Abstract
The association of platelet-to-lymphocyte ratio (PLR) with the clinicopathological features and prognosis in patients with breast cancer was evaluated. Related studies were searched from PubMed, Embase, Cochrane Library, and Web of Science up to July 1, 2021. Then, basic characteristic and prognostic data were extracted from the included studies. We synthesized and compared primary outcomes such as overall survival. Subgroups analyses in pathology, geographical area, follow-up time, and sample size were conducted. The pooled hazard ratio (HR), odds ratio (OR), and 95% confidence interval (CI) served as measures to assess the relationship of PLR with prognosis and clinicopathological features of breast cancer patients. After literature retrieval and selection, 20 studies with 7484 patients were included in this meta-analysis. High PLR was significantly related to poor overall survival (HR = 1.88; 95% CI 1.61, 2.19; P < 0.001) in breast cancer patients. Also, high PLR was associated with lymph node metastasis (LNM) (OR = 1.82; 95% CI 1.32, 2.52; P < 0.001), advanced tumor-node-metastasis (TNM) stage (OR = 1.89; 95% CI 1.25, 2.87; P = 0.003), and distant metastasis (OR = 1.76; 95% CI 1.14, 2.72; P = 0.01) in breast cancer. The stability and reliability of results in this meta-analysis were confirmed by sensitivity analysis. Elevated PLR is related to a poor prognosis and a higher risk of LNM, advanced TNM stage, and distant metastasis in breast cancer patients. Therefore, PLR can be identified as a biomarker with potential prognostic value in breast cancer.
Introduction
Breast cancer is the second leading cause of cancer death among women. 1 Recent studies have confirmed that tumor-related inflammation is a critical component of the tumor microenvironment, and inflammatory cells may contribute to the occurrence, development, and prognosis of cancer.2,3 Systemic inflammatory response is associated with tumor progression and affects the human host immune response to malignant tumors. 4
Platelets adhere to tumor cells to form clumps, protecting tumor cells from the high flow shear stress and immune attack. 5 In addition, they can also release vascular endothelial growth factors and a variety of cytokines, promote tumor progression and metastasis, and assist tumor cell immune escape.5,6 Lymphocytes play a crucial role in inducing cytotoxic death, tumor cell proliferation and migration, and producing the host anti-tumor immune response. A decrease in the number of lymphocytes will lead to the decline of lymphocyte-mediated anti-tumor immune response. 7
Platelet-to-lymphocyte ratio (PLR) is an informative marker reflecting changes in platelet and lymphocyte counts and predicting worse prognosis in various tumors.8–10 However, current studies on the prognostic prediction of breast cancer by PLR are inconsistent, and the sample sizes are small.11–14 Considering the heterogeneity and potential value of PLR, existing literature on PLR and breast cancer was retrieved and further analyzed to explore the prognostic value of this parameter in patients with breast cancer.
Methods
Search strategy
The search was conducted in PubMed, Embase, Cochrane Library, and Web of Science from inception to July 1, 2021. The published studies in English on the relationship between PLR and prognosis of breast cancer were selected. Following keywords for the online search in these databases were included: ("platelet-to-lymphocyte ratio" OR "PLR" OR "platelet-to-lymphocyte") AND ("breast carcinoma" OR "breast cancer" OR "breast tumor" OR "breast neoplasm") AND ("prognosis" OR "prognostic" OR "survival" OR "outcome" OR "recurrence”). Two researchers independently searched the literature and finally cross-checked to reach an agreement.
Inclusion and exclusion criteria
The inclusion criteria were as follows: (a) the subjects were breast cancer patients; (b) studies with participants divided into two groups of high and low PLR level; (c) articles that explored the association between PLR expression and of overall survival (OS) of breast cancer; (d) each trial with sufficient data such as hazard ratio (HR) and corresponding 95% confidence interval (CI) were used for the computation of pooled HR and 95% CI.
Exclusion criteria were as follows: (a) reviews, letters, case reports, and non-human subject research; (b) articles with missing and ambiguous data; (c) Newcastle–Ottawa Scale (NOS) score <6; (d) follow-up <60 months.
Quality assessment
The quality of the included study was evaluated according to the NOS. 15 The study was scored from object selection, comparability, outcome, and exposure with a maximum score of 9 points. A score of ≥6 points was regarded as high-quality literature. Two researchers discussed in case of disagreement, and a third researcher was invited to participate in the discussion and achieve a consistent result.
Data extraction
Studies were screened strictly according to inclusion and exclusion criteria. Two researchers then extracted data and evaluated study quality independently. The following information was extracted from the original literature: (a) basic characteristics: first author, country, publication year, sample size, pathology, follow-up time; (b) clinicopathologic parameters: case number in high and low PLR level groups after stratification by age, lymph node metastasis (LNM), tumor-node-metastasis (TNM) stage, and tumor size; (c) measures of prognosis: HR and its 95% CI of OS.
Statistical analysis
For the meta-analysis, pooled HR and OR was calculated by Review Manager 5.3 software (The Cochrane Collaboration, Copenhagen, Denmark) and STATA 12.0 (College Station, TX, USA). If the eligible study only provided Kaplan–Meier survival curve data, the Engauge Digitizer 4.1 was used to extract survival information such as HR and 95% CI from the graph.16,17 Heterogeneity among studies was assessed by according to the size of statistic I2. If I2 ≥ 50%, or P ≤ 0.05, there was significant heterogeneity among the included studies, and then a random-effects model was used. If I2 < 50%, or P > 0.05, there was no significant heterogeneity that existed among the pooled data, and a fixed-effects models was selected. 18 For determining the source of heterogeneity, subgroup analyses were conducted in terms of pathology (human epidermal growth factor receptor 2 (HER2)-positive vs. -negative groups), geographical region (Asian vs. non-Asian groups), follow-up time (>120 vs. <120 months) and sample size (n > 375 vs. <375). Pooled odds ratio (OR) and 95% CI were used to evaluate the relationship of PLR with LNM, TNM stage, and distant metastasis.
Publication bias was assessed by examining the funnel plot of each outcome. Then, Egger's test was used to further examine the results in which the natural logarithmic relative risk was plotted with SE. To evaluate the impact of each study on the estimated HR, a sensitivity analysis was performed to re-evaluate the pooled HR by omitting each study. P < 0.05 was considered statistically significant.
Results
Characteristics of eligible studies
A flow diagram of the literature screen and selection is shown in Figure 1. A total of 3422 studies were retrieved initially according to the retrieval strategy, and then 2980 pieces (repeated publications, reviews, abstracts, case reports, non-prognostic- and non-malignant-tumor-related literatures) were excluded. After reading the full text, 18 articles without relevant indicators of survival outcome were excluded; finally, 20 articles were included.19–38 The characteristics of the included studies are summarized in Table 1. The 20 studies19–38 were published between 2014 and 2021 with the sample size ranging from 62 to 1083; 12 studies were carried out in Asian countries and 8 studies in other regions. Pathological types included HER2 positive and negative. Multivariate analysis was adopted in 19 studies.19–38 The follow-up months ranged from 60 to 200 months in these studies, with NOS scores of 6–8 points (Table S1).
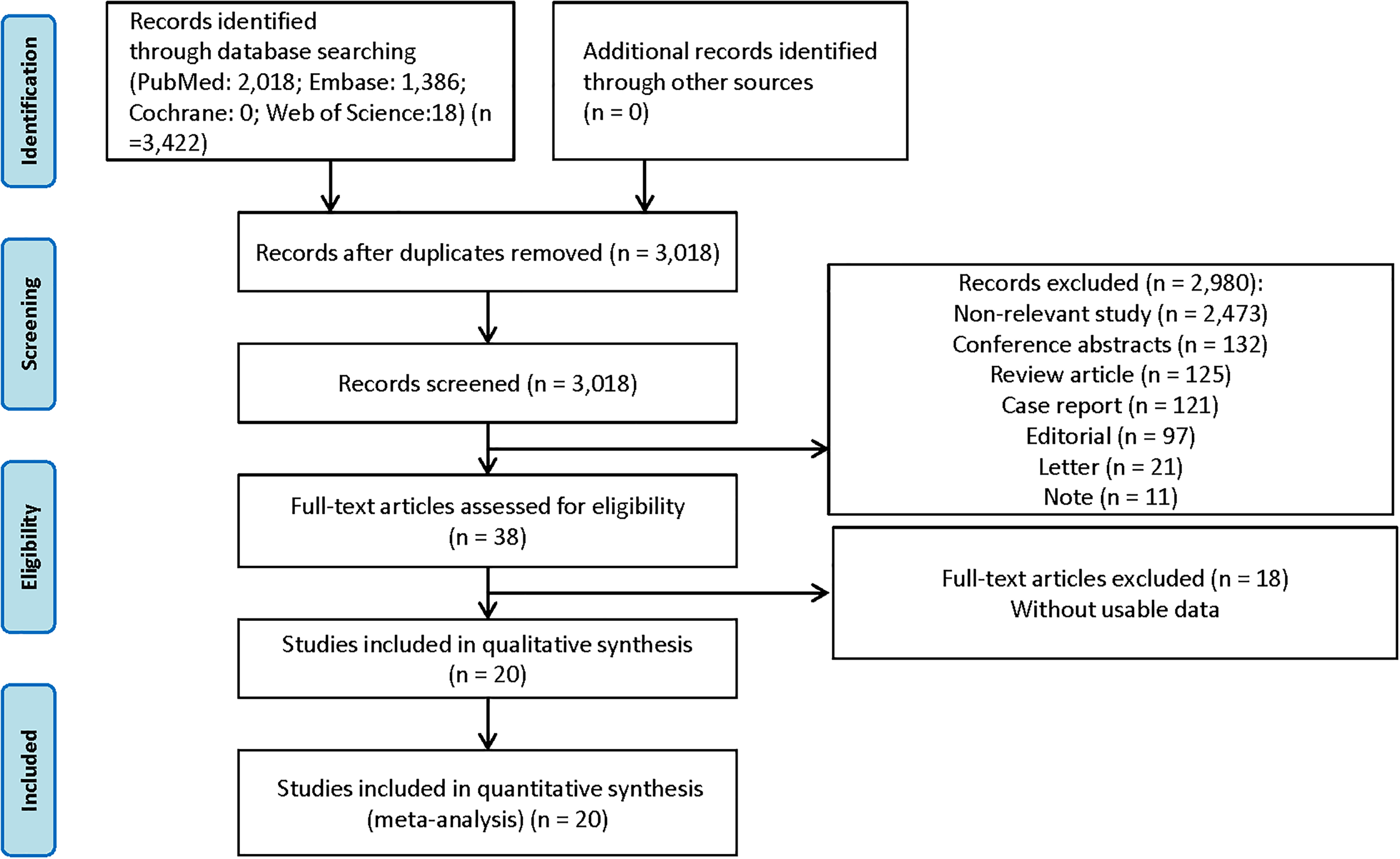
The flow diagram of literature selection.
Characteristics of studies in this meta-analysis.
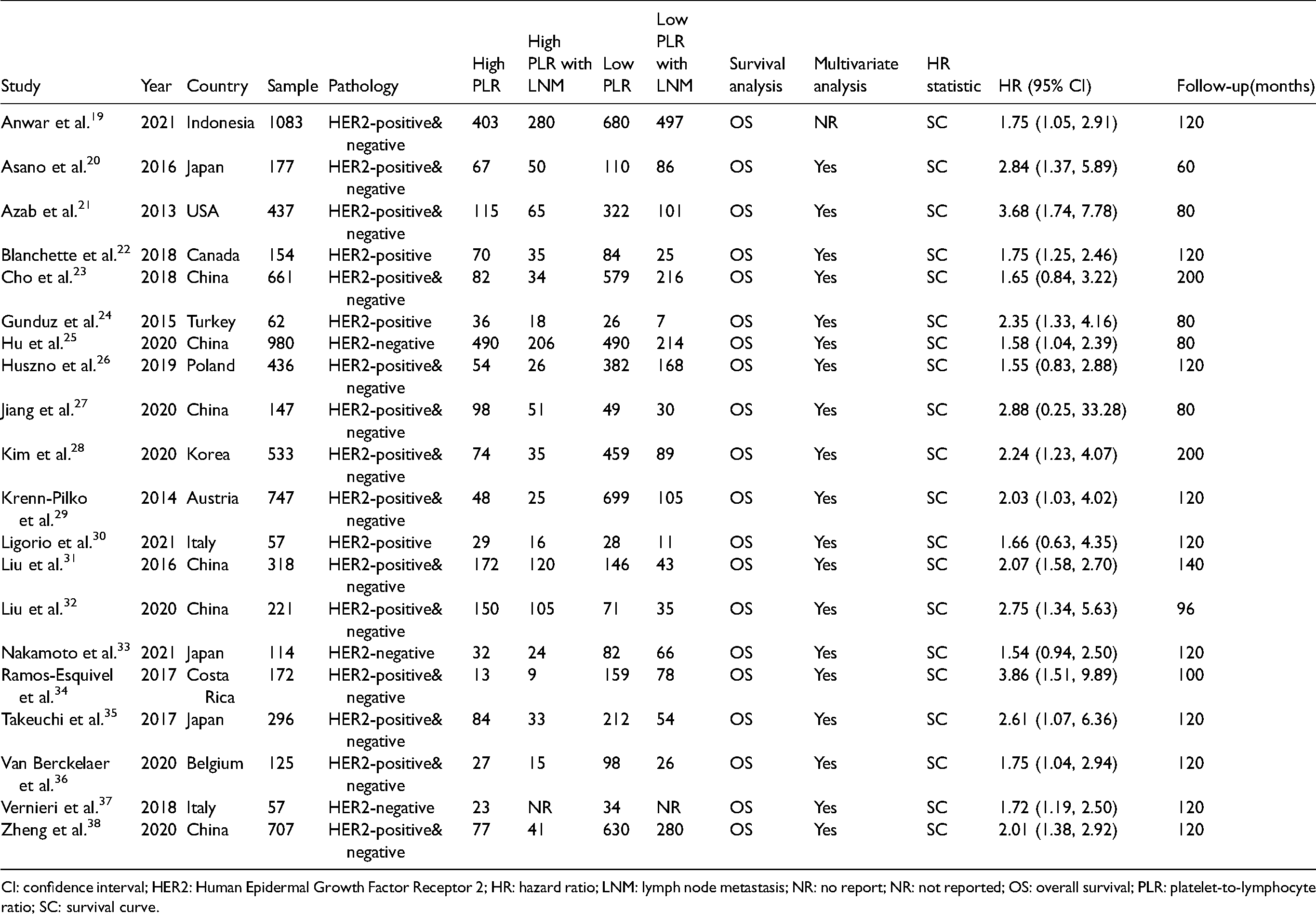
CI: confidence interval; HER2: Human Epidermal Growth Factor Receptor 2; HR: hazard ratio; LNM: lymph node metastasis; NR: no report; NR: not reported; OS: overall survival; PLR: platelet-to-lymphocyte ratio; SC: survival curve.
Association between PLR and prognosis of breast cancer patients
The 20 studies had OS as the endpoint, and 7484 patients reported the correlation between PLR level and OS in breast cancer.19–38 Pooled HR and 95% CI of OS data were collected from the included studies. Using the a random-effects model (I2 = 36%, P = 0.05), the result indicated that a high level of PLR in breast cancer patients was related to poor OS (pooled HR = 1.88; 95% CI 1.61, 2.19; P < 0.001) (Figure 2). In other words, high PLR was an adverse factor for the prognosis of breast cancer patients.
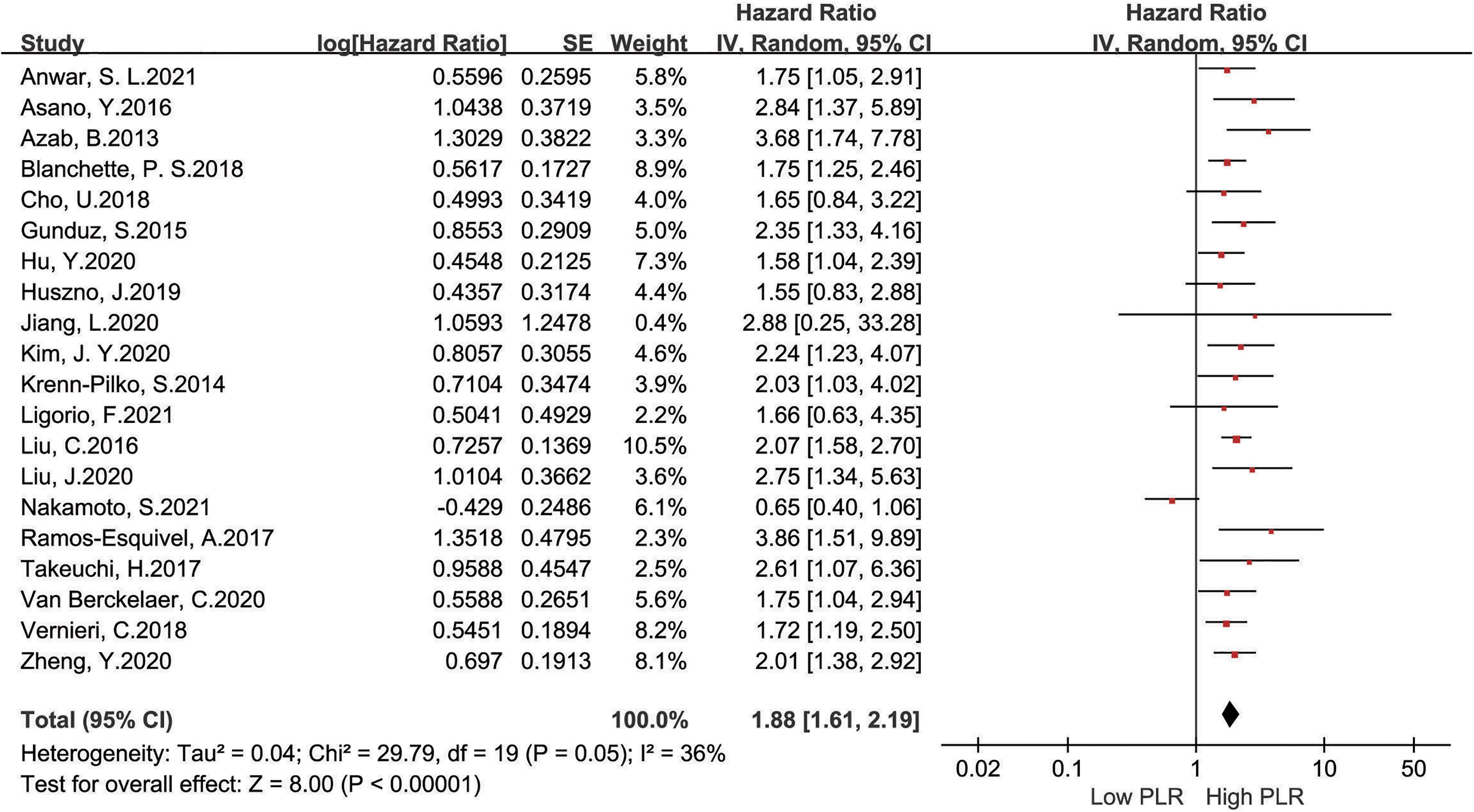
Forest plots of PLR expression and overall survival risk in breast cancer.
Subgroup analysis
The above results confirmed that high PLR in cancer tissues was a significant biomarker for the prognosis of breast cancer. As the moderate heterogeneity, we also analyzed the pathology, geographical region, follow-up time, and sample size by subgroup stratification (Table S2). In term of pathology, PLR was a prognostic factor in HER2-positive (HR = 1.87; 95% CI 1.42, 2.47; P < 0.001) and HER2-positive and -negative groups (HR = 2.10; 95% CI 1.81, 2.43; P < 0.001) with no heterogeneity (I2 = 0.0%; P > 0.1). PLR also showed a predictive value in the non-Asian groups (HR = 1.88; 95% CI 1.56, 2.27; P < 0.001) with no heterogeneity (I2 = 0.0%; P > 0.1). Subsequently, we found that PLR could act as a prognostic factor in groups with a follow-up time of < 120 months (HR = 2.28; 95% CI 1.78, 2.91; P < 0.001) and sample size > 375 cases (HR = 1.90; 95% CI 1.57, 2.29; P < 0.001) with no heterogeneity (I2 = 0.0%; P > 0.1).
Risk of bias and sensitivity analysis
The result of Egger’ test (Figure 3(a)) indicated no statistical significance in publication bias (P = 0.302). Further, after excluding any studies on the relationship between PLR and OS in breast cancer patients, no obvious changes of the pooled HR were found, suggesting that the findings were relatively stable and reliable in this meta-analysis (Figure 3(b)).
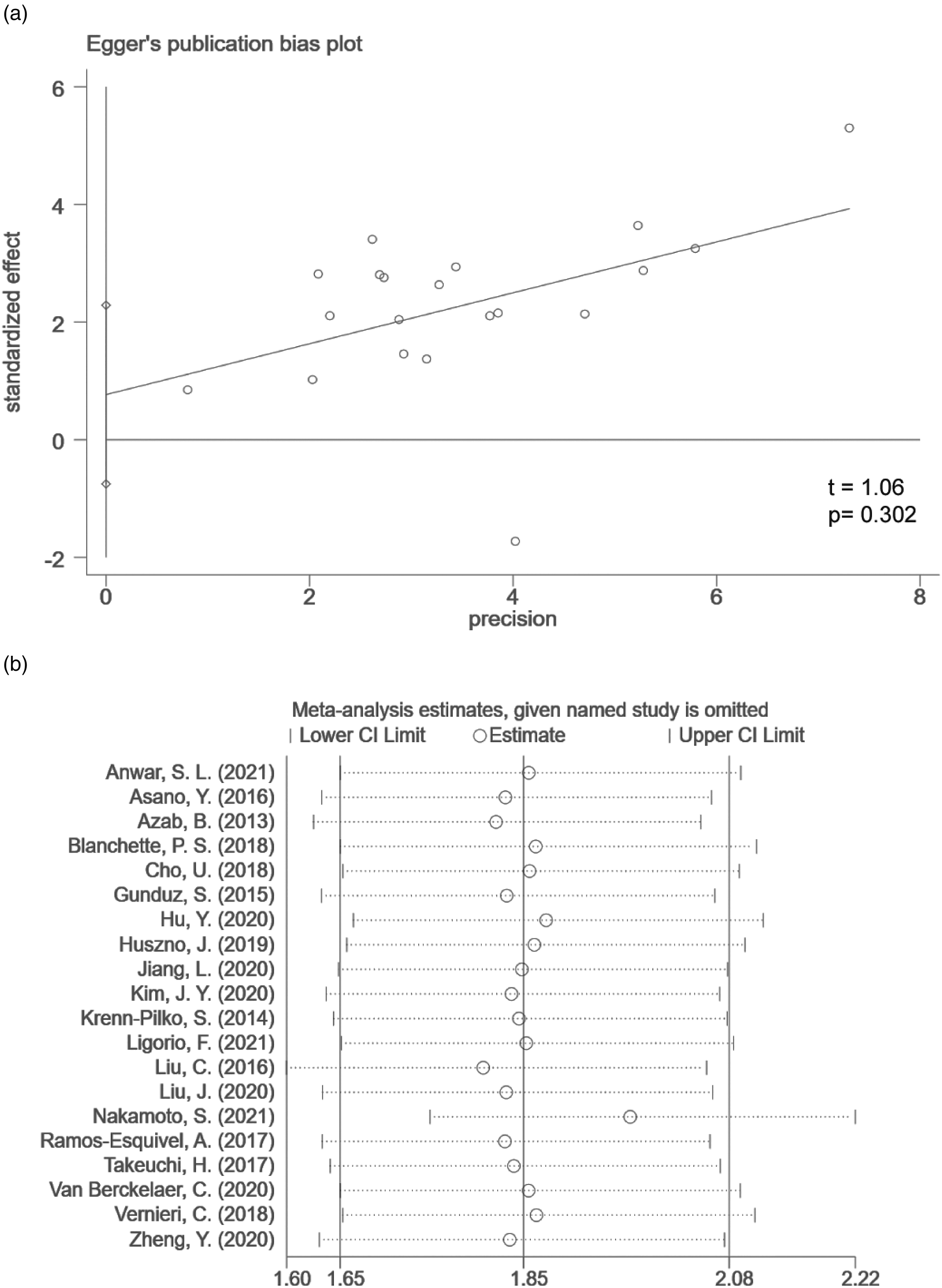
Publication bias and sensitivity analysis of PLR expression and overall survival risk in breast cancer. (a) Egger's test for publication bias; (b) sensitivity analysis for the robustness of the findings.
Association between PLR and clinicopathological features in breast cancer patients
To analyze the association between PLR and clinicopathological characteristics in breast cancer patients, we pooled the results in studies with stratification of age,19,20,25,27,29–32,35–37 LNM,19–37 TNM stage,19,20,22,24,25,27,29–32,34–37 tumor size,19,20,24,29,31,32,35,37 and distant metastasis19,20,24,29,31,32,35,37 (Table S3). The obtained results indicated no significant association of PLR with age (OR = 0.87; 95% CI 0.66, 1.16; P = 0.34) (Figure 4(a)), and tumor size (OR = 1.41; 95% CI 0.98, 2.02; P = 0.06) (Figure 4(c)). Remarkably, high PLR was significantly correlated with LNM (OR = 1.82; 95% CI 1.32, 2.52; P < 0.001; I2 = 85%; P < 0.01) (Figure 4(b)), advanced TNM stage (OR = 1.89; 95% CI 1.25, 2.87; P = 0.003; I2 = 84%; P < 0.01), and distant metastasis(OR = 1.76; 95% CI 1.14, 2.72; P = 0.01; I2 = 69%; P < 0.01) (Figure 4(c) and (d)) with significant heterogeneity.
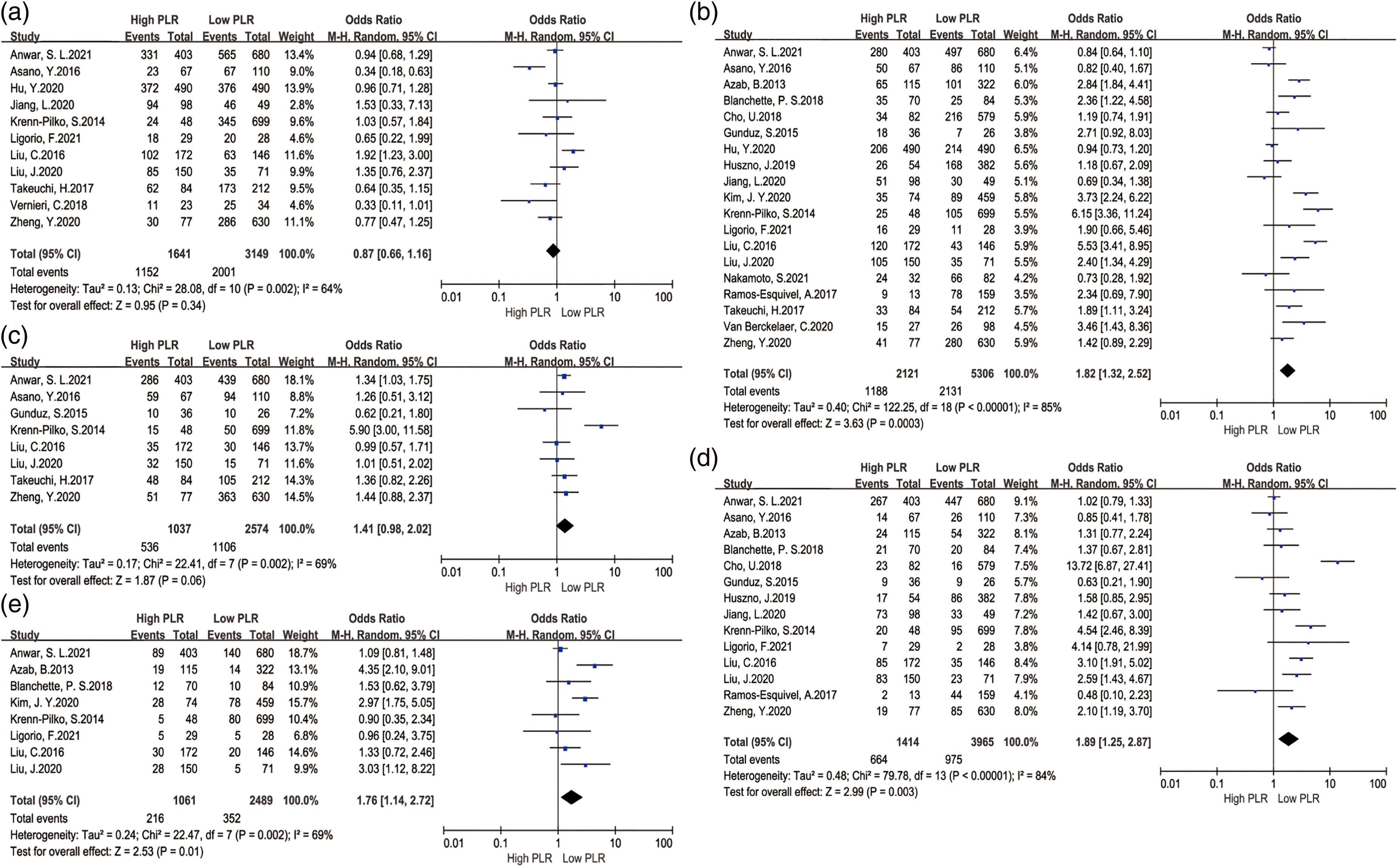
Forest plots of PLR expression and clinicopathological features in breast cancer. (a) Age ( > 50 years old vs. < 50 years old); (b) LNM (Yes vs. No); (c) tumor size (>2cm vs.<2 cm); (d) TNM stage (III–IV vs. I–II); (e) distant metastasis.
For significant heterogeneity, we also conducted a subgroup analysis stratified for geographical region, follow-up time, and sample size (Figure S1). After stratification by geographical region, we observed that PLR was a prognostic factor in LNM in the non-Asian group (HR = 2.00; 95% CI 1.46, 2.75; P < 0.001) with low heterogeneity (I2 = 34.6%; P > 0.1) (Figure S1(a)). However, heterogeneity did not change in the subgroups of follow-up time and sample size (Figure S1(b) and (c)). Taking the limited number of included studies into consideration, the subgroup analysis regarding distant metastasis was not performed.
Additionally, the funnel plot was used for testing publication bias between PLR level and clinicopathologic features in breast cancer, and presented in a roughly symmetrical manner, without obvious evidence of asymmetry (Figure S2).
Discussion
Inflammation plays a vital role in the occurrence and development of tumors. 39 Neutrophils, monocytes, platelets, and lymphocytes in peripheral blood are closely related to tumor cell invasion, metastasis, and angiogenesis. 40 During inflammation, the secretion of cytokines can stimulate the release of neutrophils, thus enhancing the invasion, proliferation, and metastasis of cancer cells.40,41 In addition, neutrophils are involved in tumor proliferation and metastasis by releasing inflammatory factors. 42 Lymphocytes influence the growth of tumor cells and improve the prognosis of patients with malignant tumors by secreting interferon-gamma and tumor necrosis factor-α. 43 Platelets protect the circulating tumor cells in the peripheral blood from flow shear stress, induce the epithelial-mesenchymal transition, and promote the tumor cells to extravasate to the metastatic site. 44 Studies have demonstrated that cancer cells overproduce proinflammatory mediators, such as IL-6 and IL-10, and increase the count of neutrophils and platelets in peripheral blood.45,46 Changes in these parameters are widely classified as signs of systemic inflammation and have been confirmed as prognostic indicators in many cancer types, such as absolute platelet count, neutrophil-lymphocyte ratio (LNR), lymphocyte-monocyte ratio (LMR), and PLR.47,48 As easy-to-measure, simple, and non-invasive markers of subclinical inflammation, the LNR and PLR have prognostic effects on many cancer types. 48 However, other studies have shown that LNR cannot be regarded as an independent prognostic factor for breast cancer. 49 Similarly, it has been reported that LMR is associated with the survival rate of several cancers, and higher LMR levels contribute to better disease-free survival rate among breast cancer patients receiving neoadjuvant chemotherapy. 50 In a study to predict the long-term survival rate of triple negative breast cancer patients, LNR was superior to LMR, but this study did not compare the prediction effect of PLR and LNR. 51 In other studies, the superiority of LNR was proved in predicting poor prognosis of metastatic colorectal cancer after hepatectomy; 52 as an independent prognostic factor, the increase in preoperative PLR was also superior to LNR and LMR in predicting the clinical outcome in breast cancer patients.23,53 However, the relationship between PLR and the prognosis of patients with breast cancer is still controversial.54,55
This meta-analysis included 20 articles on the association between PLR and OS of breast cancer patients. According to the results, breast cancer patients with high PLR had a higher proportion of TNM, distant metastasis, and LNM than those with low PLR (P < 0.05). The OS of breast cancer patients with high PLR was shorter than those with low PLR (pooled HR = 1.88; 95% CI 1.61, 2.19; P < 0.001), which is consistent with previous studies. Compared with previous meta-analyses, 56 this study embraced more comprehensive literature retrieval and a larger sample size. Publication bias has a great influence on the reliability and authenticity of meta-analysis results, which often leads to over-estimation of the comprehensive effect of the analysis. 57 The funnel plot is the most commonly used method to determine whether there is publication bias 58 and the linear regression model (Egger's test) is adopted to test the symmetry of the inverted funnel plot 59 If the P-value corresponding to the test statistic t is greater than 0.1, it can be inferred that the funnel plot is symmetrical, otherwise it is asymmetrical. The funnel plot in a symmetrical manner suggests no publication bias in the studies included in the meta-analysis, or at least the potential bias has no substantial influence on the final result. According to the funnel plot of this meta-analysis (Figure 3 and Figure S2), our obtained results were credible and reliable. Obviously, it is meaningful to determine the reasons for the inconsistency of research results; the quality heterogeneity between different studies may be an important reason for the heterogeneity.
However, the results of this study should be interpreted carefully, and there are still several limiting factors that need to be considered. First, the cut-off value of PLR in this study is inconsistent among the studies, which may be the introduction of selection bias in the meta-analysis. Second, the breast cancer patients involved in the study may receive different treatments, which may have an impact on survival rates. Third, patients included in the studies received follow-up at different time periods, which may have some influence on survival data. Finally, an international multi-center study with a larger sample size is required for further confirmation in the future.
Conclusion
The increase of PLR is related to poor prognosis, higher risk of LNM, advanced TNM stage, and distant metastasis in breast cancer patients. Further subgroup analysis confirms the negative prognostic value of PLR for the HER2-negative breast cancer patients. Therefore, PLR, as a clinically accessible index, should be identified as a biomarker with potential prognostic value in breast cancer.
Supplemental Material
sj-jpg-1-jbm-10.1177_03936155221118098 - Supplemental material for Platelet-to-lymphocyte ratio associated with the clinicopathological features and prognostic value of breast cancer: A meta-analysis
Supplemental material, sj-jpg-1-jbm-10.1177_03936155221118098 for Platelet-to-lymphocyte ratio associated with the clinicopathological features and prognostic value of breast cancer: A meta-analysis by Zhixun Gong, Ruomei Xin, Long Li, Liping Lv and Xinni Wu in The International Journal of Biological Markers
Supplemental Material
sj-jpg-2-jbm-10.1177_03936155221118098 - Supplemental material for Platelet-to-lymphocyte ratio associated with the clinicopathological features and prognostic value of breast cancer: A meta-analysis
Supplemental material, sj-jpg-2-jbm-10.1177_03936155221118098 for Platelet-to-lymphocyte ratio associated with the clinicopathological features and prognostic value of breast cancer: A meta-analysis by Zhixun Gong, Ruomei Xin, Long Li, Liping Lv and Xinni Wu in The International Journal of Biological Markers
Supplemental Material
sj-docx-3-jbm-10.1177_03936155221118098 - Supplemental material for Platelet-to-lymphocyte ratio associated with the clinicopathological features and prognostic value of breast cancer: A meta-analysis
Supplemental material, sj-docx-3-jbm-10.1177_03936155221118098 for Platelet-to-lymphocyte ratio associated with the clinicopathological features and prognostic value of breast cancer: A meta-analysis by Zhixun Gong, Ruomei Xin, Long Li, Liping Lv and Xinni Wu in The International Journal of Biological Markers
Supplemental Material
sj-docx-4-jbm-10.1177_03936155221118098 - Supplemental material for Platelet-to-lymphocyte ratio associated with the clinicopathological features and prognostic value of breast cancer: A meta-analysis
Supplemental material, sj-docx-4-jbm-10.1177_03936155221118098 for Platelet-to-lymphocyte ratio associated with the clinicopathological features and prognostic value of breast cancer: A meta-analysis by Zhixun Gong, Ruomei Xin, Long Li, Liping Lv and Xinni Wu in The International Journal of Biological Markers
Supplemental Material
sj-docx-5-jbm-10.1177_03936155221118098 - Supplemental material for Platelet-to-lymphocyte ratio associated with the clinicopathological features and prognostic value of breast cancer: A meta-analysis
Supplemental material, sj-docx-5-jbm-10.1177_03936155221118098 for Platelet-to-lymphocyte ratio associated with the clinicopathological features and prognostic value of breast cancer: A meta-analysis by Zhixun Gong, Ruomei Xin, Long Li, Liping Lv and Xinni Wu in The International Journal of Biological Markers
Supplemental Material
sj-docx-6-jbm-10.1177_03936155221118098 - Supplemental material for Platelet-to-lymphocyte ratio associated with the clinicopathological features and prognostic value of breast cancer: A meta-analysis
Supplemental material, sj-docx-6-jbm-10.1177_03936155221118098 for Platelet-to-lymphocyte ratio associated with the clinicopathological features and prognostic value of breast cancer: A meta-analysis by Zhixun Gong, Ruomei Xin, Long Li, Liping Lv and Xinni Wu in The International Journal of Biological Markers
Footnotes
Author contributions
GZX, XRM, WXN: Critical revision of the manuscript; GZX, XRM, WXN: Substantial contribution to the conception and design of the work, manuscript drafting; GZX, XRM, LL, LLP: Acquisition, analysis, and interpretation of the data; GZX, XRM, LL, LLP, WXN: Revising the manuscript critically, final approval of the version to be published. All authors have read and approved the final manuscript. Zhixun Gong and Ruomei Xin: equal contribution.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the Hainan Health and Family Planning industry scientific research project, (grant number 20A200399).
Availability of data and material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
