Abstract
BACKGROUND:
Axillary lymph node metastasis (LNM) affects the progression of breast cancer. However, it is difficult to preoperatively diagnose axillary lymph node status with high sensitivity. Therefore, we hypothesized that platelets/lymphocytes ratio (PLR) and lymphocytes/ red blood cells ratio (LRR) might help in the prognosis of lymph node metastasis in T1-T2 breast cancer.
METHODS:
166 patients (Chang Ning Maternity & Infant Health Institute) were included in our study, and the associations of PLR and LPR with lymph node metastasis were investigated. Peripheral blood was collected one week before the surgery, and the patients were divided into different categories based on their PLR and LRR.
RESULTS:
The incidence of LNM was significantly increased in the high PLR group (
CONCLUSIONS:
PLR and LRR could be identified as predictors of LNM in patients with T1/T2 breast cancer.
Introduction
Breast cancer is the most common cancer in females [1]. According to GLOBOCAN 2020 data, the global age-standardized incidence rate (ASIR) for breast cancer was 47.8 per 100,000, while in China, it stood at 39.1 per 100,000. The worldwide age-standardized mortality rate (ASMR) was 13.6 per 100,000, whereas the ASMR for breast cancer in China was notably lower, at 9.5 per 100,000 [2]. Patients with breast cancer have a high axillary lymph node metastasis rate. Metastasis to axillary lymph nodes is the most significant prognostic factor for patients with breast cancer [3]. In early breast cancer, sentinel lymph node biopsy (SLNB) has been extensively used to evaluate axillary lymph node status [4]. However, some patients undergo axillary lymph node dissection (ALND) irrespective of SLNB results due to concerns over false-negative SLNB. However, no preoperative markers exist that can successfully evaluate axillary lymph node status with high sensitivity and specificity.
Study design chart. A total of 170 patients with breast cancer diagnosed with cT1/T2N0M0 preoperatively from 2019 to 2021 at the China Chang Ning Maternity & Infant Health Institute. However, 4 patients who showed T3/T4 staged breast cancer after the histological examination were excluded.
Systemic inflammatory response affects cancer progression [5]. Inflammation is divided into two categories: acute and chronic inflammation. Acute inflammatory responses are involved in immune surveillance, whereas chronic inflammatory responses sometimes result in increased tumor growth and metastasis via the release of inflammatory mediators. Persistent chronic inflammation may also induce infiltration of inflammatory cells, which secrete cytokines and chemokines, leading to activation of oncogenes or inactivation of tumor suppressor genes and immune tolerance [6]. Both LRR and PLR are indicators of systemic inflammation [7, 8]. Platelets promote tumor growth by secreting various angiogenic growth factors, including platelet-derived growth factor (PDGF) and transforming growth factor-
Here, we hypothesized that PLR and LRR are differ based on axillary lymph node status in T1-T2 breast cancer and tested whether preoperative PLR and LRR are predictive markers of LNM in breast cancer patients.
Patients
Patients were enrolled in this study from January 2019 to September 2021 at the China Chang Ning Maternity & Infant Health Institute (Fig. 1). All enrolled patients were devoid of rheumatic or hematological disorders. They had tested negative for nucleic acid 2 days prior to surgery and remained free from coronavirus disease infection for a minimum of 6 months. All patients received breast cancer surgery and axillary lymph node dissection. Immunohistology was used to assess the status of estrogen receptor (ER), progesterone receptor (PR), Ki67, and human epidermal growth factor receptor 2 (HER2). The Immuno-histological expression of estrogen receptor or progesterone receptor was considered positive when
Receiver operating characteristic (ROC) curve analysis. PLR cut-off value for lymph node metastasis was identified as 154.17. Area under curve 0.604, sensitivity 0.645, specificity 0.606 and 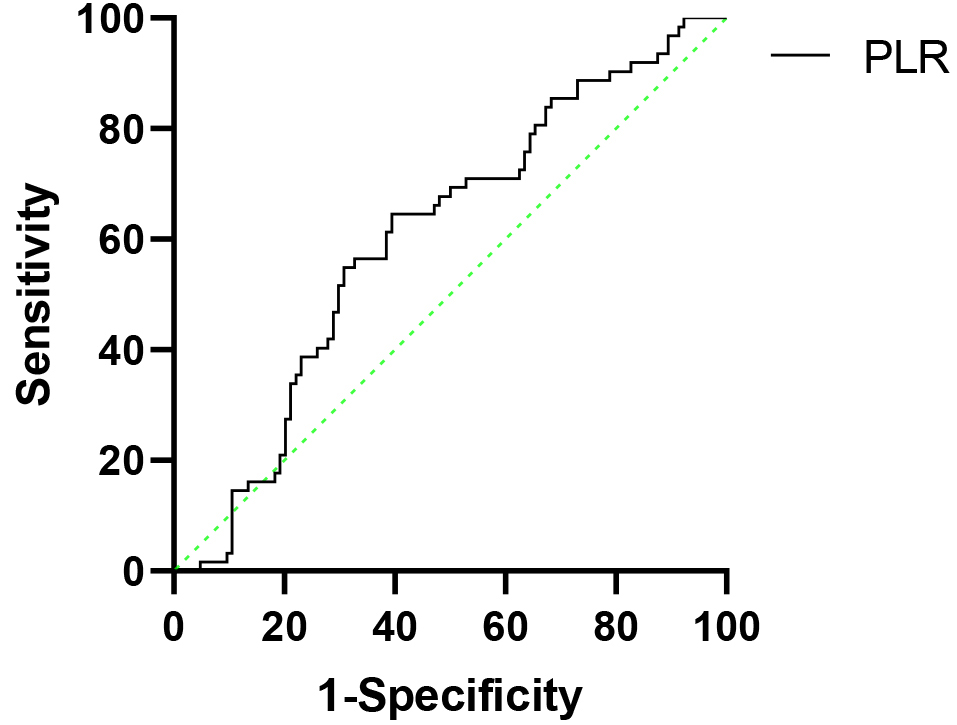
The patient clinical database was evaluated retrospectively, and the data include age, body mass index (BMI), tumor size, pathologic stage, and surgery type. Patients’ peripheral whole blood was collected one week before the surgery with a hemocytometer. Platelet lymphocytes and red blood cells were counted using the Coulter XN-1000 Hematology Analyzer (Sysmex Corporation, JPN) and PLR and LRR were calculated for each patient. Preoperative PLR was calculated as the ratio of the absolute platelet count to the absolute lymphocyte count. Preoperative LRR was calculated as the ratio of the absolute lymphocyte count to the absolute red blood cell count. The PLR cut-off value for predicting axillary lymph node metastasis was set at 154.17. The LRR cut-off value for predicting axillary lymph node metastasis was set at 0.423. Both cut-off values were defined with Receiver-operator curve (ROC) curve analysis (Fig. 2). We stratified the patients into different groups based on their LRR or PLR and analyzed the associations between PLR or LRR levels and axillary lymph node status.
The experiments were undertaken with the understanding and written consent of each subject, and that the study conforms with The Code of Ethics of the World Medical Association (Declaration of Helsinki), printed in the British Medical Journal (18 July 1964). The study was granted approval by the Ethics Management Committee of Shanghai Changning Maternity and Infant Health Hospital.
Data were analyzed using the SPSS statistical software (SPSS 26.0, Chicago, IL, USA). Nominal crosstabs were compared using the Chi-square test retrospectively. The student’s paired t-test was used for parametric two-group analyses, and the Mann-Whitney U test was used for non-parametric two-group comparison. ROC analysis was used to identify the cut-off values. PLR and LRR were assessed using univariate, multivariate logistic regression analysis. Multivariate models included age (
Clinicopathological characteristics of 166 patients
Clinicopathological characteristics of 166 patients
HER2, human epidermal growth factor receptor 2; BMI, body mass index; LRR, lymphocyte to red cells ratio; PLR, platelet to lymphocyte ratio.
Association between lymph node metastasis and clinicopathological factors
HER2, human epidermal growth factor receptor 2; BMI, body mass index; LRR, lymphocyte to red cells ratio; PLR, platelet to lymphocyte ratio.
Clinical characteristics
166 patients with T1 or T2 breast cancer were included in this study (Table 1). The average age was 55.32 years (range
Association of LRR and PLR with LNM
The relationships of clinicopathological features with LNM are summarised in Table 2. Based on the test for homogeneity, we used the Mann-Whitney U test to evaluate the association of LRP and PLR with LNM. The mean PLR of patients with metastatic lymph nodes was 168.75, and the mean PLR of patients without metastatic lymph nodes was 155.61 (
Receiver operating characteristic (ROC) curve analysis. LRR cut-off value for lymph node metastasis was identified as 0.423. Area under curve 0.593, sensitivity 0.839, specificity 0.308 and 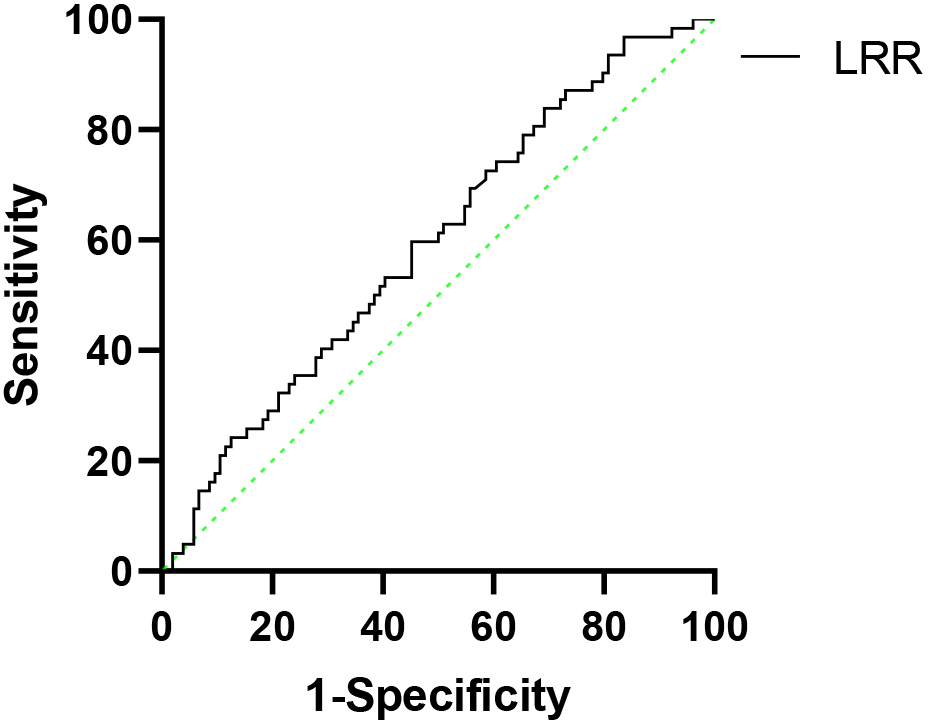
There were no statistically significant differences in age between the different groups. LNM was significantly associated with tumor size (
Association between platelet to lymphocyte ratio (PLR), lymphocyte to red cell ratio (LRR) and clinicopathological factors
HER2, human epidermal growth factor receptor 2; BMI, body mass index; LRR, lymphocyte to red cell ratio; PLR, platelet to lymphocyte ratio.
Univariate and multivariate analysis of predictors for lymph node metastasis
HER2, human epidermal growth factor receptor 2.
Breast cancer is the most prevalent cancer in women worldwide and accounts for 19.5% of newly diagnosed cancer in China [15]. The most significant prognostic factor in invasive breast cancer is axillary lymph node status. Therefore, dissection of suspected metastatic lymph nodes is necessary. 30% of patients with T1-T2 invasive breast cancer show axillary lymph node involvement [16, 17]. In our study, the positivity rate of ALND was 37.3% which is consistent with previous findings. As surgical techniques underwent improvement, SLNB widely replaced ALND [18]. However, some patients worry about false-negative sentinel lymph nodes and opt for ALND.
Our study shows that lymph node metastasis is highly associated with tumor size and clinical T stage, which is similar to previous studies [19, 20]. We found that expressions of ER, PR, and HER2 were not associated with metastasis which is also consistent with other reports [21]. However, KLAR et al. reported that patients with HER-2-positive breast cancer were likely to develop lymph node metastasis [22]. HAN et al. also reported that ER-positive breast cancer tended to metastasize more frequently [23]. These conflicting observations may be due to the differences in race, tumor stage, and tumor size.
Our ultrasound sensitivity result (40.3%) exhibited a lower value in comparison to findings from other studies. This disparity arises from the fact that our research focused on patients in the early stages of breast cancer, where lymph node metastases tend to be more subtle. In contrast, prior studies often concentrated on patients with advanced breast cancer, which could lead to differences in detectability.
PLR and LRR have been established as biomarkers for inflammation [7, 13, 14]. Lymphocytes play an important role in anti-tumor responses and can either directly kill tumor cells or indirectly activate immune responses and induce tumor cell apoptosis. But, the absolute lymphocyte numbers may be affected by nutritional status, infection, and other factors. The complex interaction between tumor cells and the microenvironment has received renewed attention due to the development of molecular biology. Tumor cell-mediated tissue destruction results in chronic inflammation. Inflammatory cells secrete chemokines and cytokines, leading to DNA damage and genomic instability, which may result in the inactivation of cancer suppressor genes or oncogene activation. Inflammatory cells also induce immune tolerance and immune escape and promote tumor progression and metastasis [5, 6]. We previously showed that LRR is an independent prognostic factor for locally advanced breast cancer [7]. In this study, we used red blood cells to reduce variation and showed that pretreatment LRR might be a predictor of axillary lymph nodes metastasis.
An increase in PLR indicates a relative increase in platelets or a decrease in lymphocytes. When a patient experiences inflammation, the release of interleukin 1, interleukin 3, and interleukin 6 increases platelets which are an integral component of blood coagulation and are one of the indicators of the inflammatory response [24]. Platelets secrete immunosuppressive factors, such as TGF-
Recently, higher PLR has been related to worse prognosis in patients with pancreatic carcinoma, stomach cancer, and ovarian carcinoma [27, 28, 29]. An elevated PLR may result from tumor cells mediated destruction of platelets leading to reactive thrombocytosis in breast cancer. Patients with high PLR are more likely to have metastatic lymph nodes and display poor DFS and OS [13, 14]. Our study shows that PLR is also a good bio-predictor of axillary lymph nodes metastasis. TAKADA et al. reported that a high PLR was associated with tumor metastasis to the sentinel lymph node in T1 Breast Cancer [30]. Morkavuk et al. previously also reported that early breast cancer patients with high PLR showed a higher frequency of sentinel axillary lymph node metastasis [31].
Our study has some limitations that are inherent in its retrospective design. The accuracy of prediction may be low because the preoperative PLR and LRR are easily affected by other diseases such as hematopoietic disease and cerebrovascular diseases. The sample size of our study was relatively small. However, we found statistically significant differences in the two biomarkers.
Conclusion
In summary, PLR and LRR could be identified as predictors of LNM in patients with T1/T2 breast cancer.
Footnotes
Acknowledgments
We would like to thank the Changning Maternity and Infant Health Hospital and Shanghai Municipal Health Commission for financial support.
Author contributions
Conception: Haofeng Wang.
Interpretation or analysis of data: Haofeng Wang, Jing Cui, Haiyan Zhao.
Preparation of the manuscript: Haofeng Wang, Jinling Yu, Weida Shen.
Revision for important intellectual content: Haofeng Wang, Beimin Gao.
Supervision: Beimin Gao.
