pathways of its anti-cancer effect in bladder cancer have not been studied. Therefore, in this study, we investigated whether cordycepin from C. militaris hot water extracts (cordycepin-CMHW) may have anti-cancer effects on human bladder carcinoma.
Materials and methods
Study approval
All experimental protocols were approved by the Research Bureau of Shandong University.
Cell culture and transfection
A human BC cell line T24 was purchased from APCC (American Type Culture Collection, Manassas, VA, USA) and has been widely used in BC research. T24 was generated from an 81-year-old female Caucasian 27 and was cultured in in RPMI1640 medium (Invitrogen, Carlsbad, CA, USA) supplemented with 15% fetal bovine serum (FBS; Sigma-Aldrich, St Louis, MO, USA) in a humidified chamber with 5% CO2 at 37°C. A3AR and short hairpin small interfering RNA for A3AR (shA3AR) were purchased from GeneCopoeia (Rockville, MD, USA). The transfection was performed with 50 nmol/L plasmids, using Lipofectamine 2000 (Invitrogen). The transfection efficiency was more than 95%, based on expression of a green fluorescent protein (GFP) reporter.
Preparation of cordycepin-CMHW
Cordycepin was isolated from the mycelium of C. militaris. Briefly, the seed culture of C. militaris was first prepared using Potato Dextrose Agar (PDA) (Nissui Pharmaceutical Co., Ltd., Japan) plate, after which the inoculant was transferred to 200 mL of Potato Dextrose Broth (PDB) medium (Nissui Pharmaceutical Co., Ltd.) and inoculated at 22°C on a rotary shaker incubator at 150 r/min for 7 days. Mycelium of C. militaris was then developed in a jar fermenter containing wheat-based medium consisting of 43.7% wheat, 5% yeast powder, 0.1% CaCO3, 0.05% MgSO4, 0.1% NaH2PO4, 0.05% KH2PO4 1% glucose, and 50% H2O and inoculated at 22°C for 28 days. The fermenters were dried at 50°C for 24 h, homogenized, and dissolved in water at ratio of 1:5 (w/v); the fermenters/water mixtures were then incubated at 100°C oven for 24 h (CMHW). Cordycepin amounts in CMHW were analyzed by high-performance liquid chromatography.
Quantitative polymerase chain reaction
Total RNA was extracted from resected tissue specimens or from the cultured cells, using RNeasy kit (Qiagen, Hilden, Germany). Reverse transcription quantitative polymerase chain reaction (RT-qPCR) was performed in duplicates using QuantiTect SYBR Green PCR Kit (Qiagen), with the primers designed by Qiagen. A 2−ΔΔCt method was used to analyze and quantify the transcript levels. Values of gene transcripts were first normalized against housekeeping gene α-tubulin, and then compared to the experimental controls.
Cell survival quantification
For assay of cell survival, cells were seeded into 24-well plate at 104 cells per well and subjected to a Cell-Clock Kit (Biocolor, Carrickfergus, UK), according to the instruction of the manufacturer. Cell-Clock assay is a live cell detection and measurement system that can be employed to monitor the four major phases of the mammalian cell cycle during in vitro culture. The assay uses a redox dye that is imported by cycling cells. Following dye uptake and incubation, a distinct color change occurs within cells, with particular color changes being associated with cells in the G0–G1, S, G2, and M Phases. The number of the cells as well as the ratio of cells within the different phases can be calculated using this assay supplemented by visible evidence from photomicrographs. Experiments were performed three times.
Western blot
Protein was extracted from the patient specimen or cultured cells by radioimmunoprecipitation assay (RIPA) buffer (Sigma-Aldrich) for Western Blot. The supernatants were collected after centrifugation at 12,000×g at 4°C for 20 min. Protein concentration was determined using bicinchoninic acid (BCA) protein assay, and whole lysates were mixed with 4× sodium dodecyl sulfate (SDS) loading buffer (125 mmol/L Tris–HCl, 4% SDS, 20% glycerol, 100 mmol/L dithiothreitol, and 0.2% bromophenol blue) at a ratio of 1:3. Samples were heated at 100°C for 5 min and were separated on SDS-polyacrylamide gels. The separated proteins were then transferred to a polyvinylidene difluoride (PVDF) membrane. The signals were recorded using X-ray film. Primary antibodies for Western Blot are anti-A3AR, anti-Akt, anti-cleaved caspase-3, and anti-α-tubulin (all purchased from Cell Signaling, San Jose, CA, USA). Secondary antibody is horseradish peroxidase (HRP)-conjugated anti-rabbit (Jackson Labs, Bar Harbor, ME, USA). Images shown in the figure were representative from five repeats.
Statistics
All statistical analyses were carried out using the SPSS 18.0 statistical software package. All values are depicted as mean ± standard deviation and are considered significant if p < 0.05. Two-tailed Student’s t test was used for comparisons of two groups and one-way analysis f variance (ANOVA) with a Bonferroni correction was used for comparisons of three or more groups.
Results
Cordycepin-CMHW dose-dependently decreases survival of bladder cancer cells
We examined the effects of cordycepin-CMHW on the survival of a human bladder cancer cell line T24. Thus, cordycepin-CMHW was given to cultured T24 cells at a dose of 0, 10, 30, and 90 µg/mL. The cells were then analyzed in a Cell-clock assay, showing that cordycepin-CMHW dose-dependently decreased survival of bladder cancer cells, by quantification (Figure 1(a)) and by representative images (Figure 1(b)). Moreover, the cells in the activated cell cycle (G1, S and M phases) appeared to decrease. These data suggest a cell growth inhibitory effect of cordycepin-CMHW on bladder cancer cells.

Cordycepin-CMHW dose-dependently decreases survival of bladder cancer cells. We examined the effects of cordycepin-CMHW on the survival of a human bladder cancer cell line T24. Thus, cordycepin-CMHW was given to cultured T24 cells at a dose of 0, 10, 30, and 90 µg/mL. (a–b) Cell survival was then analyzed in a Cell-clock assay, shown by (a) quantification and (b) representative images (N = 5).
Cordycepin-CMHW activates A3AR to suppress Akt, resulting in increases in apoptosis of bladder cancer cells
Previous studies have shown that cordycepin-CMHW is a ligand for A3AR, the activation of which directly suppresses Akt and increases apoptosis in some type of cells. Thus, we examined whether this mechanism also function in bladder cancer cells. Hence, we analyzed the protein from cordycepin-CMHW-treated T24 cells by Western blot. We found that cordycepin-CMHW dose-dependently activated A3AR in T24 cells, dose-dependently suppressed total Akt, and dose-dependently increased the levels of cleaved caspase-3, a marker for apoptosis (Figure 2). These data confirm that the mechanism found in other cell type also function in bladder cancer cells, in which cordycepin-CMHW may activate A3AR to suppress Akt, resulting in increases in apoptosis of bladder cancer cells.
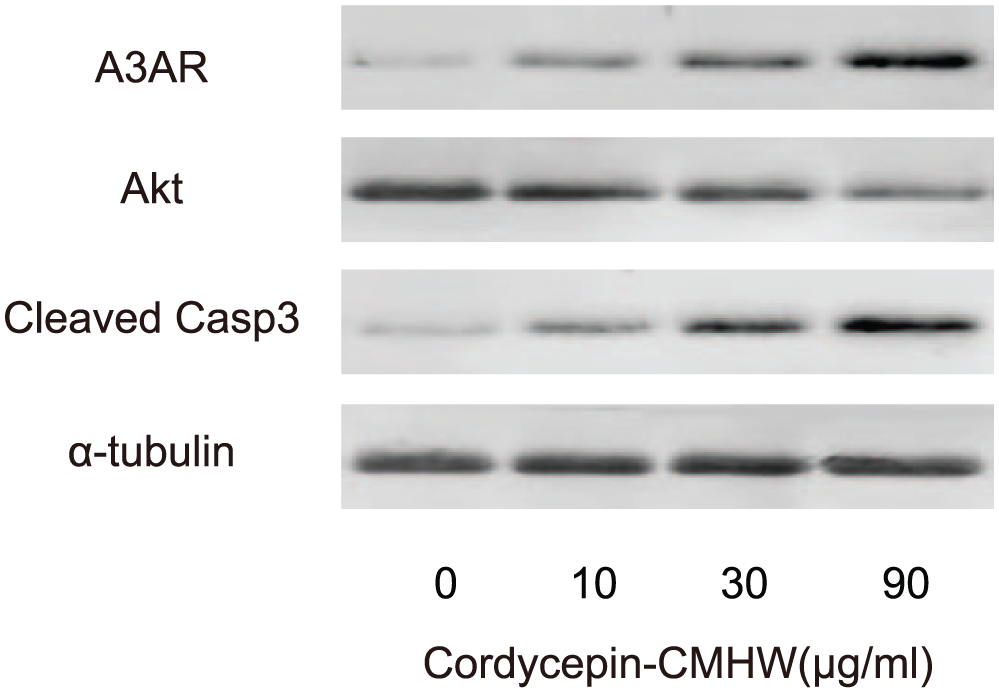
Cordycepin-CMHW activates A3AR to suppress Akt, resulting in increases in apoptosis of bladder cancer cells. We analyzed the A3AR, Akt, and cleaved caspase-3 protein from cordycepin-CMHW-treated T24 cells by Western blot, shown by representative blots (N = 5).
A3AR overexpression mimics the effects of Cordycepin-CMHW on bladder cancer cells
We then examined whether the effects of cordycepin-CMHW on the survival of bladder cancer cells were exactly through A3AR. Thus, T24 cells were transfected with either an A3AR-overexpressing plasmid or a plasmid carrying short hairpin RNA (shRNA) for A3AR. Transfection of T24 cells with a plasmid carrying a scrambled sequence was used as a control. First, the modulation of A3AR levels in transfected T24 cells was validated by RT-qPCR (Figure 3(a)) and by Western blot (Figure 3(b)). We found that overexpression of A3AR in T24 cells significantly decreased cell survival, while A3AR depletion significantly increased cell survival in the presence of 30 µg/mL cordycepin-CMHW, as a baseline challenge to the cells, shown by quantification (Figure 3(c)) and by representative images (Figure 3(d)). Moreover, the cells in the activated cell cycle (G1, S, and M phases) appeared to increase in A3AR-depleted cells and to decrease in A3AR-overexpressing cells (Figure 3(d)). These data suggest that A3AR overexpression mimics the effects of Cordycepin-CMHW on bladder cancer cells.

A3AR overexpression mimics the effects of Cordycepin-CMHW on bladder cancer cells. (a–b) T24 cells were transfected with either an A3AR-overexpressing plasmid, or a plasmid carrying shRNA for A3AR. Transfection of T24 cells with a plasmid carrying a scrambled sequence was used as a control. The modulation of A3AR levels in transfected T24 cells was validated by (a) RT-qPCR, and (b) Western blot. (c–d) Cell survival was then analyzed in a Cell-clock assay, shown by (c) quantification and by (d) representative images (N = 5).
A3AR overexpression suppresses Akt and increases apoptosis of bladder cancer cells
Next, we examined the effects of A3AR modulation on Akt and apoptosis by Western blot. We found that A3AR overexpression suppressed Akt and increased the levels of cleaved caspase-3, while A3AR depletion increased Akt and decreased the levels of cleaved caspase-3 in bladder cancer cells (Figure 4). Together, here we demonstrate that cordycepin-CMHW may be a promising treatment for bladder cancer via A3AR/Akt/Caspase-3 (Figure 5).

A3AR overexpression suppresses Akt and increases apoptosis of bladder cancer cells. We examined the effects of A3AR modulation on Akt and cleaved caspase-3 by Western blot, shown by representative blots (N = 5).

Schematic of the model. Cordycepin-CMHW activates A3AR to suppress Akt, resulting in increases in Caspase-3 and apoptosis in bladder cancer cells.
Discussion
A3AR is known to regulate cell survival and cell death. In line with this notion, A3AR has been reported to be highly expressed in many tumors. Since the activation of A3AR appears to be detrimental to tumor cell survival, possibly through Akt5 signaling, A3AR activation may be a compensation for control the outgrowth of the tumor mass.
Apoptosis has been demonstrated as a mechanism of anti-cancer activity triggers by A3AR. For example, A3AR agonists have been reported to inhibit tumor cell growth and induce cancer cell apoptosis. Here, treated with the cordycepin-CMHW, T24 cells underwent apoptosis, consistent with previous reports elsewhere.
Caspases are a family of protease enzymes, which play essential roles in the processes of apoptosis. Specifically, the initiator caspase-8 and caspase-9 motivate the executioner caspase-3 via cleavage for activation, leading to the proteolysis of poly(ADP-ribose) polymerase (PARP) and apoptosis through impairment of DNA repair. Here, we show that treatment of T24 cells with cordycepin-CMHW induced caspase-3 activation to enhance apoptosis.
The C. militaris is a critical deck for natural drugs to revitalize the various physiological processes. Cordycepin is a major bioactive component in C. militaris. Previous studies have shown that cordycepin has a potential to inhibit cell proliferation in human bladder carcinoma and found that the molecular pathways of its anti-cancer effect appear to be through the inhibition of cell proliferation and the promotion of cell apoptosis.5,6 Interestingly, those studies have shown that cordycepin binds the A3AR to activate G protein to inhibit cyclic adenosine monophosphate (cAMP) formation, to decrease glycogen synthase kinase-3beta/beta-catenin activation, and to suppress cyclin D1 and c-myc expression. Moreover, there are evidence showing that cordycepin may induce cell apoptosis via binding the DR3 receptor which consequently activates caspase-8 and caspase-3. 28 Here, we show that these mechanisms also function in bladder cancer cells. Taken together, cordycepin may be used as a natural medicine for treating bladder cancer, as it can not only control tumor cell proliferation but also induce cancer cell apoptosis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
