Abstract
Objective
The objective of this study was to explore correlations between red blood cell distribution width, neutrophil-to-lymphocyte ratio, platelet distribution width, and colorectal adenomas.
Methods
Retrospective analysis of 120 patients undergoing colonoscopy (from January to December 2024) divided into adenoma (n = 60) and control (n = 60) groups. Demographic characteristics, habits, and blood parameters were compared.
Results
Significant differences in neutrophil-to-lymphocyte ratio, platelet distribution width, and red blood cell distribution width between groups (P < 0.05) were noted. Spearman analysis showed positive correlations: neutrophil-to-lymphocyte ratio and platelet distribution width (r = 0.434), neutrophil-to-lymphocyte ratio and red blood cell distribution width (r = 0.461), and red blood cell distribution width and platelet distribution width (r = 0.424) (P < 0.001). Logistic regression identified neutrophil-to-lymphocyte ratio (odds ratio = 5.027), platelet distribution width (odds ratio = 1.553), and red blood cell distribution width (odds ratio = 9.081) as independent risks.
Conclusion
Elevated neutrophil-to-lymphocyte ratio, platelet distribution width, and red blood cell distribution width are independent risk factors. Early colonoscopy and intervention may serve as non-invasive biomarkers for early screening.
Keywords
Introduction
Colorectal adenoma is the most significant precancerous lesion of colorectal cancer. Its risks extend beyond potential malignancy to include long-term impacts on digestive system function, patients’ quality of life, and societal healthcare resources. A close biological link exists between colorectal adenoma and colorectal cancer, with the core theory being the “adenoma-carcinoma sequence.” The adenoma-carcinoma sequence is as follows: approximately 70% to 80% of colorectal cancers develop through a gradual malignant progression from adenoma, involving stages including normal mucosa, hyperplasia, adenoma (low/high-grade dysplasia), and ultimately cancer development, typically spanning 10 to 15 years. Recent studies have focused more on the correlation between neutrophil-to-lymphocyte ratio (NLR), red blood cell distribution width (RDW), and colorectal cancer, whereas fewer investigations examine their association with colorectal adenoma. This study conducted a retrospective analysis by collecting clinical data from both observation and control groups. Statistical software was used to analyze correlations between NLR, RDW, platelet distribution width (PDW), and colorectal tumors. By comparing hematological marker differences between adenoma and control groups, we calculated odds ratios (ORs) using logistic regression analysis to define the association strength of NLR, RDW, and PDW with colorectal adenoma, with elevated levels considered positive risk factors.
Materials and methods
General data
From January to December 2024, a total of 120 patients were selected from the Gastroenterology Department of Anqing First People’s Hospital for hospitalization, divided into a control group and an observation group. The control group included 60 patients with normal colonoscopy results, whereas the observation group comprised 60 patients diagnosed with colorectal adenomas through colonoscopy. Inclusion criteria included the following: (a) all required clinical data could be fully collected from the Hospital Information System (HIS); (b) colonoscopy had been completed and documented in medical records, with reports meeting study requirements; and (c) age range: ≥18 years and ≤80 years. Exclusion criteria included the following: (a) missing required data in HIS; (b) no colonoscopy report available; (c) patients with malignant or endocrine tumors; (d) patients with inflammatory bowel disease, familial polyposis, or hereditary nonpolyposis colorectal cancer family history; (e) severe dysfunction in vital organs (heart, brain, lungs, kidneys), or blood disorders (e.g. leukemia, myelodysplastic syndrome); (f) pre-hospital use of glucocorticoids or antibiotics; and (g) unexplained fever or potential localized infections.
Method
By comparing hematological parameters between adenoma and control groups, OR values were calculated using logistic regression analysis to define the association strength of NLR, RDW, and PDW with colorectal adenomas. Elevated levels of these indicators were considered positive risk factors. Through Anqing First People’s Hospital’s HIS management system, eligible colorectal adenoma patients were selected as the observation group, whereas those without colonic lesions identified during colonoscopy were assigned to the control group. All enrollment data were obtained through simple random sampling using retrospective study methods. The reporting of this study complies with Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 1 Clinical data collected included gender, age, height, weight, body mass index (BMI), smoking history, alcohol consumption history, and relevant blood parameters such as neutrophils (NEUs), lymphocytes (Lyms), RDW, red blood cell (RBC) count, hemoglobin (Hb), platelets (PLTs), and PDW.
Data processing and analysis
After completing data collection, the database was established in Excel (Microsoft, Redmond, Washington, USA) with input values. Data organization and analysis were performed using SPSS Statistics 27 software (IBM Inc., Armonk, New York, USA). The Chi-squared test was employed for categorical data comparisons, whereas t-tests were used for normally distributed measurements and Mann-Whitney U tests for non-normal distributions. Based on univariate analysis, binary logistic regression was conducted to identify independent risk factors for colorectal adenoma development, with OR values and 95% confidence intervals (CIs) calculated. A P-value of <0.05 indicates statistically significant differences.
Results
General data comparison between groups
According to inclusion and exclusion criteria, a total of 120 patients were enrolled, comprising 60 cases in the observation group (32 males, 28 females; age, 48–74 years, mean age, 60.25 ± 6.38 years) and 60 cases in the control group (34 males, 26 females; age, 46–74 years, mean age, 60.12 ± 6.49 years). No statistically significant differences were observed between the groups in terms of gender, age, smoking history, BMI, or alcohol consumption history (p > 0.05) (Table 1).
Comparison of general data between observation group and control group.
Comparison of NEU, Lym, PLT, RBC, and Hb indicators between the observation group and the control group
Normality tests were performed on NEU, Lym, PLT, RBC, and Hb. PLT and Hb were normally distributed and analyzed using t-tests. The results showed no statistically significant differences in PLT and Hb between the two groups (P > 0.05). NEU, Lym, and RBC were non-normally distributed and analyzed using the non-parametric Mann-Whitney U test. Differences in NEU and Lym were statistically significant (P < 0.05), whereas differences in RBC were not statistically significant (P > 0.05) (Table 2).
Comparison of NEU, Lym, PLT, RBC, and Hb indicators between the observation group and the control group.

Hb: hemoglobin; Lym: lymphocyte; NEU: neutrophil; PLT: platelet; RBC: red blood cell.
Comparison of NLR, PDW, and RDW between observation group and control group
Normality tests were conducted on NLR, PDW, and RDW, revealing all three to be skewed distributions. Subsequent analysis employed the Mann-Whitney U test. The observation group demonstrated statistically significant higher levels of NLR, PDW, and RDW compared with the control group (P < 0.05), as shown in Table 3.
Comparison of NLR, PDW, and RDW between observation group and control group.
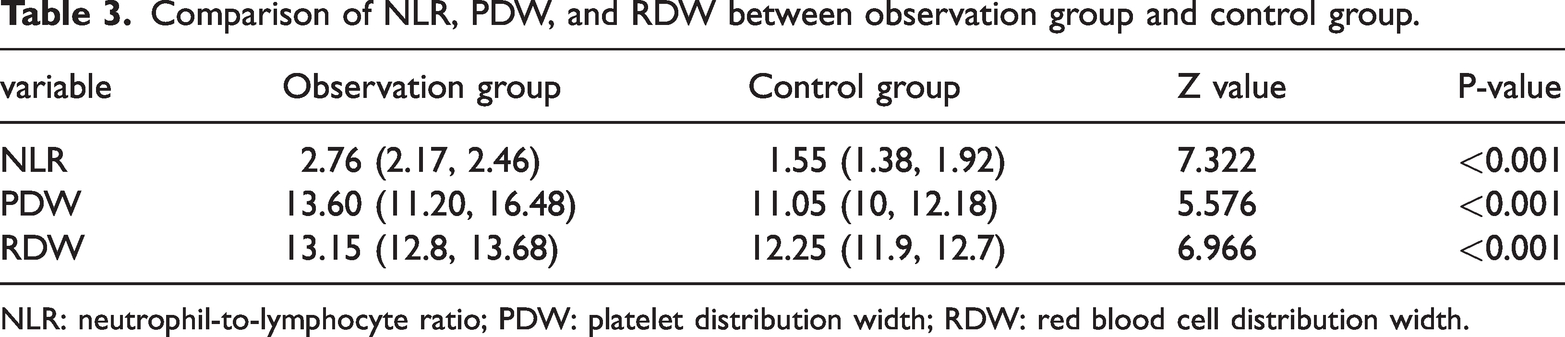
NLR: neutrophil-to-lymphocyte ratio; PDW: platelet distribution width; RDW: red blood cell distribution width.
Correlation analysis of NLR, PDW, and RDW
The Spearman correlation analysis was used to examine the relationships between NLR, PDW, and RDW. The results showed that NLR was positively correlated with PDW (r = 0.434, P < 0.001), NLR was positively correlated with RDW (r = 0.461, P < 0.001), and RDW was positively correlated with PDW (r = 0.424, P < 0.001) (Table 4).
Correlation analysis of NLR, PDW, and RDW.

NLR: neutrophil-to-lymphocyte ratio; PDW: platelet distribution width; RDW: red blood cell distribution width.
Multivariate binary logistic regression analysis of factors associated with colorectal adenoma occurrence
The statistically significant indicators (NLR, PDW, RDW) identified in the univariate analysis were incorporated into a logistic regression model for multivariate analysis. Pre-hypothesis tests for multicollinearity showed that NLR (variance inflation factor (VIF) = 1.34), PDW (VIF = 1.29), and RDW (VIF = 1.26) all remained below the 5% threshold. The ORs and 95% CIs were calculated. Results were considered statistically significant when P < 0.05 in two-tailed tests. The data demonstrated positive correlations between NLR, PDW, and RDW levels and the occurrence of colorectal adenomas, as detailed in Table 5 and Figure 1.
Binary logistic regression analysis of factors related to the occurrence of colorectal adenomas.

NLR: neutrophil-to-lymphocyte ratio; PDW: platelet distribution width; RDW: red blood cell distribution width; OR: odds ratio; CI: confidence interval.
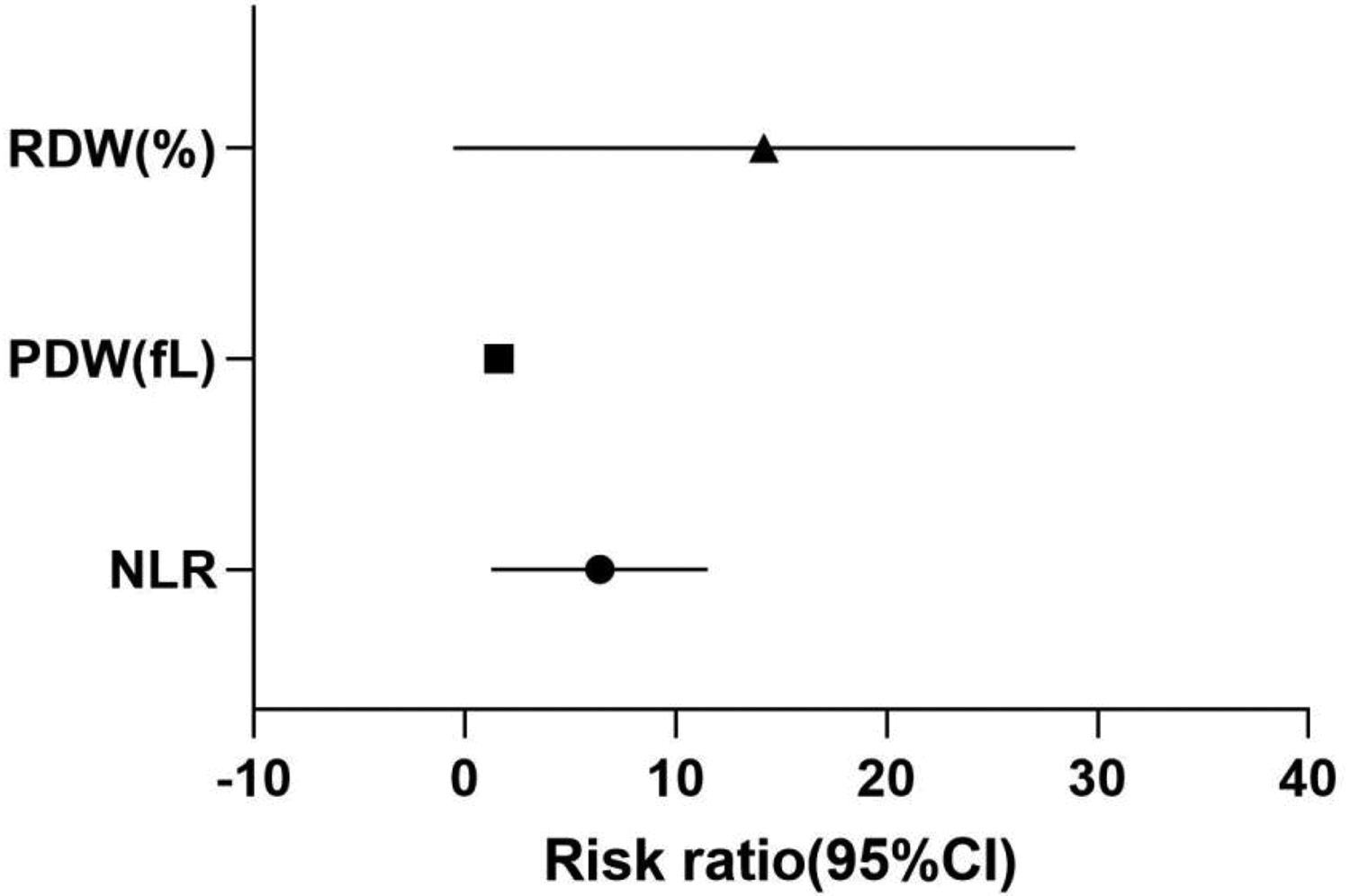
95% confidence interval of odds ratio.
Analysis of the subject-characteristic (receiver operating characteristic) curve for predictive models of colorectal adenoma development. The receiver operating characteristic (ROC) curve was used to evaluate predictive models. Results showed that for NLR, area under the curve (AUC) = 0.866 (0.8231–0.9463), P < 0.001; for PDW, AUC = 0.795 (0.7162–0.8738), P < 0.001; for RDW, AUC = 0.866 (0.8033–0.9290), P < 0.001. For the combined model combining NLR+PDW+RDW, AUC = 0.948 (0.9088–0.9862), P < 0.001 (Table 6 and Figure 2).
Forecast model parameters.

AUC: area under the curve; CI: confidence interval; NLR: neutrophil-to-lymphocyte ratio; PDW: platelet distribution width; RDW: red blood cell distribution width.

Receiver operator characteristic curve analysis of predictive model for colorectal adenoma occurrence.
Discussion
Colorectal adenoma is the most significant precancerous lesion of colorectal cancer, closely associated with genetic and environmental factors. Studies indicate that most colorectal cancers originate from precancerous adenomas or serrated polyps, 2 with approximately 85% of colorectal adenocarcinomas developing from precancerous adenomas. Preventing colorectal cancer through early detection and removal of precancerous lesions before malignancy progression and metastasis holds significant importance for reducing cancer incidence and improving quality of life. This study demonstrated statistically significant differences in NLR, PDW, and RDW between the observation group and control group (P < 0.05). Additionally, logistic regression analysis revealed that NLR (OR: 5.027, 95% CI: 2.096–12.061), PDW (OR: 1.553, 95% CI: 1.056–2.285), and RDW (OR: 9.081, 95% CI: 2.677–30.802) were independent risk factors for colorectal adenoma development. These findings suggest that systemic inflammatory responses and hematological component heterogeneity may play crucial roles in the occurrence and progression of colorectal adenomas.
Chronic inflammation serves as a central driver in the development of colorectal adenomas. White blood cells, including neutrophils and lymphocytes, act as key indicators of systemic inflammatory responses. Neutrophils induce DNA damage in intestinal epithelial cells through the release of reactive oxygen species. They also directly promote genetic instability and cellular proliferation, thereby accelerating colorectal tumor progression. 3 Blood tests measuring neutrophil and lymphocyte counts provide cost-effective insights into systemic inflammation status and the balance between these cell types—crucial for immune regulation. The ratio of these counts (known as the NLR) has emerged as a more predictive indicator than individual parameters. A Korean study revealed that patients with an NLR of ≥2.5 showed significantly higher adenoma detection rates compared with those with an NLR of <2.5, demonstrating a strong correlation between elevated NLR and colorectal adenoma risk. 4 As a biomarker of systemic inflammation, NLR reflects the equilibrium in human tumor immune responses dominated by NEUs and lymphocytes. Lymphocytes, particularly T cells, 5 are considered central to anti-tumor immunity. Tumor-infiltrating lymphocytes play vital roles in immune landscapes, 6 whereas CD4+ T cells are essential for developing and maintaining effective anti-tumor immunity. Live CD4+ T cells secrete interleukin (IL), which directly activates CD8+cytotoxic T lymphocytes and collaboratively inhibits tumor development by driving their effector function, differentiation, and proliferation. 7 An elevated NLR is significantly associated with a higher risk of colorectal adenomas.
RDW reflects the heterogeneity of RBC volume. Elevated RDW is closely associated with chronic inflammation, oxidative stress, and nutritional deficiencies. In chronic inflammatory states, pro-inflammatory factors inhibit erythropoietin activity, leading to impaired erythrocyte maturation and increased heterogeneity. Elevated RDW may be linked to oxidative damage in the intestinal mucosa, promoting adenoma formation. Oxidative stress can cause erythrocyte membrane damage, further increasing RDW. 8 The combination of RDW and NLR may provide more effective diagnostic value for colorectal tumors. 9 RDW has been demonstrated to have a significant correlation with colorectal cancer development. 10 Although existing studies have not directly explored the relationship between RDW and colorectal adenomas, it may influence the progression of adenomas through inflammatory and oxidative stress mechanisms. The combination of high RDW and high NLR may indicate more severe immune-metabolic disorders, collectively increasing the risk of adenoma recurrence. During chronic inflammation, IL-6 produced by inflammatory cells can elevate heparin levels. 11 Since iron serves as a raw material for erythrocyte production, RDW also increases. Moreover, in certain systemic inflammatory diseases such as rheumatoid arthritis and chronic kidney disease, 12 both indicators may rise simultaneously, suggesting they could serve as biomarkers related to inflammation and oxidative stress.
PDW is a hematological parameter reflecting platelet size heterogeneity. Elevated PDW typically indicates platelet activation and abnormal coagulation status. Tumors can promote platelet production and activation through IL-6 signaling, which manifests as increased PDW. Activated platelets stimulate angiogenesis in adenomas by releasing vascular endothelial growth factor and platelet-derived growth factor, whereas simultaneously secreting TGF-β that suppresses the anti-tumor function of CD4+ T cells. 13 Tumors may utilize platelets to facilitate their growth and metastasis. Conversely, proliferating tumors can enhance platelet production and activation, potentially creating a positive feedback loop that accelerates tumor progression.
Multiple studies have demonstrated that elevated RDW, increased NLR, and abnormal PDW are significantly associated with the occurrence, progression, and carcinogenic risks of colorectal adenomas. In chronic inflammatory states, NLR levels tend to rise. Furthermore, colorectal tumor patients often exhibit abnormal nutrient metabolism, iron metabolism disorders, or folic acid deficiency—factors that impair erythropoiesis and promote abnormal platelet activation. 14 The simultaneous activation of IL-6 in neutrophils suppresses lymphocyte proliferation, inhibits erythrocyte maturation, and enhances platelet activation, thereby creating a microenvironment conducive to adenoma development. This imbalance between pro-inflammatory and anti-inflammatory mechanisms plays a crucial role in cancer formation and progression.15–17 These three factors may contribute to the pathological process of adenomas through mechanisms involving inflammatory responses, oxidative stress, and platelet activation.
The ROC curve analysis of the predictive model based on NLR showed: For NLR, AUC = 0.888 (0.827–0.948), Youden index = 0.684, with sensitivity of 91.7% and specificity of 76.7%. The ROC curve analysis of the predictive model based on PDW indicated: AUC = 0.795 (0.716–0.874), Youden index = 0.484, sensitivity of 51.7%, and specificity of 96.7%. The ROC curve analysis of the predictive model based on RDW revealed: For RDW, AUC = 0.868 (0.806–0.931), Youden index = 0.584, with sensitivity of 76.7% and specificity of 81.7%. These results confirm that NLR, PDW, and RDW possess independent predictive value for colorectal adenoma.
Compared with endoscopic examinations, these biomarkers are more accessible, non-invasive, highly reproducible, non-invasive, and cost-effective, making them suitable for large-scale population screening. The combined detection of RDW+NLR+PDW may help identify high-risk patients. As a preliminary screening method, individuals with abnormal indicators should be prioritized for colonoscopy to reduce unnecessary invasive procedures. Dynamic monitoring of NLR, PDW, and PDW can provide early warnings of adenoma recurrence or malignant transformation (every three months).
Additionally, this study has the following limitations: (a) selection bias may occur in retrospective studies; (b) factors such as diet, lifestyle, and gut microbiota were not included; (c) non-specificity: infections and autoimmune diseases may interfere with results; (d) population differences: baseline values in elderly populations are relatively higher, requiring threshold adjustments; and (e) this study only demonstrates predictive value for colorectal adenomas without exploring related mechanisms, and experimental group stratification was not conducted due to sample size limitations. Future research directions are as follows: (a) multimodal integration: combining blood parameters, gut microbiota, and metabolomics to build an adenoma risk prediction model; (b) targeted intervention: attempting anti-inflammatory drug therapy (e.g. COX-2 inhibitors) for patients with high NLR to prevent adenoma development; and (c) artificial intelligence (AI)–driven dynamic monitoring: developing AI-based early warning systems using RDW/NLR/PDW to assess adenoma progression risks in real-time.
Summary: RDW, NLR, and PDW are clinically accessible blood parameters closely associated with colorectal adenomas, reflecting inflammation, oxidative stress, and pre-thrombotic states. Clinically, patients with elevated NLR, PDW, or RDW levels should be prioritized for attention. Early colonoscopy and intervention should be implemented to reduce the incidence of colorectal adenomas. Although non-specific limitations exist, their combined application can optimize adenoma screening and monitoring processes. Future efforts should integrate multidimensional data to enhance predictive accuracy.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251379263 - Supplemental material for Correlation between neutrophil-to-lymphocyte ratio, red blood cell distribution width, platelet distribution width, and colorectal adenoma: A retrospective cohort study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251379263 for Correlation between neutrophil-to-lymphocyte ratio, red blood cell distribution width, platelet distribution width, and colorectal adenoma: A retrospective cohort study by Runhan Liu and Fanglai Zhu in Journal of International Medical Research
Footnotes
Acknowledgments
The authors would like to thank Zhongsen Dou whose discussions and suggestions greatly contributed to the manuscript and for his valuable support.
Agreed to publish
Not applicable.
Conceptualization
Runhan Liu, Fanglai Zhu.
Contributions by authors
FZ conceived the case report and completed the protocol. RL prepared the initial draft of the paper. RL and FZ conducted the research, edited it, and finalized the manuscript. RL and FZ contributed equally. All authors read and approved the final version of the manuscript.
Data availability statement
De-identified data supporting this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
This study has been approved by the Ethics Committee of Anqing First People’s Hospital. The study is in accordance with the Helsinki Declaration (2024 Revision) and approved by the Ethics Committee of Anqing First People’s Hospital (approval number: AQYY–YXLL–LWLL–24). The report complied with the STROBE guidelines (von Elm et al., Ann Intern Med 2007).
Funding
Fanglai Zhu.
Investigator
Runhan Liu.
Method
Runhan Liu.
Resource
Fanglai Zhu.
Instructor
Fanglai Zhu.
Written–first draft: Runhan Liu.
Writing—reviewed and edited by Fanglai Zhu.
Express ones thanks
Not applicable.
Informed consent form of the patient
The authors declare that they have obtained all necessary consent forms from the patients. In these consent forms, the patients have agreed that their images and other clinical information will be published in the journal. The patients understand that their names and initials will not be published and will be kept confidential to the extent possible, but complete anonymity cannot be guaranteed.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
