Abstract
BACKGROUND:
A growing body of studies have demonstrated the aberrant expression of microRNAs (miRNAs) contributes to human tumor metastasis. MicroRNA-124-3p (miR-124-3p), which is down-regulated in various cancers, has been found to be involved in several signaling pathways relevant to tumor cell migration and invasion. However, the roles of miR-124-3p in human bladder cancer remain unclear. This study aims to investigate the functional significance of miR-124-3p and to understand how it targets the integrin receptor, and thus affects the progression of human bladder cancer.
METHODS:
Clinical specimens from 36 patients and three human bladder cancer cell lines were analyzed for miR-124-3p and integrin
RESULTS:
The down-regulation of miR-124-3p and up-regulation of ITGA3 were observed in clinical specimens and bladder cancer cell lines. Overexpression of miR-124-3p or silencing ITGA3 inhibited tumor cell migration and invasion. Luciferase assay confirmed miR-124-3p directly targets ITGA3, and western blot suggested that miR-124-3p plays a crucial role in the EMT and metastasis of human bladder cancer through FAK/PI3K/AKT and FAK/Src signaling mechanism. Also, by targeting ITGA3, miR-124-3p can modulate the expression of N- and E-cadherin, and thus inhibit the EMT.
CONCLUSIONS:
By targeting ITGA3 and downstream FAK/PI3K/AKT and FAK/Src signaling pathways, miR-124-3p suppresses cell migration and invasion in bladder cancer. Our study reasonably speculates that miR-124-3p can be potentially developed as a therapeutic target and prognostic biomarker for bladder cancer.
Introduction
Bladder cancer (BC) ranks the ninth most common cancer with particularly high mortality worldwide [1, 2]. Although approximately 75% of patients newly diagnosed with BC suffer from non-muscle invasive bladder cancer (NMIBC), the recurrence rate remains high (50%–70%) within 5 years following the bladder-sparing surgery as well as intravesical chemotherapy or immunotherapy [1]. Moreover, up to 20% of the recurrence cases progress to muscle-invasive bladder cancer (MIBC) [3]. Therefore, research on BC at the molecular and genetic level is highly required to update the diagnostic approaches and therapeutic strategies. In recent decades, a large number of studies showed that the dysregulation of microRNAs (miRNAs) are deeply involved in the clinical progression of bladder cancer. MiRNA expression profile has also revealed that more than hundred various miRNAs are dysregulated in the BC tissues in comparison to the normal ones [4]. Increasingly, the bladder cancer-related miRNAs (most recent cases include miRNA-101, -150, -430, etc.) are appreciated as potential tumor biomarkers as well as promising therapeutic targets for treating human BC [5, 6, 7].
MiRNAs are a group of conserved non-coding RNAs that typically consist of 20-22 nucleotides. As negative regulators of gene expression, miRNAs bind to the 3’-untranslated region (3’UTR) of target messenger RNAs (mRNAs), leading to the cleavage of mRNAs with subsequent degradation or inhibition of protein translation [8]. Gene silencing induced by miRNAs plays a significant role in many cellular processes, such as cell differentiation, proliferation, apoptosis, tumor development and metabolism [9]. Among those cancer-related miRNAs, miR-124-3p has been widely reported as a tumor suppressor in the malignant transformation and development of human glioma, nasopharyngeal cancer, and breast cancer [10, 11, 12, 13, 14]. Moreover, the pathologically down-regulation of miR-124-3p is observed in bladder cancer tissues as well [13, 15]. Recent reports have demonstrated that miR-124-3p regulates cell proliferation and motility of bladder cancer through targeting a variety of gene products, such as cyclin-dependent kinase (CDK4) [15], Rho-associated, coiled- coil containing protein kinase 1 (ROCK1) [13], and ubiquitin-like, containing PHD and IRNG finger domains 1(UHRF1) [16].
Integrins are the major transmembrane receptors for cell adhesion to extra-cellular matrix (ECM) proteins, and integrin-mediated signaling is crucial for cell survival, proliferation, migration and invasion in normal and malignant cells [17]. Integrins are type I
This study seeks to investigate the functional significance of miR-124-3p and to identify its integrin targets as well as the downstream signaling pathways in BC cells. Our data demonstrated that the miR-124-3p was pathologically down-regulated in both bladder cancer tissues and cell lines, and that restoration of miR-124-3p suppressed migration and invasion of BC cells. Moreover, we explored the possible antitumor effects of miR-124-3p in BC through silencing ITGA3, and thus regulating the focal adhesion kinase (FAK) signaling and its downstream pathways. In the present study, we uncover a new integrin-mediated tumor-suppressive mechanism of miR-124-3p in human BC. Our study may shed light on the future investigation of miR-124-3p as an effective therapeutic target and prognostic biomarker for BC.
Materials and methods
Human clinical samples and cell lines
Thirty-six paired BC tissues and adjacent non-tumorous bladder mucosal tissues were obtained from patients undergoing radical cystectomy at The Third Xianya Hospital of Central South University, Hunan, China, from March to December in 2016. All the patients signed informed consent and the study was approved by the Ethics Committee of the hospital, and the clinical characteristics of these patients were summarized in Table 2. Tissue samples were immediately frozen in liquid nitrogen until RNA extraction. The human non-malignant cell line SV-HUC-1, human BC cell lines T24, J82 and 5637 cells were obtained from ATCC (Manassas, VA, USA) and were cultured in RPMI 1640 medium supplemented with 10% fetal bovine serum under humidified air atmosphere of 5% CO
Cell transfection
The miRNA mimics, negative control duplex with random sequence (denoted as NC) and small interfering RNA (siRNA) were all synthesized by GenePharma (Shanghai, China). Cells were transfected using Lipofetamine 2000 (Invitrogen) according to the manufacturer’s protocol. The mixture containing miRNA mimics, NC or medium (blank control group) and Lipofetamine 2000 reagent (Invitrogen, Carlsbad, CA, USA) were prepared and added directly to cells at a final oligonucleotide concentration of 50 nM in gain-of-function experiments. ITGA3 siRNA and negative control siRNA were applied in the same manner in loss-of-function experiments. The sequences are listed in Table 1.
Oligonucleotide sequences
Oligonucleotide sequences
Clinicopathological characteristic of patients with bladder cancer
Total RNA from tissue samples and cultured cells was isolated using Trizol reagent (Invitrogen) according to the manufacture’s protocol. For cDNA synthesis, 1
MTT assay
Approximately 3
Colony-formation assay
For colony-forming activity, the J82 and T24 cells post-transfection were seeded into 12-well plates at low density (100 cells per well) in complete medium. The procedure was suspended until visible colonies appeared (14 days), and then cells were stained with 0.1% crystal violet and the colonies were counted. Each treatment was performed in triplicate.
Cell apoptosis detection using flow cytometry
The transfected cells were collected by trypsinization and washed in PBS. Then cells were re-suspended at a concentration of 1
Transwell invasion assay
After 48 h of transfection, cells in each group were digested using pancreatin to single-cell suspension; cells were washed twice with serum-free medium, and counted, then cell concentration was adjusted to 1
Wound healing assays
Cells were seeded into 6-well plate, and after being transfected with the corresponding RNAs, they were allowed to grow to around 100% confluence. The cell layers were wounded by scraping with 200
Dual-luciferase reporter assay
Two oligonucleotide pairs that contained the desired miRNA target region (ITGA3 wild type) and ITGA3 mutant were designed and ordered from GenePharma (Shanghai, China). Two oligonucleotide pairs were annealed at 90
Western blotting analysis
Cells were harvested at 48 h following various treatments as described above, lysed and quantified by a BCA kit (Thermo, USA). Proteins were separated by 10% SDS-PAGE and transferred to polyvinylidene difluoride (PVDF) membranes. The membranes were then blocked with 5% non-fat milk and incubated overnight with corresponding antibodies at dilutions specified by the manufacturer’s protocol. Antibodies applied in the western blot included GADPH antibodies (1:1000, Santa Cruz) and other antibodies against FAK (1:200, Invitrogen), phospho-FAK (pY397) (1:200, Invitrogen), AKT (1:200, Invitrogen), phospho-AKT (pS473) (1:200, Invitrogen), Src (1:200, Invitrogen), phosphor-Src (pY416) (1:200, Invitrogen), PI3K (1:200, Abcam), phosphor-PI3K (pY607) (1:200, Abcam), E-cadherin (1:200, Invitrogen), N-cadherin (1:200, Invitrogen) and ITGA3 (1:200, Invitrogen). After washed in TBS for three times, the membranes were incubated for 1 h with horseradish peroxidase-conjugated goat anti-mouse secondary antibody at the 1:1000 dilution (Santa Cruz). After another three times of washing in TBS, the bound secondary antibody was detected using an enhanced chemiluminescence (ECL) system (Pierce, Biotechnology Inc., Rockford, IN, USA). Each western blot was repeated for three times in the following experiments.
Statistical analysis
Differences in measurement data were expressed as mean
Expression levels of miR-124-3p and ITGA3 in BC clinical specimens and human BC cell lines T24, J82 and 5637. (A, B) QRT-PCR showed that the expression levels of miR-124-3p and ITGA3 were significantly lower and higher in BC tissues than in normal bladder tissues. (C, D) The expression levels of miR-124-3p and ITGA3 were also analyzed in non-malignant cell line SV-HUC-1 and human BC cell lines T24, J82 and 5637. It was found that the expressions of miR-124-3p and ITGA3 were decreased and increased significantly in T24, J82 and 5637 than in non-malignant cell line SV-HUC-1. (E) The expression levels of ITGA3 in BC or normal tissues and cells were further analyzed by western blot assay. The tissue samples analyzed here are the mixed cancer tissues or adjacent tissues from 10 BC patients, and western blots were repeated for three times. *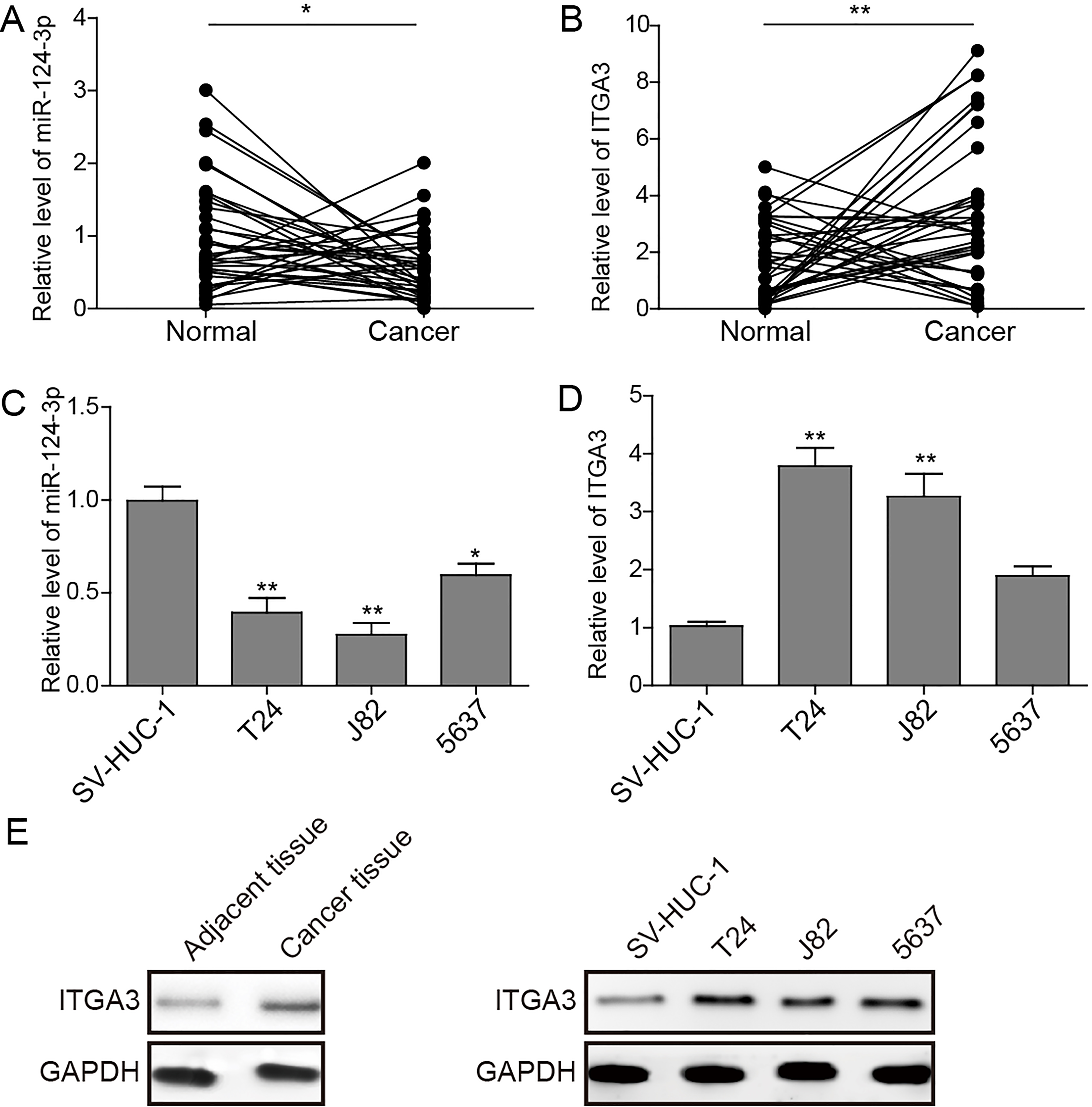
Down-regulation of miR-124-3p and up-regulation of ITGA3 were observed in clinical samples and cell lines
In the present study, we evaluated the expression levels of miR-124-3p and ITGA3 in 36 patients with bladder urothelial carcinoma, whose clinicopathological characteristics were summarized in Table 2. The results of qRT-PCR showed that the miR-124-3p expression was significantly down-regulated in BC tissues, while the ITGA3 expression was up-regulated compared to the adjacent non-cancerous tissue samples (Fig. 1A,
MiR-124-3p induces apoptosis and inhibits clonogenicity and proliferation in BC cells. (A, B) The proliferation of cells was detected by MTT assay over 24 h, 48 h and 72 h after transfection, A for T24 cell, B for J82 cell. (C) The apoptotic ratios of cells were detected by Annexin V FITC- based flow cytometry. (D) The vitality of BC cells after transfection of miR-124-3p was assessed by colony formation assays. *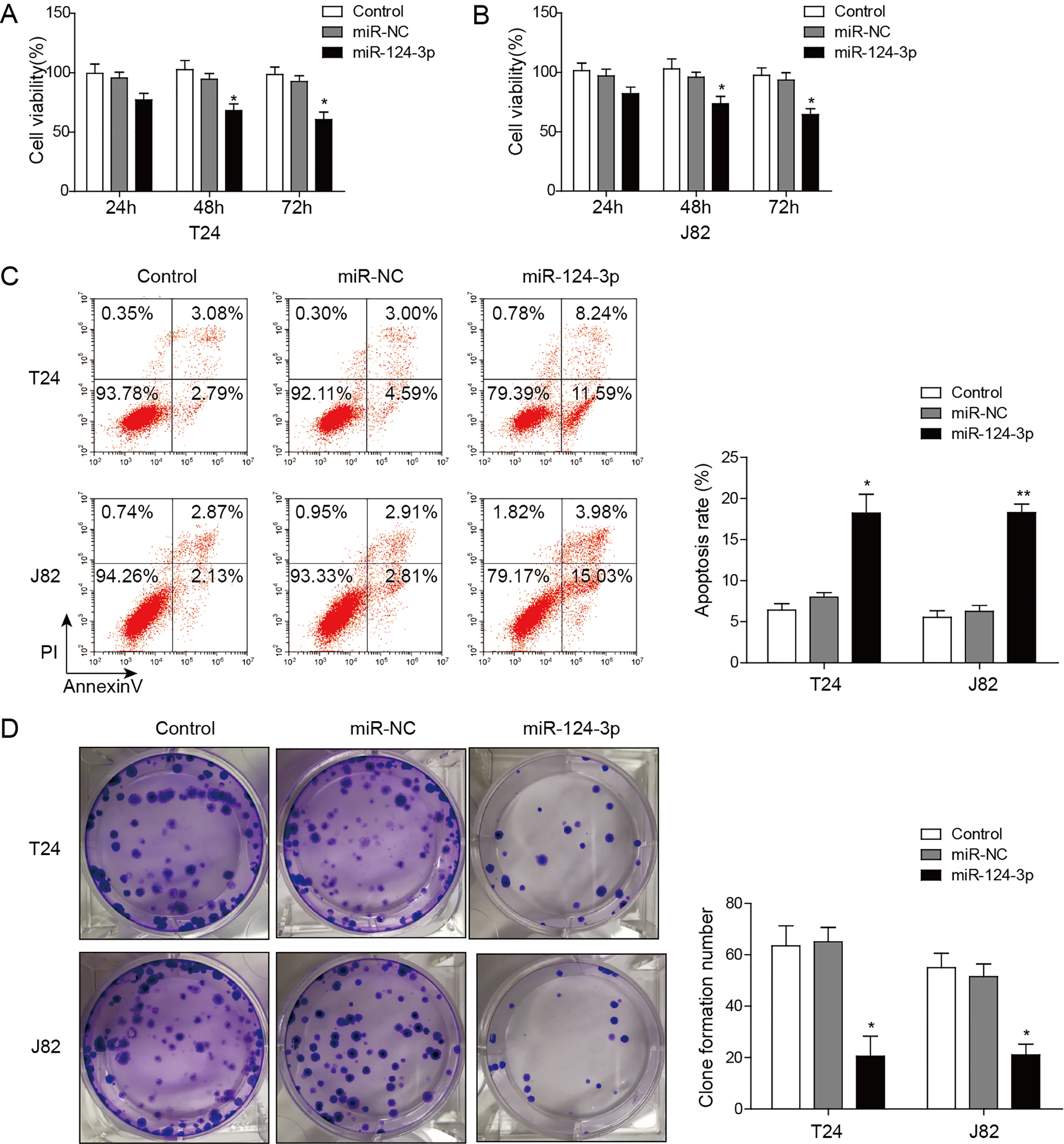
Cell migration and invasion of bladder cancer cells are inhibited by miR-124-3p in wound healing and transwell assays. (A, B) T24(A) and J82 (B) cells were transfected with miR-NC or miR-124-3p mimics, and were performed wound healing assays with a 24-h recovery period. (C) T24 and J82 cells were transfected with medium, miR-NC and miR-124-3p mimics. Cell invasion were assessed after 24 h incubation by transwell assay. *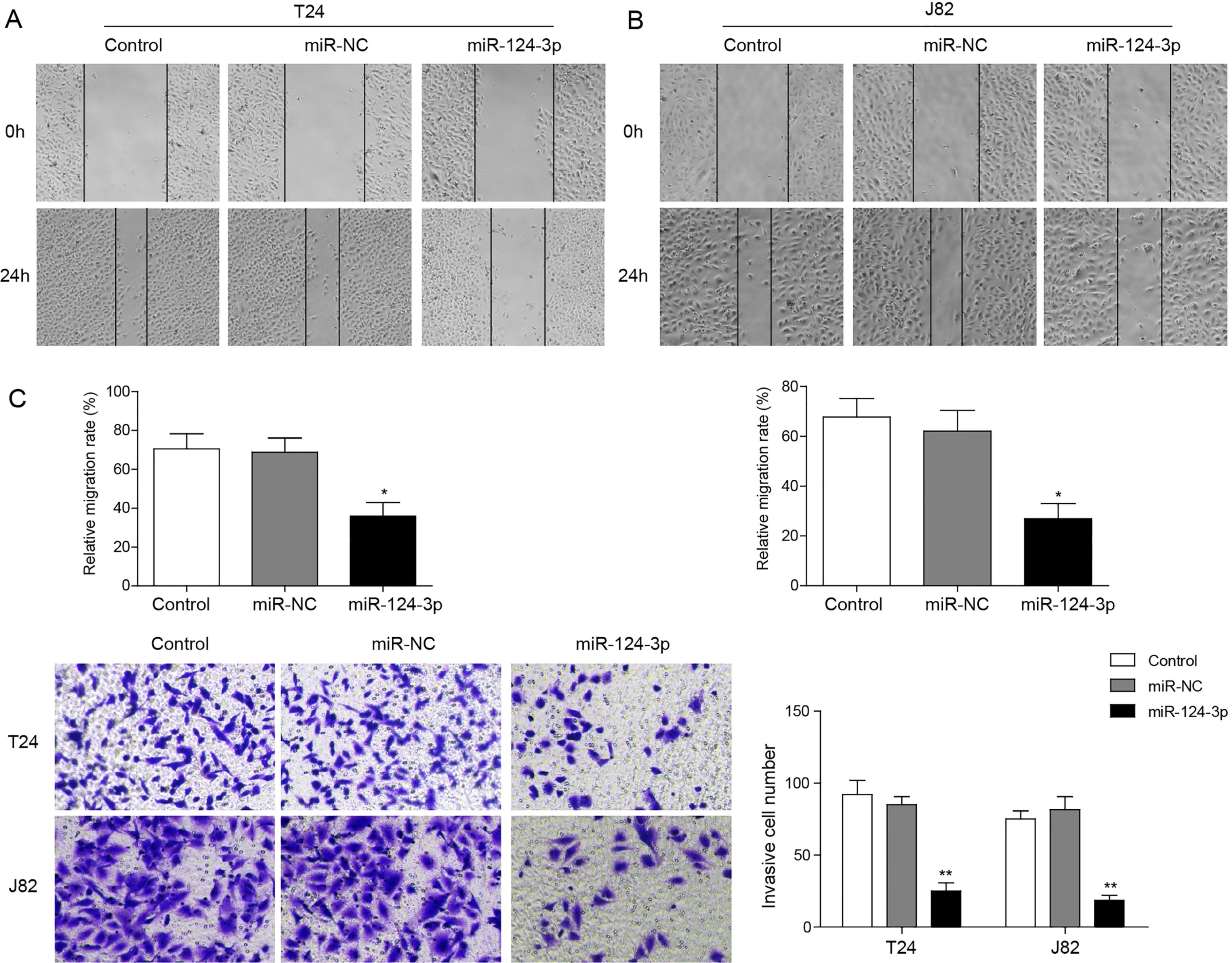
MTT assay revealed the reduced cell viability in T24 and J82 cells within 24 h, 48 h and 72 h after transfection with miR-124-3p mimics, compared to control or miR-NC group (Fig. 2A and B). By flow cytometry, we found that the apoptosis ratios in T24 and J82 cells with restored miR-124-3p were significantly enhanced (T24,
MiR-124-3p directly targets ITGA3 in bladder cancer cells. (A) Oligonucleotides containing the predicted miR-124-3p binding sites in the 3’-UTR of ITGA3 mRNA were synthetized. Alignment between the miR-124-3p mimics and 3’-UTR of ITGA3 mRNA (WT or MUT) was shown. (B) HEK293T cells were co-transfected with 50 nM of either miR-124-3p mimics or NC and 100 ng pmirGLO Dual-Luciferase miRNA Target Expression Vector with WT or MUT 3’-UTR of ITGA3. The relative luciferase activity was measured 48 h after transfection.(C) QRT-PCR analysis confirmed the elevated expression level of miR-124-3p after transfection. (D) QRT-PCR analysis confirmed the down-regulation of ITGA3 after overexpression of miR-124-3p. (E) Western blot assay confirmed the reduced expression of ITGA3 after miR-124-3p were overexpressed in BC cells. *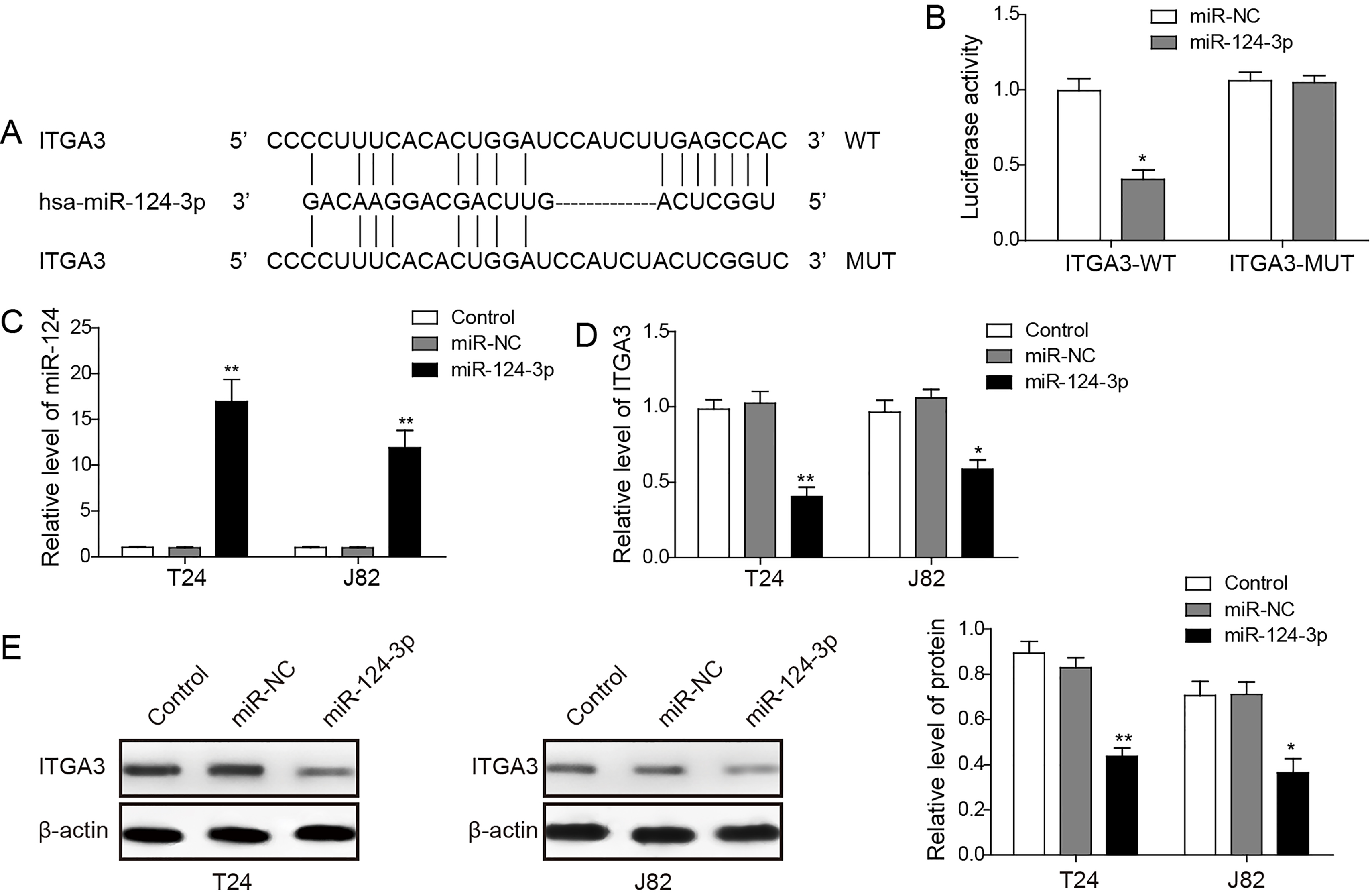
To investigate whether the restored level of miR-124-3p has any impact on migration and invasion capacity of BC cells, we performed the wound healing assays in T24 and J82 cells. The result showed that the spread of wound closure in T24 and J82 cells with forced expression of miR-124-3p was retarded significantly (
MiR-124-3p directly targets ITGA3
Functional mechanism of miRNAs relies on their specific binding to the 3’-UTR of the downstream genes. Previous studies have suggested that miR-124-3p down-regulates ITGA3 in podocytic cells, and it is involved in capacity damage during the pathological progression of diabetic nephropathy (DN) [26, 27]. In our study, since the negative correlation between the expression levels of miR-124-3p and ITGA3 has been validated in clinical specimens and BC cell lines, we speculated that ITGA3 may serve as a direct target of miR-124-3p. To discover whether the cell migration and invasion capacity is regulated by miR-124-3p through targeting ITGA3 3’-UTR, the luciferase reporter assay was carried out. In this assay, annealed oligonucleotides with the wild type (WT) or mutated (MUT) sequence of ITGA3 3’-UTR was cloned into pmirGLO Dual-Luciferase Vector (Fig. 4A). HEK293T cells were transiently transfected with these constructs and miR-124-3p mimics or miR-NC. miR-124-3p mimics significantly suppressed the luciferase activity of reporter genes containing wild type 3’-UTR of ITGA3, while the inhibition was almost rescued for the mutated target sites (Fig. 4B).
Silencing ITGA3 enhances apoptosis and decreases viability and proliferation of BC cells. (A) QRT-PCR analysis showed that the expression of ITGA3 was significantly repressed in si-ITGA3 transfectants in comparison with that in blank control and NC groups in T24 and J82 cells. (B) Western blot showed that the protein expression of ITGA3 was reduced after transfection with si-ITGA3. (C) The proliferation of cells was detected by MTT assay over 24 h, 48 h and 72 h after transfection of si-ITGA3. (D) The apoptotic ratios of cells were then detected by flow cytometry. (E) The vitality of BC cells after transfection of si-ITGA3 was assessed by colony formation assays. *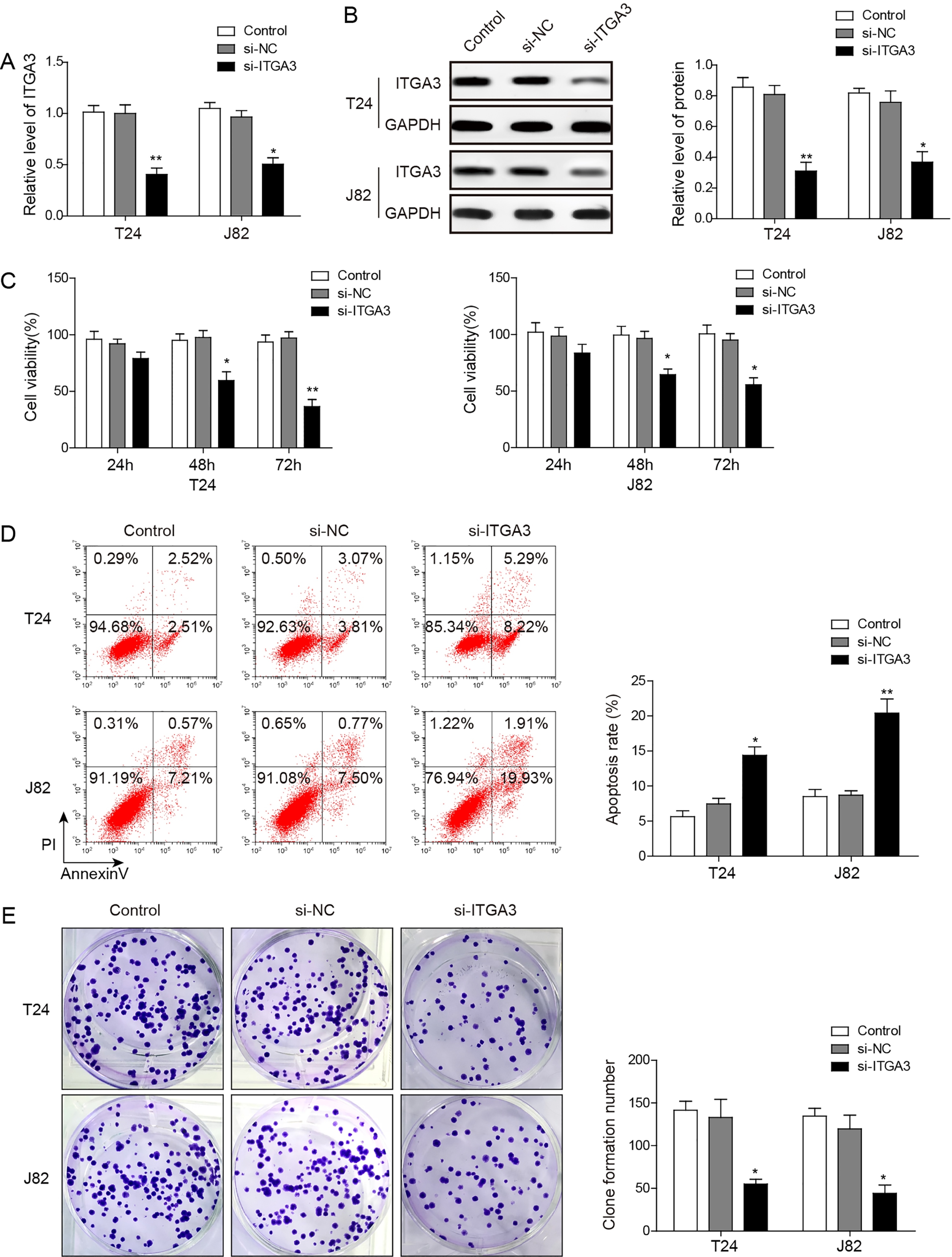
ITGA3 is involved in migration and invasion of bladder cancer cells. (A, B) T24 (A) and J82 (B) cells were transfected with si-ITGA3, and wound healing assays were performed with a 24-h recovery period. (C) T24 and J82 cells were transfected with medium, miR-NC and miR-124-3p mimics. Cell invasion was assessed after 24 h incubation by transwell assay. *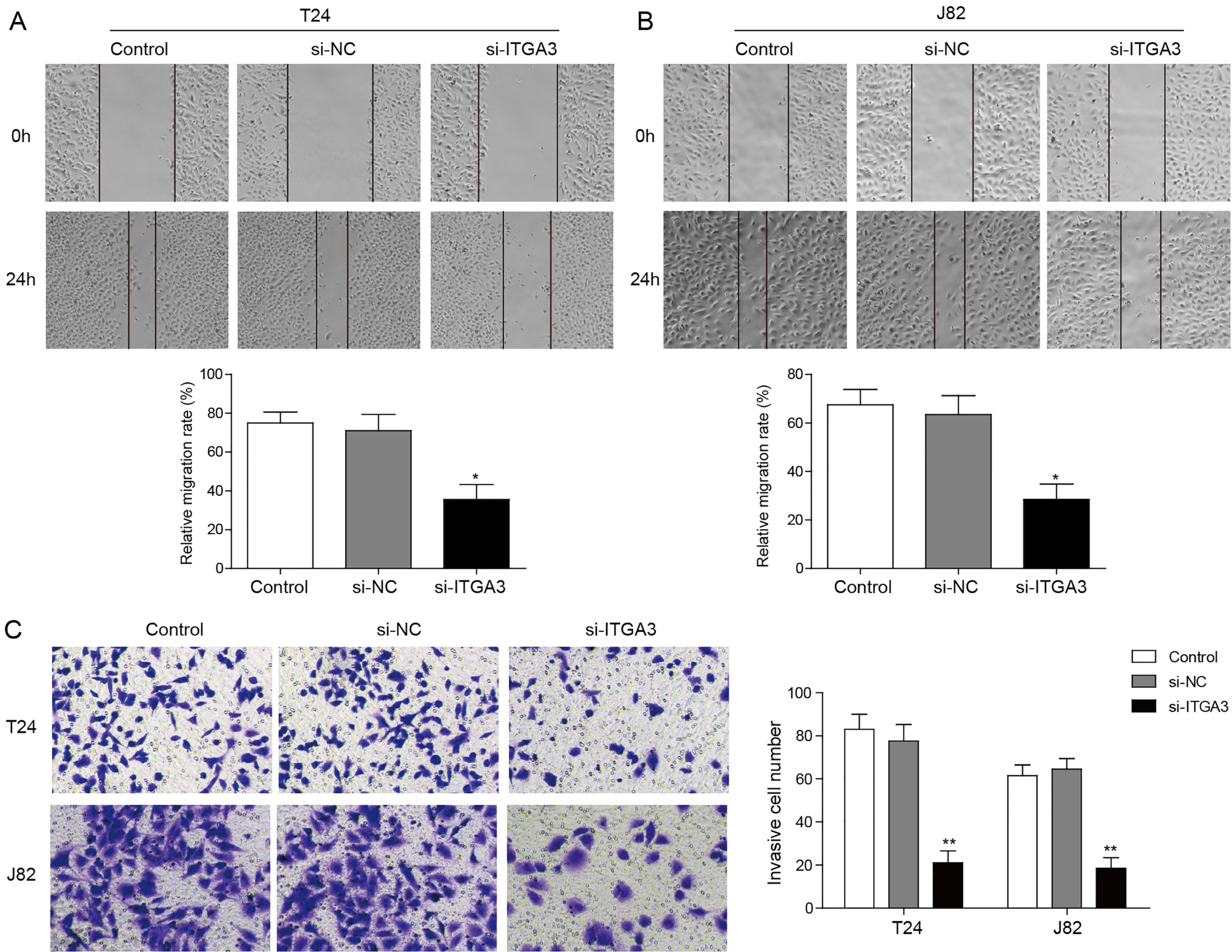
To examine functional effects of miR-124-3p in the development of BC, the miR-124-3p mimics were transfected to T24 and J82 cells. The restoration of miR-124-3p was subsequently confirmed by qRT-PCR analysis (Fig. 4C). We then analyzed the expression level of ITGA3 in T24 and J82 cells after miR-124-3p was overexpressed. The qRT-PCR showed that the mRNA levels of ITGA3 were remarkably reduced in the miR-124-3p treated groups in contrast to the blank control and NC groups (Fig. 4D; T24,
Next, to analyze the function of ITGA3, we silenced ITGA3 in BC cells. T24 and J82 were transfected by the small interfering RNA against the ITGA3 (si-ITGA3). QRT-PCR and western blot were applied to verify that ITGA3 expression was greatly reduced after transfection by si-ITGA3 (Fig. 5A and B). Then MTT assay and colony-formation assay were carried out. Our results showed that silencing ITGA3 decreased the cell viability and proliferation of T24 and J82 (Fig. 5C and E). Moreover, flow cytometry revealed that the apoptosis rate was significantly enhanced after the cells were transfected with si-ITGA3 (Fig. 5D; T24,
ITGA3 is involved in migration and invasion of bladder cancer cells
In the following experiments, we investigated whether loss of ITGA3 can also function similarly as restoration of miR-124-3p in BC cells migration and invasion. To achieve this aim, wound healing assay and transwell assay were carried out. The results showed that silencing of ITGA3 expression retarded up to 50% of the wound closure in T24 and J82 cells 24 h after transfection with si-ITGA3 (Fig. 6A and B). Moreover, transwell assay revealed that the invasion activities of the BC cells were significantly reduced after silencing ITGA3 (Fig. 6C,
MiR-124-3p inhibits FAK/PI3K/AKT and FAK/Src pathways, and meanwhile regulates EMT via ITGA3 in bladder cancer cells. (A, C) Examination of the phosphorylation of FAK, Src, AKT and PI3K after cells were transfected by miR-124-3p mimics or si-ITGA3 in comparison with blank control and NC groups in T24 (A) and J82 (C). (B, D) Examination of epithelial markers and mesenchymal markers after cells were transfected by miR-124-3p or si-ITGA3 in comparison with blank control and NC groups in T24 (B) and J82 (D). Histogram indicated the relative expression intensity of EMT markers after normalization by the expression intensity of GADPH. *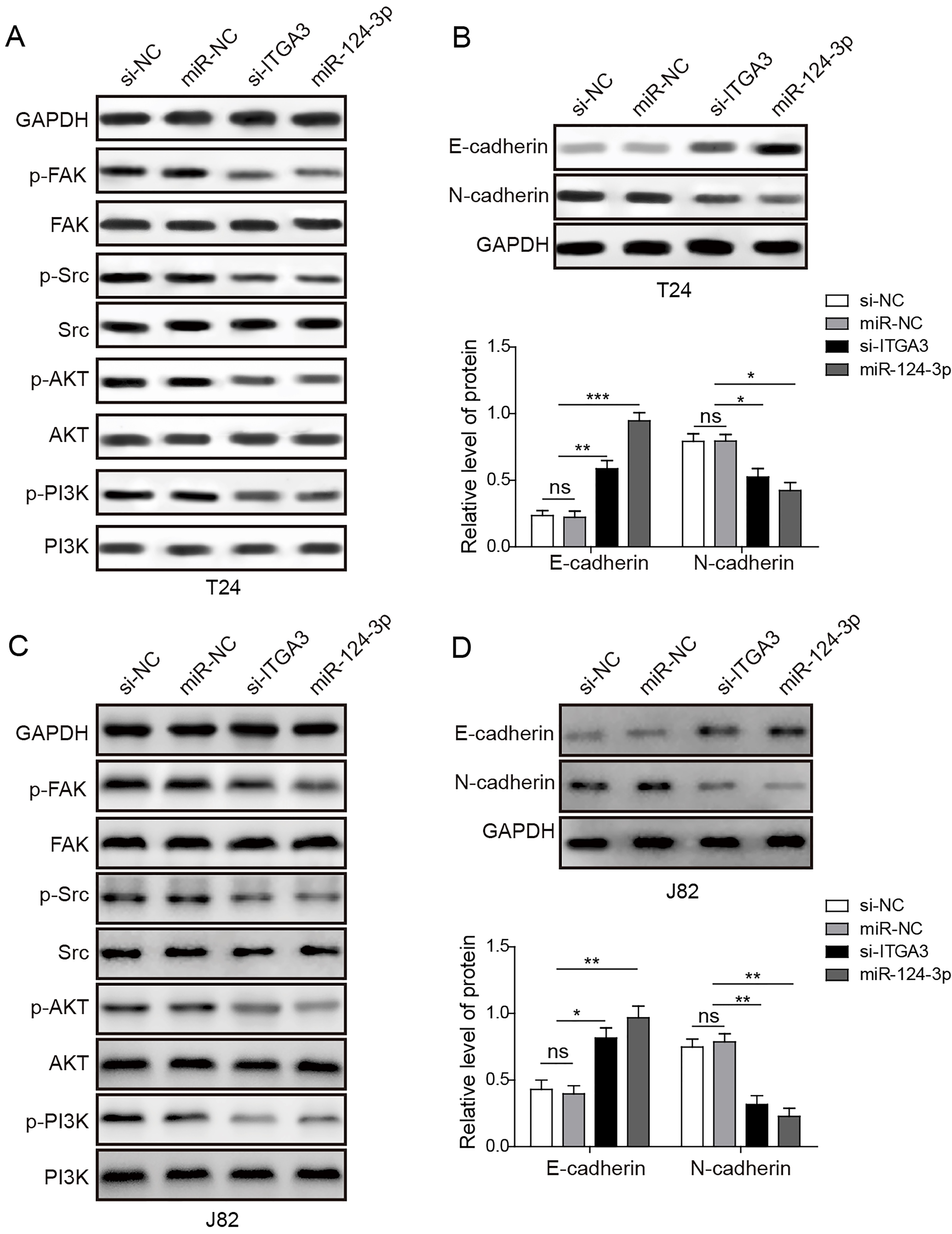
Given integrin family is deeply involved in the migration and invasion capacities of BC cells, we next investigated which signal pathway was regulated by miR-124-3p via targeting ITGA3 accordingly. Integrin is generally known as an activator of multiple FAK signaling pathways (e.g., FAK/Src, FAK/PI3K/AKT, FAK/Grb7, FAK/N-WASP) that regulate cell migration and invasion [29]. We therefore focused on two major integrin-mediated pathways, FAK/Src pathway and FAK/phosphatidylinositol-3-kinase (PI3K)/AKT pathway, both of which are well-established in previous studies [30, 31]. The results of western blot assays showed that restoration of miR-124-3p or inhibition of ITGA3 significantly suppressed FAK phosphorylation, and thus reduced the phosphorylation level of PI3K, AKT, and Src protein in T24 and J82 cells (Fig. 7A and C).
In addition, western blot assays also showed that elevated expression level of miR-124-3p led to up-regulation of the epithelial marker, E-cadherin, and down-regulation of mesenchymal marker, N-cadherin in T24 and J82 cells (Fig. 7B and D). Similarly, knockdown of ITGA3 up-regulated the epithelial marker and down-regulated mesenchymal marker (Fig. 7B and D). These results revealed that miR-124-3p may inhibit migration and invasion through modulating EMT progress via ITGA3 in BC cell lines.
Discussion
Despite advancements in cancer diagnosis and treatment, bladder cancer remains a common disease with a relatively stable incidence and a high recurrence rate world-widely [2]. Due to the limits of available biomarkers for bladder cancer, it is urgent to identify better diagnostic and prognostic molecular targets. Previous studied showed that miR-124-3p is frequently down-regulated in several human cancers, and forced restoration of it leads to the inhibition of proliferation and invasion of cancer cells. For example, it was reported that abnormal lower expression of miR-124-3p is correlated with prognosis of lung cancer, and thus miR-124-3p could exert its function as a potential biomarker for further risk stratification [31]. Another study showed that overexpression of miR-124-3p suppressed the proliferation and migration of breast cancers [2, 32]. In the context of research in bladder cancer, the methylation of miR-124-3p presumably contributes to its down-regulation [31, 33], and several signaling pathways it modulates was found to be involved in cell migration and invasion [13, 15, 16, 32]. However, how miR-124-3p specifically functions in bladder cancer progression remains elusive.
This study aims to further clarify the antitumor mechanism of miR-124-3p and its downstream target genes in the pathogenesis of human bladder cancer. By investigating the clinical samples from 36 patients with BC, we found significant lower expression level of miR-124-3p in tumor tissues and cells with comparison to the normal ones. In addition, the BC cells with forced expression of miR-124-3p exhibited reduced proliferation activities in the colony-formation assay. Furthermore, the results of transwell assay and wound healing assay suggested overexpression of miR-124-3p significantly inhibited the migration and invasion capacity of BC cells. Altogether, our study confirmed the crucial role played by miR-124-3p in tumorigenesis and progression of bladder cancer.
It was reported that ITGA3 played a significant role in migration and invasion for various types of cancer cells [22, 28], and also previous studies suggested that ITGA3 was directly targeted by miR-124-3p in the diabetic nephropathy [26, 27]. Therefore, we speculated ITGA3 may also serve as a direct target of miR-124-3p in BC cells. The qRT-PCR revealed the decreased expression of miR-124-3p and the increased expression of ITGA3 in BC tumor tissues and cells, in comparison to normal tissues and cells. In addition, by western blot we showed that the protein expression level of ITGA3 was remarkably higher in bladder cancer tissue than that in normal adjacent tissue. To investigate whether ITGA3 was a potential target for miR-124-3p in BC, luciferase reporter assay was deployed, in which we demonstrated the binding between miR-124-3p and the complementary 3’-UTR sites of ITGA3. In addition, we found that silencing ITGA3 could induce apoptosis and inhibit proliferation of BC cells by flow cytometry, MTT assay and colony-formation assay. Furthermore, wound healing and transwell assays also confirmed silencing ITGA3 achieved similar results as restoring miR-124-3p in inhibition of cancer cell migration and invasion. These data provide the evidence that miR-124-3p exerts its function in BC cell migration and invasion via regulating the expression of ITGA3.
Integrins are the major cell adhesion receptors that transduce the extracellular matrix (ECM) cues into the intracellular signal pathways. Accumulating evidence support a vital role of integrin in many facets of metastatic cascade, particularly in the initial phase of tumor dissemination which requires EMT [21, 34, 35]. In this study, we analyzed the functional effects of ITGA3 and miR-124-3p on downstream signaling in BC cells. Our data revealed that restoration of miR-124-3p and silencing ITGA3 significantly decreased the phosphorylation levels of FAK, and moderately reduced the phosphorylation of PIK3, AKT and Src. Since FAK/PI3K/AKT and FAK/Src phosphorylation cascades function to promote cell migration and invasion, our findings offer further insight into the mechanism of miR-124-3p in BC development [29]. As a key step of tumor progression, EMT is generally thought to be paralleled with a ‘cadherin switch’ process, in which the downregulation of E-cadherin is balanced by increased expression of N-cadherin [33]. Our study showed that either forced expression of miR-124-3p mimics or silencing ITGA3 led to higher expression of E-cadherin and lower expression of N-cadherin [35]. Considering the cross-talk between integrin-based and cadherin-based adhesion structures [34], this result may suggest that miR-124-3p regulates the migration and invasion of BC cells through inhibiting EMT and integrin-mediated signaling pathways in a cooperative manner.
In conclusion, our study confirmed the decrement of miR-124-3p contributes to the tumorigenesis and progression of human BC. To our best knowledge, this is the first report showing miR-124-3p directly targeted ITGA3 in BC cells. Moreover, we established the integrin-mediated mechanism for miR-124-3p to affect the epithelial-mesenchymal transfer, cell migration and invasion. Therefore, miR-124-3p may reasonably be a prognostic and diagnostic biomarker for patients with BC, and a potential therapeutic target in the BC treatment.
Footnotes
Conflict of interest
The authors declare no conflict of interest.
